Abstract
To the Editor,
Recently in this journal, Nijns et al. 1 provided convincing data that polytetrafluoroethylene (PTFE) devices loaded with stem cell-derived pancreatic endodermal (hu-iPSC-PE) cells implanted in the subcutaneous (SC) tissue of athymic, immune-compromised (nude) rats engendered dense fibrotic capsule formation resulting in virtually no functional beta cells. This observation was remarkable in two aspects. First, their observations in the nude rat were in stark contrast to their earlier reports that the same type of implant (device and cells) did not provoke an FBR in both the athymic nude mouse 2 using human islets and in the severe combined immunodeficiency (SCID)/beige mouse3,4 with hu-iPSC-PE cells; instead the cells had proven capable of establishing a functional beta cell mass (FBM). Because similar devices and cells have led to fibrotic reactions in man 5 , they concluded that observations from the nude rat model are more clinically relevant than findings in either the nude or SCID mouse 1 . Second, and more broadly, they have demonstrated that the athymic nude rat is indisputably capable of mounting a foreign body response (FBR) to implanted devices containing cells. In the past, this assumption has often been challenged despite the knowledge that nude rats, while being T-cell deficient 6 , have normal B-cell populations. The FBR does not require T-cells7–9.
Here we write in support of the Pipeleers group’s conclusion regarding the use of the nude rat model for preclinical investigations of biocompatibility of encapsulated cells 1 . Furthermore, we extend their observations to biomaterials and devices devoid of cellular contents. As part of a more expansive study of membranes potentially useful for cell encapsulation, we implanted PTFE membranes in the dorsal SC tissues of male Crl:NIH-Foxn1 rnu athymic nude rats. The membranes or devices did not contain cells of any kind. After 3 weeks to 3 months, the membranes and surrounding SC tissue were explanted; formalin-fixed; paraffin-embedded; and stained with hematoxylin and eosin (H&E) or Masson’s trichrome stain. The presence or absence of a fibrotic capsule was noted and its extent quantified histologically.
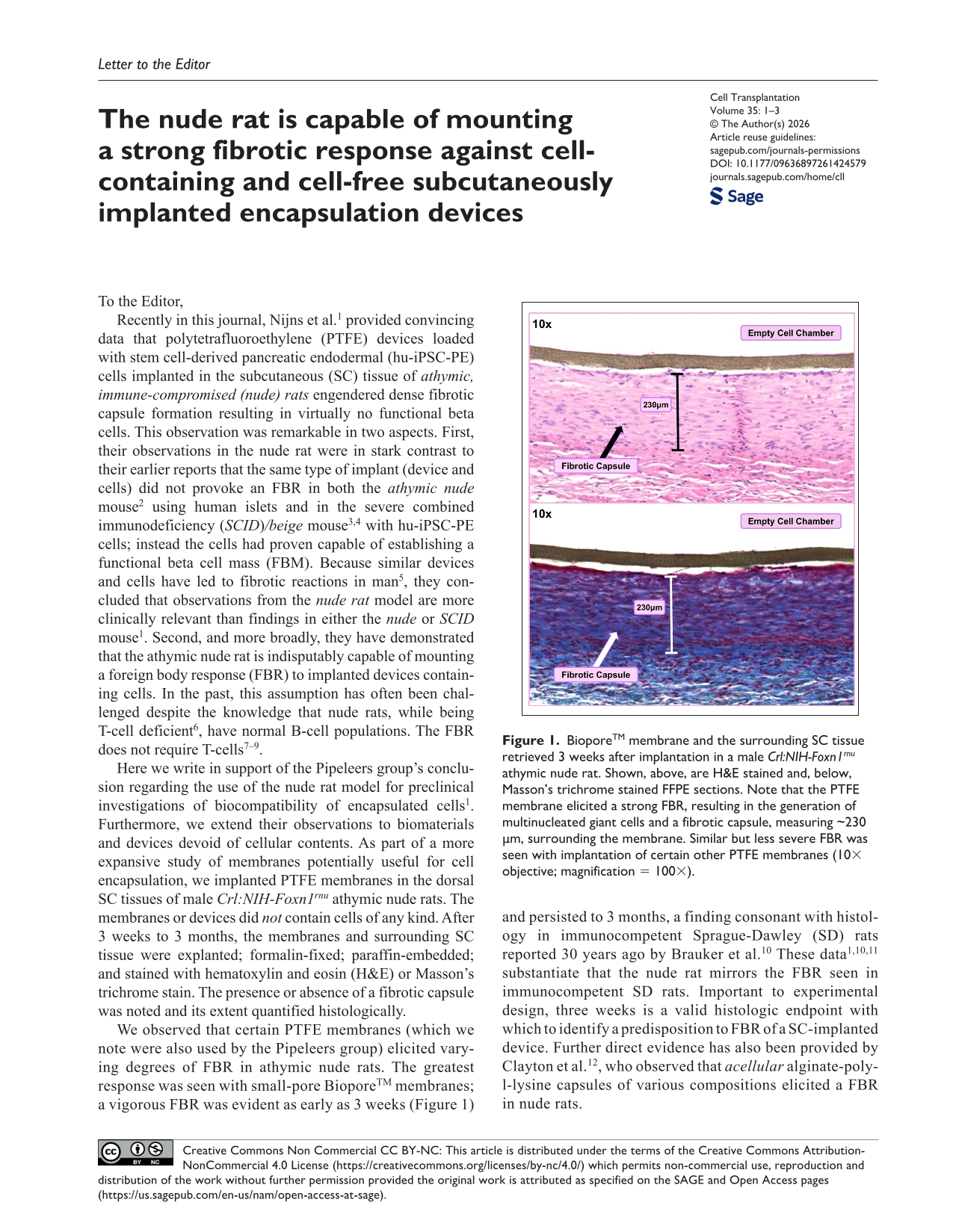
We observed that certain PTFE membranes (which we note were also used by the Pipeleers group) elicited varying degrees of FBR in athymic nude rats. The greatest response was seen with small-pore BioporeTM membranes; a vigorous FBR was evident as early as 3 weeks (Figure 1) and persisted to 3 months, a finding consonant with histology in immunocompetent Sprague-Dawley (SD) rats reported 30 years ago by Brauker et al. 10 These data1,10,11 substantiate that the nude rat mirrors the FBR seen in immunocompetent SD rats. Important to experimental design, three weeks is a valid histologic endpoint with which to identify a predisposition to FBR of a SC-implanted device. Further direct evidence has also been provided by Clayton et al. 12 , who observed that acellular alginate-poly-l-lysine capsules of various compositions elicited a FBR in nude rats.

BioporeTM membrane and the surrounding SC tissue retrieved 3 weeks after implantation in a male Crl:NIH-Foxn1 rnu athymic nude rat. Shown, above, are H&E stained and, below, Masson’s trichrome stained FFPE sections. Note that the PTFE membrane elicited a strong FBR, resulting in the generation of multinucleated giant cells and a fibrotic capsule, measuring ~230 µm, surrounding the membrane. Similar but less severe FBR was seen with implantation of certain other PTFE membranes (10× objective; magnification = 100×).
Fibrosis is the principal host defense against implanted devices, whether or not they contain cells 10 . Consequently, strategies to abrogate the FBR and animal models to evaluate the success—or failure—of these approaches, are critical elements of preclinical development of implantable devices (reviewed in Papas et al. 13 and Anderson et al. 14 ). We echo the findings reported by the Pipeleers group lending strong support to the choice of the athymic nude rat model rather than the nude or SCID mouse to foretell whether an adverse foreign body response (FBR) will occur in humans to either cell-containing or acellular implanted devices.
Footnotes
Acknowledgements
Not applicable.
Research Ethics
All experiments were performed with the written approval of and in accordance with the guidelines established by the Institutional Animal Care and Use Committee at The University of Arizona, Approval number 12-392, August 8, 2024.
Patient Consent
Not applicable.
Author Contributions
Made a significant contribution to the concept, design, acquisition, analysis or interpretation of data: All authors. Drafted the article or revised it critically for important intellectual content: KMP,TL, RCJ, CWP, KKP. Approved the final version of the article for publication: KMP, CWP, KKP. Agreed to be accountable for all aspects of the work and resolved any issues related to its accuracy or integrity: All authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Breakthrough T1D (formerly JDRF) [3-SRA-2023-1437-M-B].
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.K.P. declares that he is an Associate Editor of Cell Transplantation. R.C.J., T.L, and K.K.P. declare that they are co-founders of and have financial interests in Procyon Technologies LLC. All other authors declare that they have no conflicts of interest.
Data Availability Statement
All relevant data are included in the manuscript.
Statement of Human and Animal Rights
This article does not contain any studies with human subjects. The use of animals is addressed in “Research Ethics” (above).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
