Abstract
Chronic active Epstein–Barr virus (CAEBV) infection is a rare and highly lethal lymphoproliferative disorder. The pathological basis of this condition involves Epstein–Barr virus (EBV) persisting in hematopoietic stem cells, driving clonal expansion of T cells or natural killer (NK) cells, and subsequently triggering systemic inflammatory responses and multi-organ failure. Current treatment modalities, encompassing antiviral medications, immunosuppressants, and cytotoxic chemotherapy, offer only transient remissions, with the majority of patients ultimately experiencing relapse. Recent single-cell sequencing and chimera studies have confirmed that EBV-infected hematopoietic stem cells constitute the “seed” cell population for CAEBV initiation and maintenance. This finding indicates that allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only treatment known to date that can fully eradicate viral reservoirs and restore normal immunity, suggesting that it may represent a curative strategy. Nevertheless, transplantation timing, donor matching, conditioning intensity, and transplant-related complications have been shown to have a significant impact on long-term prognosis. The clinical decision-making process necessitates a high degree of individualization, incorporating molecular risk factors, disease activity, and comorbidities. Advancing research into the latent-lytic cycle regulation mechanisms of EBV, in addition to the clinical translation of small-molecule inhibitors targeting viral proteins and EBV-specific adoptive cell therapies, holds great promise for the future. One such potential avenue for future research is the development of an integrated “pre-transplant viral load reduction-post-transplant relapse prevention” strategy. This approach shows great potential in reducing transplant-related mortality and continuously improving survival outcomes for CAEBV patients.

Keywords
Introduction
Epstein–Barr virus (EBV), also known as Human Herpesvirus 4, is one of nine known human herpesviruses and one of the most common viruses. EBV is a double-stranded DNA virus surrounded by a lipid bilayer embedded with membrane glycoproteins that play a key role in viral infection and host-cell integration 1 . Research indicates that the global EBV infection rate is exceedingly high, with approximately 90% of the population becoming infected at some point in their lives 2 . The positivity rate of serum tests for EBV tends to increase with age, particularly in developing countries where the infection rate is notably high among children and immunocompromised populations 3 . Studies have shown an increase in the prevalence of EBV infection among children and adolescents in China between 2017 and 2022 4 . EBV is primarily transmitted through bodily fluids, particularly saliva, which facilitates its spread among individuals in close contact. Although EBV infections are often asymptomatic, most individuals mount an immune response to effectively control the virus. Consequently, many infected individuals remain asymptomatic carriers in adulthood. However, under certain circumstances, such as immune deficiency or specific genetic predispositions, chronic active Epstein–Barr virus (CAEBV) infection may occur. CAEBV infection is a rare condition characterized by the continuous proliferation of EBV-infected T or natural killer (NK) cells, leading to multi-organ involvement and systemic inflammatory responses. The clinical manifestations of CAEBV infection vary and include fever, hepatosplenomegaly, lymphadenopathy, skin lesions, and hematological disorders. Given the complex pathological mechanisms and limited efficacy of traditional treatment options, the prognosis of patients with CAEBV infection is generally poor. Recently, researchers investigating the origin of CAEBV infection employed flow cytometry combined with fluorescence in situ hybridization to label cells in the bone marrow and peripheral blood of patients with CAEBV infection and utilized quantitative polymerase chain reaction to analyze EBV-DNA copy numbers. The findings demonstrated that EBV infection extends beyond lymphocytes to include the bone marrow cells. Moreover, by tracing the differentiation pathways of EBV-positive cells within the bone marrow, the study revealed that cells across all lineage branches were infected with EBV, thereby further confirming that the infection originated from hematopoietic stem cells (HSCs) 5 . Thus, allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only therapeutic approach to effectively eradicate EBV-infected hematopoietic and immune cells in patients with CAEBV infection, thereby improving patient outcomes. However, allo-HSCT, which involves resetting the immune system, is associated with significant risks. Thus, selecting the appropriate timing and conditioning regimens is of paramount importance in the implementation of allo-HSCT. This review discusses the pathogenesis, diagnostic criteria, limitations of conventional therapies, and role of allo-HSCT in CAEBV infection management, including its current applications, therapeutic efficacy, and challenges.
Pathological mechanism of CAEBV infection
Infection and latency of EBV
During the initial infection, EBV enters B lymphocytes by binding its glycoprotein gp350 to CD21 or CD35 receptors on the surface of host cells, thereby facilitating infection 6 . Because T/NK cells do not express CD21, EBV does not directly infect these cells. Instead, the infection usually occurs through the coupling of EBV-infected B lymphocytes (Figure 1). Once inside the host cell, the EBV membrane fuses with the host cell membrane, releasing the viral genome into the nucleus where replication begins 7 . This process activates the proliferation and growth of B cells, allowing EBV to persist in a latent state within the host cell 8 . EBV exhibits various latency types, classified by distinct sequences of EBV nuclear antigen (EBNA)2. EBV latency is categorized into types I, II, and III, each of which influences the behavior of the virus within host cells and contributes to the development of various diseases 9 . Type I latent infections involve the expression of EBV proteins, including EBNA1 and BamHI-A rightward transcripts (BARTs), and are commonly linked to Burkitt’s lymphoma. In contrast, type II latent infection primarily involves expression of EBNA1, latent membrane protein (LMP)1, LMP2, BARTs, and Epstein–Barr encoding regions (EBERs) and is associated with Hodgkin’s lymphoma. All latent infection genes are expressed during type III latency, which is linked to lymphoproliferative diseases in immunosuppressed patients (Figure 1).
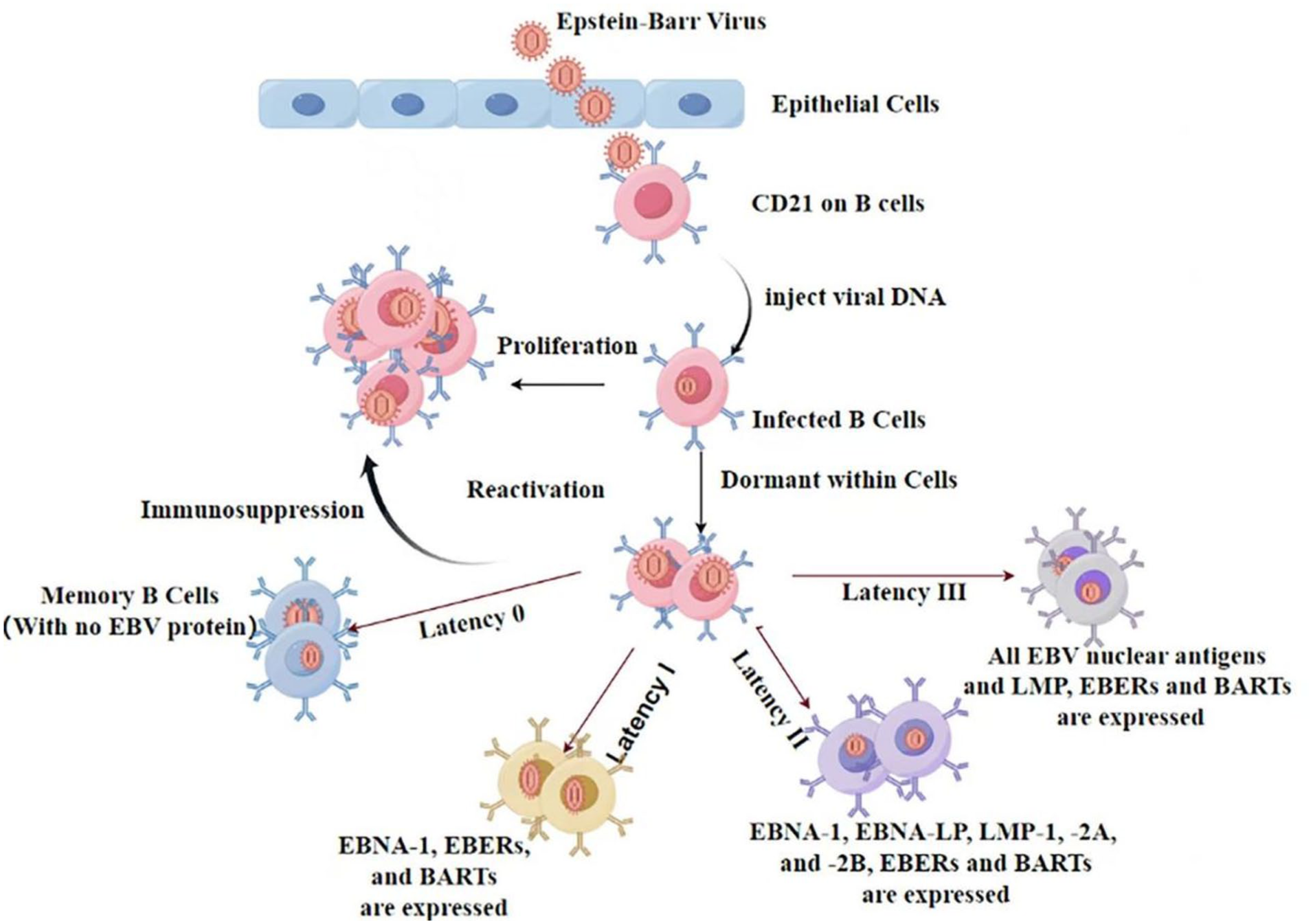
The schematic diagram illustrates the mechanisms of EBV infection and the characteristics of latency. During the primary infection, Epstein–Barr virus (EBV) binds to the CD21 receptor on host B lymphocytes via its glycoprotein gp350, thereby mediating cellular infection. EBV displays characteristic gene expression profiles across distinct latency stages: Latency Type 0 is characterized by the absence of viral protein expression. Latency Type I, which is observed in endemic Burkitt’s lymphoma, results in the expression of EBNA-1, EBERs, and BART transcripts. The latent phase II, evident in Hodgkin’s lymphoma, leads to the expression of EBNA-1, EBNA-LP, LMP-1/2A/2B, and EBERs/BARTs. Finally, the latent phase III, present in lymphoproliferative disorders of immunocompromised patients, expresses all latent infection-associated genes. This schematic diagram methodically unveils the pathogenetic link between disparate latent types and the clinical manifestations of associated diseases. Created with Figdraw.com.
It is noteworthy that recent studies have further elucidated the pathogenesis of CAEBV, demonstrating that it involves not only lymphocytes but is also closely associated with infection of HSCs at an earlier stage. Research has indicated that CAEBV may have its origins in infected hematopoietic HSCs 5 . During the process of differentiation, these EBV-latently infected HSCs have the capacity to transmit the virus to various blood cell lineages, including T cells, NK cells, and B cells. This process establishes a multilineage, persistent state of viral latency and activation. This mechanism provides a comprehensive explanation for the widely observed systemic inflammatory response, clonal proliferation, and multi-organ involvement in CAEBV. Furthermore, it offers a cellular origin-level explanation for the disease’s clinical characteristics of being difficult to cure and prone to recurrence. It is evident that the pathophysiology of CAEBV encompasses not only classic B-cell latent infection and subsequent immune cell infection, but more crucially, is founded upon systemic, persistent viral–host interactions established through hematopoietic stem cell infection. This contributes to a more profound comprehension of the origins and progression of the disease.
Immune escape mechanism of EBV
Establishes latency in B lymphocytes, during which time it expresses a limited number of antigens to minimize its detection by the host immune system. Downregulation of LMP2A facilitates the evasion of infected B cells from cytotoxic T lymphocyte (CTL) attack, as demonstrated in prior studies 10 . During the lytic phase, EBV expresses >80 proteins, including immunomodulatory proteins that regulate cytokine expression and suppress host immune responses, thereby facilitating immune evasion and viral particle production11–13. EBV has also been demonstrated to modulate host cell apoptosis through latent proteins, such as LMP1 and LMP2, which regulate multiple molecular and transcriptional pathways to promote viral latency. The LMP1 accumulation promotes cell survival while compromising immune-mediated clearance of the infection14,15. In patients with CAEBV infection, the number of LMP2-specific CTLs and the activity of EBV-specific CTLs are often suppressed. EBV-infected B lymphocytes express EBNA1, which inhibits the NK cell receptor ligand (ULBP1), thereby enabling infected B cells to evade recognition and attack by NK cells 13 . Furthermore, EBV induces cytokines and chemokines that suppress T-cell activation and proliferation, thereby weakening the host immune response. The pathogenesis of CAEBV infection is closely linked to immune evasion mechanisms of EBV-infected T or NK cells, and impaired immune function in patients with CAEBV infection further contributes to the inability to effectively eliminate EBV-infected cells.
Clinical manifestations and diagnostic criteria of CAEBV infection
CAEBV infection primarily affects children and adolescents, with ages ranging from 5 to 31 years and an average age of 8.3 years 16 . CAEBV infection is characterized by a persistent state of EBV infection, leading to systemic inflammation and malignant clonal proliferation of T or NK cells that is pathologically defined as lymphocytic tumors 17 . The main clinical manifestations of CAEBV infection include inflammation, persistent or recurrent fever, liver insufficiency, thrombocytopenia, anemia, and rashes. Fever is the most common symptom, affecting approximately 91% of patients with CAEBV infection 18 . Owing to the clonal proliferation of T/NK cells, CAEBV infection can also cause invasive enlargement and functional failure of several vital organs, including the lymphatic system, liver, spleen, and central nervous system17,19. In contradistinction to acute EBV infection, chronic active EBV infection is characterized by a duration that is typically longer than 3 months. The disease progresses rapidly, often leading to severe cytokine release syndrome, hemophagocytic lymphohistiocytosis, and multiple organ dysfunction syndrome. This condition is associated with an elevated risk of both tumor progression and mortality, as established in prior studies.
CAEBV infection is potentially fatal and requires appropriate diagnostic and therapeutic interventions. Currently, the diagnosis of CAEBV infection relies on clinical characteristics and laboratory results for virus detection. According to the international consensus, the primary clinical criteria include the following: (1) continuous or recurrent symptoms of EBV infection (such as fever, hepatosplenomegaly, and rash) persisting for >3 months; (2) detection of EBV-DNA load in peripheral blood or infected tissues (EBV-DNA load ≥10,000 IU/mL); (3) confirmation of infiltrating EBV-infected T or NK cells in tissues using immunohistochemistry or flow cytometry; and (4) presence of chronic diseases that cannot be explained by other known disease processes at the time of diagnosis 17 . Furthermore, all four clinical criteria must be satisfied simultaneously for a CAEBV infection diagnosis. Commonly used ancillary diagnostic criteria include serological tests utilizing fluorescent antibodies to identify EBV-specific antibodies, such as VCA-IgG and EA-IgG. However, in some patients, EBV-specific antibodies may not be present at detectable levels, indicating high EBV activity. Therefore, the presence of EBV-specific antibodies is not essential for the diagnosis of CAEBV infection. Histopathological examination and flow cytometry can be used to assess T and NK cell infiltration in EBV-infected tissues. In addition, molecular biological techniques, such as quantitative polymerase chain reaction, can be used to measure the EBV-DNA load 17 .
Traditional treatment methods
Antiviral treatment
Viral reactivation is closely associated with CAEBV infection, and antiviral therapy remains a crucial approach for managing CAEBV infection. Commonly employed antiviral agents include nucleoside analogs, such as acyclovir, valacyclovir, ganciclovir, and valganciclovir, which exert their antiviral effects by inhibiting viral DNA synthesis 20 . Although these drugs can moderately inhibit the early replication of viral infections, their application in the treatment of CAEBV infection has not been widely accepted primarily because CAEBV infection predominantly involves the latent EBV infection.
Chemotherapy
Chemotherapy is the mainstay of treatment for CAEBV infection. Current strategies often follow a “three-step therapeutic approach” that integrates immunomodulation, multiagent chemotherapy, and allo-HSCT 21 . Step 1: Immune modulation with corticosteroids and immunosuppressants to attenuate hyperinflammation and address immune dysregulation. Step 2: Multiagent chemotherapy to reduce the burden of EBV-positive T/NK cells, minimize transplant-related complications, and optimize post-transplant outcomes. A modified CHOP regimen is commonly used as the first-line therapy. Step 3: Allo-HSCT, which is critical for eradicating EBV-infected cells and restoring the immune system because chemotherapy alone is insufficient for disease eradication. The sequential approach aims to balance symptom control, disease reduction, and curative intent in the management of CAEBV infection.
Immunotherapy
Interferon
In the management of CAEBV infection, immunomodulatory therapy is critical to regulate the patient’s immune status and enhance anti-EBV responses. Although immunosuppressive agents, such as corticosteroids and cyclosporine, can temporarily alleviate CAEBV infection symptoms, their long-term use may suppress immune responses against EBV and potentially promote the proliferation of virus-infected cells 22 . Interferon (IFN)-alpha has demonstrated some efficacy in inhibiting viral DNA replication 23 , and both IFNs (IFN-α and IFN-γ) and interleukin (IL)-2 have been reported in the management of CAEBV infection (Figure 2). However, currently, there is no evidence that these agents can achieve clinically satisfactory therapeutic outcomes 22 .

Cytokines (IL-6, IL-12, IFN-γ) boost antiviral signaling; JAK inhibitors dampen hyperactive JAK/STAT; PD-1 blockade mitigates T-cell exhaustion; mTOR inhibitors curb aberrant proliferation; gp350 CAR-T cells potently eliminate EBV-infected cells. IL, interleukin; IFN, interferon; EBV, Epstein–Barr virus; CTL, cytotoxic T lymphocyte; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; IFNGRs, interferon-γ receptors; EGFR, epidermal growth factor receptor; MHC, major histocompatibility complex; TCR, T-cell receptor; CAR-T cell, chimeric antigen receptor T-cell. Created with Figdraw.com.
JAK1/JAK2 inhibitors
It is evident that JAK1 and JAK2 play pivotal roles in the regulation of immune cell functions, including the development and activity of T and B cells. These cells mediate immune modulation through the facilitation of cytokine signals, such as IL-6, IL-12, and IFNs24,25. During viral infections, cytokines, such as IFN-α and IFN-β, activate the JAK/STAT signaling pathway, leading to the expression of antiviral genes and enhancement of the host’s resistance to viruses 26 . Consequently, JAK1/JAK2 inhibitors, such as ruxolitinib, have demonstrated significant therapeutic potential for CAEBV infection. A retrospective study evaluated the efficacy of ruxolitinib in 9 patients with CAEBV infection 27 . Of these patients, a total of seven exhibited a positive response to ruxolitinib therapy. It is worthy of note that a total of six patients successfully attained fever control within a 48-h period. Furthermore, aspartate transaminase and alanine transaminase levels were found to decrease in 75% of patients, and two patients suffering from cytopenia experienced a recovery. The median duration of sustained remission was 7.1 weeks (range, 3.4–101.0 weeks). In a similar manner to the pharmaceutical compound tofacitinib, which is an oral Janus kinase (JAK) inhibitor primarily employed in the treatment of autoimmune diseases, the JAK enzyme is inhibited, thus preventing the phosphorylation and activation of signal transducer and activator of transcription (STAT) proteins. This process serves to disrupt the JAK-STAT pathway. This inhibition reduces inflammatory responses 28 , indicating tofacitinib’s potential for treating inflammatory diseases including CAEBV. However, these agents remain under investigation for the treatment of CAEBV, with only small-sample, non-randomized studies available at present. Their long-term efficacy and safety require validation through larger, multicenter trials.
Programmed cell death protein 1 inhibitors
Programmed cell death protein 1 (PD-1) is an immune checkpoint protein primarily expressed on T cells, whereas its ligand, programmed death ligand 1 (PD-L1), is predominantly found on the surface of tumor cells. The interaction between PD-1 and PD-L1 induces lymphocyte apoptosis, enabling tumor cells to evade immune surveillance. In patients with CAEBV infection, upregulation of PD-L1 expression in infected cells leads to exhaustion of effector T cells, allowing the virus to escape host immune detection. Blocking PD-1 can restore T-cell activity and enhance the immune response against EBV (Figure 2). Retrospective studies on patients with CAEBV infection have revealed significant efficacy of PD-1 inhibitors, and among 16 patients with CAEBV disease, 12 demonstrated a response to PD-1 inhibitors. The median progression-free survival was 11.1 months (range, 4.9–54.8 months). Three patients achieved clinical complete response (CR) and molecular CR, whereas five patients achieved and maintained partial response 29 .
Furthermore, the combination of PD-1 inhibitors with agents such as lenalidomide has been studied for treating CAEBV infection. In a multicenter prospective study involving 34 patients with CAEBV infection, the overall response rate was 54.2%, with a CR rate of 45.8% and a partial response rate of 8.3%. A substantial decrease in EBV-DNA copies in the peripheral blood was observed. However, the question of whether the early therapeutic response can be further translated into long-term progression-free survival remains to be answered and will require further observation. A comparative analysis revealed that responders showed a significant post-treatment increase in effector memory CD8+ T cells and CTL activation, evidenced by elevated cytokines (IFN-γ, CD27, CD30, MIG, IP-10). These changes may be mediated by enhanced regulatory T cell (Treg) activity 30 .
mTOR inhibitor
Sirolimus, an mTOR inhibitor initially developed to prevent rejection following organ transplantation, has recently demonstrated potential for the treatment of CAEBV infection 31 . mTOR is a critical cellular signaling protein that plays a pivotal role in regulating cell growth, proliferation, and metabolism. By targeting this pathway, sirolimus inhibits the abnormal proliferation of lymphocytes, thereby modulating the pathological progression of CAEBV infection. Furthermore, sirolimus exerts its effects by inhibiting the synthesis of cell cycle–related proteins, impeding lymphocyte G1-to-S phase transition, and promoting the activation of apoptosis-related pathways. This process enhances the apoptosis of infected cells and reduces the survival of pathological lymphocytes 32 . Although the effects of sirolimus on CAEBV infection have not been examined in clinical studies, case reports of EBV-related diseases have highlighted the close association between aberrant activation of the Akt/mTOR signaling pathway and disease progression. Consequently, mTOR inhibitors emerge as a novel therapeutic modality for EBV-associated conditions. The potential of combining sirolimus with IFN-α or IL-2 is also under investigation, as these combinations may enhance antiviral effects by boosting overall immune system activity 33 . Notwithstanding its encouraging therapeutic potential, long-term sirolimus use is associated with an elevated risk of infection and renal impairment, as established in clinical experience. Consequently, further research is warranted to enhance our understanding of the immunomodulatory role of sirolimus in treating CAEBV infection and to optimize its clinical application.
Cellular immunotherapy
In recent years, the field of cellular immunotherapy for hematological malignancies has advanced considerably. This approach has also attracted significant attention in the treatment of chronic active EB virus–associated diseases (CAEBVD), encompassing strategies such as CAR-T cell therapy and EBV-specific CTL therapy (Figure 2). In animal models of CAEBV infection treated with CAR-T cell therapy, 75% of mice exhibited controlled or reduced EBV dissemination following treatment with autologous CD8+ gp350 CAR-T cells. The present study demonstrated low frequencies of EBER+ B-cell lymphoproliferation and associated inflammatory responses in these mice. The results of this study provide robust evidence supporting the potential of CAR-T cell therapy for the treatment of CAEBV infection. In clinical applications, a case study of a patient with CAEBV infection treated with CAR-T cells targeting the gp350 epitope demonstrated a significant increase in gp350-reactive cytotoxic T cells, thereby amplifying the immune response against EBV 34 . These findings underscore the clinical efficacy of CAR-T cells in the treatment of CAEBV infection. However, further research is required to fully elucidate their therapeutic potential and optimize their application in this field.
EBV-specific CTLs have demonstrated potential as a therapeutic modality for the treatment of EBV-associated diseases. Tabelecleucel, the first approved allogeneic EBV-specific T-cell therapy, is indicated for recurrent or refractory EBV-positive post-transplant lymphoproliferative disorders (PTLD) 35 . Clinical studies have demonstrated the safety of infusion of autologous EBV-specific CTLs, and moreover, have evidenced its capacity to ameliorate the clinical symptoms associated with CAEBV infection. A 2022 trial revealed that autologous EBV-targeted CTL therapy induced specific immune responses, particularly against LMP2. These responses demonstrated positive effects on disease control 36 . In light of the ongoing advancements in EBV-specific CTL therapy, it is anticipated that further treatment protocols and clinical trials will be developed in the future. This approach has the potential to emerge as a novel treatment option for CAEBV and other EBV-related diseases. Researchers are currently engaged in endeavors to optimize the preparation and infusion techniques for CTLs with the objective of enhancing their therapeutic efficacy and minimizing potential adverse effects.
Furthermore, EBV-specific T-cell receptor–engineered T (TCR-T) cell therapy has shown promising results in preclinical studies of EBV-associated tumors. Wang et al. 37 were the first to identify a CD4+TCR (TCR135) targeting EBNA1 and restricted by HLA-DP5. TCR-T cells engineered with this receptor significantly inhibited tumor growth both in vitro and in an HLA-DP5+ nasopharyngeal carcinoma humanized mouse model, providing a first-in-class CD4+TCR-T candidate for EBV-associated solid tumors. With ongoing research, EBV-TCR-T therapy holds the potential to address unmet needs in the treatment of CAEBVD and emerge as a novel, precise immunotherapeutic option.
Despite the encouraging results observed with the aforementioned immunotherapies in the treatment of CAEBV, it is important to note that the majority of these therapies remain in the preliminary stages of investigation, with the majority of studies being case reports and lacking validation from large-sample, randomized controlled trials. Consequently, these strategies should be regarded as transitional therapies to HSCT, as opposed to established curative regimens. It is evident that further multi-center, large-sample, randomized controlled trials are required to elucidate their long-term efficacy, safety, and optimal positioning in the HSCT bridging pathway.
Other drugs
Histone deacetylase inhibitors have considerable potential for the treatment of CAEBV infection, particularly for facilitating the transition from latent infection to the lytic cycle. These inhibitors alleviate transcriptional repression in the latent state, subsequently promoting the transcription and expression of EBV lytic genes, which aid in eliminating EBV-infected cells 38 . Studies have shown activation of NF-κB and STAT3 in EBV-positive T/NK cells 18 . Bortezomib, a specific proteasome inhibitor, prevents excessive activation of NF-κB induced by LMP1 by inhibiting the NF-κB pathway. It interferes with the intracellular protein degradation process, leading to alterations in several critical cell signaling pathways, ultimately inhibiting cell growth and promoting apoptosis. Studies have shown that bortezomib can inhibit the survival of EBV-positive T and NK cells, induce apoptosis, and reduce the spread and dissemination of EBV 39 . Notably, following the combination of bortezomib and ganciclovir for the treatment of T-CAEBV infection, EBV-DNA levels decreased to <100, indicating a significant therapeutic response. This finding suggests that bortezomib not only directly inhibits the survival of EBV-positive cells but also enhances the efficacy of other antiviral agents.
C-C motif chemokine receptor 4 (CCR4) is a G protein–coupled receptor that belongs to the CC chemokine receptor family. It is predominantly expressed on the surface of immune cells, such as Th2 cells and Tregs. CCR4 is also expressed on the surface of most EBV-positive T/NK cells. By binding to ligands such as CCL17 and CCL22, CCR4 regulates the migration and function of immune cells, playing a crucial role in various inflammatory diseases, autoimmune diseases, and tumors. Mogamulizumab is a monoclonal antibody that targets CCR4. Studies have shown that mogamulizumab can eliminate EBV-positive T cells and CCR4-expressing Tregs from the peripheral blood, thereby reducing their immunosuppressive effects on tumor immunity and positively impacting the tumor immune microenvironment. In animal experiments, mogamulizumab exerts targeted effects on CCR4-positive cells through antibody-dependent cellular cytotoxicity, thereby inhibiting the growth of EBV-positive NK cells. Thus, it provides a novel strategy for immunotherapy against CAEBV infection. However, the lack of clinical trials to date limits the validation of its broad application in the treatment of CAEBV infection. Therefore, further investigation is required.
The pathogenesis of CAEBV infection is complex and has not been fully elucidated. Increasing evidence indicates that genetic mutations play critical roles in its development. In one study 40 , a patient with CAEBV was found to harbor a missense mutation in the PIK3CD gene (E1021K), leading to overactivation of the PI3Kδ signaling pathway, which promotes abnormal cell development and proliferation and increases susceptibility to neoplastic diseases. In addition, mutations in ADA and CD3D were detected in the same patient, suggesting that these genetic mutations may contribute to the persistence of EBV infection and the onset of CAEBV by impairing normal immune system. Moreover, mutations in genes such as UNC13D, STXBP2, SH2D1A, BIRC4, and MAGT1, which encode proteins involved in vesicle fusion, perforin function, and SLAM-associated signaling, have been shown to compromise the cytotoxic activity of T and NK cells, thereby facilitating EBV persistence and disease progression 41 . Nevertheless, it is important to note that some of these mutations—particularly in PIK3CD, ADA, and UNC13D—are also well-recognized causes of primary immunodeficiency syndromes. Given that the diagnosis of CAEBV requires the presence of a chronic EBV-related disorder that cannot be explained by other underlying diseases, patients carrying such mutations may, in certain cases, be more appropriately classified as having EBV-associated disease secondary to immunodeficiency rather than primary CAEBV infection. Further research is therefore needed to delineate the clinical and molecular boundaries between CAEBV and immunodeficiency-related EBV diseases, and to elucidate the mechanisms by which these genetic defects contribute to EBV persistence and lymphoproliferation, ultimately paving the way for the development of targeted therapeutic strategies.
Allogeneic-HSCT
CAEBV infection is fundamentally characterized by a persistent EBV infection state marked by systemic inflammation and clonal proliferation of EBV-infected T or NK cells. Therefore, allo-HSCT is considered the only curative treatment for CAEBV infection 17 . This approach aims to eradicate EBV-infected cells and reconstitute the new immune system, thereby enabling long-term survival. Studies have shown that the 5-year survival rate of patients with CAEBV infection undergoing allo-HSCT is approximately 80% 42 . In contrast, the prognosis of patients with CAEBV infection without transplantation is notably poor, with many patients succumbing to the disease within 5 years. These findings highlight the significant improvement in outcomes conferred by allo-HSCT 43 . Although HSCT is a reliable curative treatment, it is associated with a treatment-related mortality of approximately 10%. This underscores the fact that while transplantation can cure the disease, it also carries inherent risks. Therefore, careful and individualized consideration should be given to the timing of allo-HSCT intervention, conditioning intensity, and donor selection, as these factors critically influence treatment outcomes. Representative clinical studies investigating allo-HSCT in CAEBV infection are summarized in Table 1.
Representative clinical studies of allo-HSCT transplantation for CAEBVD.
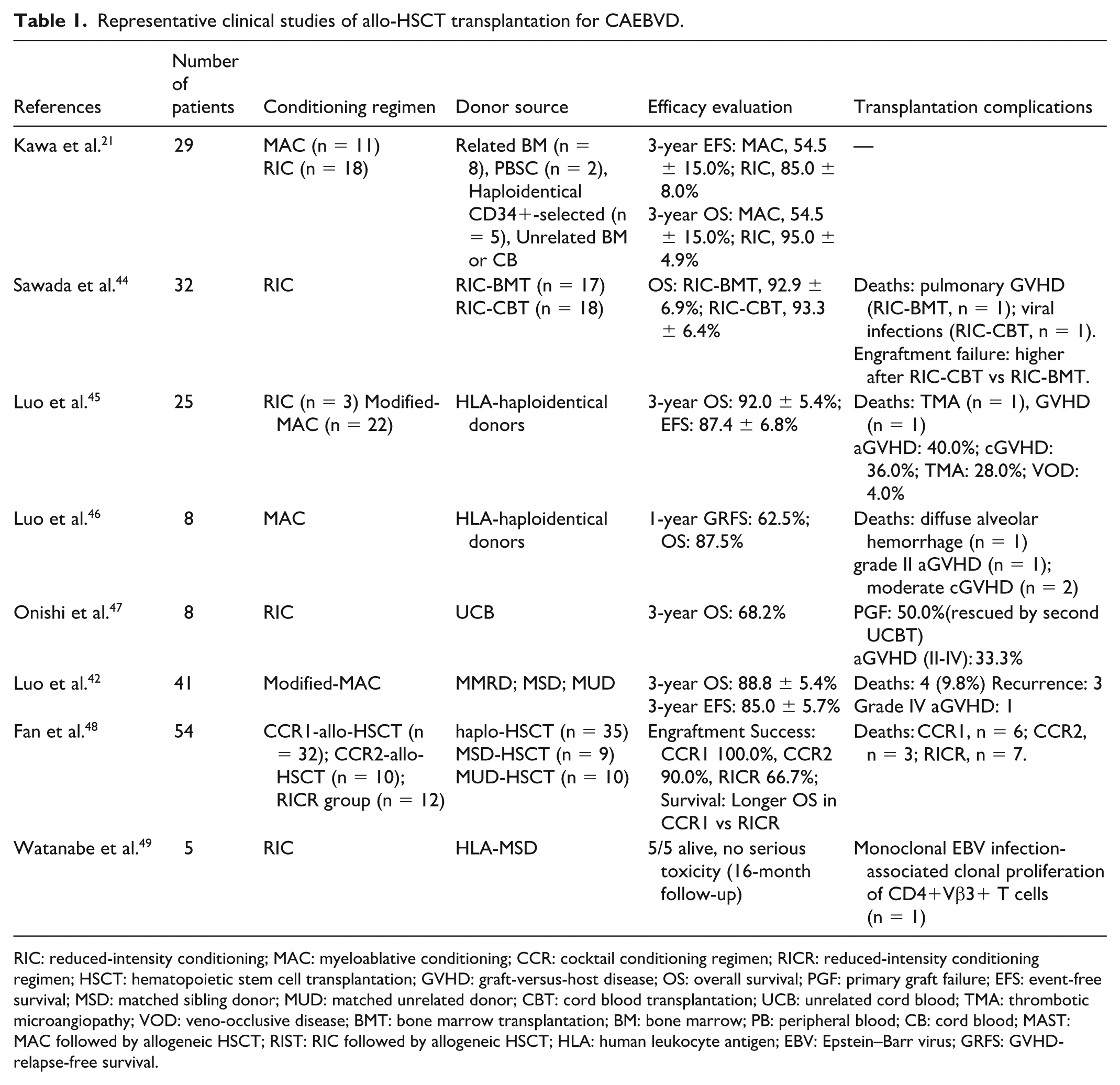
RIC: reduced-intensity conditioning; MAC: myeloablative conditioning; CCR: cocktail conditioning regimen; RICR: reduced-intensity conditioning regimen; HSCT: hematopoietic stem cell transplantation; GVHD: graft-versus-host disease; OS: overall survival; PGF: primary graft failure; EFS: event-free survival; MSD: matched sibling donor; MUD: matched unrelated donor; CBT: cord blood transplantation; UCB: unrelated cord blood; TMA: thrombotic microangiopathy; VOD: veno-occlusive disease; BMT: bone marrow transplantation; BM: bone marrow; PB: peripheral blood; CB: cord blood; MAST: MAC followed by allogeneic HSCT; RIST: RIC followed by allogeneic HSCT; HLA: human leukocyte antigen; EBV: Epstein–Barr virus; GRFS: GVHD-relapse-free survival.
Timing of HSCT
Early intervention during the initial stages of CAEBV infection can effectively control viral replication, slow disease progression, and prepare patients for allo-HSCT. A study involving 102 patients with CAEBV infection who underwent allo-HSCT showed that early transplantation significantly improved survival rates and quality of life, with short intervals between disease onset and transplantation correlating with low post-transplant mortality 50 . Furthermore, the pretransplant disease status significantly affects transplant outcomes. Patients with active disease manifestations, such as fever, liver dysfunction, progressive skin lesions, vasculitis, or uveitis, tend to have worse outcomes than those with non-active disease states 18 . Therefore, for patients with CAEBV infection, transplantation should be performed as early as possible after disease activity is controlled.
Donor selection
In patients with chronic CAEBV infection undergoing HSCT, donor selection significantly affects the prognosis. Donor type, particularly the distinction between related and unrelated donors, can influence transplant success rates and overall survival (OS). Human leukocyte antigen (HLA) matching is the primary factor in donor selection. High HLA compatibility is associated with a low risk of graft rejection and graft-versus-host disease (GVHD), thereby improving transplantation success rates. Related donors, such as siblings, typically exhibit better HLA compatibility with recipients, resulting in a significantly lower risk of GVHD and higher OS rates than unrelated donors 51 .
Rethinking genetic background and donor strategies
Patients with CAEBV (particularly those of Asian descent) frequently possess germline or somatic mutations in genes such as UNC13D, STXBP2, PRF1, CTPS1, and PIK3CD, resulting in immune dysregulation and elevated post-transplant relapse rates52,53. Consequently, in cases of CAEBV that have been confirmed to have immune-related germline mutations, human leukocyte antigen-matched unrelated donors (HLA-URDs) should be given priority for identification. In the event that no URD is identified, kinship screening should be initiated to assess the risk of relapse.
Related donors
Among the related donors, HLA-matched sibling donors remain the preferred choice for allo-HSCT. However, it should be noted that only approximately 25%–30% of patients can identify a fully HLA-matched sibling donor 54 , thus rendering haploidentical and unrelated donors important alternative options. Haploidentical donors are typically available for most patients, often with multiple potential donors, allowing for optimal selection. The recommended hierarchy for haploidentical donor selection is as follows: children, male siblings, fathers, siblings with non-inherited maternal antigen mismatches, siblings with non-inherited paternal antigen mismatches, mothers, and other collateral relatives55,56. In the context of haploidentical donation, the screening for donor-specific anti-HLA antibodies is of paramount importance. In instances where robust donor-specific anti-HLA antibody positivity is identified and alternative donors are available, these should be prioritized 57 . It is imperative to acknowledge that when selecting a related donor, it is strongly recommended that the donor undergo EBV-DNA testing (whole blood or PBMC+plasma), with priority given to EBV-DNA-negative donors.
In addition to HLA matching, several other factors must be considered when selecting a donor, including overall health status, age, sex, and willingness to donate. Advanced donor age may compromise the quality and quantity of HSCs, thereby increasing the risk of post-transplant complications 58 . Therefore, donors aged >60 years should be avoided. Donor sex also plays a role in transplantation outcomes. According to the Chinese consensus on the diagnosis and treatment of chronic GVHD (cGVHD), stem cells from female donors may increase the risk of cGVHD in male recipients. However, the impact of donor–recipient sex mismatch varies across different diseases and transplant protocols, warranting further investigation. The EBV serostatus of both donors and recipients significantly influences transplant outcomes. A large meta-analysis revealed that EBV-seropositive donors had a 17% increased risk of cGVHD and a 5% increased risk of acute GVHD (aGVHD) 59 . However, it is hypothesized that they may also confer a protective effect against EBV-related complications via donor immunity, potentially improving prognosis 59 . Furthermore, a mismatch in cytomegalovirus (CMV) serostatus between the donor and recipient is a known risk factor for early post-transplant viral infections, including Epstein–Barr virus (EBV) reactivation 60 . Other factors, such as donor blood type and killer cell immunoglobulin-like receptor genotype, may serve as additional criteria for donor selection. Therefore, the selection of an optimal donor requires a comprehensive evaluation of multiple factors to ensure the best possible transplant outcomes.
Unrelated umbilical cord blood
Unrelated umbilical cord blood transplantation (UCBT) is a readily available alternative for patients without HLA-matched or haploidentical donors. Small-scale retrospective studies have shown promising outcomes, with a 3-year OS rate of 83.3% in eight patients with CAEBV infection undergoing UCBT, accompanied by a significant reduction or undetectable levels of EBV-DNA load 47 . Therefore, unrelated cord blood can be an alternative source for HSCT for CAEBV. However, there are few reports of UCBT for the treatment of adult CAEBV cases. Further studies are required to evaluate the superiority of CBT over other stem cell sources for HSCT.
Conditioning regimen
The conditioning regimen plays a critical role in allo-HSCT for patients with CAEBV infection because its design and selection directly influence transplant success rates, incidence of complications, and long-term patient outcomes. Traditional myeloablative conditioning (MAC) regimens employ high-intensity chemotherapy or radiotherapy to completely eradicate the recipient’s hematopoietic and immune systems, thereby creating a favorable environment for donor stem cell engraftment. These regimens effectively eliminate EBV-infected lymphocytes, thereby reducing the risk of post-transplant EBV reactivation and disease relapse. In addition, they are associated with high donor stem cell engraftment rates and stable chimerism after transplantation. However, the significant toxicity of myeloablative regimens increases the risk of transplant-related complications, such as infections and GVHD, making them suitable for younger patients or those in good physical condition. In contrast, non-MAC regimens are less intensive than MAC regimens and are associated with reduced transplant-related toxicity, thereby minimizing damage to normal tissues. However, their ability to eliminate EBV-infected cells is comparatively low, potentially leading to a high risk of EBV reactivation and disease relapse after transplantation. In addition, these regimens may result in low donor stem cell engraftment rates and unstable chimerism, making them appropriate for older patients and those with comorbidities. To address these limitations, modified conditioning regimens are being explored for the treatment of patients with CAEBV infection. These regimens aim to balance the reduced transplant-related toxicity with effective control of CAEBV infection, thereby minimizing the risk of disease relapse while optimizing patient outcomes.
In a retrospective study involving 29 patients with CAEBV infection, the efficacy of MAC followed by allo-HSCT (MAST) was compared with that of reduced-intensity conditioning (RIC) followed by allo-HSCT (RIST). The study showed that the 3-year event-free survival rate was 54.5 ± 15.0% in the MAST group and 85.0 ± 8.0% in the RIST group, while the 3-year OS rate was 54.5 ± 15.0% in the MAST group and 95.0 ± 4.9% in the RIST group. These findings suggest that RIC regimens offer greater clinical benefits to patients than MAC regimens 21 . However, previous studies have indicated that for patients with active disease, high-intensity chemotherapy is required to effectively eliminate EBV-infected lymphocytes, making MAC regimens advantageous 61 . In a retrospective study involving 41 pediatric patients with CAEBV infection, the efficacy and safety of allo-HSCT were evaluated using a modified MAC regimen. The results demonstrated that allo-HSCT with a modified MAC regimen is safe and effective for pediatric patients with CAEBV infection and benefits patients with hemophagocytic lymphohistiocytosis or active disease 42 . Another retrospective study of 54 pediatric patients with CAEBV infection compared the effectiveness of cocktail conditioning regimen-allo-HSCT with that of RIC regimen-allo-HSCT. The findings revealed that the cocktail conditioning regimen significantly improved engraftment success rates and OS rates in pediatric patients with CAEBV infection compared with the RIC regimen 48 . Therefore, the selection of a conditioning regimen should be based on comprehensive consideration of the patient’s disease status and overall physical condition.
Furthermore, for patients lacking an HLA-matched donor, RIC followed by cord blood transplantation can achieve long-term survival, with an OS rate of 93.3 ± 6.4%. Unrelated cord blood can serve as an alternative source of blood for RIC-HSCT. However, the rate of graft failure after RIC-cord blood transplantation remains higher than that after RIC-bone marrow transplantation 44 , necessitating vigilance for potential graft failure or poor engraftment.
Transplantation complications
Allo-HSCT markedly improves prognosis in CAEBV; however, transplantation-related complications exert a substantial impact on survival and quality of life. The primary complications arising from this treatment include GVHD, infections, organ dysfunction, and poor engraftment. Bone marrow-depleting conditioning-induced tissue injury is particularly pronounced in patients with chronic active EBV (CAEBV), given that the disease itself frequently involves organs such as the liver, spleen, and bone marrow. Hepatic veno-occlusive disease (VOD) is one of the typical complications observed in 10%–15% of cases 62 .
From a clinical perspective, the condition is characterized by the following symptoms: weight gain, hepatomegaly, jaundice, and ascites. In severe cases, the condition may progress to coagulation disorders and hepatic encephalopathy63,64.
The underlying pathophysiology of this condition involves the induction of damage to the hepatic sinusoidal endothelium by radiation and chemotherapy, accompanied by local microcirculatory impairment and the release of pro-inflammatory factors such as TNF-α and IL-1. These factors serve to further exacerbate inflammation and tissue injury65,66. The primary treatment for VOD involves supportive care and defibrotide, a medication that reduces mortality by enhancing microcirculation 64 . Research indicates that the one-year survival rate for patients with isolated VOD is 66.7%, while it drops to 60% for those with concomitant transplant-associated thrombotic microangiopathy (TA-TMA) 67 .
TA-TMA represents a further severe complication, with an incidence rate of approximately 10%–25% and a mortality rate as high as 50%68–70. The clinical manifestations of this condition include microangiopathic hemolysis, thrombocytopenia, and microthrombosis in various organs. Treatment requires prompt discontinuation of calcineurin inhibitors, with eculizumab or plasma exchange as potential options 71 .
AGVHD manifests in approximately 50% of cases, primarily due to donor T cells attacking host tissues, with implications for the skin, gastrointestinal tract, and liver72,73. Current grading systems include the Glucksberg criteria and its revised version. In recent years, the MAGIC consensus has further refined organ assessment and severity grading (I–IV). The five-year survival rate for patients diagnosed with severe aGVHD is less than 25% 72 .
The National Institutes of Health (NIH) have established a set of criteria by which to classify cGVHD as mild, moderate, or severe 74 . The 5-year OS rate for patients with severe cGVHD is only 52%75,76, and the condition is often accompanied by non-malignant late complications that adversely affect long-term quality of life 77 .
Infection is a prevalent issue following transplantation. CAEBV patients are particularly susceptible to early and high-risk EBV reactivation 78 . In addition, CMV and adenovirus infections can cause pneumonia, gastroenteritis, hemorrhagic cystitis, and other manifestations 79 , while immunosuppression elevates the risk of bacterial and invasive fungal infections80,81.
Graft failure or poor engraftment is associated with a similarly deleterious effect on prognosis. The failure to achieve donor cell engraftment within 28 days is defined as implantation failure, with patients exhibiting long-term survival rates below 10% 82 . The mechanisms involved include immune rejection, disruption of the bone marrow microenvironment, and inadequate conditioning intensity83,84. Prevention and treatment strategies have been established, and these include optimizing donor selection, increasing stem cell dose, enhancing immunosuppression, or performing a second transplant.
EBV reactivation after transplantation
Post-HSCT EBV reactivation is a critical factor influencing the prognosis of patients with CAEBV infection. The incidence of EBV reactivation in patients with CAEBV infection following HSCT is as high as 30% 85 . The use of immunosuppressive agents after HSCT significantly impairs the host immune system, leading to the reactivation of latent EBV. This immunosuppression not only affects B-cell proliferation but also compromises the function of NK and T cells, thereby weakening the immune surveillance mechanisms against EBV. Furthermore, in patients with CAEBV infection, the interaction between CD27 and CD70 is considered a key mechanism for inducing protective EBV-specific T-cell immunity. These interactions are crucial for controlling EBV activity. However, this interaction may be disrupted in immunosuppressive environments, leading to EBV reactivation 86 . In the case of patients diagnosed with CAEBV, the behavior of the EBV is more complex. In addition to the challenge of disease recurrence, vigilance is required against EBV-induced abnormal B-cell proliferation and malignant transformation under immunosuppression, such as progression to lymphoma (e.g., EBV-associated lymphoproliferative disorders, PTLD) and leukemia. Consequently, the regular monitoring of EBV-DNA levels in the blood is of particular importance for the early detection of EBV reactivation following HSCT. The majority of centers undertake high-frequency monitoring (weekly to biweekly) during the initial 1–6 months post-transplant. Thereafter, the frequency of monitoring is either extended or relaxed, contingent on patient risk factors 87 . In cases where elevated EBV-DNA is detected, a tiered intervention strategy may be employed. The initial measures typically include the assessment and reduction of immunosuppression (RIS) where feasible, with the aim of restoring host T-cell immune surveillance against the virus. In cases where the viral load remains persistently high or where clinical and imaging findings suggest disease progression, the administration of anti-CD20 monoclonal antibodies (rituximab) as preemptive therapy or prophylactic treatment is frequently recommended. A substantial body of research, evidenced by numerous cohort studies, has demonstrated that the administration of rituximab leads to a significant reduction in the risk of PTLD and a decrease in viral load 88 . For patients who are refractory to such measures or who have biopsy-confirmed EBV-driven lymphoproliferation, adoptive cellular immunotherapy using donor- or third-party derived EBV-specific cytotoxic T lymphocytes (EBV-CTLs) is increasingly used. Clinical trials report complete or partial responses in a substantial proportion (e.g., ~60%–90%) of treated patients, with favorable safety profiles 89 . Thus, effective management in this population should emphasize early and systematic EBV-DNA monitoring, risk-adapted intervention (immunosuppression modulation, anti-CD20 therapy, EBV-CTLs), and integration of clinical, virologic and, when indicated, pathologic evaluation to differentiate relapse of CAEBV versus EBV-PTLD and guide personalized therapy.
Conclusion
CAEBV infection is a lymphoproliferative disorder driven by EBV-specific infection of T cells or NK cells. Recent mechanistic studies indicate that CAEBV pathogenesis may originate at an earlier hematopoietic stage, involving latent EBV infection of HSCs. During differentiation, these infected HSCs can transmit the virus to multiple lineages, including T cells, NK cells, and B cells, establishing a persistent, multilineage state of viral latency and activation. This mechanism provides a novel cellular framework for understanding the systemic pathology and therapeutic resistance characteristic of CAEBV. At present, there is no universally accepted curative treatment for CAEBV infection. Conventional monotherapies—such as antiviral agents, immunosuppressive drugs, cytotoxic chemotherapy, or EBV-specific CTL infusion—generally confer only transient disease control and are associated with high relapse rates. Allo-HSCT remains the only potentially curative approach, achieving clearance of EBV-infected cells by restoring normal hematopoiesis and virus-specific immune surveillance. Effective pretransplant reduction of viral load and control of disease activity are critical for minimizing post-transplant relapse and complications, while the timing of HSCT, donor selection, and conditioning intensity remain key determinants of long-term outcomes.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable
Consent to Participate
This is a review article that does not involve any new studies with human participants performed by the authors.
Author Contributions
Yu Wang: Conceptualization, Writing—Original draft, Writing—Review and Editing. Jiaying Wu: Writing—Original draft, Writing—Review and Editing. Xiaobing Huang: Conceptualization, Supervision. Yi Xiao: Conceptualization, Supervision, Founding Acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was supported by the National Natural Science Foundation of China (No. 81873444, No. 82070213, and No. 82370196).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No datasets were generated or analyzed during the current study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
