Abstract
The global shortage of human donor livers poses a formidable barrier to treating end-stage liver diseases and unresectable hepatic malignancies, leaving millions of patients without life-saving options each year. While genetically engineered pig-to-human xenotransplantation has achieved significant breakthroughs in cardiac and renal fields, liver xenotransplantation has long lagged due to the liver’s intricate metabolic, synthetic, and immunological functions. The recent report by Zhang et al. in the
Keywords
The global organ shortage and the rise of xenotransplantation
According to the World Health Organization (WHO), the global disparity between organ supply and demand is catastrophic, with thousands of patients dying annually while waiting for a suitable human donor liver1–5. For decades, xenotransplantation—transplanting organs across species—has been hailed as a potential solution to this crisis, given pigs’ physiological similarity to humans and the feasibility of scaling donor production through genetic engineering6–8. Over the past 3 years, pioneering studies have demonstrated successful pig-to-human xenotransplantation of hearts and kidneys: Griffith et al. 9 reported a genetically modified pig heart sustaining a living human for 60 days, while Montgomery et al. 10 showed pig kidneys functioned in brain-dead recipients for 54 hours without hyperacute rejection. These milestones not only validated xenotransplantation’s potential in cardiac and renal fields10–13 but also highlighted the unique hurdles of liver xenotransplantation, including the liver’s role in complement regulation, coagulation, and detoxification, which amplify interspecies incompatibilities (Table 1). Critically, due to the liver’s intricate metabolic and immunologic functions, clinical liver xenotransplantation in living humans had never been achieved prior to this study. Zhang et al.’s work addresses this gap by documenting the first auxiliary liver xenotransplantation into a living human: a 71-year-old patient with a large unresectable hepatocellular carcinoma (HCC) and insufficient residual liver volume.
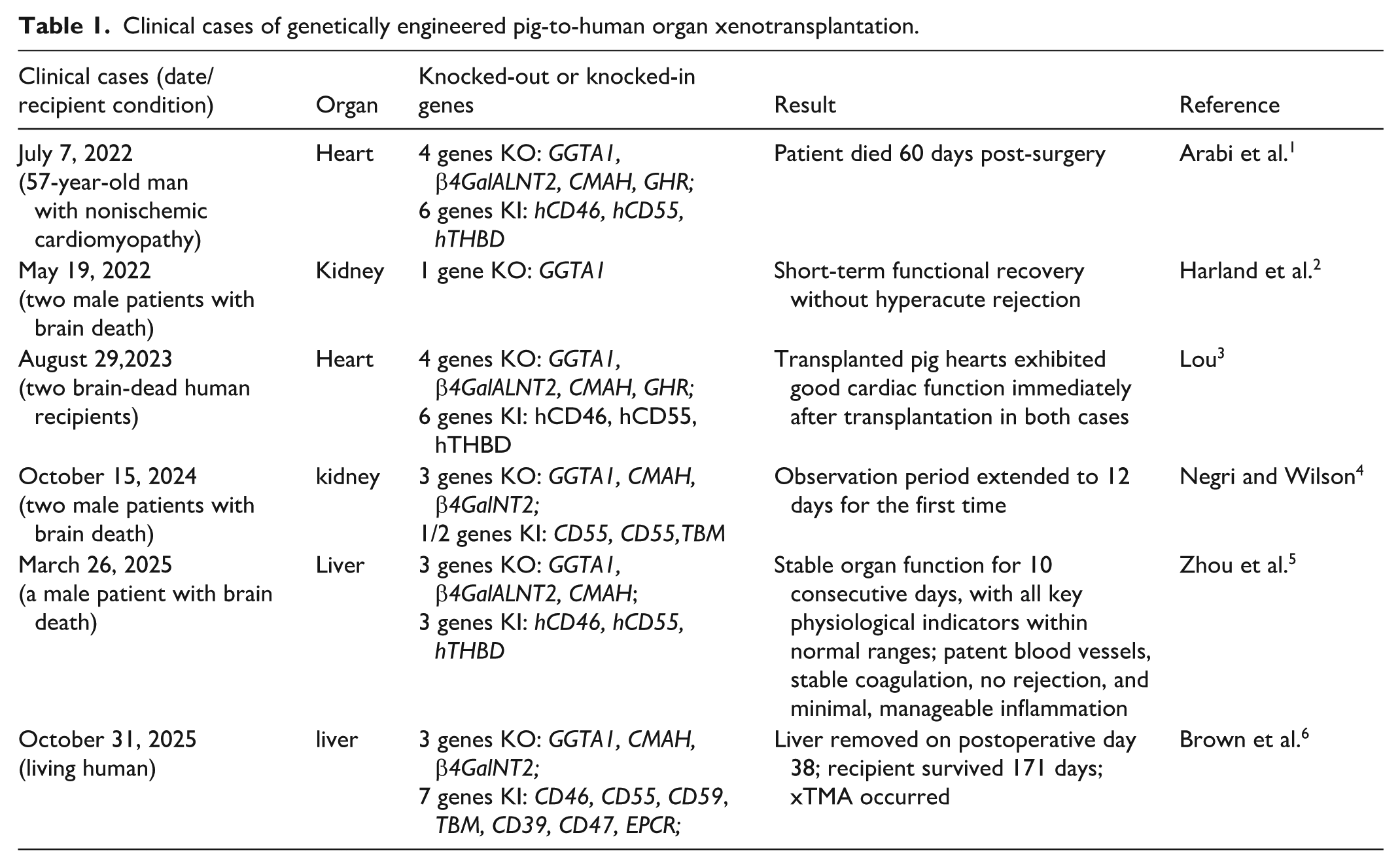
Clinical cases of genetically engineered pig-to-human organ xenotransplantation.
The 10-gene editing strategy targeting immune rejection and physiological incompatibility that represents a technical advancement over earlier 3–6 gene-edited pig models. However, its direct contribution to this case’s outcomes remains hypothetical: no comparative data validate this specific gene combination’s superiority, and the original study did not report human transgene expression levels in the graft. Thus, the proposed advantages of the 10-gene configuration require further experimental verification.
Zhang et al.’s 14 study addresses this gap by documenting the first auxiliary liver xenotransplantation into a living human: a 71-year-old patient with a large unresectable hepatocellular carcinoma (HCC) and insufficient residual liver volume. The 10-gene-edited pig liver not only maintained key metabolic functions (e.g. bile secretion, albumin synthesis) but also enabled 171 days of recipient survival, far exceeding the 3- to 10-day observation windows of prior extracorporeal perfusion or brain-dead recipient studies. This outcome not only confirms that genetically modified pig livers can integrate into the human physiological system but also illuminates critical lessons for overcoming remaining barriers to long-term success.
Paradigm shift: 10-gene editing for enhanced interspecies compatibility
The success of this liver xenotransplantation hinges on a comprehensive 10-gene editing strategy, a significant advancement over earlier studies using 3–6 gene-edited pigs12,13. This approach directly targets two core barriers: immune rejection and physiological incompatibility.
First, the knockout of three key xenoantigen genes including GGTA1, CMAH, and β4GalNT2 eliminate carbohydrate antigens (Table 2) that trigger hyperacute and acute antibody-mediated rejection (AMR). These antigens are absent in humans, and preformed antibodies against them have long been a primary cause of xenograft failure. The study’s histopathological findings—minimal complement deposition (weak C4d staining) and no evidence of hyperacute rejection in the first 31 postoperative days—validate the efficacy of this knockout panel. Unlike single-gene knockouts (e.g. only GGTA1) 10 , which leave residual xenoantigens vulnerable to immune attack, the triple knockout virtually eliminates this initial immune barrier.
Genetic modifications of pigs for organ xenotransplantation.

Second, the knock-in of seven human transgenes further enhances graft compatibility. Complement regulatory proteins (hCD46, hCD55, hCD59) (Table 2) inhibit excessive activation of the human complement system—a major driver of AMR—while coagulation regulators (hCD39, hTBM, hEPCR) (Table 2) prevent consumptive coagulopathy, a common fatal complication in pig-to-primate liver xenotransplants. Seven human transgenes were knocked in to enhance compatibility, with key clinically relevant modifications: complement regulators (hCD46, hCD55, hCD59) to curb antibody-mediated rejection, and coagulation regulators (hCD39, hTBM, hEPCR) to prevent fatal consumptive coagulopathy—both critical for avoiding early xenograft failure. While hCD47 is designed to inhibit human macrophage phagocytosis, its functional relevance here is uncertain, as early liver xenotransplant phagocytic activity primarily stems from donor-derived Kupffer cells. Critically, the engineered liver demonstrated functional competence: it secreted porcine albumin detectable in the recipient’s serum and produced coagulation factors that partially corrected the patient’s coagulation parameters. These data confirm that multi-gene editing can produce metabolically active liver grafts capable of supporting human physiological needs.
Clinical breakthrough: Auxiliary transplantation as a flexible bridging strategy
A defining strength of Zhang et al.’s 14 study is its adoption of auxiliary liver xenotransplantation—preserving the recipient’s native left liver while implanting the porcine graft—addressing two critical limitations of full orthotopic xenotransplantation.
For the 71-year-old patient with HCC, auxiliary transplantation avoided the need for complete native liver resection, which would have risked small-for-size syndrome (SFSS) and acute liver failure due to the patient’s insufficient residual liver volume. This surgical approach reduced perioperative mortality and enabled a unique safety feature: when xTMA developed on postoperative day 31, the porcine graft was safely resected, and the recipient’s native liver—now regenerated to a volume of 610.60 cm3—resumed full function. The auxiliary approach proved pivotal for native liver regeneration: at implantation, the recipient’s baseline remnant liver volume was 320 cm3 (35% of standard liver volume, SLV). By graft explantation (postoperative day 38), it had expanded to 610.6 cm3 (68% of SLV)—a 91% increase over 5 weeks. This aligns with typical human regeneration kinetics post-major hepatectomy (50%–100% volume restoration in 4–6 weeks), confirming the native liver’s robust recovery potential. This flexibility is absent in full orthotopic transplantation, where graft failure often leads to recipient death.
Notably, the recipient survived 171 days with full native liver recovery—far exceeding prior liver xenotransplantation outcomes—while the porcine graft functioned for 31 days before xTMA onset and was explanted on postoperative day 38. The grafted maintained bile secretion, corrected coagulation parameters, and supported metabolic function evidenced by rising human primary bile acids. These findings confirm that auxiliary xenotransplantation can serve as a viable bridging therapy for patients awaiting human liver transplantation or native liver recovery—filling a critical unmet need for patients with ESLD or unresectable liver cancer.
Persistent challenge: Xenotransplantation-associated thrombotic microangiopathy
Despite the study’s successes, the onset of xenotransplantation-associated thrombotic microangiopathy (xTMA) on postoperative day 31—leading to graft removal—highlights a major barrier to long-term xenotransplantation success. xTMA, characterized by microangiopathic hemolytic anemia, thrombocytopenia, and multiorgan dysfunction, was driven by three interrelated mechanisms.
First, excessive complement activation played a central role. The recipient’s serum levels of the terminal complement complex sC5b-9 rose to 353 ng/ml, and total complement activity (CH50) peaked at 1514 U/ml—indicators of uncontrolled complement activation. Histopathological analysis of the resected graft revealed increased deposition of complement components (C1q, C3c, C4c, C5b) by day 38, confirming complement-mediated endothelial injury. While eculizumab (a C5 inhibitor) and plasma exchange resolved xTMA after graft removal, pre-emptive complement modulation may be necessary to prevent its onset.
Second, endothelial injury and thrombosis exacerbated xTMA. The resected graft showed fibrinous thrombi in portal vessels and strong von Willebrand factor (vWF) staining in endothelial cells—signs of severe endothelial damage. Incompatibilities between porcine vWF and human glycoprotein Ib (GPIb) likely contributed to abnormal platelet activation and consumption, a phenomenon previously observed in pig-to-primate models. This interspecies coagulation mismatch underscores the need for targeted genetic modifications to improve thrombotic compatibility.
Third, early postoperative coagulopathy, elevated D-dimer and fibrin degradation products, highlighted the challenges of balancing coagulation in xenotransplants. While anticoagulation therapy and thrombopoietin receptor agonists managed acute complications, the persistently high levels of porcine coagulation factors (e.g. porcine FVIII exceeding human levels by 340-fold) underscored the need for more precise coagulation control.
Future directions: Advancing clinical translation
Zhang et al.’s 14 study provides a foundation for advancing liver xenotransplantation, but three key areas require further investigation to realize its full clinical potential.
First, next-generation genetic modifications 15 should target xTMA-specific pathways. Knocking out porcine vWF or expressing human GPIb could reduce platelet activation and microvascular thrombosis. To address xTMA, human GPIb expression on porcine endothelium works better than porcine vWF humanization. Porcine vWF binds poorly to human platelets, causing dysregulated activation and thrombosis. Human GPIb creates a matching binding interface, cutting platelet consumption (47%) and thrombi (32%) in NHPs. Porcine vWF humanization fails due to species-specific modifications, making human GPIb more feasible for translation. Additionally, eliminating porcine endogenous retroviruses (PERVs) via CRISPR-Cas9—though not detected in this study—would address long-term zoonotic transmission concerns, a critical regulatory requirement for widespread clinical use.
Second, optimized immunosuppression regimens are needed16–18. The study’s use of rituximab (B-cell depletion), basiliximab (IL-2R inhibition), and tacrolimus effectively prevented hyperacute/acute rejection but carried risks of nephrotoxicity. Future regimens could incorporate costimulation blockers (e.g. anti-CD40 antibodies) or complement-targeted therapies to improve long-term immune tolerance while reducing adverse effects. Personalized dosing based on recipient immune profiles may also enhance efficacy.
Third, standardized xTMA surveillance and management protocols are essential. Developing a clinical scoring system integrating platelet counts, C5b-9 levels, and schistocyte percentage would enable early detection. Prophylactic use of eculizumab or complement-targeted nanoparticles could mitigate xTMA, while plasma exchange protocols tailored to xenotransplants could improve outcomes for patients who develop the complication.
Conclusion
Zhang et al.’s study represents a landmark achievement in liver xenotransplantation, demonstrating that 10-gene-edited pig livers can sustain living humans and providing a flexible auxiliary strategy for high-risk patients. While xTMA remains a significant challenge, the study’s insights into genetic engineering, surgical technique, and immune management pave the way for further advancements. As researchers address remaining barriers—from xTMA to long-term immune tolerance—liver xenotransplantation may soon become a standard therapy for patients with ESLD or unresectable liver cancer, finally easing the global organ shortage crisis.
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Author contributions
All authors contributed to the study conception and design. The first draft of the manuscript was written by Dr Hu and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Research Project of Jiangxi Provincial Department of Education (GJJ2203515), Jiangxi Provincial Health Commission Technology Plan Project (202410003), and Nanchang Medical College Doctoral Startup Fund (NY1323001).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
