Abstract
Extracellular vesicles (Evs) act as a natural intercellular message transmitter, Evs can carry proteins, ribonucleic acid (RNA) and other bioactive substances, and have rich biological regulatory functions. Because of its low immunogenicity and high biocompatibility, it has become a popular research object in drug delivery. These remarkable properties also create new opportunities for modern therapy. However, due to the complex preparation process, there are challenges in terms of targeting accuracy, load release controllability, and pharmacokinetic optimization, and many problems may be encountered in reality. Based on real-life biomedical experiments, this paper summarizes the methods and types of Evs loading drugs, membrane modification methods, and the use of biological materials to improve the release efficiency, so as to provide reference for future research on engineered Evs.

Introduction
The core of engineered Evs is to perform targeted modification of natural Evs through synthetic biology and nanotechnology to break through their inherent biological limitations. Although natural Evs have advantages such as low immunogenicity, natural delivery ability, and crossing biological barriers, they have defects such as insufficient targeting, low concentration of therapeutic molecules, and high heterogeneity. The engineering strategy can precisely regulate its delivery pathway, load release kinetics and microenvironment responsiveness through surface ligand modification, membrane protein recombination or endogenous drug loading optimization, thereby showing application potential beyond traditional nanocarriers in the fields of tumor targeted therapy, gene editing delivery and tissue regeneration. This interdisciplinary research is promoting the paradigm upgrade of Evs from “biological messengers” to “intelligent treatment platforms.”
Evs were first discovered by Ali et al. 1 in the 1970s. The name “extracellular vesicles” was suggested in 2011 as a collective term for lipid bilayer-enclosed, cell-derived particles 2 . In 1983, a small Evs was discovered in sheep reticulocytes, which was named “exosome” by Johnstone et al. 3 Exosomes are a type of Evs. All cells, whether eukaryocyte or prokaryotic cell, can produce Evs 4 , they can modulate extracellular matrix, induce mineral formation under physiological conditions (bone formation) or ectopic calcification as in osteoarthritis and vascular calcification.
These are vesicles secreted outside the cell, Evs subpopulations share multiple surface proteins, and Evs from different sources also carry similar markers, making it difficult to accurately distinguish them in general, in addition, the most commonly used differential centrifugation and density gradient centrifugation methods mainly rely on physical properties such as density or sedimentation coefficient to separate different Evs. To determine its types, it is generally named according to its size.
Evs can be roughly divided into three types according to their size: exosomes, microvesicles, and apoptotic bodies 5 . Among these three vesicle types, the diameter of exosomes was 50-150 nm, the diameter of microvesicles was 100-1000 nm, and the diameter of apoptotic bodies was greater than 1000 nm6. In addition, there are also some non-vesicular nanoparticles outside the cell, such as albumin, supermeres 7 , and viral particles. Compared to microvesicles and apoptotic bodies, exosomes are currently an area of more extensive research.
Specifically, the formation process of exosomes is formed by the inward budding of the limiting membrane of early endosomes, which mature into multivesicular bodies (MVBs) in the process 6 . Early endosomes originate from the inward budding of the cytoplasmic membrane, and MVBs are involved in the endocytosis and transport functions of cellular materials. MVBs are eventually either sent to lysosomes for degradation along with all their components or they fuse with the cell’s plasma membrane, releasing their contents, including exosomes, into the extracellular space 8 . Structurally, exosomes are nanospheres with a double-layer membrane structure 9 . There are many protein families on the surface of exosomes, such as tetraspanins represented by CD63, CD9 and CD81, heat shock protein (Hsp70), lysosomal protein (Lamp-2b), and annexin 10 . The generation and general structure of exosomes are shown in Figure 1.
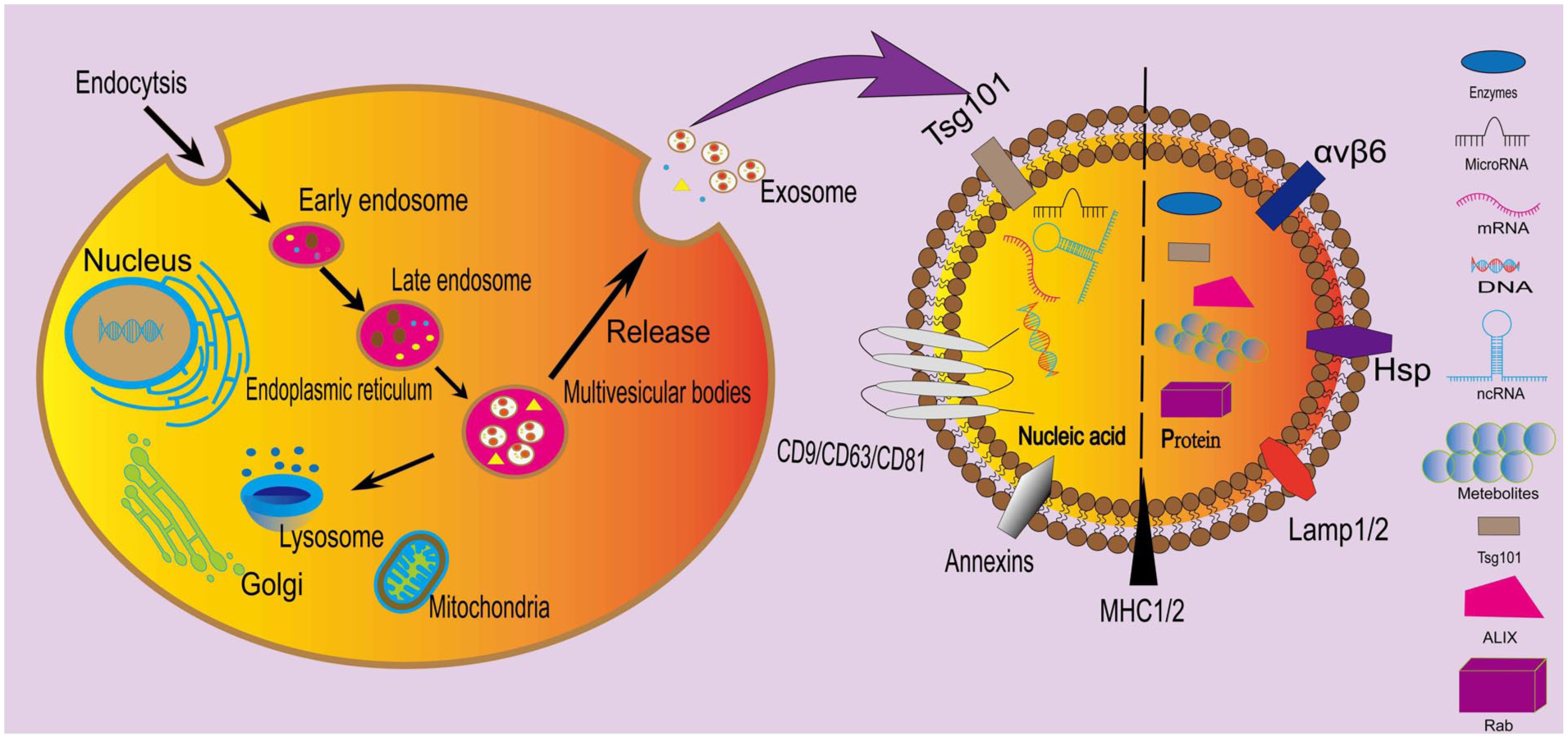
Biogenesis and composition of exosomes. On the left side of the figure is the process of generating exosomes within a eukaryotic cell; on the right side is the contents contained within the exosomes. Figure 1 was created using “Adobe Illustrator.”
Microvesicles are Evs formed by cells directly budding outward, containing cell membrane and some cytoplasmic components 11 . Specifically, they are involved in protein sorting, recovery, storage, transport, and release. Apoptotic bodies are a form of Evs released by dying cells in the last stage of apoptosis and were previously regarded as debris of dead cells 12 . There are many non-vesicular nanoparticles outside cells. Due to the overlap in the particle size of exosomes and microvesicles, it is difficult to separate highly pure exosome subpopulations using the currently used differential centrifugation and density gradient centrifugation methods. According to the recommendation of ISEV (International Society for Extracellular Vesicles), vesicles with a size range of 100-200 nm are usually called sEvs; therefore, it is more standardized to call the extracted exosomes sEvs. This concept has been adopted by more and more papers and accepted by more and more journals 13 .
In general, exosomes are also known as sEvs 14 . Increasing evidence shows that sEvs can be released from different cell types, such as macrophages, dendritic cells, tumor cells, mesenchymal stem cells (MSC), epithelial cells, mast cells, endothelial progenitor cells, platelets, lymphocytes, and fibroblasts15–17. sEvs from different cells exhibit different characteristics and functions 18 . The released vesicles enter the interior of the target cells through endocytosis or bind ligands to receptors specifically through internal biological active substances such as proteins, nucleic acids and lipids, thereby mediating the physiological state of the cells.
sEvs can not only carry bioactive substances such as MicroRNA and proteins, but also facilitate intercellular communication 19 . sEvs have unique properties, including low immunogenicity, high delivery efficiency, and ability to pass through the blood−brain barrier (BBB) 20 . However, the therapeutic potential of sEvs delivery systems is largely constrained by its uncertain loading efficiency, rapid clearance from the systemic circulation, and relatively weak targeting ability 21 . The engineering modification of sEvs can endow them with new characteristics and improve treatment efficiency in terms of carrying drugs or improving targeting capabilities 22 . Engineered sEvs can generally be categorized into three aspects. First, drugs are loaded into sEvs through endogenous loading method and exogenous loading method to deliver specific cargoes 23 ; The second is to modify the structure of sEvs so that it can better perform therapeutic effects in the body24,25, and it can specifically target tissues or cells; The third is to combine it with modern biological materials to improve its release efficiency in the body. When combined with new biomedical materials, the characteristics of both can complement each other, making it possible to achieve a more intelligent, targeted and multi-functional drug delivery platform24,26–31.
Choice of nanocarrier drug delivery
Nano drug carriers are a promising method for drug delivery. Their essence is to improve the pharmacodynamics and kinetics of drugs in the body, thereby improving the efficacy of drugs 32 . In recent years, drug delivery systems based on nanocarriers have made great progress. Among them, liposome particles have become a favorable carrier for drug delivery. However, considering the biological characteristics of liposomes (low bioavailability, toxicity, immunogenicity, etc), it shows that it has defects in clinical application 33 . Subsequently, researchers turned their attention to Evs, which are non-toxic, non-immunogenic, and have no side effects in the human body. They can become a more ideal drug delivery carrier after liposome particles. In addition, Evs can carry a wide variety of drugs, including nucleic acid drugs, large molecular proteins, compounds, and so on, which prolong the half-life after entering the human body34–37. It not only has a lipid bilayer structure, but also carries a variety of biological signals such as proteins and nucleic acids from the mother cell. These natural components make it easier for target cells to recognize and absorb them, thus having better biodistribution and targeting capabilities. For this reason, Evs are considered to be a natural drug carrier with greater potential than liposomes.
Drug loading into sEvs
Indirect sEvs engineering method
Endogenous loading method is to make the target drug appear in the donor cells of the vesicle. This is an engineered loading method based on the donor cells 38 . This method of engineered loading is based on the generation mechanism of sEvs, these donor cells sort these cargoes into the vesicles 39 . Studies have shown that the type and condition of donor cells can impact the yield of sEvs 40 , by appropriately nurturing or modifying the donor cell, it is possible to obtain sEvs with the desired characteristics. The main method to achieve this step is to co-incubate the donor cells with drugs; the expression of therapeutic substances required for research in the donor cells of the vesicles requires the introduction of exogenous nucleic acid sequences into the cells, relying on transfection methods 41 . When sEvs are formed in donor cells, the cells actively sort specific proteins, nucleic acids (such as mRNA, miRNA, and lncRNA), and other molecules into sEvs. By introducing exogenous nucleic acid sequences into donor cells, the cells’ own sorting mechanism can be used to integrate these nucleic acids and expressed proteins into sEvs. This method avoids additional modifications of sEvs after secretion and retains their natural biocompatibility and membrane integrity. The endogenous loading method is an attractive method for creating desirable sEvs. First of all, it relies on the normal process of cells producing sEvs, which means that there is no need to consider the immune reaction when entering the organism, this method is safe42,43; moreover, the conditions are controllable, and the flexibility is high in controlling the functions of engineered sEvs. The main loaded therapeutic substances include proteins, RNA, small nucleic acids, hydrophobic small molecule drugs, and so on 44 .
Co-incubation
Compounds, such as small molecule drugs, and anti-inflammatory factors, can be introduced into sEvs through co-incubation. sEvs can protect fragile drugs and deliver them to target cells 45 . In addition, sEvs can be co-incubated with growth factors to enhance the therapeutic effect of sEvs. Co-incubation appears to be the simplest and straightforward loading method and can be achieved by mixing the molecule of interest with donor cells. Small molecules can enter donor cells through the cell membrane under a concentration gradient driven passive transport mechanism 46 . Generally speaking, the co-incubation of sEvs needs to be carried out at a pH that simulates physiological conditions in vitro (usually 7.2-7.4), and the temperature is mostly around 37°C. The duration and reaction concentration need to be adjusted according to the donor cell type and drug type.
Curcumin has anti-inflammatory properties and is a potential candidate for treating osteoarthritis 47 . However, bioavailability is often poor due to its low water solubility and stability 48 . Therefore, to improve the bioavailability of the drug in the body, Li et al. 47 used dimethyl sulfoxide (DMSO) to dissolve curcumin and prepare a solution, and then incubated it with human bone marrow MSCs to isolate sEvs and obtain Cur-Evs (Evs carrying curcumin), Cur-Evs improve the metabolism of IL-1β stimulated human osteoarthritic chondrocytes, reduce cell apoptosis and increase vitality. Xu et al. 49 also obtained sEvs primed by curcumin and found that it can effectively promote the proliferation and anabolism of chondrocytes induced by TBHP (tert-butyl hydroperoxide). MSCs have a powerful anti-inflammatory mechanism and provide new hope for the treatment of various chronic inflammatory and autoimmune diseases 50 . Kim et al. 51 co-incubated IL-1β with human bone marrow MSCs, and collected the supernatant to obtain MSC-IL-sEv. They confirmed that MSC-IL-sEv inhibited the IL-1β-induced expression of IL-1β and TNFα in SW982 human synoviocytes. Wang and Xu 52 isolated exosomes from bone marrow MSCs and found that when co-incubated with transforming growth factor (TGF)-β1, the expression of MiR-135b inside them was increased, which could reduce the upregulation of pro-inflammatory factors in the serum of rats with osteoarthritis and the damage of cartilage tissue
Co-incubation with donor cells is a common technique for loading drugs into sEvs, it has the advantages of high controllability and easy operation. However, the method of using donor cells to co-incubate with drugs has constraints. The selected drugs must have low toxicity to the cells, preferably small molecule hydrophobic drugs, and the loading efficiency of this method does not meet the needs. The loading efficiency is influenced by drug properties incubation periods 53 . In addition, the stability of sEvs is limited by temperature and pH conditions, which also indicates that the method of co-incubation with donor cells in the medical field is unlikely to be large-scale. Based on this, it is necessary to conduct in-depth research on exogenous drug loading in the field of drug loading.
Transfection
Transfection is a mechanism by which appropriate exogenous sequences are found and transfected into cells to express drugs within cells, and donor cells are used to produce sEvs, thereby collecting drug-loaded sEvs. The transfection process can be accomplished through physical methods [electroporation 54 , gene gun 55 , sonication 56 , chemical methods (calcium phosphate method 57 ), biological methods (mainly virus-mediated 58 ), and so on].
In general, chemical transfection methods have lower efficiency, and the efficiency is influenced by the cell line 59 while physical methods may cause significant damage to the cells 60 . Strategies based on viral transfection have become popular in recent years; due to its stable and effective transfection ability, it has been applied in sEvs-based delivery systems 61 , this transfection method is suitable for efficient loading of a variety of cells 62 . Retrovirus, lentivirus 62 , adenovirus 63 , and adeno-associated viruses have been widely used as viral vectors for gene delivery 64 . After the viral vector is transfected into the donor cells, the expressed therapeutic substances may be loaded into sEvs, which bind to the target cells and play a regulatory role 65 . Transfection facilitates integration of non-native genetic constructs into donor cells and makes the cells acquire new phenotypes, and these sEvs produced can be modified 66 . With the continuous development of cell biology and the deepening of sEvs modification, this method has become a routine tool for controlling and studying the gene function of donor cells. However, the downside is that it is relatively complicated and cumbersome, and its effectiveness after transfection is uncertain, which may lead to abnormal gene expression in donor cells 67 . In addition, the encapsulation of drugs and the negative effects of viral transfection cannot be guaranteed, such as cell malformations, which also need to be studied in depth.
Due to the high biocompatibility and low immunogenicity of sEvs, as well as their ability to efficiently penetrate biological barriers, they have become ideal drug carriers, but their disadvantages are also very obvious. The advantages and disadvantages of the endogenous loading method are shown in Table 1. From the perspective of cytotoxicity, the co-culture method requires high concentrations of drugs to act on donor cells for a long time. The drugs will continuously and directly act on various metabolic links of the donor cells, interfering with their normal life activities. Therefore, the cytotoxicity is usually higher than that of the transfection method and is concentration-dependent. In addition, the drugs will go through two major steps: transmembrane and sorting, which limits the loading efficiency. The cytotoxicity of conventional transfection methods primarily stems from the transfection reagent itself and the interaction between the transfection complex and the cell membrane. With technological breakthroughs, an increasing number of transfection reagents are now demonstrating lower biotoxicity. Furthermore, because drug loading is a passive process, the amount of drug carried by exosomes obtained through co-culture varies greatly. Transfection, on the other hand, involves genetically modified donor cells, whose markers are relatively consistent with the cargo contents.
The advantages and disadvantages of the endogenous loading method.

Direct sEvs engineering method
Due to the limitations of sEvs’ endogenous loading methods, the development of exogenous loading methods is necessary. Different from endogenous loading method, exogenous loading method loads drugs directly into isolated sEvs 72 . Commonly used exogenous loading methods include co-incubation, extrusion, ultrasonic, electroporation, freeze−thaw cycle, and so on 73 . See Table 2 for details.
The advantages and disadvantages of the exogenous loading method.
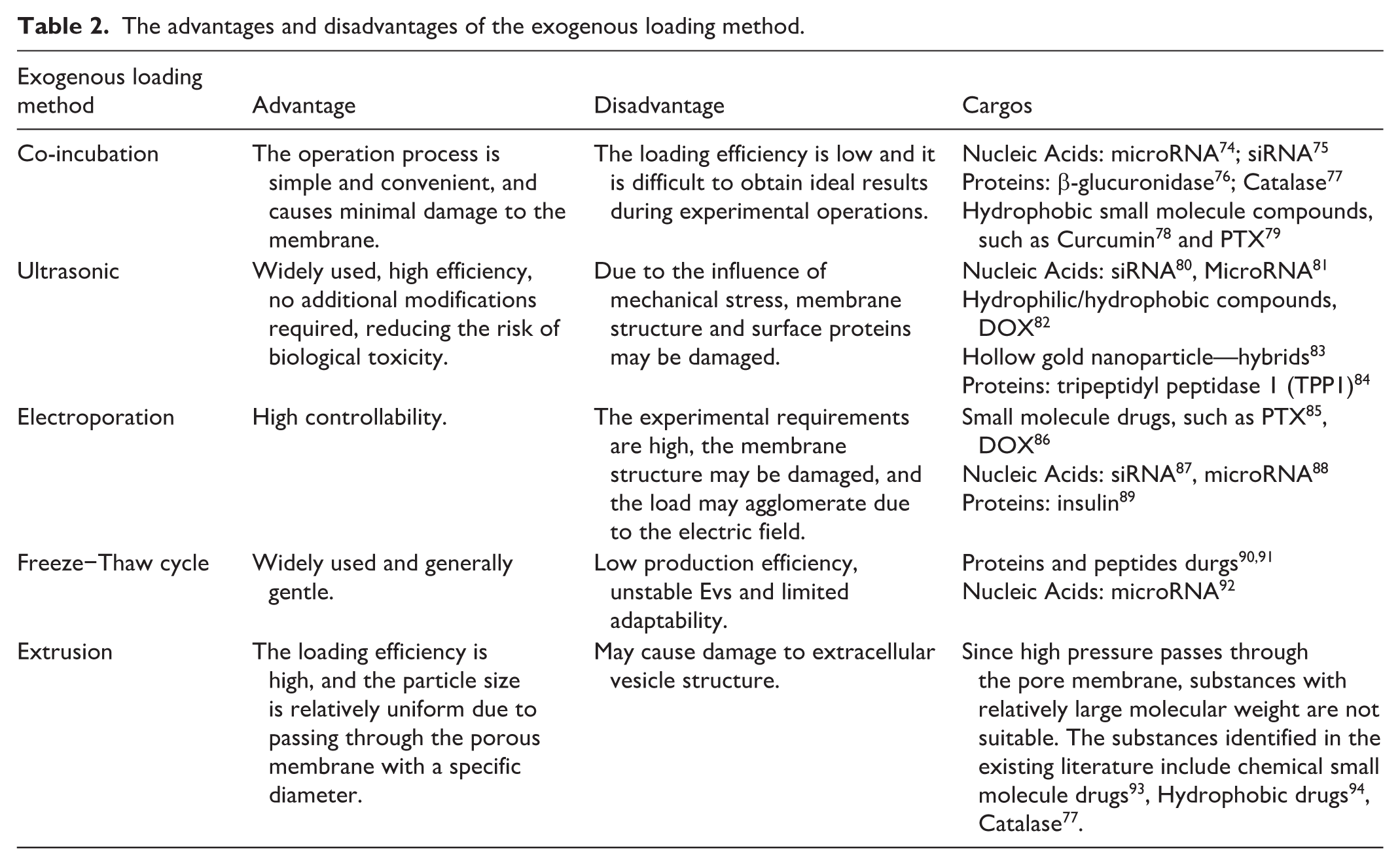
With the deepening of research, many loading methods have been developed, but unfortunately there is currently no efficient and balanced drug delivery method is an important factor restricting clinical application. Although various drug loading methods differ significantly in principle and operation, they exhibit quantifiable differences in key performance indicators such as loading efficiency, stability, and reproducibility. Loading efficiency is the primary indicator for evaluating drug loading methods. Co-incubation methods typically exhibit stable but low loading efficiency and are often used for loading hydrophilic small molecule drugs. The loading efficiency of small molecule drugs is similar to that of nucleic acid drugs and protein molecule drugs, generally speaking, the load efficiency will not exceed 15%74,75,95–97. Similar to extrusion, ultrasonic methods utilize mechanical shear force to distort the membrane structure, enabling efficient intercalation of small molecule drugs98,99. Ultrasonic loading supports a wider variety of drug types than extrusion loading, and its loading efficiency is significantly higher. Electroporation loading efficiency fluctuates considerably, varying greatly with different electric field conditions. Electroporation is suitable for loading small molecule drugs, nucleic acid drugs, and protein drugs; however, nucleic acid molecules exhibit the lowest loading efficiency among all loaded drugs89,100–102. The freeze−thaw cycle method has a relatively narrow application, mainly used for loading protein molecules. Co-incubation, because it does not damage the membrane structure, has a significant advantage in stability. Ultrasonic, electroporation, and extrusion methods cause greater damage to the membrane structure, and therefore their stability is less than satisfactory.
From a practical perspective, among the above drug loading methods, due to the safety of electroporation, this method is only suitable for small-scale preparation under safe conditions and cannot form a certain production scale in reality, and will cause the loading to aggregate 103 . The ultrasonic method is likely to disrupt the membrane structure 104 ; the extrusion method has high requirements on equipment and cost, and the extrusion method is sometimes used with saponin, which has a certain degree of toxicity and is not suitable for large-scale use 105 . Although the co-incubation method is low in efficiency, overall it is the simplest among all methods. It does not destroy the stability of the membrane, is low cost and highly safe 106 . In fact, during the co-incubation operation, reasonable temperature and pH can also be adjusted, or gentle stirring can be used to improve loading efficiency. Therefore, the co-incubation method has broad application prospects in clinical applications 107 .
Improvement of targeting ability
In addition to loading drugs into sEvs, if we want these drugs to effectively exert therapeutic effects in vivo, it is essential to modify the membrane structure of sEvs to obtain high targeting capabilities and drug delivery capabilities and increase the circulation time of sEvs in the body. Currently, the commonly used approaches for this purpose include chemical modification, genetic engineering 108 , and physical stimulation. The advantages and disadvantages related to this matter have been summarized in a table. Please refer to Table 3 for details.
The advantages and disadvantages of methods for enhancing the targeting properties of engineered Evs.

Genetic engineering
The approach of genetic engineering involves directing the fusion of a protein or polypeptide sequence to a selected membrane protein sequence 123 . This can also be referred to as targeted peptide technology. Targeted peptide technology is currently the most widely used membrane modification strategy 124 . Using genetic technology, the target gene is inserted, deleted or modified at specific sites, thereby enhancing the targeted delivery function of sEvs 125 . Protein fusion is presented through genetic techniques by editing sequences to construct fusion proteins and fuse the target protein with the membrane protein so as to improve the therapeutic ability of sEvs123,126,127. Genetic engineering is the basis for modifying the surface of exosomes to enhance their targeting ability. Specific proteins can be transplanted onto the exosome membrane by transfecting plasmid vectors. As shown in Figure 2, current research on modifying membrane structures through genetic engineering usually selects lysosomal membrane proteins represented by Lamp-2b128–132, and tetraspin proteins represented by CD63, CD9, and CD81133–136. Among them, since Lamp-2b has a signal peptide, Lamp-2b is often the subject of experiments 137 . The N-terminus of Lamp-2b is located on the extracellular side of the membrane and can be connected to the target sequence 123 . Liang et al. 128 constructed a plasmid and linked the chondrocyte affinity peptide to the membrane protein Lamp-2b to target the obtained sEvs to chondrocytes. In some studies, a glycosylation motif was added to the N-terminus of the peptide-Lamp-2b fusions to enhance the expression of the fusion protein and protect the target peptide from shearing or degradation138–140. To inhibit the loss of peptides in vivo, Hung et al. designed a fusion protein between the targeting peptide and Lamp-2b to contain glycosylation sequences at different positions. The introduction of the glycosylation motif both protected the peptide from degradation and resulted in increased overall expression of the Lamp-2b fusion protein in cells and sEvs 141 . In addition to Lamp-2b as the target for protein fusion, tetraspanin protein family is also often chosen as the target for protein fusion135,136. Based on membrane fusion technology, Li et al. 133 constructed engineered sEvs overexpressing CD63-VEGFC fusion protein, the engineered sEvs can be successfully delivered to lymphatic endothelial cells in mice, significantly improving lymphedema in mice. Liang et al. 135 introduced tumor cell targeting function by expressing CD63 transmembrane protein and Apo-A1 sequence in 293T cell hosts and delivered the generated sEvs to HepG2 cells. Genetic engineering is a modification strategy that often uses highly expressed membrane proteins in sEvs as target protein modification sites. In addition to the most commonly used Lamp-2b and tetraspanins, it also includes platelet-derived growth factor receptor, lactadherin, glycosylphosphatidylinositol, epidermal growth factor receptor, human epidermal growth factor receptor 2, and so on142–145. In addition to enhancing the targeted delivery capability, protein fusion can also enable sEvs to have a certain ability of drug polymerization. Li et al. 134 improved the RNA loading efficiency of sEvs by constructing a fusion protein in which the membrane protein CD9 was fused to HuR (an RNA-binding protein that interacts with MiR-155), in donor cells, fused CD9-HuR successfully enriched MiR-155 into sEvs when MiR-155 was overexpressed.

Genetic engineering methods improve the targeting and transport function of sEvs. In simple terms, it involves using genetic engineering to splice sequences and transfect them into donor cells, fusing the targeting peptide onto the membrane proteins of vesicles. The Evs produced by the donor cells then possess high targeting capabilities. Figure 2 was created using “biorender” (biorender.com)
Chemical modification
Chemical principles are used to modify membranes to enhance therapeutic capabilities 146 . Mainly includes covalent modification and non-covalent modification 147 . Among covalent modifications, the principle of click chemistry is the most commonly used. It was proposed by Nobel Prize winner in Chemistry K. Barry Sharpless in 2001 and refers to a type of highly efficient, highly selective, modular chemical reaction. The core features are mild reaction conditions, high yield, few by-products, and the ability to be completed quickly in a complex system, just like two molecules “clicking” together precisely. In the biomedical field, “click chemistry” can be applied in the fields of biomolecule labeling and imaging, drug development and delivery, diagnosis and biosensing. As shown in Figure 3, the principle of click chemistry which is the most common copper-catalyzed azide-alkyne cycloaddition (CuAAC) in click chemistry, is that the amino groups of membrane proteins can be easily modified with alkyne groups through the condensation reaction of EDC-NHS [1-ethyl-(carbodiimide-n-hydroxysuccinimide)] 148 . Alkyne-labeled proteins can then be bioorthogonally coupled to azide-containing reagents via a copper-catalyzed azide-alkyne cycloaddition (CuAAC) “click reaction” 149 .

Schematic diagram of chemical modification basics. (a) Covalent modification, using the principle of “click chemistry,” EDC/NHS condenses the alkyne group of the alkyne-containing compound onto the amino group of the protein; then a bioorthogonal reaction with an azide-containing reagent is carried out under copper ion catalysis. (b) Non-covalent modification, which are hydrophobic insertion, aptamer modification, and electrostatic interaction, aim to improve the uptake of vesicles by target cells or tissues. Figure 3(a) and (b) was created using “biorender” (biorender.com).
Treating Ev membranes with covalent modifications allows highly specific binding, meaning drugs or other molecules can be precisely attached to the membrane, thereby enhancing targeting and therapeutic efficacy20,150,151. To develop engineered exosomes for glioma, Jia et al. 150 used sEvs produced by RAW264.7 cells to study the NRP-1 transmembrane protein overexpressed in glioma cells, the research team used EDC-NHS to condensate 4-pentynoic acid with phosphatidylethanolamine on the exosome membrane, then, the REG with azide (NRP-1 targeting peptide) and the alkyne group undergo a cycloaddition reaction using the principle of click chemistry to couple to the membrane through chemical bonds, thereby obtaining targeting ability. Considering that high concentrations of copper ions are toxic to cells and cause physiological disturbances to sEvs, a bioorthogonal reaction without copper ion catalysis is a more suitable approach 152 . Bertozzi et al. developed a metal-free variant, strain-promoted azide-alkyne cycloaddition (SPAAC), by careful design and modification of the alkyne group, which has been used to label glycoproteins on the cell surface in vitro and in vivo without significant cytotoxicity. This also led to bioorthogonal reactions, which use the principles of click chemistry to undergo chemical reactions in vivo without interfering with their own biochemical reactions. However, some chemists were not satisfied with the second-order reaction rate of SPAAC, so Blackman et al. successfully developed the inverse electron demand Diels-Alder (iEDDA) reaction between the cycloaddition of s-tetrazine and trans-cyclooctene (TCO) derivatives, resulting in a copper-free click chemistry reaction that is faster than the SPAAC reaction. Blackman is a chemist, and his achievements were mentioned in the two articles.153,154 These reaction methods are all covalent binding, which does not lead to changes in the size and function of sEvs 155 , and also enhanced targeting capacities. “Click” chemistry usually involves fast and efficient reactions that can complete a large number of modifications in a short period of time 146 . Such efficiency is crucial for future large-scale production and applications. “Click” chemistry is different from general traditional chemistry 156 . The reaction of traditional chemistry must take into account changes in temperature and pressure, which often leads to the destruction of the Phospholipid bilayer in biomedical experiments 148 . “Click” chemistry reacts very quickly, and has no negative impact on sEvs size or bioactivity 104 . This also fully demonstrates that the application of “click” chemical principles is a suitable method for surface modification 148 .
Inserting Phospholipid molecules into the lipid bilayer (hydrophobic insertion) is not a covalent coupling process, as it does not form a covalent bond with the membrane. Since the vesicle membrane is a Phospholipid bilayer, Phospholipid molecular binding with functional groups can be inserted into Ev membranes by hydrophobic interactions 157 . It is also a method to enhance targeting and drug delivery efficiency. Kim et al. 151 used DSPE-PEG-AA to modify the surface of sEvs released by autologous macrophages. PEG (polyethylene glycol) prolonged the blood circulation time of drug carriers in vivo to target the sigma receptors overexpressed in lung cancer cells. DSPE (1,2-Distearoyl-sn-glycero-3-phosphoethanolamine) is a Phospholipid and is fused to the Phospholipid membrane, thus making a rational modification. Cheng et al. combined the receptor binding domain (RBD) targeting SARS-CoV-2 spike protein to the linking molecule DSPE-PEG-NHS to form RBD-PEG-DSPE, which was inserted onto the surface of Evs derived from lung glomus cells by co-culture, and loaded mRNA vaccine into Evs by electroporation. Develop novel vaccines targeting lung mucosal cells and alveolar cells 158 . In addition to the hydrophobic insertion method, there are many other non-covalent binding methods. For example, since the sEvs membrane carries a negative charge, a portion of positive charge is added to the sEvs membrane to enhance the targeting efficiency of the biomembrane by utilizing the principle of multivalent electrostatic interactions 159 , 160 ; and aptamers are used for surface modification 161 .
Although chemical modification of membranes allows for rapid and efficient enhancement of vesicles with high targeting capabilities and efficient drug delivery, there are still challenges and drawbacks. For example, the surface complexity of vesicles produced by different cells varies, which may affect the efficiency of the reaction and even endanger the structure and function of the carrier162–164.
In addition, the use of chemically modified vesicles may encounter issues of cytotoxicity in clinical transformation.
To enhance the targeting of therapeutic Evs, in addition to the most commonly used genetic engineering and chemical modification methods such as peptide fusion and hydrophobic insertion, and aptamer modification to obtain engineered exosomes, physical principles can also be used to achieve more precise targeting 165 .
Physical stimulation
The integration of Engineered EVs with external physical stimuli (such as ultrasound, laser, and magnetic fields), can achieve more precise targeted delivery. Villa et al. 116 used pretreated myo-exosomes, immobilized them on ferromagnetic nanotubes, to achieve controlled and precise delivery of myo-exosomes to skeletal muscle, promoting beneficial muscle responses in muscular dystrophy mice. In addition to using magnetic fields, ultrasound is also a highly efficient method to enhance the targeting of nanovesicles. In myocardial infarction research, adipose stem cells were pretreated to secrete exosomes (ADSC-EXO-SDF-1α) that highly express stromal cell-derived factor 1α (SDF-1α). A dual-modified targeted nanobubbles (TNBCD81-cRGD) were designed—using perfluoropropane (C3F8) as the gas core and phospholipid DSPC as the shell, with a cyclic arginine-glycine-aspartic peptide (cRGD) targeting the myocardium and an exosome-anchored antibody (anti-CD81) simultaneously attached to the surface. Triggered by low-intensity pulsed ultrasound (LIPUS), the nanobubbles directionally burst, releasing exosomes. The greatest advantage of this method is its high spatiotemporal control precision, allowing for non-invasive focusing on tiny deep regions within the body, achieving on-demand, targeted release 166 . Compared with gene engineering targeted peptide fusion technology and membrane modification technology based on chemical principles, the biggest advantage of physical intervention is that it makes up for the lack of targeting while achieving “spatiotemporally controllable” treatment. The physical signal can be turned on or off at any time, and the timing and duration of treatment can be artificially controlled.
Integration with materials
When sEvs loaded with drugs enter the body, their release effect will have a direct impact on the treatment 167 . In clinical therapy, people often seek to use treatment methods that can provide stable and long-lasting therapeutic effects 168 . If the drug release efficiency is inefficient, frequent administration will be required; if the dose is too large, it may affect patient compliance 168 . Therefore, efficient drug release is essential. Based on this, biomaterials born in recent years have been widely studied, which has also enriched the application of engineered sEvs. There are many studies that use biomaterials in combination with sEvs to play roles in neural tissue regeneration169,170, muscle tissue regeneration 171 and cartilage regeneration172,173 and other areas. The widely used hydrogel is a hydrophilic three-dimensional network structure that can absorb a large amount of water. Its synthetic raw materials are diverse, and the molecules of the material rely on physical bonding and chemical bonding 174 , Mixing drugs with hydrogels allows for slow release to maximize therapeutic effects 175 . The details are shown in Figure 4. In the field of cartilage regeneration, Guan et al. introduced aldehyde-functionalized chondroitin sulfate (OCS) into gelatin methyl methacrylate (GM) to form gelatin methacryloyl oxidized chondroitin sulfate (GMOCS) hydrogels to investigate the treatment of growth plate cartilage damage. They then introduced sEvs from bone marrow MSCs and observed that the sEvs were continuously released for up to two weeks, ensuring the optimal biological effect on the growth plate 172 . Sang et al. 173 cross-linked Pluronic F-127 and hyaluronic acid in situ to obtain a thermosensitive, injectable hydrogel that can serve as a sustained-release carrier to retain primary chondrocyte-derived sEvs of SD rats in the damaged cartilage for a long time, effectively amplifying its repair effect. In the field of nerve repair, Liu et al. 169 used photo-cross-linked hyaluronic acid methacrylate to make hydrogels of different Stiffness and combined them with sEvs for sciatic nerve repair, Compared with stiff hydrogels, soft hydrogels have better repair effects on damaged peripheral nerves. In muscle tissue repair, Rolland et al. 171 used PEP (purified exosomal product) produced by platelets to mix with fibrin glue in vivo to form a hydrogel. PEP promote M2 polarization by upregulating NF-κB and PD-L1 in damaged tissues, thereby promoting skeletal muscle proliferation and differentiation in a mouse skeletal muscle injury model; in a pig urinary incontinence model, it can facilitate the regeneration of damaged muscles 171 .
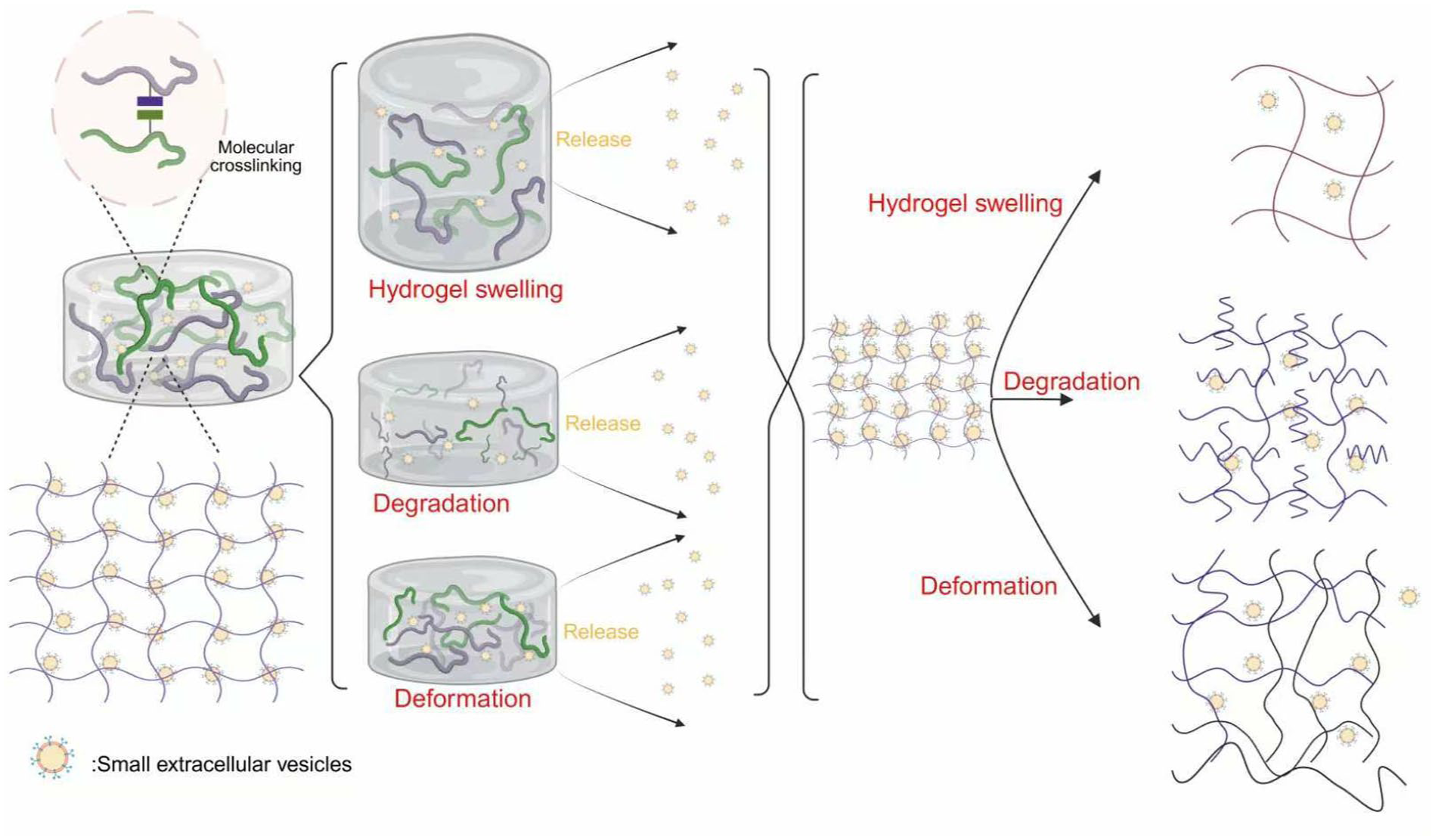
Schematic diagram of the basic hydrogel encapsulation and release technology. Figure 4 was created using “biorender” (biorender.com).
In addition to hydrogels, 3D scaffolds are another biomedical material that has been developed in recent years 176 . In addition to the most basic mechanical support, the morphology and properties of the obtained Evs can also be better than those of traditional 2D culture 177 . Regeneration of craniofacial bones after injury remains a challenge 178 . Kang et al. 179 used 3D bioprinting technology to create a dECM (decellularized extracellular matrix)/Gel (gelatin)/QCS (quaternized chitosan)/nHAp (nano-hydroxyapatite) 3D scaffold and attached sEvs to the scaffold using electrostatics, which showed increased bone and vascular regeneration in both in vivo and in vitro experiments. The osteogenesis process is closely related to the formation of systemic vascular networks 180 . The method of combining bioactive substances with scaffolds to achieve slow release while supporting the structure has been widely used181,182. Commonly used therapeutic substances include DFO (deferoxamine), simvastatin, growth factor VEGF, and so on. The commonly used carriers can be roughly divided into three categories, namely, hydrogels, microspheres, and nanoparticles (liposomes, exosomes) 183 . In addition, some new biomedical materials have great potential, such as polymer nanomaterials, metal nanomaterials, carbon nanomaterials, quantum dot nanomaterials, and so on27,28,31. These innovative materials are usually combined with hydrogels and exosomes and are presented in the form of 3D-printed scaffolds or as nanoparticles, addressing multiple mechanism issues. Although the use of biomaterials provides new ideas and methods for efficient treatment, the immune response in the body and whether they have adverse effects on drugs still require further exploration.
This picture represents the different principles of hydrogel release from top to bottom: the molecular cross-linked network inside the hydrogel swells when exposed to water, and the pores become larger and then released; the molecular cross-linked network inside the hydrogel degrades when exposed to water, and the pores become larger and released; the molecular cross-linked network inside the hydrogel deforms when exposed to water, and the pores become larger and then released.
Clinical application
Since there are few discussions on the clinical application of engineered Evs, the translation rate of results needs to be improved. Although the application of engineered exosomes in clinical treatment is still in the early exploration stage, its huge therapeutic potential based on its unique delivery mechanism and combination with new biomedical materials has driven academia and industry to actively plan its clinical transformation path.
As of 2025, there are nearly 560 exosome clinical drugs under development around the world, but most of them are in the early stages, and the field as a whole is still in the exploration and verification stage. There are two main types of Evs used in clinical trials, namely, human tissue/cell exosomes and plant exosomes 184 . So far, the complete process and results of clinical trials using exosomes from human samples have been reported; however, plant-derived exosomes are still in their infancy, and there is currently no data from clinical trials 185 . In an analysis of exosomes in human samples under different conditions, a total of 116 trials were recorded, of which 50% were biomarker applications, 28% therapeutic, 5.1% drug delivery studies, and the least related to vaccine research 184 . The Globaldata database shows that the largest clinical application area of Evs is oncology (54%), followed by central nervous system diseases (13%) and infectious diseases (8%) 186 . Because it is released under pathological and physiological conditions such as cancer, immune response, neurodegeneration, and cell death, and contains intracellular proteins, nucleic acids and other substances, it can be used as a clinical marker for disease diagnosis and assessment of the severity of the disease 187 , its internal microRNA is the most commonly used tissue-specific biomarker 165 , in cardiovascular diseases, the levels of microRNAs related to them are increased. Measuring the levels of microRNAs inside Evs can provide a strong basis for disease diagnosis 188 . In addition, in the field of extracellular vesicle diagnostic applications, most of the cancer diagnoses that account for the highest proportion adopt liquid biopsy, which is a minimally invasive and rapid method for in vitro diagnosis of tumors. Because the bioactive substances carried by tumor Evs are expressed differently in normal people and tumor patients, the detection of markers can help improve the specificity and sensitivity of early diagnosis186,189–191.
Generally, human exosomes are concentrated using differential centrifugation and ultrafiltration, and then identified through electron microscopy and detection of relevant biomarkers 185 . To ensure their bioactivity, compliance with Good Manufacturing Practices (GMP) is fundamental to successful clinical trials. In recent years, GMP-grade extracellular vesicle production methods have focused on cell type, culture environment and system, dissociation enzymes, and culture media, and further purification is required after production 185 . It generally involves three steps: removing cells and cell debris; concentrating the conditioned medium; and purifying the product185,192.
While most applications rely on biomarkers for diagnosis, a growing number of clinical trials are exploring drugs that utilize Evs as therapeutic agents. A search of clinicaltrials.gov yielded the following Table 4.
Clinical trial situation.

The clinical application and transformation rate of Evs are not ideal, and data on the clinical application of engineered Evs are severely lacking. This is because their reliable effects depend on a suitable microenvironment and a well-controlled separation system. Currently, various cell types have been used for the effective separation and fabrication of Evs, such as HEK293 cells 193 , MSCs 194 , dendritic cells 195 , and adipose-derived MSCs 196 . Downstream processing and purification methods for Evs include density centrifugation, precipitation, chromatography, and membrane filtration. These processes all affect its quantity, stability, and purity 197 , Therefore, with the expansion of clinical applications, the manufacturing processes developed under the background of academic theory need to be re-examined and updated.
Evs are at a crossroads in clinical application. Numerous academic publications and substantial research funding in the field have spurred exploration and research into their applications. Various strategies to optimize their therapeutic effects are under development, and regulatory frameworks are being established to ensure safe and successful clinical trials.
Conclusion and perspective
As a new generation of bio-nano delivery platform, engineered sEvs have demonstrated revolutionary potential in the fields of disease treatment, tissue repair and precision medicine. Through gene editing, chemical modification and bioengineering strategies, researchers have successfully overcome the limitations of natural sEvs such as insufficient targeting and low drug loading efficiency, and endowed them with characteristics such as precise delivery, efficient drug loading, and intelligent response. At present, engineered sEvs have made breakthrough progress in scenarios such as tumor immunotherapy, neurodegenerative disease intervention, bone/cartilage regeneration, and cross-barrier drug delivery.
However, currently engineered sEvs still face multiple technical bottlenecks: low efficiency of large-scale production and standardized quality control, low yield and high heterogeneity of mainstream methods (such as ultracentrifugation), and lack of international unified quality control standards; insufficient targeting accuracy, complex in vivo microenvironment (such as dynamic receptor expression or barrier penetration requirements) lead to single-target modification failure and significant off-target effects; drug delivery technology is limited, existing methods (electroporation, freeze−thaw) are prone to damage the capsule membrane and limit the loading efficiency of macromolecules (such as CRISPR system), while endogenous drug delivery relies on parent cell modification, with a long cycle and reduced cell activity; in vivo metabolism and safety assessment system is imperfect, long-term immunogenicity risks (such as modified protein-induced antibody accumulation) and cross-species clearance mechanisms have not yet been clarified, restricting the progress of clinical transformation.
For the application of engineered Evs, high standards and regulations must be followed, with rigorous quality control over vesicle loading efficiency and safety, and strict receptor clinical trials. Close attention must be paid to adverse reactions after drug administration, and the conversion rate of experimental results into clinical applications must be accelerated; this is crucial for promoting clinical development. Due to the low yield and high heterogeneity of traditional separation and preparation methods, in addition to established preparation techniques such as ultrafiltration, polymer precipitation, and immunomagnetic beads, emerging technologies have further expanded the application scope, including microfluidics, tangential flow filtration, and asymmetric flow field-flow fractionation, to fully explore their biomedical potential 198 . In the field of biomedicine, 3D printing technology has brought spatial structural control to the research of engineered extravesical vesicles, mainly in two aspects: first, the donor cell culture microenvironment used for vesicle production; and second, implantable scaffolds used for delivery. In the near future, it will be possible to print hydrogel scaffolds with specific physicochemical properties to simulate different tissues in the human body and guide cells to secrete “customized” EVs with specific tissue orientation and function 199 . The core of artificial intelligence lies in its ability to process complex data and make intelligent predictions. It can effectively address challenges such as heterogeneity and functional complexity in EV research. Currently, the preparation and characterization of engineered Evs are highly dependent on the operational data of laboratory personnel, resulting in strong subjectivity and significant bias. Future breakthroughs lie in developing intelligent analysis platforms that integrate machine learning and computer vision. For example, by analyzing nanoparticle tracking analysis data and high-resolution microscopic images using deep learning algorithms, AI can automatically identify the size distribution, concentration, and surface markers of EVs, achieving high-throughput, standardized characterization at the single-particle level and significantly improving the reproducibility and reliability of the data 200 . Synthetic biology involves genetic recombination of donor cells to enable them to continuously and stably produce Evs with specific functions. For example, hybridization of MSCs and neutrophils (NEs) has demonstrated that MSCs-NEs hybrid cells (HCs) possess the biological characteristics of both MSCs and NEs, and their exosomes H/Exos inherit the rapid inflammatory response of NEs and the tissue repair properties of MSCs. Even after multiple passages, the phenotypes of HCs and their exosomes remain stable. This opens a new avenue for the large-scale production of EVs from non-passaged cell sources 201
As nanotherapy becomes increasingly important in biomedicine, it is foreseeable that technological innovation will foster a highly integrated “design-manufacturing-application” platform, ultimately driving engineered Evs from basic research to clinical translation and large-scale production, thus opening a new chapter for next-generation biomedical applications.
Footnotes
Acknowledgements
The authors thank all participants enrolled in the present study. Furthermore, Figures 2–4 in this manuscript were drawn by the biorender platform.
Ethical Considerations
Since the manuscript is a review article and does not include case reports or clinical trials, there was no need for informed consent or patient consent. This article does not contain any studies with human or animal participants. There are no human participants in this article and informed consent is not required.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by [Haoyu Zhang]. The first draft of the manuscript was written by [Haoyu Zhang] and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Family Planning Research Fund of Wuhan City (WX18M01 and WZ22Q13); Knowledge Innovation Special Project of Wuhan Municipal Bureau of Science and Technology (2022020801010547); Hubei Provincial Health Commission Medical Research Achievement Transformation Project (No. WJ2023ZH0025).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
