Abstract
The optimal intensity of conditioning regimens for allogeneic hematopoietic stem cell transplantation (allo-HSCT) in patients diagnosed with myelodysplastic syndromes with excess blasts (MDS-EB) remains debated. We retrospectively analyzed the clinical outcomes of 102 MDS-EB patients who received an individualized conditioning protocol based on age and comorbidity, including busulfan, fludarabine, cytarabine, and low-dose total body irradiation (TBI). All patients achieved successful engraftment, with a median time to neutrophil and platelet engraftment of 12 and 13 days, respectively. The cumulative incidence of grade II-IV acute graft-versus-host disease (GVHD) at 180 days was 16.9%, while moderate-to-severe chronic GVHD at 2 years occurred in 15.0% of patients. After a median follow-up of 23 months, the 2-year relapse incidence was 11.5%. The 2-year overall survival (OS), progression-free survival (PFS), and GVHD-free/relapse-free survival (GRFS) were 81.9%, 77.3%, and 66.4%, respectively. The very high-risk subgroup of the International Prognostic Scoring System for MDS (IPSS-M) exhibited significantly higher relapse rates compared with the lower-risk groups (17.8% vs 2.4%, P = 0.03). Multivariate analysis identified IPSS-M as the only independent predictor of PFS (hazard ratio (HR) = 3.30, P = 0.04), whereas transplant conditioning intensity (TCI) showed no association with survival outcomes. These findings suggest that the age- and comorbidity-adjusted conditioning regimen achieves high engraftment rates, low relapse, and favorable survival in MDS-EB patients, with disease biology (IPSS-M) outweighing conditioning intensity in prognostic relevance.

Keywords
Introduction
Myelodysplasia syndrome (MDS) is a neoplastic disease originating from hematopoietic stem cells. It is characterized by morphological dysplasia, ineffective hematopoiesis, peripheral blood cytopenia, and a high risk of malignant transformation1–3. Currently, allo-hematopoietic stem cell transplantation (allo-HSCT) remains the only potentially curative treatment for MDS, particularly for patients with MDS-EBs4–6.
Myeloablative conditioning (MAC) regimens are known to reduce relapse risk but are often correlated with elevated treatment-related mortality, making them unsuitable for elderly patients. Conversely, reduced-intensity conditioning (RIC) regimens offer a lower toxicity option, relying more on the graft-versus-tumor effect for disease elimination7–12. However, despite numerous studies comparing MAC and RIC, the optimal conditioning intensity for patients with MDS remains under active debate. The conflicting results underscore the need for further studies to refine the selection criteria for conditioning intensity in MDS patients undergoing allo-HSCT. This challenge is further complicated by the demographic shift toward an aging transplant population. Real-world data have shown a steady increase in the number of older patients—particularly those older than 65 years—receiving allo-HSCT for MDS13,14. This trend highlights the urgency of developing dose-adjusted conditioning regimens tailored to age and comorbidities, to minimize toxicity while maintaining efficacy 15 . The traditional RIC/MAC classification lacks granularity in measuring conditioning intensity. To address this, the transplant conditioning intensity (TCI) score has been proposed as a more standardized and quantitative system. A previous study validated the TCI score in patients with acute myeloid leukemia (AML) and demonstrated that it outperformed the conventional RIC/MAC dichotomy in predicting outcomes16,17. However, to date, no studies have assessed the applicability of the TCI system for MDS patients.
To evaluate the efficacy and safety of an age- and comorbidity-adjusted conditioning regimen consisting of busulfan, fludarabine, cytarabine, and TBI, we conducted a retrospective analysis of MDS-EB patients who received allo-HSCT at our center, with the additional aim of validating the TCI score in this population.
Materials and methods
Patients
From January 2016 to July 2023, MDS-EB patients diagnosed according to the 2016 World Health Organization (WHO) classification underwent allo-HSCT at our center 18 . Risk stratification was performed at the time of MDS diagnosis using International Prognostic Scoring System for MDS (IPSS-M) 19 . Genomic DNA was extracted from bone marrow (BM)-derived cells at diagnosis and mutations were assessed using next-generation sequencing (NGS) with the core-AML mutation panel (Rightongene, Shanghai, China) that includes 42 genes frequently mutated in AML and related myeloid malignancies. The research adhered to the principles of the Declaration of Helsinki and was by the Ethics Committee of Shanghai General Hospital (Approval ID: 2024-217). Written informed consent was obtained from all participants or their authorized guardians.
Transplant procedures
High-resolution HLA typing at the HLA-A, HLA-B, HLA-C, HLA-DRB1, and HLA-DQB1 loci was used to select donors. Donor types included matched sibling donors (MSDs) and haploidentical family donors (HIDs), and unrelated donors (URDs), sourced from the China Marrow Donor Program. Two age- and comorbidity-adapted pre-transplant conditioning regimens were applied. For patients younger than 55 years old, the regimen included intravenous busulfan (3.2 mg/kg/day for 3 days), fludarabine (30 mg/m²/day for 5 days), cytarabine (1–1.5 g/m²/day for 5 days), and TBI at a dose of 3 Gy. Patients aged 55 or older, or those with a hematopoietic cell transplant comorbidity index (HCT-CI) score ≥2, received a modified protocol with reduced busulfan (3.2 mg/kg/day for 2 days) and cytarabine (1 g/m²/day for 5 days) doses, while fludarabine and TBI were administered at the same doses. TBI (3 Gy) was delivered as a single fraction on day −1. Conditioning intensity was assessed using the TCI scoring system 8 , which was designed to standardize the evaluation of TCI. The TCI score assigns intensity-weighted scores to commonly used conditioning regimen components, which are then summed to calculate the TCI score. The intensities of conditioning regimen were categorized into low (1–2), intermediate (2.5–3.5), and high (4–6) groups 8 .
For HID transplantation, two regimens were used: anti-human thymocyte globulin (ATG, Thymoglobin) or low-dose ATG/post-transplant cyclophosphamide (PTCy)-based20,21. The ATG-based regimen included ATG (2.5 mg/kg/day on days −4 to −1) and cyclosporine A (CsA) (2 mg/kg/day from day −5 with a nadir range of 200–300 ng/ml), short-term methotrexate (MTX) (10 mg/m2 on day +1; 5 mg/m2 on day +3 and +6), and mycophenolate mofetil (MMF) (15 mg/kg, administered three times daily from day +1 to +30). In the low-ATG/PTCy-based group, patients received ATG (2.5 mg/kg/day on days −2 and −1), PTCy (50 mg/kg on day +3), CsA (2 mg/kg/day from day +4), and MMF (15 mg/kg three times daily from day +4 to +34). For MSD transplantation, GVHD prophylaxis included ATG (2.5 mg/kg/day on days −2 and −1), CsA, short-term MTX (10 mg/m² on day +1 and 5 mg/m² on days +3 and +6), and MMF (15 mg/kg twice daily from day +1 to +30). Patients undergoing URD transplantation received the same low-dose ATG/PTCy protocol. Granulocyte colony-stimulating factor (G-CSF) was initiated on day +5 and maintained until neutrophil recovery. Ganciclovir was given to all patients from day −7 to −1 for antiviral prophylaxis. Posaconazole treatment began with conditioning and was continued for at least 3 months after transplantation, including during the administration of cyclophosphamide.
Definition of engraftment and GVHD
Neutrophil engraftment was determined by achieving an absolute neutrophil count >0.5 × 109/l for three consecutive days, without the use of G-CSF. Platelet engraftment was defined as the first day on which platelet counts exceeded 20 × 109/l for seven consecutive days, without platelet transfusions. Chimerism was evaluated using short tandem repeat (STR) PCR on BM aspirates. Full donor chimerism was defined as ≥95% donor-derived T cells in BM samples 22 . Acute graft-versus-host disease (aGVHD) was assessed using the modified Glucksberg criteria 23 , while chronic graft-versus-host disease (cGVHD) was evaluated based on the 2014 NIH consensus guidelines 24 .
CMV-DNA and EBV-DNA detection
CMV-DNA and EBV-DNA levels in peripheral blood were monitored weekly or biweekly using quantitative PCR until day +100 or beyond. Reactivation was defined as two consecutive tests with viral DNA levels exceeding 1000 copies/ml25,26. Pre-emptive therapy with ganciclovir (5 mg/kg twice daily) or sodium foscarnet (50 mg/kg twice daily) was initiated if CMV-DNA levels exceeded 1000 copies/ml, regardless of serological status or post-transplant timing. Rituximab (375 mg/m² once) was administered when EBV DNA increased by one log or more within 1 week, or exceeded 1 × 105 copies/ml in high-risk patients with EBV reactivation.
Statistical analysis
The Kaplan–Meier method, complemented by the log-rank test, was used to estimate the probabilities for OS, PFS, and GRFS. The cumulative incidence of events such as relapse, non-relapse mortality (NRM), relapse-related mortality (RRM), CMV and EBV infections, as well as aGVHD and cGVHD was calculated using Fine-Gray’s method, treating relapse, and NRM as competing events. The Cox model assumes proportional hazards, meaning that the hazard ratio (HR) between any two groups remains constant over time. This assumption was validated by examining Schoenfeld residuals and conducting tests for non-proportionality to ensure the model’s appropriateness. The Cox and Fine-Gray models were evaluated using Stata/SE (version 14.0). Data analysis was performed with SPSS software (version 23.0). Cumulative incidence, Kaplan–Meier, and forest plots were created using the cmprsk, survival, and forestplot packages in R (version 4.0). A P-value of <0.05 was considered statistically significant.
Results
Patient characteristics
Between January 2017 and July 2023, 102 patients with MDS-EB underwent all-HSCT at our institution. The median age was 45 years, ranging from 16 to 68. The TCI scores of the conditioning regimens ranged from 2.5 to 4.5 (median = 3.5). The characteristics of the patients, donors, and transplantation procedures are summarized in Table 1. The distribution and functional classification of common MDS-related gene mutations in this cohort are shown in Fig. 1.
Patient characteristics.


Frequency of each gene mutation. Colors linked to the bars represent the gene function.
OS, PFS, and GRFS
Until follow-up, 17 patients died, including 6 from relapse and 11 from non-relapse causes, such as 6 infections, 2 respiratory failures, 1 cerebral hemorrhage, 1 myocardial infarction, and 1 gastrointestinal bleeding. For all patients, the 2-year OS, PFS, and GRFS were 81.9% (95% CI, 73.8%–88.1%), 77.3% (95% CI, 71.3%–85.2%), and 66.4% (95% CI, 59.7%–72.1%), respectively (Fig. 2a–c). Univariate analysis revealed that TCI did not significantly influence survival (Fig. 2d–f). A comparison of 2-year OS, PFS, and GRFS between patients with the intermediate and high TCI groups showed no significant differences: 78.3% versus 85.4% (P = 0.30) for OS, 72% versus 83.1% (P = 0.17) for PFS, and 64.5% versus 76.6% (P = 0.28) for GRFS. Instead, the IPSS-M score significantly influenced survival (Fig. 2g–i). The 2-year OS and PFS of the very high-risk IPSS-M group were markedly reduced compared with the other risk categories, with 73.1% versus 94.3% (P = 0.02) for OS, and 67.7% versus 91.1% (P = 0.01) for PFS. Multivariate analysis showed that the IPSS-M score was statistically correlated with PFS (HR, 3.30 (95% CI, 1.08–10.10); P = 0.04), but not with OS (HR, 2.48 (95% CI, 0.79–7.79), P = 0.12) or GRFS (HR, 1.28 (95% CI, 0.57–2.87); P = 0.54) (Fig. 3). To assess the prognostic significance of specific mutations and cytogenetic abnormalities, we performed univariate Cox regression for OS, shown as a volcano plot (Supplemental Fig. S1). TP53 mutations and complex karyotypes were associated with a trend toward poorer OS and were largely clustered in the very high-risk IPSS-M group. While most other mutations were not statistically significant due to limited events, these findings underscore the role of molecular and cytogenetic features in post-transplant risk assessment. All other factors, including recipient age and gender, disease status before transplant, ECOG performance status, HCT-CI score, TCI, donor type, and GVHD prophylaxis, did not significantly associate with OS, PFS, or GRFS.

Survival outcomes of all patients: (a) OS, (b) PFS, (c) GRFS. Survival outcomes stratified by TCI score: (d) OS, (e) PFS, (f) GRFS. Survival outcomes stratified by IPSS-M risk groups: (g) OS, (h) PFS, (i) GRFS. OS, overall survival; PFS, progression-free survival; GRFS, graft-versus-host disease-free/relapse-free survival; TCI, transplant conditioning intensity.

Forest plots showing results of multivariable analyses for OS, PFS, and GRFS based on the Cox proportional hazards model. HR and 95% CI values are listed in Supplementary Tables 1–3. HR: hazard ratio; CI: confidence interval.
NRM RRM and relapse
We used a competing risks model to compare the 2-year rates of RRM and NRM. Across all patients, the 2-year RRM was 6.3% (95% CI, 1.4%–11.1%), while the 2-year NRM was 10.4% (95% CI, 3.8%–17.1%) (Fig. 4a). Univariate competing risk analysis showed that IPSS-M had no effect on NRM or RRM (Fig. 4b). Subsequently, multivariate competing risk analysis was conducted to evaluate the factors affecting NRM and RRM. Our analysis revealed that factors such as recipient age and gender, disease status before transplantation, ECOG performance status, HCT-CI score, TCI, donor type, and GVHD prophylaxis regimen did not significantly impact RRM or NRM (Fig. 5).

Cumulative incidences of non-relapse mortality (NRM) and relapse-related mortality (RRM): (a) All patients; (b) Comparison between very high-risk and other IPSS-M groups. NRM: non-relapse mortality; RRM: relapse-related mortality; TCI: transplant conditioning intensity.

Forest plots showing results of multivariable analyses for the cumulative incidence of relapse, RRM, and NRM based on the competing risk regression model. SHR and 95% CI values are listed in Supplementary Tables 4–6. SHR: subdistribution hazard ratio; CI: confidence interval.
After a median follow-up of 23 months, 10 patients relapsed, with a median relapse time of 7.1 months. The estimated 2-year cumulative relapse rate (CIR) was 11.5% (95% CI, 4.6%–18.4%) (Fig. 6a). The IPSS-M very high-risk subgroup showed a substantially increased estimated 2-year CIR of 17.8% (95% CI, 6.9%–28.7%) compared with 2.4% (95% CI, 0.2%–7.1%) in other subgroups (P = 0.03) (Fig. 6b). Stratification by other variables, including TCI, age, and donor characteristics, showed no significant differences. Multivariate competing risk analysis also found no significant predictors of relapse. However, IPSS-M remained a borderline factor, showing a non-significant trend toward higher relapse in the very high-risk subgroup (SHR, 6.52 (95% CI, 0.70–60.45); P = 0.09) (Fig. 5).

Cumulative incidence of relapse: (a) all patients and (b) comparison between very high-risk and other groups based on IPSS-M score.
Engraftment
All patients engrafted successfully. The median time to neutrophil engraftment was 12 days (range, 10–25), while platelet engraftment occurred at a median of 13 days (range, 11–37). Chimerism monitoring showed that all patients achieved full donor chimerism by day 28 post-transplant, with a median donor chimerism of 99.69% (range, 96.21%–100%).
CMV and EBV reactivation
CMV reactivation occurred at a median of 64 days (range, 29–813), whereas EBV reactivation occurred later, at a median of 170 days (range, 36–226) post-transplant. The 1-year cumulative incidence rates for CMV and EBV reactivation were 55.3% (95% CI, 42.7%–66.2%) and 20.7% (95% CI, 13.9%–24.6%), respectively.
Cumulative incidences of GVHD
The cumulative incidences of aGVHD grades I–IV and II–IV within 180 days after transplantation were 28.0% (95% CI, 22.1%–34.3%) and 16.9% (95% CI, 11.6%–20.3%), respectively. Two years post-transplant, the incidence of cGVHD was 24.8% (95% CI, 21.4%–33.5%), whereas moderate-to-severe cGVHD occurred in 15.0% (95% CI, 11.1%–20.4%) (Fig. 7).
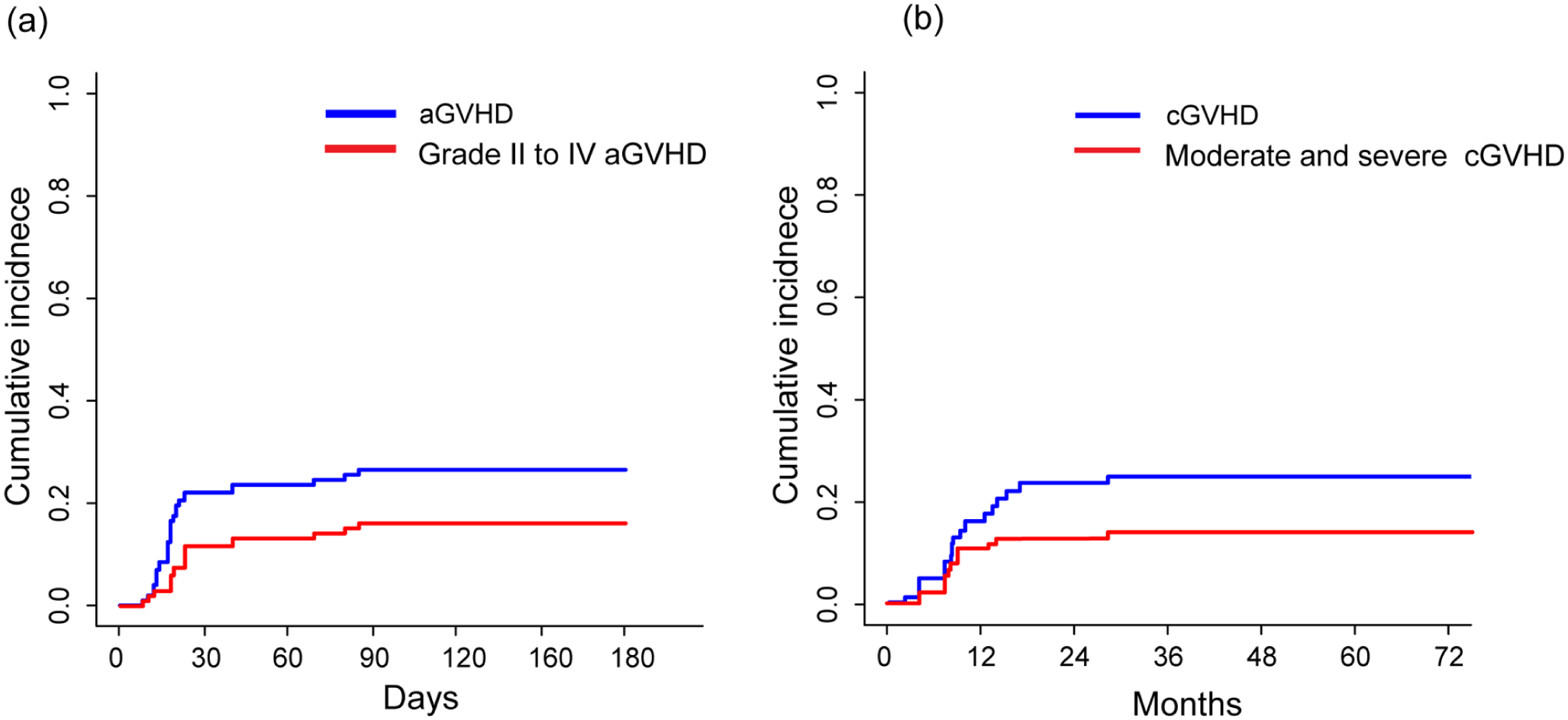
Cumulative incidence of (a) aGVHD and (b) cGVHD.
Discussion
In our study, we retrospectively evaluated the busulfan-, fludarabine-, cytarabine-, and TBI-based (BuFluAraC-TBI) conditioning regimen in MDS-EB patients receiving allo-HSCT. Our results demonstrate that this age- and comorbidity-adapted regimen achieved high engraftment rates and favorable 2-year OS, supporting its feasibility in balancing efficacy and safety.
With advancements in treatment modalities, the prognosis for patients with MDS has significantly improved11,27. However, the ideal pretransplant conditioning strategy for MDS patients remains contentious. To lower the post-transplant relapse rate, many centers have developed innovative intensified conditioning regimens. Cao et al. 28 added decitabine (5 days) to the Bu/Cy/Flu/AC regimen for 44 high-risk and very-high-risk MDS patients, achieving 2-year OS rates of 74% and 86%, respectively. Xia et al. used a decitabine-intensified BuCy2 regimen for high-risk MDS patients receiving allo-HSCT, reporting 3-year OS and disease-free survival (DFS) rates of 70.2% and 64.9%, respectively 29 . However, as MDS is most commonly diagnosed in individuals around 70 years of age, intensified conditioning regimen might not be tolerated by these older patients. Therefore, it is important to appropriately adjust the conditioning regimen intensity for aged and frail patients to balance efficacy and tolerability.
Busulfan, when administered at a reduced total dose of 6.4 mg/kg (3.2 mg/kg/day for 2 days), offers myeloablative efficacy while reducing toxicity. This dose adjustment is particularly beneficial for elderly and frail patients 30 . Based on a study by Potter et al. 31 , the 6.4 mg/kg busulfan-based conditioning regimen resulted in 5-year OS of 44%, event-free survival (EFS) of 33%, and NRM of 26% in patients with MDS and AML secondary to MDS. Moreover, previous studies have shown that fludarabine combined with myeloablative busulfan dosing yields comparable outcomes to fludarabine combined with a reduced dose of busulfan 32 , supporting the clinical utility of dose modification in selected patient populations. Fludarabine effectively reduces tumor burden in MDS patients while providing strong immunosuppressive effects to ensure successful engraftment. Conditioning regimens combining fludarabine with alkylating agents and immunosuppressants have become a key focus in RIC-HSCT research. Busulfan, treosulfan, and melphalan are commonly used alkylating agents33–35. In our study, fludarabine (30 mg/m²/day for 5 days) replaced cyclophosphamide as the lymphodepletion backbone, reducing the risk of hemorrhagic cystitis while preserving full donor engraftment rates. Importantly, the sequential administration of fludarabine followed by cytarabine may contribute to improved disease control due to their known synergistic interaction 36 . Fludarabine inhibits ribonucleotide reductase, thereby increasing the intracellular concentrations of Ara-CTP, the active metabolite of cytarabine. This potentiates DNA synthesis inhibition and promotes apoptosis in leukemic cells. This pharmacologic synergy has been supported by preclinical studies and forms the rationale for regimens such as FLAG in high-risk myeloid malignancies37,38. We believe that this interaction may partly explain the favorable outcomes observed in our BuFluAraC-TBI cohort, though further prospective studies are warranted to confirm this hypothesis. TBI has long been the standard MAC regimen, effectively suppressing the recipient’s immune system and facilitating engraftment 39 . Reduced-dose TBI was incorporated into conditioning regimens, balancing immune suppression with lower toxicity. Low-dose TBI (e.g. 2 Gy), when combined with immunosuppressive agents such as fludarabine, forms the basis of RIC regimens that promote chimerism and reduce the risk of graft rejection without excessive toxicity 40 . When combined with chemotherapy, TBI in RIC-HSCT has shown clinical efficacy41,42. Supporting literature indicates that patients who received 2 Gy TBI had lower transplant-related mortality (TRM) and longer EFS than those who received 4 or 6 Gy 43 . In our study, a 3Gy TBI dose was included as a component of the conditioning regimen21,44,45, demonstrating clinical efficacy in promoting chimerism while minimizing toxicity. Given the high-risk nature of MDS patients in our cohort, many of whom underwent haploidentical transplants—correlated with higher rates of GVHD and relapse—we hypothesize that the inclusion of 3Gy TBI significantly contributed to the accelerated engraftment and the higher-than-expected donor chimerism levels observed by day 28 post-transplant. The combination of 3Gy TBI, chemotherapy, and immunosuppressive agents likely played a crucial role in achieving stable, early donor chimerism, which is essential for optimal post-transplant outcomes, particularly in high-risk populations. This approach supports the idea that 3Gy TBI may facilitate a more robust immune tolerance and engraftment, contributing to early and stable chimerism in the context of RIC. In our protocol, for patients aged 55 or older, or having an HCT-CI score ≥2, busulfan was reduced to 6.4 mg/kg total, and cytarabine was reduced to 1 g/m²/day for 5 days. The results demonstrated both the safety and efficacy of this tailored conditioning protocol. In our study, the BuFluAraC-TBI regimen integrates pharmacologically optimized components to address the dual challenges of disease control and regimen tolerance in elderly MDS-EB patients undergoing allo-HSCT.
Achieving successful engraftment remains the primary goal in allo-HSCT for MDS patients. In 2020, Baumgart et al. reported an engraftment rate of 96% in 476 MDS patients receiving treosulfan or busulfan plus fludarabine-based conditioning for HID HSCT 46 . Similarly, Shin et al. 47 reported a 95% engraftment rate in 60 MDS patients treated with RIC non-T-cell-depleted HID HSCT. In our cohort, all patients achieved successful engraftment, with HID transplants comprising 56% of the cohort. When grouped by conditioning intensity, 55% of patients received an intermediate-intensity TCI regimen, suggesting that our conditioning regimen effectively ensures successful engraftment.
With the expanded application of ATG-based or PTCy–based protocols, the cumulative incidence of GVHD has been notably reduced across donor types48–50. Our previous study also showed that low-dose ATG/PTCy prophylaxis significantly reduced both aGVHD and cGVHD in HID and URD transplantation settings20,44,51,52. In our study, low-dose ATG/PTCy-based prophylaxis for GVHD was administered in 50% of the cohort. The results indicate that the cumulative incidences of grade II–IV aGVHD, as well as overall and moderate-to-severe cGVHD, were comparable to those observed with ATG-based prophylaxis in URD transplants. Several recent studies have shown that both ATG and PTCy, when properly used, may reduce GVHD without compromising the GVL effect. For example, a European Group for Blood and Marrow Transplantation (EBMT) analysis of MSD HSCT in MDS patients suggested a trend toward lower relapse in the PTCy group, with similar OS and GRFS outcomes 53 . A large registry-based study also demonstrated that PTCy is safe in URD settings, with no increased risk of graft failure 54 . Furthermore, ATG-based prophylaxis was correlated with better OS and reduced NRM compared with non-ATG regimens in MDS, without higher relapse risk 55 . Compared with ATG, PTCy may offer additional advantages in mismatched donor transplantation, including significantly lower cGVHD and NRM 56 . In our study, we combined ATG and PTCy, leveraging the strengths of both protocols. These findings support the application of low-dose ATG/PTCy in our cohort as a strategy that effectively modulates GVHD while potentially maintaining the GVL effect.
In our study, the 1-year incidences of CMV and EBV reactivation were 55.3% (95% CI, 42.7%–66.2%) and 20.7% (95% CI, 13.9%–24.6%), respectively. A study conducted in Japan, which included 1082 MDS patients, found that 57.5% of patients experienced CMV reactivation within the first 100 days after allo-HSCT, with an estimated 1-year cumulative incidence exceeding 60%, CMV reactivation occurred independently of conditioning intensity 57 . In our cohort, a few patients developed clinical CMV infections, primarily involving the lungs and bladder (e.g., CMV pneumonitis and cystitis). These cases responded well to antiviral and supportive therapy, and no CMV-related mortality occurred. The incidence of EBV DNAemia varies significantly across transplant centers, ranging from 27% to 63%58,59. A study on MDS patients reported that the 1-year incidence of EBV reactivation after allo-HSCT of 17.9%, which was independent of the conditioning regimen’s intensity 60 . Regarding EBV reactivation, no patients developed clinical EBV disease or post-transplant lymphoproliferative disorder (PTLD). Preemptive interventions, including reduction of immunosuppressive agents, intravenous immunoglobulin, and rituximab administration as needed, were promptly implemented and effectively controlled viral replication.
In our study, the 2-year relapse incidence was 11.5%. Our study demonstrated significantly lower relapse rates. Notably, subgroup analysis revealed no significant differences in CIRs (P = 0.57) or RRM (P = 0.61) between patients with high versus intermediate TCI scores. A retrospective analysis by EBMT reported a 10-year relapse rate of 25.2% in MDS patients receiving RIC allo-HSCT, compared with 25.7% in those receiving MAC 11 , another analysis reported a 2-year relapse incidence of 17% for RIC and 15% for MAC 61 . The EBMT study showed OS between RIC and MAC groups remained similar, suggesting that while RIC is associated with higher relapse rates, it does not necessarily translate into reduced survival 61 . In contrast, data from the BMT CTN 0901 prospective randomized trial indicated that MAC provided superior outcomes in certain subgroups, particularly among younger patients 62 . Our results suggest that for MDS-EB patients undergoing allo-HSCT, the age- and comorbidity- adjusted conditioning regimen might offer the potential to lower relapse risk without increasing treatment-related toxicity.
To further clarify the impact of conditioning intensity on clinical outcomes, we conducted a subgroup analysis. our results showed that TCI did not significantly affect OS, PFS, or GRFS, suggesting that conditioning intensity may not be as crucial as previously thought. Moreover, by using competing risks analysis, our study revealed that TCI did not significantly affect relapse, RRM or NRM. These findings suggest that for older or frail patients, reducing the dose of busulfan and appropriately lowering the TCI may help minimize treatment-related toxicity without compromising outcomes. Meanwhile, intermediate-intensity conditioning may serve as a promising option for younger patients with limited comorbidities, compared with full-intensity regimens. Specifically, a regimen based on reduced-dose busulfan (6.4 mg/kg) in combination with fludarabine, cytarabine, and TBI may provide a favorable balance between antitumor efficacy and reduced toxicity. This approach could help optimize transplant outcomes while avoiding excessive regimen-related complications in medically fit individuals. However, to validate these results, further prospective studies are essential. In addition, we found that IPSS-M score was significantly correlated with PFS and showed a trend toward significance for OS, highlighting the critical role of underlying disease biology in determining post-transplant outcomes. Previous studies typically included only traditional prognostic scores such as IPSS and IPSS-R, which did not account for genetic mutations. By incorporating the IPSS-M score, which includes genetic mutation data, our study provided a more comprehensive assessment of patient prognosis and highlighted the importance of genetic factors in MDS-EB. In the present study, only one relapsed patient was classified as high risk, whereas the remaining nine relapsed patients were categorized in IPSS-M very high-risk group. This group exhibited an elevated CIR in the univariate analysis. Multivariate analysis also supported a potential trend toward higher relapse rates in the very high-risk group, although this was not statistically significant. This finding confirmed the powerful predictive capability of the IPSS-M score in identifying the most relapse-prone patients6,63. These results reinforced the notion that the inherent characteristics of the disease, rather than the conditioning regimen intensity, represent the most influential factors in determining outcomes following transplantation.
However, our study has several inherent limitations. As a retrospective analysis from a single center, it is subject to potential selection bias and limited external applicability. Furthermore, the median follow-up duration was only 23 months, which limits our ability to reliably assess survival outcomes beyond the 2-year time point. Consequently, late-phase clinical events, particularly cGVHD progression and delayed relapse patterns, may not be fully captured. Future studies with extended follow-up periods are necessary to validate and complement our findings. Although statistically adjusted, heterogeneity in GVHD prevention strategies across donor types persists as a confounding variable. In addition, the relatively high mutation frequencies of GATA2, FLT3, and NPM1 observed in our study may be partly due to selection bias, as patients with these high-risk genetic features are more likely to be referred for allo-HSCT. Our relatively small sample size may further amplify this bias.
Conclusion
Our study demonstrated the age- and comorbidity-adjusted BuFluAraC-TBI-based conditioning regimen for patients with MDS-EB can achieve high engraftment rates and improved 2-year survival. Patients with very high IPSS-M score had a significantly high incidence of relapse even after allo-HSCT, indicating the need for effective maintenance therapy or immunotherapy post-transplant.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251380928 – Supplemental material for Age- and comorbidity-adjusted busulfan, fludarabine, cytarabine, and low-dose TBI-based conditioning regimen is effective for myelodysplastic syndromes with excess blasts undergoing allogeneic peripheral blood stem cell transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897251380928 for Age- and comorbidity-adjusted busulfan, fludarabine, cytarabine, and low-dose TBI-based conditioning regimen is effective for myelodysplastic syndromes with excess blasts undergoing allogeneic peripheral blood stem cell transplantation by Jie Gao, Huixian Wu, Huiying Qiu, Yin Tong, Chongmei Huang, Jun Yang, Baoxia Dong, Jiahua Niu, Kun Zhou, Yu Cai, Chang Shen, Xinxin Xia, Xianmin Song and Liping Wan in Cell Transplantation
Footnotes
Ethical approval
This study was approved by the Ethics Committee of Shanghai General Hospital (Approval ID: 2024-217).
Informed consent
Written informed consent was obtained from all participants or their authorized guardians.
Author contributions
Liping Wan and Xianmin Song conceptualized and supervised the study. Jie Gao and Huixian Wu analyzed the data and plotted the figures. Huiying Qiu, Jun Yang, Yin Tong, Kun Zhou, Baoxia Dong, Jiahua Niu, Liping Wan, Chang Shen, Xinxin Xia, Yu Cai provided patients’ clinical data. Jie Gao wrote the manuscript. Liping Wan and Xianmin Song revised the manuscript. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a 3-year development project from Shanghai Shen Kang Hospital Development Center (SHDC2020CR1012B for Xianmin Song), the National Natural Science Foundation of China (81570148 for Xianmin Song), the Bethune Charitable Foundation (YDTR-046 for Jie Gao).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
