Abstract
γδ T lymphocytes and NK cells are effective to kill tumors or viral-infected cells avoiding graft versus host disease (GvHD), thus they have attracted high interest as potential tool for adoptive cell therapy. We generated an advanced therapy medicinal product (ATMP) composed of mature γδ T and NK cells to provide an innovative tool to protect patients against tumor relapse and life-threatening infection after haploidentical hematopoietic stem cell transplantation. The ATMP was manufactured and validated in a GMP facility and was obtained from leukapheresis stimulated with zoledronic acid and IL-2, afterward depleted of αβ T lymphocytes using the CliniMACS Prodigy. The ATMP is characterized by high homogeneity, cell viability, cytotoxic abilities, stability after cryogenic preservation, and it was virtually free of αβ T and B lymphocytes. Both NK and γδ T cells were activated and characterized by high expression of cytotoxic and activating receptors including NKG2D, CD16, NKp30, NKp44, and NKp46. Furthermore, γδ T lymphocytes and NK cells were cytotoxic against myeloid leukemia or neuroblastoma cells. In conclusion, we implemented a novel ATMP to be shortly translated into clinical practice, which may be used in the post-transplant phase as efficacious immunotherapy in neuroblastoma and leukemic pediatric patients.

Introduction
Gamma-Delta (γδ) T lymphocytes and NK cells represent killer cells bridging the innate and adaptive immunity1,2 that are attracting growing interest as adoptive cell therapy, based on their unique, multi-functional, biological properties1–4.
First, NK and γδ T cells kill virally infected cells and tumors through a wide panel of receptors and ligands, such as NK group 2D (NKG2D), NK protein (NKp)30, NKp44, and NKp465–8, preventing immunological evasion driven by selective antigen loss2,9–14.
The mechanisms underlying the γδ T cell functions are primarily based on γδ TCR-mediated target recognition of phosphoantigens expressed on cancer or virally infected cells, with additional contributions from receptors and ligands including DNAX accessory molecule-1 (DNAM-1) or natural killer group 2 member D (NKG2D)15,16, consistently expressed also by NK cells. More recently, butyrophilin (BTN)2A1 and 3A1 have been reported as key mediators of phosphoantigen signaling and, for this reason, one of the main activators of γδ T cell functions17–19. By contrast, NK cell function is regulated by a delicate balance of signaling mediated by inhibitory and activatory receptors. Among the latter, the most important are defined “killer cell immunoglobulin-like receptors” (KIRs) involved in missing self-recognition and alloreactivity20,21. Furthermore, NKG2A is another receptor shared by NK and γδ T cells, acting as inhibitory mediator on NK cells, through the recognition of HLA-E molecule 22 , and as activating receptor on γδ T cells 23 .
Second, different from conventional αβ T lymphocytes, these killer cells recognize antigens in an HLA-independent manner 9 and do not induce graft versus host disease (GvHD), while supporting graft versus leukemia (GvL) effects24–27, a feature of particular importance in patients affected by leukemia receiving allogeneic hematopoietic stem cell transplantation (HSCT).
Third, they produce interferon (IFN)-γ and mediate antibody-dependent cellular cytotoxicity (ADCC) through CD16, a mechanism underlying the anti-tumor effects of combined therapy in solid and hematological malignancies28,29.
Next, γδ T lymphocytes may act as antigen-presenting cells toward αβ T elements, in the context of tumor antigens and microbial peptides 30 and also develop immunological memory 31 , as demonstrated by the finding that they may form long-lived memory cells able to robustly respond to secondary or tertiary exposure to bacterial agents and viruses. In addition, γδ T lymphocytes may respond to non-peptidic antigens by inducing a cascade of immune reactions, leading to αβ T and B cell activation, cytokine release, and cytotoxicity 32 . This memory-like feature is shared with NK cells 33 .
In the setting of allogeneic HSCT, the αβ T- and CD19+ B-depleted grafts have been proven superior to unmanipulated mismatched unrelated HSCT in terms of (1) faster immune reconstitution, (2) improved acute and chronic GvHD, (3) reduced transplant-related mortality, and (4) relapse-free survival34,35. The main reason associated with this success is related to the presence of mature γδ T and NK cells in the graft exerting a protective role against tumor relapse and life-threatening infections27,36–38. Indeed, a meta-analysis on clinical studies revealed that a high level of γδ T lymphocytes post-allogenic HSCT was associated with unaltered acute GvHD incidence, fewer episodes of viral infections, lower risk of relapse, improved disease-free survival (DFS) and overall survival (OS) 14 . Similarly, the administration of donor-derived expanded NK cells after HSCT were proven safe and associated with NK cell-dominant immune reconstitution in early phases post-transplantation, with preserved T-cell reconstitution, and improved DFS 39 . Notably, no clinical studies are currently available treating patients with a combination of NK and γδ T cells. Both cells can be expanded ex vivo from donors using different approaches and cell sources. Several groups included zoledronic acid (ZOL) and IL-2 stimulation to deplete αβ and enrich/expand γδ T cells40,41. The resulting cell formulation was predominantly composed of Vγ9Vδ2 T lymphocytes highly cytotoxicity, resilient to the suppressive activity of programmed cell death-1 (PD-1) and co-activating NK cells10,42. Importantly, the content of αβ T cells remained below the clinically acceptable standards for HSC therapy applications (5 × 105/kg). Although donor variability may affect efficiency on cell expansion 43 , this cell product was clinically administered to adult patients with different type of cancers, it was well tolerated, and a robust clinical response observed. It is also known that the donor variability may influence also the profile of NK cells 44 , thus many efforts are focusing to select the best donor to expand both γδ T and NK cells. In this context, a recent work by Jonus et al. 43 reported as the ZOL+IL-2-expanded leukocyte formulation, generated by healthy donors, was characterized by two predominant populations, the CD3+γδTCR+CD56−/+ γδ T cells and CD3−CD56+ NK lymphocytes. In the same study, the presence of NK cells within γδ T cell expansions did not warrant safety concerns. Furthermore, clinical trials based on NK infusion demonstrated the feasibility and safety of this approach and paved the way for advanced immunotherapies 45 . In patients affected by hematological tumors, a reduced leukemia relapse was observed in recipients of HLA haplotype-mismatched grafts with KIR ligand incompatibility, supporting beneficial effects in T-cell-depleted allogeneic HSCT 46 . Notably, recipients of αβ T-cell-depleted graft suffered from suboptimal immune-reconstitution and combinatory approaches boosting post-HSCT immune reconstitution, increasing the GvL and avoiding GvHD, are needed.
With this background, we developed a novel advanced therapy medicinal product (ATMP) composed of activated/cytotoxic γδ T lymphocytes and NK cells (GADEKILL), in accordance with good manufacturing practice (GMP), to be infused into pediatric patients receiving an haploidentical grafts with beneficial effects on leukemia relapse and life-threatening infections.
Methods
Manufacturing in GMP facility and process validation
Three leukaphereses (LA), harvested with Spectra-Optia system (Terumo BCT) and 1 buffy coat (BC) from healthy donors were obtained after informed consent (CER Liguria: 593/2021—DB id 11884), according to the Declaration of Helsinki. The processed volume was 51 ml (BC #1), 70 ml (LA #2), 113 ml (LA #3), and 103 ml (LA #4). Mononuclear cells (MNC), separated by Ficoll-Paque gradient (Cytiva) and CliniMACS Prodigy (Miltenyi), were seeded (5 × 106/ml) in gas-permeable differentiation bags (Miltenyi) and stimulated with ZOL (25 μM; Tillomed by Clinigen) in TexMACS medium (Miltenyi), 5% human AB serum, 1000 U/ml IL-2 (Proleukin by Novartis) for 4 days at 37°C, 5% CO2. The concentration of IL-2 and ZOL has been selected in accordance with previous results 47 . Afterward, cells were transferred to a ZOL-free culture medium (2 × 106/ml) for additional 10 days. Medium was replaced every 3–4 days. At the end of culture, αβ T cells were depleted by CliniMACS Prodigy and TCRαβ biotin and anti-biotin reagents (all from Miltenyi). All the afore-listed reagents were clinical grade. The flow chart of the process is summarized in Figure 1.

The production process of the GADEKILL in GMP facility. The flow chart of the process with timing, intermediate and final quality controls are shown.
Quality assessment
The entire process was checked by environmental monitoring of viable/non-viable contamination, air pressure differential and temperature in class A, B, and C areas. Class D was monitored only for viable contamination by microbiological plates 48 . Operators and instruments were microbiologically monitored in class A and B.
Starting material and MNC were counted, as described in European Pharmacopeia (EP) 2.7.29, and evaluated for sterility (EP2.6.27), viability (EP2.7.29), and phenotype, by flow cytometry, following validated methods 47 . At every medium change, cells were counted and sterility assessed.
Before αβ T-cell depletion, sterility was assessed and cell count and phenotypic analysis performed. Sterility in the starting material and the in-process samples was analyzed using the BactAlert system (Biomerieux) 49 . Cryogenic preservation of the final product was performed before quality assessment of cell viability, identity (phenotype), sterility, endotoxin analysis, and presence of mycoplasma. The Lymulus Amebocyte Lysate (LAL) test was performed using the Kinetic QCL Kit, Biotek EL808 reader, and WinKQCL software (all by Lonza), prior to internal validation.
The entire process was validated in asepsis, by performing three media-fill runs simulating all the operations carried out by operators, although with a reduced incubation time (3 days instead of 15 days).
Asset qualification
Instruments were qualified by Installation Qualification, Operation Qualification, and Performance Qualification. Micropipettes were calibrated yearly. The MACSQuant Analyzer10 (Miltenyi) flow cytometer was calibrated and compensated, using compensation kit and calibration beads (Miltenyi).
Identity of GADEKILL
Immunophenotype was assessed on at least 50,000 cells by flow cytometry using fluorochrome-conjugated monoclonal antibodies (mAb): FITC-CD3, PerCP-CD14, Viogreen-CD19, PEVio770 anti-TCRαβ, APC anti-TCRγδ, Viogreen-TCRVδ1, Vioblue-TCRVδ2, and APC-CD56 (all from Miltenyi). T lymphocytes (CD3+), NK (CD3−CD56+), B cells (CD19+), and monocytes (CD14+) were analyzed and percentage calculated in gated lympho-mononuclear cells. γδ and αβ T cells were identified in gated CD3+ T lymphocytes, whereas the Vδ1 and Vδ2 subsets in gated γδ T lymphocytes. Validation of the method was reported 47 . Viable cells were identified as 7-AAD-negative cells. Analyses were performed using Kaluza Software (Beckman Coulter).
Cryogenic preservation, final formulation, and plasma recalcification
In accordance with validated procedures for clinical use, plasma was cryopreserved using the rapid “Shock freezing” freezer KPFF24B (KW), that guarantees the achievement of −30°C in less than 1 h. Plasma bags were transferred into blood bank/freezer at −30°C ± 2°C. AB plasma was supplemented with 0.75% of 100 mg/ml clinical grade calcium gluconate (S.A.L.F) and placed in thermostat bath at 37°C ± 2°C for 2 h. A clot formed inside the bag and recalcified plasma (from now called AB serum) transferred into a new bag then incubated at 56°C ± 2°C for 30 min. Aliquots were divided and frozen.
Cell batches were cryopreserved in physiological water containing 9% human albumin and 6% DMSO (Wak-Chemie Medical) in 50 ml bags (concentration 108/ml) placed in pre-cooled to 0°C Planner Kryo 560-16, then in vapor phase of liquid nitrogen and finally into liquid nitrogen Cryo-Bank dewars.
The bags were thawed by immersion in a 37°C ± 2°C thermostat bath and the cell product was washed before additional studies.
The stability of GADEKILL
The stability of the GADEKILL was studied in terms of maintenance of cell viability and percentage of γδ T and NK cells after cryopreservation, by flow cytometry. Batch #1 was thawed 192 days after freezing, batch #2 after 165, batch #3 after 164, and batch #4 after 118 days. Release criteria required cell viability superior to 70% and deviations between % of γδ T and NK cells, before and after thaw, below 10%, as validated by internal procedures.
γδ T cell differentiation
The differentiation status of γδ T cells was tested after cryopreservation, by flow cytometry. Double staining with PEVio770-CD45RA (BD) and APC-CD27 (EBioscience) identified naȉve (CD45RA+CD27+), central memory (CD45RA−CD27+), effector memory (CD45RA−CD27−), and terminally differentiated (CD45RA+CD27−).
Expression of activating/inhibitory receptors and immune checkpoint molecules
Flow cytometric analyses were performed on the GADEKILL in gated CD3+ (γδ T) and CD3− (NK) cells using Vioblue-CD3, APCVio770-CD16, PE anti-NKG2A, PEVio770 anti-NKG2D, PEVio770 anti-NKp30, PEVio770 anti-NKp44, PEVio770 anti-NKp46, PEVio770 anti-PD1, and PE anti-TIM (all from Miltenyi).
Potency
Cytotoxicity was assessed by degranulation assay, as previously validated 47 . Briefly, effector cells (i.e. GADEKILL) were cocultured in the presence of PE-CD107a mAb (Miltenyi) for 3 h at 37°C and 5% CO2 to equal amount of target cells. Targets were human myeloid leukemia cell line K562 (Cell bank at Ospedale-Policlinico San Martino, Genova, Italy) and two human neuroblastoma cell lines (SK-N-AS and SK-N-SH) provided by ECACC (UK Health Security Agency). Cytotoxic γδ T and NK cells were identified as CD45+CD3+TCRγδ+CD107a+ and CD45+CD3−CD56+CD107a+, respectively. Cytotoxicity of Vδ2+ and Vδ2− lymphocytes was identified in gated CD45+CD3+TCRγδ+ cells. At least 50,000 events were acquired.
Proliferation assay
Cell proliferation was assessed using CellTrace™ Carboxyfluorescein diacetate N-succinimidyl ester (CFSE) Cell Proliferation Kit (Thermo-Fisher), according to manufacturer’s instructions. Briefly, once thawed, cells were resuspended in TexMACS without serum and stained with 2 μM CFSE for 15 min at 37°C. After 6-day culture in TexMACS with 5% AB serum and IL-2 (1000 UI/ml), cells were washed, stained with PE-CD3 mAb, and analyzed for CFSE dilution.
Comparison between γδ T lymphocytes and NK cells in the GADEKILL and in the starting material
Additional cryopreserved samples were studied to compare γδ T and NK cells in our cell product and in the starting material in terms of activation phenotype, differentiation status, cytotoxicity, and proliferation ability. Three paired samples (i.e. starting material and the derived GADEKILL) of one buffy coat and 2 leukapheresis were used. The process to obtain the final product was performed in laboratory and not in GMP facility, thus the αβ T cell depletion was achieved using immune-magnetic beads (Miltenyi). All the other reagents and materials were the same of those used in the GMP procedure. Phenotypic and functional analyses in the starting materials were performed after αβ T cell depletion. Cell proliferation was assessed by staining with PE-CD3, ECD-CD56, and APC-TCR γδ mAbs.
Statistical analysis
Statistical analysis was performed using Prism software, version 5.03 (GraphPad Software Inc.). Mann–Whitney test was used to compare data sets and significant differences were considered when P value was ≤ 0.05. Results are presented as mean ± standard deviation.
Results
Production of GADEKILL under GMP conditions
The monitoring of the clean room resulted compliant for air particles, pressure differential, and temperature in all classes. Few values measured over the limits by operators (14/1220, 1.1%) in class A and B and the environment (B and C), but such uncompliant values did not affect the sterility of the cell products. The batches were free of endotoxins (<0.5 EU/ml) and mycoplasma.
The starting materials comprise a variable number of cells (mean = 4.89 × 109; Figure 2A), with a mean cell loss of 56.7% during MNC separation. At the beginning of γδ T-cell expansion, we detected 0.37 × 109 cells in the BC and a mean of 2.57 × 109 cells in the LAs (Figure 2A shows data in individual samples). Flow cytometric analyses on MNC revealed the presence of viable cells above 99.5%. The percentages of monocytes, B lymphocytes, NK cells, γδ T cells, and Vδ1/Vδ2 subsets are shown in Figure 2A. Before αβ T-cell depletion, γδ T cells ranged from 67 × 106 to 673 × 106, and NK cells from 32.1 × 106 to 1530 × 106, in BC and LA respectively (Figure 2A). The fold expansion of γδ T cells (normalized on the total cell number before and after expansion) was 20.3 in BC, 3.4, 8.8, and 55 in LAs, respectively. A representative flow cytometric analysis of the process is reported in Figure 2B–D.

Flow cytometric analysis of cell populations during the process. Panel A. Number of cells and proportion of populations obtained at the different steps of the process starting from buffy coat (BC) or leukapheresis (LA). Panels B–D. One representative experiment using LA (#3) as staring material is shown. Panel B. CD14+ monocytes (a), CD19+ B cells (b) CD3+ T cells (c), CD3−CD56+ NK cells (d), γδ and αβ T lymphocytes (e) as well as Vδ1 and Vδ2 subsets (f), present after Ficoll-Paque gradient in gated lympho-mononuclear cells. Panels C and D. CD14+ monocytes (a), CD19+ B cells (b) CD3−CD56+ NK cells (c), γδ and αβ T lymphocytes (d) as well as Vδ1 and Vδ2 subsets (e), gated mononuclear cells at the end of expansion (day 14) (Panel C) and harvested after αβ T-cell depletion (Panel D). Panel E shows γδ T cell differentiation, by APC-CD27 and PEVio770-CD45RA staining.
Identity and purity
The GADEKILL had consistent cell viability (98.6% ± 1.6%), was predominantly composed of NK cells, with variable percentages of γδ T cells, which are mainly Vδ2 (Figure 2A). Differences in NK proportion were observed between BC (20.4%) and LAs (mean 68.8% ± 7.9%), suggesting that the starting material may affect γδ expansion and presence of NK. All batches satisfied viability criteria and were composed of differentiated γδ T lymphocytes, as witnessed by predominant CD45RA−/CD27− γδ T effector memory cells (>68.9%, one representative experiment is reported in Figure 2E). The majority of γδ T cells were effector memory (78.6% ± 7.1%), with subsets of naïve (1.4% ± 0.8%), central memory (1.7% ± 1.4%), and terminally differentiated cells (18.2% ± 10.2%). The differentiation status of γδ T cells in all batches produced is reported in Supplemental Figure 1.
In lymphoproliferative disorders and GvHD, the presence of B and αβ T lymphocytes in the final product may gamble safety. Due to the consistent presence of monocytes before γδ expansion, we analyzed the resting in the final product. As reported in Figure 2A, monocytes were virtually absent, whereas B lymphocytes and αβ T cells were present at very low percentages (Figure 2A). Figure 2B shows phenotypic analyses on MNC before γδ T-cell expansion, with monocytes (a), B cells (b), T lymphocytes (c), NK cells (d), γδ/αβ T lymphocytes (e), and Vδ1/Vδ2 subsets (f). Figure 2C reports analyses of the same populations at the end of γδ T-cell expansion. Similar phenotypic analysis on GADEKILL, revealing a successful eradication of αβ cells and enrichment of γδ and Vδ2 cells, is reported in Figure 2D.
Stability
Comparison of the final product before and after cryogenic preservation is illustrated in Figure 3. Both cell viability and NK/γδ T lymphocytes have been efficiently preserved. In all batches (panel A), the cell viability was consistently higher than 80%, and the differences between the percentage of γδ T lymphocytes (both Vδ1 and Vδ2) or NK cells before and after freezing were not significantly different. One representative batch before (panel B) and after thawing (panel C) is shown. These results support the stability of the GADEKILL product, with at least a 4-month shelf life.
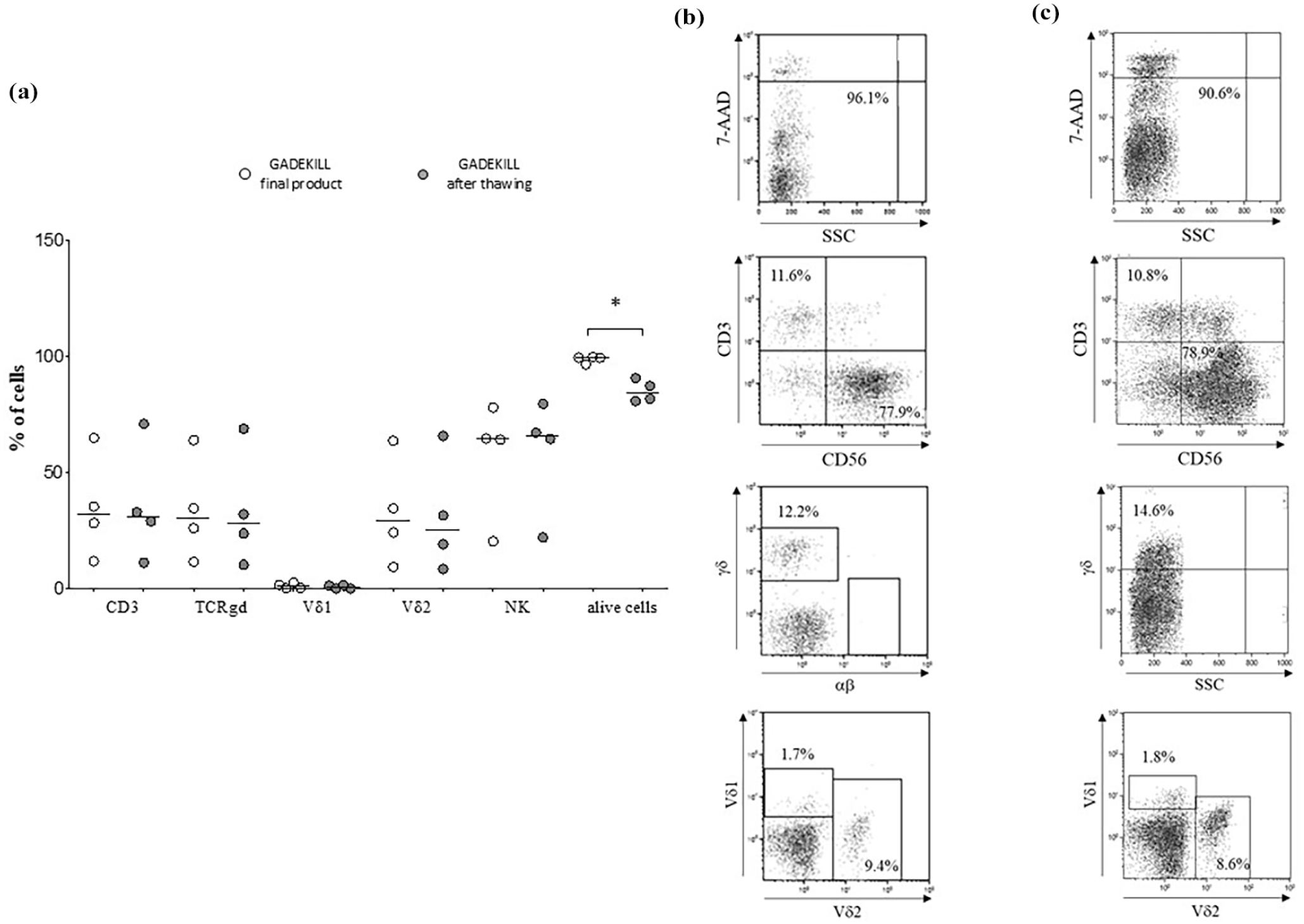
Stability of the GADEKILL. Panel a. Results from flow cytometric analyses on the four batches produced at the end of the process (white circles) and 165 days after cryopreservation (gray circles). Analyses were performed in gated lymphomononuclear cells. Percentages of T cells, NK, γδ T cells, Vδ1 and Vδ2 subsets and live cells (7AAD negative) are reported. Horizontal bars indicated medians. Asterisks indicated statistically significant differences. Panel b (end of the process) and c (after thawing) show one representative experiment performed on the GADEKILL obtained from LA#2.
Expression of activating/inhibitory receptors and immune checkpoint molecules
To investigate whether the GADEKILL expresses receptors potentially involved in activation and cytotoxicity, we analyzed NKG2A, NKG2D, NKp30, NKp44, and NKp46 on γδ T lymphocytes and NK cells after cryopreservation. In addition, we evaluated the CD16 expression, due to its involvement in ADCC. As shown in Figure 4a, both γδ T lymphocytes (CD3+) and NK cells (CD3−) consistently expressed, although at different levels, CD16, NKG2A, NKG2D, NKp30, and NKp44. Conversely, NKp46 was expressed only by NK cells. The expression of these markers in each individual GADEKILL batch is reported in Figure 4b.

Expression of CD16, natural cytotoxic receptors and exhaustion markers. Panel a. Expression of CD16, NKG2A, NKG2D, NKp30, NKp44 and NKp46 was tested by flow cytometry and analyzed in gated lympho-mononuclear cells. Experiments performed using batch from LA#4 is reported. NK and γδ T cells were discriminated by the different expression of CD3 (negative in NK and positive in γδ T lymphocytes). Results from all batches produced are reported in Panel b. Expression of PD-1 and TIM-3 in γδ (gated in CD3+ cells) and NK cells (gated in CD3−) present in batch #4 is shown in Panel c. Percentages of the two exhaustion markers expression in all batches produced are detailed in Panel d.
Finally, the expression of two immune checkpoint molecules (i.e. PD-1 and TIM-3) involved in cell exhaustion was studied in the four batches. Flow cytometric analysis revealed that both PD-1 and TIM-3 were expressed in both NK cells and γδ T cells although at different levels (Figure 4d). One representative experiment is reported in Figure 4c. The impact of PD-1/TIM-3 expression on the exhaustion of NK and γδ T cells was addressed by cytotoxic and proliferation assays.
Potency and cell proliferation
The potency of the GADEKILL was evaluated in terms of cytotoxic ability of γδ T and NK cells against the leukemia cell line K562 and two different NB cell lines (SK-N-AS and SK-N-SH). Surface CD107a was expressed on γδ T (CD45+CD3+TCRγδ+) and NK cells (CD45+CD3−CD56+) in the absence of target tumor cells (37.2% ± 1.7% and 21.7% ± 5.3%, respectively) and significantly increased in the presence of K562 (γδ T cells: P = 0.05, NK: P = 0.05) and SK-N-AS (γδ T cells: P = 0.05, NK: P = 0.05), but not of SK-N-SH cells. Indeed, cytotoxicity against K562, SK-N-AS, and also SK-N-SH cells was ascribed exclusively to the Vδ2+ cells. In details, CD107a surface expression on Vδ2+ cells (38% ± 3.1%) was significantly increased in the presence of K562 (55.6% ± 4.7%, P = 0.05), SK-N-AS (46.7% ± 3.4%, P = 0.05), and SK-N-SH (45.6% ± 4%, P = 0.05) cells. Surface expression of CD107a on the Vδ2− population did not significantly increase upon coculture with any tumor cell lines. Results from each individual batch are reported in Figure 5a, c, and e. One representative experiment using batch from LA#2 in the absence (panel b) or in the presence of K562 (panel d) and SK-N-AS cells (panel f) is shown in Figure 5, whereas data from LA3 and 4 are reported in Supplemental Figure 2.

Cytotoxic activity of the GADEKILL against target tumor cells. CD107a surface expression on γδ T lymphocytes (Panel a), Vδ2 cells (Panel b), and NK cells (Panel c) in the absence (ctr) or presence of target cells (K562, SK-N-AS and SK-N-SH) have been analyzed on the four batches. γδ T cells and Vδ2 cytotoxic subsets were analyzed in gated CD45+CD3+ cells, by staining with APC-TCRγδ, Vioblue anti-Vδ2. NK were analyzed on gated CD45+CD3− cells, by staining with APC-CD56 mAb. CD107a degranulation assay was performed using a 1:1 E:T ratio. Horizontal bars indicated medians. Asterisks indicated statistically significant differences. One representative experiment from batch #2 is shown in panels d, e and f. Degranulation of effector cells in the absence of the targets (Panel d) and against the myeloid leukemia K562 cells (Panel e) and the neuroblastoma SK-N-AS cell line (Panel f) is reported.
Finally, we asked whether the cryopreserved batches proliferated, once cultured in the presence of IL-2 for 6 days. The proliferation assay revealed that both NK cells and γδ T lymphocytes were able to perform 2 and 4-cell divisions, respectively (not shown).
Differences between γδ T lymphocytes and NK cells in the GADEKILL and in the starting material
Finally, we analyzed whether GADEKILL formulation, in regard to NK and γδ T cells, was superior to unmanipulated counterparts in the starting material in terms of activation, cytotoxicity, and proliferative ability. As reported in Figure 6a left panel, γδ T cells in the GADEKILL showed significant (P = 0.05) up-regulation of NKG2A (50.3% ± 4.8% and 16.9% ± 7.3%, respectively), CD16 (46.8% ± 4.3% vs 4.3% ± 6.8%), NKp30 (10.4% ± 2.7% vs 2.6% ± 0.6%), NKp44 (16.2% ± 4.9% vs 7.2% ± 1.1%), NKp46 (0.99% ± 0.18% vs 0.43% ± 0.1%), and TIM-3 (32.5% ± 6.3% vs 4.1% ± 1.2%). Similarly, NK cells (Figure 6a, right panel) in the cell product revealed significant higher expression (P = 0.05) of NKG2D (78.7% ± 2% vs 9.3% ± 13.1%), NKp44 (42.3% ± 8.3% vs 1.5% ± 1%), PD-1 (42.1% ± 5.8% vs 2.1% ± 1.5%), and TIM-3 (58.2% ± 4.8% vs 8.8% ± 5.2%).

Comparison between γδ T lymphocytes and NK cells in the GADEKILL and in the starting material. Panel a. The expression of activating/inhibitory receptors and immune checkpoint molecules has been analyzed in three additional samples. γδ T cells (left panel) and NK cells (right panel) were analyzed from starting materials (white circles) and corresponding cell products (gray circles). Panel b shows the percentages of terminally differentiated (TD), naïve, central memory (CM) and effector memory (EM) γδ T cells in starting materials (white circles) and corresponding cell products (gray circles).
Analysis of the differentiation status (Figure 6b) highlighted that the starting material was mainly composed of TD γδ T cells (58.8% ± 5.8%) followed by EM (26.1% ± 5%), naïve (9.1% ± 3.8%), and CM (7.9% ± 4.7%). Our process significantly (P = 0.05) induced a CM phenotype (83% ± 16.1%) paralleled by a virtual absence of TD and naïve cells (<1% each).
The higher cytotoxic abilities in γδ T and NK cells in the cell product was witnessed by the following results (Figure 7a and b). First, degranulation of γδ T cells in the GADEKILL (21% ± 2.6%, gray circles) significantly increased when co-cultured with K562 (38.3% ± 1.7%) or SK-N-AS (38% ± 2.5%) cells, a feature not observed in the same unmanipulated cells (white circles, left panel). Second, NK cells in both GADEKILL (gray circles) and starting material (white circles) up-regulated CD107a expression in response to K562 cells (24.3% ± 3.4% vs 45% ± 3.7% in the product and 12.3% ± 3.8% vs 37.2% ± 1.8% in the starting material, right panel). In contrast, NK cells in GADEKILL, but not in starting material, up-regulated CD107a expression in the presence of SK-N-AS (44.7% ± 5.7%). The cell lysis induced by the GADEKILL on target cells was documented by the 7-AAD+ staining on K562 and SK-N-AS cells (Figure 7b). Of note, the cell product (gray circles) was more effective to induce lysis on the NB cell line compared to the unmanipulated counterpart (white circle) whereas was comparable against K562 cells.

Cytotoxicity of γδ T lymphocytes and NK cells in the GADEKILL and in the starting material. Cytotoxic activity of γδ T cells (left panel) and NK cells (right panel) was evaluated as CD107a+ cells on effector cells (Panel a) in the absence (ctr) or presence of target cells (K562, SK-N-AS and SK-N-SH) in starting materials (white circles) and corresponding cell products (gray circles). Lysis of target cells was evaluated as 7AAD+ cells (Panel b). Proliferation of γδ T cells (left panel) and NK cells (right panel) was analyzed as number of cell division assessed by CFSE dilution in starting materials (white circles) and derived cell products (gray circles) (Panel c). Horizontal bars indicated medians. Asterisks indicated statistically significant differences.
In terms of proliferative ability, as shown in Figure 7c, γδ T cells in GADEKILL displayed a significantly (P = 0.05) lower percentage of 0 (5.9% ± 2.1% vs 23.1% ± 3.6%) or 1 (46.8% ± 0.9% vs 71.2% ± 4.8%) cell divisions and a higher percentage of cells with 2 or more divisions (48.3% ± 2.7% vs 6.9% ± 2.1%). In contrast, proliferation of NK cells is similar in GADEKILL and starting material.
Discussion
During the past years, several studies highlighted the importance of human γδ T and NK cells in antitumor surveillance and antiviral immunity pinpointing their potential use as adoptive cell therapy, particularly in the context of cancer3,50,51. Differently from conventional αβ T lymphocytes, these immune cells exert effector functions by (1) recognition of target cells in HLA-independent manner avoiding GvHD; (2) cytotoxic activities mediated by binding of receptors and ligands; (3) anti-viral responses when conventional adaptive immunity is insufficient; and (4) resilient activity of immune checkpoint molecules, such as PD-12, 42 . In the context of HSCT, high amount of γδ T and NK cells is typically associated with favorable clinical outcomes 46 .
In such scenario, we developed a novel ATMP composed of expanded γδ T lymphocytes and NK cells, with the primary intent of providing an immunotherapeutic tool to counteract tumor relapse after HSCT, taking advantage of the complementary effects played by these two populations. We revised current technology40,41 and, moving a method previously validated in our lab 47 , optimized such GMP production. Notably, the final formulation in our ATMP presented some interesting differences in terms of cell subsets compared to previous reports 41 . The percentage of NK cells was unambiguously superior, especially when leukapheresis was used as starting material.
Several novel ATMP technologies are commonly designed and optimized, but very few innovative products have been afterward validated in full GMP conditions, facing risks with large-scale manufacturing and cell expansion. The optimization and production of an advanced medicinal product, like the one here described, has been generated according to the most stringent requirements, supporting a rapid and safe translation to the clinic in the shortest time. Quality controls were implemented and applied to the entire process production and media-fill steps. The absence of endotoxin, mycoplasma, aerobic, and anaerobic microorganisms certified sterility of the whole process and release for clinical use.
Another critical pillar in cell production for clinical use is the final cell viability and the absence of both αβ T and B lymphocytes. We analyzed and confirmed integrity maintenance and cell viability in the final product after cryogenic preservation, highlighting the feasibility of an off-the-shelf therapy, a feature of particular relevance in clinical perspectives.
Characterization of GADEKILL revealed the unique characteristics of the composition of matter, where both effector/cytotoxic NK and γδ T cells were present, enhancing quality and specific biological effects. The ATMP generated starting from LA resulted in a composition of NK cells consistently higher than 60%, a feature never documented in any other similar technology, supporting the characteristic of unicity in our formulation (GADEKILL), compared to other published data41,43.
The novelty of our method includes the (1) use of decalcified human AB serum, (2) the use of clinical reagents, (3) the time of αβ T-cell depletion and cell expansion, (4) the use of biobags as cell culture system, and finally (5) the cell product obtained, which is composed of both NK and γδ T cells, although with high predominance of NK cells especially starting from leukapheresis (>64%). Although consistently higher percentages of NK cells were found in the starting material when LA was used, it is difficult to gain more insight into the mechanisms underlain, due to the small number of processes performed using the BC.
As expected, the γδ T cells were predominantly Vδ2 (>76%), but the marginal presence of Vδ1, similar to physiological conditions, may be clinically relevant. It has been largely documented that, in recipients of allogenic HSCT or healthy individuals, the Vδ1 subset may be expanded in vivo upon CMV or viral reactivation36,52. Furthermore, Vδ1 cells have been reported cross-reacting with hematopoietic tumor cells, explaining the protective effect of CMV reactivation in eventual leukemia relapse 53 .
Another relevant observation is that γδ T lymphocytes present in GADEKILL express activating receptors, such as NKG2A, NKG2D and, to a lesser extent, NKp30 and NKp44. Of note, the expression of NKG2A classifies a subset of Vδ2 T cells licensed with an intrinsic hyper-responsiveness against cancer, capable of counterbalancing the inhibitory signals delivered by HLA-E expressed on malignant cells 23 . NK cells also consistently expressed NCR, except for NKp46, revealing a cytotoxic potential. Although both NK and γδ T cells expressed the immune checkpoint molecules TIM-3 and PD-1, this was not associated with an exhausted functionality, as supported by their documented cytotoxicity against tumor cell lines and the ability to proliferate. This finding may be conceivably related to the concept that the effector and cytotoxic functions of immune cells result from a balance of complex interactions between different receptors and ligands present on the effector and target cells 5 . Importantly, NK and γδ T cells (both Vδ2+ and Vδ2− subsets) exerted cytotoxic activities against the myeloid leukemia K562 and NB cell lines. The choice of these cell lines is based on the consideration that, in our Institute, the GADEKILL will be firstly used in pediatric patients receiving a haploidentical HSCT, fully characterized for NK alloreactivity, and such transplant procedure is currently applied to children affected by neuroblastoma and leukemias. However, it is to keep in mind that this cell product may be used also as universal cell therapy.
Regarding the cytotoxic ability, we selected the degranulation assay since the cryopreserved GADEKILL will be infused into the patients immediately after thawing, thus a rapid and reliable test in support of biological activity in the final product is needed. However, specific lysis of target cells, especially of neuroblastoma type, has been reported at 1:1 effector:target ratio after 3 h culture. A result that deserves attention is related to the consistent expression of CD16, with high levels especially on NK cells, in our cell product. Accordingly, other previous results 43 reported that NK cells, present after stimulation of blood MNC with ZOL and IL-2 for 12 days, were as CD3−CD56brightCD16+ different from the NK populations (CD56dimCD16+ and CD56brightCD16−/dim) typically identified within the peripheral blood. Jonus and coworkers previously reported, the anti-tumor activity of a proprietary cell formulation in pre-clinical NB studies, highlighting an enhanced tumor killing effect offered by either NK- or γδ-dominant cell expansion in combination with dinutuximab 43 . Such an ADCC effect was mediated by CD16 on the immune cells. In such a preliminary study, the authors concluded the key activity mainly mediated by NK and proposed that high expression of CD16 on immune cells during the expansion phase may be used as biomarker of an advanced, ex vivo expanded, γδ T cell therapy.
An issue that deserves improvement is the cell recovery observed, but it is to mention that we here reported a validation of a GMP procedure starting from a quite low number of MNC for ethical reasons. For clinical purposes, we plan to start from higher volume of leukapheresis and cells (e.g. 40–60 × 109) in order to obtain a better cell recovery after the CliniMACS Prodigy separation.
Conclusion
We implemented a novel GMP-manufactured medicinal product, named GADEKILL, containing both NK and γδ T cells that are pure, activated, and cytotoxic against leukemia and neuroblastoma. The NK/γδ T-cell formulation is virtually free of αβ T lymphocytes and B cells, supporting use in clinical settings, especially in the context of haploidentical HSCT. GADEKILL can be efficiently and routinely manufactured in accordance with GMP requirements, supporting a translation into clinical practice in the shortest time. Indeed, we envisage infusion of the GADEKILL in the post-transplant phase as immunological support to NB and leukemic pediatric patients, counteracting tumor relapse and life-threatening infections.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251374248 – Supplemental material for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897251374248 for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation by Fabio Morandi, Martina Della Lastra, Gino Tripodi, Federica Sabatini, Claudia Cocco, Marco Risso, Roberto Gramignoli, Federico Zara and Irma Airoldi in Cell Transplantation
Supplemental Material
sj-jpg-2-cll-10.1177_09636897251374248 – Supplemental material for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation
Supplemental material, sj-jpg-2-cll-10.1177_09636897251374248 for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation by Fabio Morandi, Martina Della Lastra, Gino Tripodi, Federica Sabatini, Claudia Cocco, Marco Risso, Roberto Gramignoli, Federico Zara and Irma Airoldi in Cell Transplantation
Supplemental Material
sj-jpg-3-cll-10.1177_09636897251374248 – Supplemental material for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation
Supplemental material, sj-jpg-3-cll-10.1177_09636897251374248 for A novel GMP-manufactured medicinal product candidate composed of NK and γδ T cells as adjunct immunotherapy for hematopoietic stem cell transplantation by Fabio Morandi, Martina Della Lastra, Gino Tripodi, Federica Sabatini, Claudia Cocco, Marco Risso, Roberto Gramignoli, Federico Zara and Irma Airoldi in Cell Transplantation
Footnotes
Acknowledgements
Ethical Considerations
Leukaphereses and buffy coats from healthy donors were obtained after written informed consent, according to the Declaration of Helsinki. The study has been approved by Regional Ethic Committee (CER Liguria: 593/2021—DB id 11884) on 9 November 2021.
Consent to Participate
Written informed consent has been obtained from donors.
Consent for Publication
Not applicable.
Author Contributions
F.M. designed the study, performed experiments, analyzed and interpreted data, wrote and revised the manuscript; M.D.L. performed the experiments, contributed to writing the manuscript and revised the manuscript; G.T. provided AB serum and contributed to writing the manuscript; F.S. and C.C. produced the ATMP in GMP facility; M.R. provided leukapheresis, F.Z. revised the first version of the manuscript; R.G. revised the first version of the manuscript and contributed to the second revision; I.A. designed the study, performed the experiments, analyzed and interpreted data, and wrote and revised the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Italian Ministry of Health Ricerca Corrente and 5x1000 project year 2017 code 5M-2017-23684161.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Fabio Morandi and Irma Airoldi are the inventors of and have filed a patent request “Prodotto per la terapia cellulare avanzata” number 102024000014212 related to the protocol presented in this manuscript. Roberto Gramignoli is a cofounder of Tandem Biotherapeutics, Inc., with no competing financial interests in the technology here presented. The remaining authors declare no competing financial interests.
Data Availability
Data are available upon request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
