Abstract
Despite extensive research and increased public awareness regarding the adverse health effects of tobacco, the prevalence of cigarette smoking continues to rise, resulting in millions of annual deaths attributed to its use. Consistently, cigarette smoke contains harmful toxins that adversely affect all organs, tissues, and cells. As regenerative medicine is developing with the successful application of mesenchymal stem cells (MSCs) in numerous diseases, it is mandatory to elaborate on the factors that may impair or change the regenerative potential of MSCs. It is well established that cigarette smoke negatively affects the function of MSCs; this impairment has critical implications for regenerative medicine, which emphasizes the need to consider patient smoking history prior to application of MSCs. The review elaborates on primary toxic compounds of cigarette smoke as well as its impact on MSC functions such as proliferation and viability, migratory potential, differentiation, and finally immunomodulation, suggesting avenues for further research.

Keywords
Introduction
In the 1970s, Friedenstein et al. 1 identified mesenchymal stem cells (MSCs) as spindle-shaped, adherent, non-hematopoietic stem cells located in bone marrow. 2 Subsequent research has identified the presence of MSCs in various tissues, including adipose tissue, umbilical cord, and dental pulp3,4. Afterward, Pittenger et al. 5 found that MSCs can be induced to differentiate specifically into adipocytes, chondrocytes, or osteoblasts. In addition to their notable self-renewal abilities, MSCs demonstrate significant potential for differentiation into diverse cell lineages (mesoderm lineages and, under certain conditions, into ectoderm and endoderm tissues), demonstrating significant developmental plasticity 6 . Studies indicate that MSCs demonstrate a reduced risk of tumorigenesis, as their differentiation pathway generally avoids malignant transformation, somewhat mitigating concerns related to the safety and side effects of stem cell therapies7,8. Besides, MSCs demonstrate low immunogenicity attributed to the absence of MHC class I molecules and the costimulatory molecule CD40, as well as limited expression of MHC class II molecules, thereby decreasing the likelihood of rejection post-transplantation9,10. Of note, a recent research has shown that MSCs possess considerable immunomodulatory capabilities11–13. This positions them as an excellent tool in regenerative medicine and highly promising candidates for the treatment of autoimmune inflammatory disorders. The immunomodulatory mechanisms of MSCs are primarily mediated through interactions with immune cells, involving both direct cell contact and paracrine signaling 14 . Consequently, MSCs demonstrate significant potential for cell therapy in the treatment of diverse diseases in both human and animal models, contributing importantly to immunoregulation, anti-inflammatory responses, and tissue regeneration15,16. In particular, concerning the role of MSCs in tissue homeostasis, it has been proposed that indirect effects, particularly those involving paracrine signaling that regulates neovascularization, proliferation, differentiation of adjacent stem cells, and immune responses, are more significant in tissue regeneration than direct differentiation in alignment with surrounding tissues17–19. Likewise, experimental evidence indicates that paracrine effects are mediated by extracellular vesicles (EVs), which encompass a range of intracellular bioactive and signaling molecules secreted by MSCs. Therefore, the use of EVs or secretomes has surfaced as a viable alternative to mitigate the risks associated with live-cell therapies20–23. Considering all of the mentioned traits of MSCs and their excellent potential in regenerative medicine, it is of paramount importance to appraise the impact of environmental factors that may impede their regenerative capacity. Despite a decline in global smoking prevalence, tobacco use remains widespread, contributing to a substantial health burden globally, with one billion individuals smoking regularly and resulting in 7.0 million deaths in 2020 24 . Cigarette smoke contains toxic components that are absorbed into the bloodstream via the lungs and subsequently distributed to various organs, impairing their function 24 . Furthermore, cigarette smoke, which contains toxins that induce oxidative stress and inflammation, plays a significant pathogenic role in the progression of various diseases 25 . Besides, cigarette smoke contains hazards that are distributed to the bone marrow, negatively affecting hematopoiesis by impairing the proliferation and differentiation of hematopoietic stem cells (HSCs) 26 . Like HSCs, MSCs located in the lungs, liver, kidneys, gut, brain, and bone marrow are similarly impacted by cigarette smoke due to their continuous exposure to toxins present in cigarette smoke. The research consistently indicates that exposure of MSCs to cigarette smoke may impair proliferation, differentiation potential, migratory capacity, immunomodulatory properties and finally, overall therapeutic efficacy of MSCs23,27–33. These findings have serious repercussions for regenerative medicine and underscore the necessity of evaluating patient exposure history in cell-based therapies. Hence, the purpose of this review is to provide an up-to-date understanding of the impact of cigarette smoke on MSCs function.
Chemical composition of cigarette smoke
Tobacco smoke contains numerous complex combustion compounds. Research indicates the presence of 7357 chemical constituents, of which approximately 70 compounds have been validated as having carcinogenic activity34–36. It is impracticable to measure all 7357 components for product monitoring and regulatory compliance. Therefore, it is imperative to choose a list of smoke constituents that has a suitably extensive chemical, pharmacological, and toxicological profile 37 . Hoffmann et al. have released multiple lists enumerating diverse toxicologically and physiologically active components of mainstream smoke, recognized as Hoffmann analytes by both the tobacco industry and regulatory bodies38–40. The Hoffmann analytes comprise 44 hazardous smoke components, excluding tar, nicotine, and carbon monoxide, categorized into nine classes: ammonia, aromatic amines, polycyclic aromatic hydrocarbons (PAH), phenols, carbonyls, hydrocyanic acid, nitrosamines, inorganic elements, and other volatile organics38,40. Moreover, a subsequent study by Talhout et al. 37 enumerates 98 hazardous smoke components. Hence, these constituents include many chemical types such as alkaloids, aldehydes, nitrosamines, heavy metals, volatile organic compounds, and free radicals37,39,41,42. The concentration and biological activity of these compounds differ based on tobacco type, cigarette design, and smoking patterns; however, their cumulative effect consistently indicates significant cellular toxicity43,44. All of these ingredients are generated during combustion and include gas-phase and particulate-phase, interacting synergistically to elicit biological effects on all of the tissues and cells27,37,45. Consequently, the gaseous phase can permeate the pulmonary alveolar epithelium, enter the bloodstream, and potentially engage directly with MSCs present in the circulation and tissues 45 . Toxic Compounds in cigarette smoke and their effects on MSCs were summarized in Table 1.
Toxic compounds in cigarette smoke and their effects on MSCs.

nAChRs: nicotinic acetylcholine receptors; MAPK/ERK: Mitogen-Activated Protein Kinase Extracellular Signal-Regulated Kinase; PI3K/AKT: Phosphoinositide 3-kinase/Protein kinase B; JAK/STAT: Janus kinase/signal transducer and activator of transcription.
Primary toxic compounds of cigarette smoke affecting MSCs function
Nicotine
Nicotine is the primary chemical constituent of cigarette smoke, existing as acid salts that are absorbed in the lungs and regarded as a key factor in cigarette smoking addiction54,55. Moreover, prior research has demonstrated that nicotine enhances proliferation, invasion, and angiogenesis while inhibiting apoptosis in cancer cells56–58. On top of that, nicotine has been shown to adversely impact stem cells. Thus, whole cigarette smoke extract (CSE) and nicotine have been demonstrated to adversely affect the regenerative capacity of MSCs48,59–62. The mechanisms involve direct cellular contact through nicotinic acetylcholine receptors, leading to altered intracellular signaling pathways that compromise vital cellular functions63,64. Li et al.’s research underscores the adverse effects of nicotine on human umbilical cord mesenchymal stem cells (hUC-MSCs), illustrating that nicotine induces toxicity and apoptosis while simultaneously augmenting their migratory and stem-like characteristics. The alterations in hUC-MSCs, including elevated IL-6 levels and modifications in epithelial-mesenchymal transition markers, indicate the potential role of nicotine in promoting cancer progression 63 . A study by Alkam and Nabeshima 65 displayed that nicotine, along with its oncogenic derivatives NNK and NNN, activates various intracellular signaling pathways by binding to nicotinic acetylcholine receptors (nAChRs) and β-adrenergic receptors (β-ARs). The homomeric α7 and heteromeric α4β2 subunits of nAChR are regarded as the principal receptors facilitating nicotine’s effects on the cell cycle 65 . The expression of both subunits is significantly elevated in response to tobacco smoke-specific nitrosamine or nicotine 65 . MSCs express the functional α7 subunit of the nicotinic acetylcholine receptor (nAChR). Nicotine exposure significantly elevates intracellular calcium levels in MSCs, resulting in the activation of calcium-dependent intracellular pathways and modifications to the cell cycle in MSCs exposed to nicotine 66 . The interaction of nitrosamine or nicotine with nAChRs activates the MAPK/ERK, PI3K/AKT, and JAK/STAT signaling pathways, facilitating the development and proliferation of MSCs 66 . Additionally, nicotine binds to nAChRs, activating Src kinase in a β-arrestin-1-dependent manner, which subsequently inactivates the Rb protein. Also, E2F1 regulation enhances the activity of proliferative genes 67 . The nicotine/nAChR axis regulates the growth and proliferation of MSCs through its impact on the MAPK/ERK, PI3K/AKT, JAK/STAT, and Src/E2F1 signaling pathways 63 . Shen et al. 60 noted a significant enhancement in the proliferation rate of bone marrow-derived mesenchymal stem cells (BM-MSCs) upon exposure to 50 to 100 nM nicotine over duration of 7 days. Kim et al. 59 demonstrated that nicotine concentrations ranging from 1 to 100 µM did not promote the proliferation of alveolar BM-MSCs. Consequently, this study displayed that only nicotine concentrations between 1 and 2 mM could enhance proliferation, while higher concentrations exceeding 5 mM failed to induce optimal proliferation of BM-MSCs 59 . Zeng et al. 68 documented similar findings, indicating a dose-dependent decrease in proliferation and an elevation in apoptosis of human UC-derived MSCs subsequent to exposure to 0.5 to 1.5 mg/ml nicotine (3–9 mM). Nicotine’s negative effects on the growth of MSCs are due to its ability to cause oxidative stress and G0 cell cycle arrest in rapidly growing cells at high concentrations (>3 mM) 63 . A significantly increased proliferation rate was observed in nicotine-exposed MSCs cultured in growth medium supplemented with antioxidant vitamin C 60 . Ng et al. 69 conducted a later study indicating that the proliferation rate of MSCs derived from chronic smokers was 2.5 times lower than that of MSCs from non-smokers. Furthermore, a diminished proliferation ability was noted in MSCs after 3–5 passes, signifying that the detrimental effects of extended nicotine exposure were enduring and transmitted throughout successive generations. The findings indicate that a comprehensive history of nicotine use must be accurately assessed for all potential MSC donors to prevent the transplantation of MSCs with diminished proliferative capacity.
Formaldehyde and aldehydes
Among the numerous chemicals, nine toxicants have been prioritized for reduction by the WHO Study Group on TobReg37,70. Components include those in the particulate phase, such as specific nitrosamines and benzo(a)pyrene, as well as those in the volatile phase, including aldehydes (e.g. acetaldehyde, acrolein, formaldehyde), volatile organic compounds (e.g. benzene, 1,3-butadiene), and carbon monoxide37,70. The predominant chemical class of aldehydes includes acetaldehyde, acrolein, and formaldehyde. Short-chain aldehydes are formed during the pyrolysis and combustion of tobacco and are characterized as highly reactive electrophilic compounds due to their chemical structure. These aldehydes exhibit analogous mechanisms of formation, molecular structures, and chemical properties, potentially representing the wider chemical class of aldehydes found in smoke49,50,71. The risk assessment of the three individual aldehydes, utilizing computer models that consider inhalation exposure risk factors, led to the establishment of a hazard ranking. The ranking determined that acrolein, the most extensively researched aldehyde, is the most detrimental component among the three72,73. According to Talhout et al., 74 the concentration of aldehydes in cigarette smoke is influenced by various factors, including the inclusion of added sugars in tobacco, smoking techniques, and the design of the cigarette. A study by Pauwels et al. has demonstrated the influence of these features on the quantity of aldehydes present in each cigarette74–78. Research indicates a significant correlation between the sugar content in tobacco, particularly sucrose and glucose, and the production of formaldehyde74,75. Higher sugar levels in tobacco may result in increased formaldehyde release, thereby enhancing the toxicity of MSCs. Also, numerous studies displayed that prolonged exposure to the formaldehyde elevates the production of ROS42,79,80 resulting in sustained oxidative stress that may adversely affects the viability and functionality of MSCs 81 . Nonetheless, it is important to acknowledge that there is no research that directly investigates the impact of aldehydes on MSCs, so future research is mandatory to comprehensively understand this interaction.
Tobacco-specific nitrosamines
Tobacco and tobacco smoke include over 80 carcinogens. The tobacco-specific N-nitrosamines (TSNAs) 4-(N-nitrosomethylamino)-1-(3-pyridyl)-1-butanone (NNK) and N′-nitrosonornicotine (NNN) are significant carcinogens responsible for lung, oral cavity, and esophageal cancers in smokers 82 . Nitrosamines often present in tobacco products and smoke, such as N-nitrosodimethylamine (NDMA) and N-nitrosopyrrolidine (NPYR), typically exhibit low concentrations. NNN and NNK, conversely, are consistently present in significantly greater quantities. NNK and NNN are primarily synthesized by nitrosating their precursor amines, specifically tobacco pseudooxy nicotine and nornicotine51,83. Nitrosation of nicotine 1, a prevalent tobacco alkaloid, can also lead to their formation 84 . In the United States, tobacco consumption is the primary route of N-nitrosamine exposure. Individuals who smoke daily acquire significantly more than those who consume alcohol, food, or water 85 . The main tobacco-specific N-nitrosamines, such as NNN, NNK, NAT, and NAB, are associated with the formation of DNA adducts via their metabolism 84 , resulting in irreversible genetic changes that may impair the functionality of MSCs over time. It is crucial to acknowledge that contemporary research lacks direct examinations of the precise effects of tobacco-specific nitrosamines on MSCs. Consequently, subsequent investigations concentrating on this link may produce conclusive results.
Heavy metals
Cadmium, nickel, lead, and chromium are hazardous heavy metals present in elevated concentrations in cigarette smoke52,86,87. These metals accumulate in tobacco smoke condensate and cigarette ash with their concentration being contingent upon its origin. A research by Ajab et al. 41 indicates that imported brands contain higher levels of nickel compared to domestic brands. Wu et al. 52 demonstrate that the concentrations among various brands differ markedly, and that there exists a statistically significant difference among different types of cigarettes. Pourkkhabbaz and Pourkhabbaz 86 observed that imported products may contain greater concentrations of certain metals compared to local brands. Nonetheless, this does not apply universally to all metals or in all locations. Anyways, the presence of these metals in tobacco smoke condensate and cigarette ash is a documented phenomenon, with research indicating notable bioaccumulation in diverse biological tissues after exposure53,87. The presence of these metals in mainstream smoke guarantees direct exposure of respiratory tract tissues and subsequent systemic distribution to MSCs populations across the body. Although direct research connecting heavy metals in cigarette smoke to MSC damage is scarce52,53. Recent research by Abnosi et al. that focused on the impact of cadmium on bone marrow mesenchymal stem cells (BM-MSCs) showed that cadmium impedes the osteogenic development of BM-MSCs, influencing ALP activity and collagen synthesis, with these effects being facilitated by oxidative stress and metabolic disturbances, therefore offering a potential mechanism by which heavy metals may impede the function of MSCs42,53.
The impact of cigarette smoke on MSCs functions
Numerous studies indicate that exposure to cigarette smoke markedly disrupts the functionality of MSCs23,29,31,88,89. Principal research findings regarding the effects of cigarette smoke on MSCs were summarized in Table 2. Exposure to cigarette smoke results in diminished regenerative capacity, characterized by reduced proliferation and viability, migration, and differentiation potential of exposed MSCs as well as detrimental impact on their immunomodulatory properties as shown in Fig. 1, indicating that smoking is a significant risk factor frequently neglected in cell-based therapeutic screening protocols, therefore raising significant concerns for clinical applications.
Principal research findings regarding the effects of cigarette smoke on MSCs.
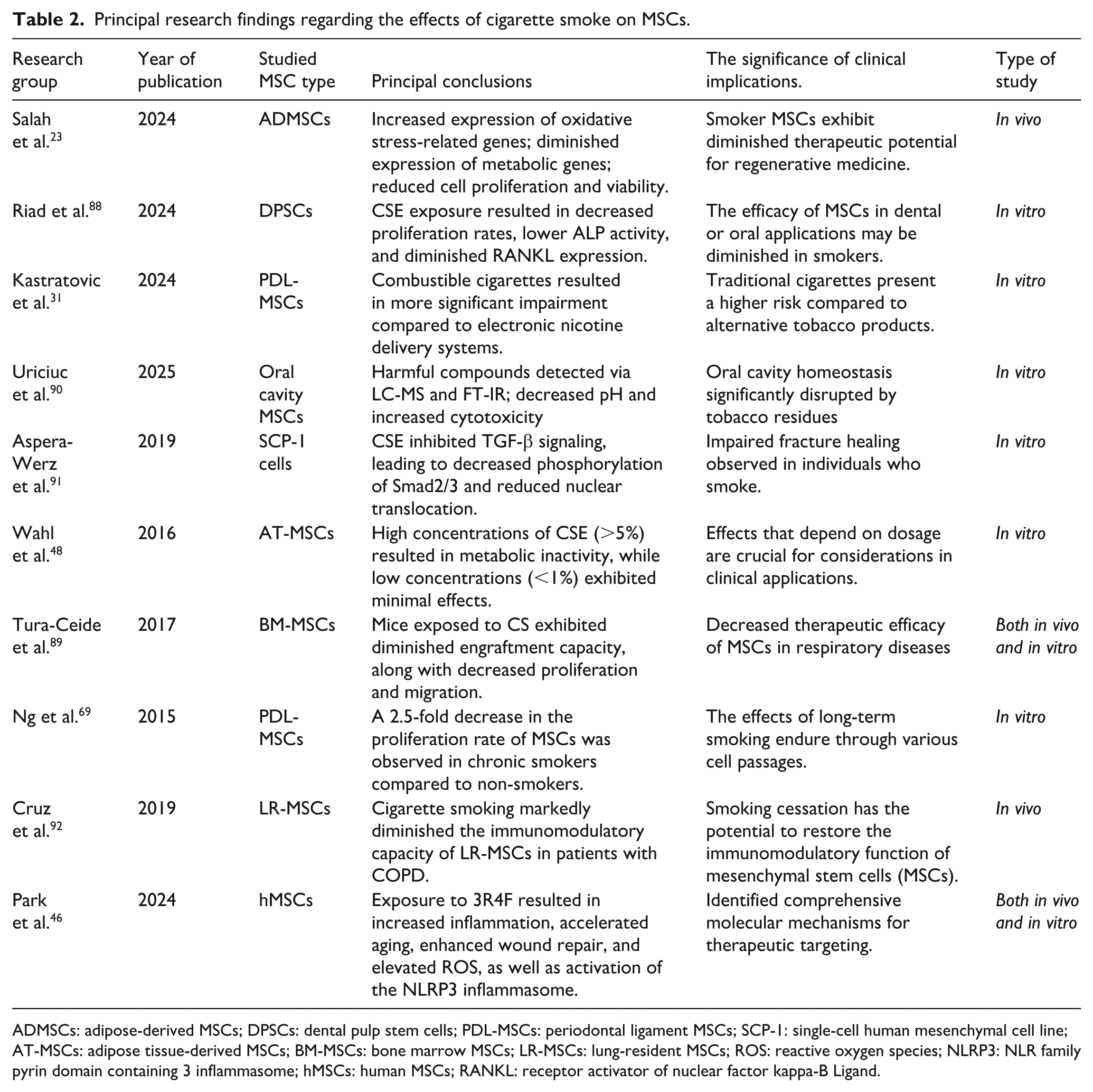
ADMSCs: adipose-derived MSCs; DPSCs: dental pulp stem cells; PDL-MSCs: periodontal ligament MSCs; SCP-1: single-cell human mesenchymal cell line; AT-MSCs: adipose tissue-derived MSCs; BM-MSCs: bone marrow MSCs; LR-MSCs: lung-resident MSCs; ROS: reactive oxygen species; NLRP3: NLR family pyrin domain containing 3 inflammasome; hMSCs: human MSCs; RANKL: receptor activator of nuclear factor kappa-B Ligand.

The impact of cigarette smoke on MSCs functions. This figure illustrates how exposure to cigarette smoke results in diminished regenerative capacity, characterized by reduced proliferation and viability, migration, and differentiation potential of exposed MSCs as well as detrimental impact on their immunomodulatory properties. MSCs: mesenchymal stem cells; TGF-β: Transforming Growth Factor beta; CTTN: cortactin; BIRC5: surviving; CXCL9: C-X-C motif chemokine ligand 9; NLRP3: NLR family pyrin domain containing 3 inflammasome; Created with CorelDraw.
The impact of cigarette smoke on MSCs’ proliferation and viability
Numerous studies are consistent among MSCs subtypes, indicating that cigarette smoke universally impairs the proliferative potential of MSCs88,89,93. These studies demonstrate that both acute and chronic exposure hinder proliferation of MSCs, with proliferation impairment being directly tied to smoke concentration88,89,93. A recent study by Salah et al. 23 demonstrates that cigarette smoke significantly impacts the proliferation, survival, and functionality of adipose-derived mesenchymal stem cells (ADMSCs) in smokers relative to non-smokers. ADMSCs obtained from smokers exhibited elevated expression of oxidative stress response genes (e.g. HMOX1, SOD2) and reduced expression of metabolic genes (e.g. PPARG, CAT), hence hindering proliferative pathways 23 . Moreover, the same study indicates a marked reduction in cell proliferation and viability among smokers, accompanied by evidence of apoptosis and DNA damage while analysis of gene expression indicates an increased expression of oxidative stress response genes and a decreased expression of genes associated with inflammation and metabolism 23 . Of note, mitochondrial membrane potential (MMP) was stable in ADMSCs from smokers; however, apoptosis assays indicated decreased viability through alternative pathways, including p53/p21-mediated cell cycle arrest as shown in Fig. 1. A recent study conducted by Riad et al. 88 on dental pulp stem cells (DPSCs) substantiates this finding, demonstrating that exposure to cigarette smoke extract (CSE) led to diminished proliferation rates in comparison to control cells. Further, a recent study by Kastratovic et al. 31 found that combustible cigarettes induced viability and proliferation impairment in PDL-MSCs, which was greater when compared to electronic nicotine delivery systems (ENDS). Thus, the latest study by Uriciuc et al. 90 demonstrates the negative effects of cigarette smoking on oral cavity homeostasis, particularly through an analysis of tobacco residues and their influence on MSCs. The detection of harmful compounds in CSEs, demonstrated through LC-MS and FT-IR analyses, is associated with a decrease in pH and an increase in cytotoxicity in MSCs, resulting in notable morphological alterations and diminished cell viability 90 . In addition, a study by Tura-Ceide et al. 89 demonstrated that exposure to CSE diminished the proliferative capacity of guinea pig BM-MSCs, exhibiting dose-dependent effects in vitro. Besides, a study by Aspera-Werz et al. 91 demonstrates the substantial influence of CSE on TGF-β signaling in MSCs, clarifying a key mechanism that contributes to the impaired fracture healing seen in smokers. CSE exposure diminished the phosphorylation of Smad2/3 and the nuclear translocation of Smad3/4, which are essential for TGF-β-mediated proliferation and differentiation 91 . This effect occurred downstream of the ALK5 receptor, indicating direct interference with cell cycle regulators 91 Molecular pathways and biomarkers affected by cigarette smoke in MSCs were summarized in Table 3, though it should be noted that that the list in is not exhaustive, while specific mechanistic details in MSCs require more targeted research. Besides, a study by Abate et al. 94 found that the structure and strength of cartilage deteriorates following prolonged exposure to cigarette smoke. In accordance with that, a study by Chen et al. 95 found that cigarette smoke reduces chondrocyte proliferation and adversely impacts cartilage metabolism through the downregulation of COL1A1 expression. COL1A1 is highly expressed in MSC-derived chondrocytes, which plays a big role in MSC-mediated repair and regeneration of damaged cartilage 96 . Wahl et al. 48 utilized a metabolic activity assay to evaluate the viability of adipose tissue-derived mesenchymal stem cells (AT-MSCs) after exposure to different concentrations of CSE. No changes in metabolism or cell viability were observed in AT-MSCs exposed to low (<1%) concentrations of CSE for 48 h 48 . Nearly all AT-MSCs subjected to 5% or 10% CSE exhibited metabolic inactivity or non-viability. In the aftermath, this research provided strong evidence that cigarette smoking, under certain conditions, adversely impacts the metabolism and viability of MSCs.
Molecular pathways and biomarkers affected by cigarette smoke in MSCs.

TGF-β: Transforming Growth Factor beta; MAPK/ERK: Mitogen-Activated Protein Kinase Extracellular Signal-Regulated Kinase; PI3K/AKT: Phosphoinositide 3-kinase / Protein kinase B; JAK/STAT: Janus kinase/signal transducer and activator of transcription; NLR family pyrin domain containing 3 inflammasome; RUNX2: Runt-related transcription factor 2; CTTN: cortactin; BIRC5: surviving; CXCL9: C-X-C motif chemokine ligand 9; Cx43: Connexin 43.
The impact of cigarette smoke on MSCs’ migration
Numerous studies have displayed that cigarette smoke has detrimental effects on the migratory potential of MSCs48,89,93, which is essential for their homing to sites of tissue injury and inflammation (Fig. 1). Thus, a study by Wahl et al. 48 found that constant exposure to high concentrations of CSE resulted in lethal effects on AdMSCs, whereas lower concentrations of CSE hindered cell migration relative to control conditions. Notably, the concentration of CSE exceeding 5% markedly reduced the expression of chemokine receptors on AT-MSCs and compromised their homing capabilities 48 . However, the migratory properties of AT-MSCs remained unchanged when exposed to low concentrations (below 1%) of CSE. Tura-Ceide et al. 89 conducted additional research that demonstrated BM-MSCs derived from CS-exposed mice had compromised engraftment capacity, along with reduced proliferation and migration rates. In vitro, BM-MSCs treated with CS extract exhibited a marked decrease in their capacity for proliferation, differentiation, and migration 89 . The research conducted by Aspera-Werz et al. 91 underscores the critical function of CSE in modulating the TGF-β signaling pathway, particularly for its influence on phospho-Smad2/3 levels and the nuclear translocation of Smad3/4. These findings elucidate the mechanisms through which TGF-β signaling dysregulation partially contributes to the adverse consequences reported in MSC migration. Recent research by Heikkinen et al. 93 shows that MSCs exposed to CSE had less effective wound healing and higher cell death rates at higher concentrations of cigarette smoke. At lesser concentrations, CSE also halted osteogenic differentiation, migration, and proliferation 93 (Fig. 1 and Table 3). Ouhtit et al.’s 98 research elucidates the significance of CD44 in preventing the migration and invasion of MSCs in cigarette users. It further demonstrates that CD44 simultaneously regulates the transcription of cortactin (CTTN) and survivin (BIRC5). This study demonstrated that cigarette smoke condensate reduces the expression of CD44 and its downstream targets, CTTN and BIRC5, in MSCs, with CTTN specifically recognized as a sign of cellular motility 98 . Hence, the disruption of these pathways directly impacts the cellular mechanisms required for appropriate chemotactic responses. These findings indicate that one must exercise caution when considering smokers’ MSCs for cell therapy, as they may be linked to the development of conditions such as osteoporosis, lung injury, and emphysema. Additionally, the identification of CD44/CTTN and CD44/BIRC5 signaling pathways provides fresh opportunities to develop therapeutic strategies targeting cancer and other diseases associated with CD44. Besides CD44, other molecules involved in the migration of MSCs must be considered. Consequently, C-X-C motif chemokine ligand 9 (CXCL9) is expressed by circulating BM-MSCs and is recognized for its significant role in their migration through the endothelium103,104. In line with that, a study by Tura-Ceide et al. 89 found that CXCL9 expression levels were diminished in the bone marrow of animals exposed to cigarette smoke when compared to the control group. Therefore, this signaling pathway plays a role in the homing of BM-MSCs and is negatively affected by cigarette smoke. Furthermore, a recent study conducted by Park et al. 46 demonstrated that direct ex vivo and systemic in vivo exposure of MSCs to 3R4F cigarettes resulted in compromised engraftment in a humanized mouse model. Moreover, transcriptomic profile analysis demonstrated a pronounced up regulation of signaling pathways related to ROS, inflammation, and aging in MSCs exposed to 3R4F cigarettes, while ingenuity pathway analysis revealed the activation of the NLRP3 inflammasome signaling pathway in 3R4F-treated MSCs 46 . Withal, pretreatment with the NLRP3 inhibitor MCC950 restored the hematopoietic stem and progenitor cells (HSPCs)-supporting capacity of 3R4F-treated MSCs 46 . These findings indicate that exposure to CSE reduces HSPCs’ supportive function of MSCs by inducing robust ROS production and subsequent NLRP3 activation, thereby impairing HSPC engraftment.
The impact of cigarette smoke on MSCs’ differentiation potential
It is widely recognized that cigarette smoke significantly impairs the capacity of MSCs to differentiate into various cell types as shown in Fig. 1; yet, acute exposure does not influence adipogenic differentiation48,64,91,93. Various research teams have effectively illustrated that the adverse impacts of cigarette smoke and nicotine on the osteogenic differentiation of MSCs are mostly responsible for the impaired osteogenesis observed in cigarette smokers61,62,100. Ng et al. 62 reported a notable reduction in alkaline phosphatase (ALP) expression, reduced calcium accumulation, and altered miRNA-1305 expression in MSCs derived from smokers’ PL-MSCs. They hypothesized that the diminished osteogenic ability of smokers’ MSCs results from the detrimental effects of cigarette smoke on the miRNA-1305/Runt-related transcription factor 2 (RUNX2) axis, given that miRNA-1305 regulates RUNX2 activity, which is crucial for osteoblast development 62 . Additionally, the latest study by Hekkinen et al. 93 assessed ALP activity and calcium deposition in the extracellular matrix to evaluate the effects of nicotine and CSE exposure on the osteogenic differentiation of MSCs (as shown in Fig. 1 and Table 3). They found that CSE diminished osteogenic differentiation of MSCs in vitro 93 . Also, the latest research by Raid et al. 88 analyzed how CSE affects the proliferation and osteogenic differentiation of dental pulp stem cell (DPSCs). It is well established that RANKL plays a vital role in bone remodeling by regulating the activities of osteoblasts and osteoclasts involved in bone production and resorption. Thus, they demonstrated that DPSCs treated with IC50 TE-p and e-Cig-LV exhibited a significant reduction in ALP activity levels and a significant reduction in RANKL expression relative to control cells 88 . The results align with the findings of Sreekumar et al., 105 which indicated that treatment of human MSCs with CSE significantly inhibited RANKL production, potentially impacting overall bone stability. CSE may also impede bone healing by inhibiting the early osteogenic differentiation of MSCs and disrupting the equilibrium between mature osteoblasts and osteoclasts, thus affecting the bone remodeling processes in fractured bones. Notably, Zhang et al. 101 assessed that the overproduction of ROS inhibits the osteogenic differentiation of BM-MSCs treated with smoke. Besides, data from various studies indicate that the reduction in osteogenic differentiation may be attributed to Cx43, the principal connexin found in MSCs, which plays a direct role in osteogenic remodeling 106 . The deficiency of Cx43 following exposure to CSE is associated with a decrease in osteogenic differentiation potential and disruption of cell communications via gap junctions, which are closely tied to the modulation of repair mechanisms. Numerous studies have demonstrated that gap junctions are critical for the prevention of oxidative stress-induced cell death 107 . As such, exposure to cigarettes may impair tissue repair 108 . Wahl et al. 48 found that cigarette smoke notably impacted the osteogenic and chondrogenic differentiation capabilities of MSCs. Appropriately, they demonstrated the negative impact of cigarette smoke on the chondrogenic differentiation of MSCs. Exposure to 0.5% CSE significantly altered the expression of cartilage-specific proteoglycan core protein (CSPCP) and the chondrogenic transcription factor SOX9 in MSCs 48 . The down-regulation of COL1A1 expression in CSE-exposed MSCs suggests that the adverse effects of cigarette smoke on the chondrogenic potential of MSCs may contribute significantly to increased cartilage loss and delayed healing in injured cartilage, commonly seen in cigarette smokers48,94. However, it should be noted that MSCs exposed to CSE exhibited no significant changes in adipogenic differentiation 48 , suggesting that the signaling pathways regulating adipogenic differentiation in MSCs remained largely unaffected by cigarette smoke. Not just osteogenic, chondrogenic, but also cardiac differentiation of MSCs can be affected by cigarette smoke. Particularly, a recent study by Gheisari et al. 64 displayed that nicotine exerts an inhibitory effect on cardiac differentiation of MSCs. Thus, the expressions of two cardiac-specific markers were examined. Accordingly, GATA4, which was expressed at the early stages of cardiac differentiation, was significantly downregulated on day 3 in MSCs 64 . This was the case for troponin as well, which is a late cardiac marker. However, increased expression of both GATA4 and troponin on day 7 may be explained by the decreasing effect of nicotine after 7 days. It seems that, similar to proliferation, there is a threshold of nicotine concentrations for affecting the differentiation potential of MSC. This factor holds significant importance for smokers eligible for stem cell therapy due to cardiovascular disease.
The impact of cigarette smoke on MSCs’ immunomodulatory potential
MSCs do not exhibit constitutive immunosuppressive properties 109 . The phenotype and function of MSCs can be influenced by endogenous factors and environmental hazards27,109. When MSCs are transplanted into inflamed tissues characterized by elevated levels of inflammatory cytokines, they acquire an immunosuppressive phenotype and secrete anti-inflammatory cytokines, leading to a reduction in ongoing inflammation. On the other hand, when MSCs are exposed to low levels of inflammatory cytokines, they adopt a pro-inflammatory phenotype, resulting in the production of significant amounts of TNF-α and IFN-γ, which exacerbates inflammation 109 . Hence, the local tissue microenvironment, particularly the concentration of inflammatory cytokines affecting MSCs exposure, influences the phenotype and immunoregulatory characteristics of MSCs. Similar to tissue microenvironments, environmental hazards, especially cigarette smoke, may influence the phenotype and function of MSCs 27 , diminishing their therapeutic effects. In line with that, using α-galactosylceramide (α-GalCer)-induced liver injury, a recognized murine model of fulminant hepatitis, we investigated the molecular mechanisms underlying the adverse effects of cigarette smoke on MSC-dependent immunomodulation 28 . MSCs that were cultured in cigarette smoke-exposed medium (MSC-WS-CM) induced a pro-inflammatory phenotype, resulting in suboptimal production of hepatoprotective and immunosuppressive cytokines (TGF-β, HGF, IL-10, NO, KYN) 28 . Additionally, it led to significantly higher secretion of inflammatory cytokines (IFN-γ, TNF-α, IL-17, IL-6) compared to MSCs cultured in standard medium without cigarette smoke exposure (MSC-CM) as shown in Fig. 2. In contrast to MSC-CM, which effectively reduced α-GalCer-induced hepatitis, MSC-WS-CM did not prevent hepatocyte injury or liver inflammation. MSC-WS-CM exhibited diminished capacity to suppress liver-infiltrated inflammatory macrophages, dendritic cells (DCs), and lymphocytes. In α-GalCer+MSC-CM-treated mice, there was a significantly lower number of IL-12-producing macrophages and dendritic cells, as well as TNF-α, IFN-γ, or IL-17-producing CD4+ and CD8+ T lymphocytes, NK, and NKT cells in the liver compared to α-GalCer+saline-treated animals 28 . However, this phenomenon was not observed in α-GalCer-injured mice that received MSC-WS-CM. The presence of inflammatory, IFN-γ and IL-17-producing NK cells and T lymphocytes was significantly reduced in the livers of α-GalCer+MSC-CM-treated mice. This reduction is attributed to MSC-derived TGF-β, which suppresses the activation of the Jak-Stat signaling pathway in activated T and NK cells, induces G1 cell cycle arrest, and inhibits the expansion of these hepatotoxic cells 99 (as shown in Table 3). We propose that the decreased production of TGF-β by MSC-WS-CM primarily contributed to the heightened presence of inflammatory NK cells and T lymphocytes in the livers of α-GalCer+MSC-WS-CM–treated animals, leading to subsequent NK and T cell-mediated hepatocyte injury. Also, MSC-WS-CM failed to promote the expansion of anti-inflammatory IL-10-producing FoxP3+CD4+ and CD8+T regulatory cells and did not establish an immunosuppressive microenvironment in the liver, unlike MSC-CM 28 . As observed in mice, MSC-WS-CM did not effectively inhibit the production of inflammatory and hepatotoxic cytokines in activated human Th1/Th17 and NKT1/NKT17 cells 28 . This supports the hypothesis that cigarette smoke significantly reduces the therapeutic potential of MSCs in cell-based immunotherapy for inflammatory liver diseases, as shown in Fig. 2. In line with that, Cruz et al. 92 identified cigarette smoking as a significant behavioral risk factor that markedly reduces the immunomodulatory and therapeutic potential of MSCs. Cruz et al. 92 analyzed the phenotype and function of lung-resident MSCs (LR-MSCs) obtained from current and former smokers with COPD, demonstrating that cigarette smoking significantly reduces the immunomodulatory capacity of LR-MSCs in these patients. Given that the transplantation of MSCs and their secretome effectively mitigates COPD in mice by inhibiting harmful T cell-mediated lung inflammation 110 , the impact of cigarette smoke on LR-MSC-mediated T cell inhibition was examined. CSE-exposed LR-MSCs demonstrated an inability to effectively suppress the proliferation of CD4+T helper cells and CD8+CTLs, as well as to inhibit the production of inflammatory cytokines in activated T lymphocytes in vitro 92 . Cruz et al. hypothesized that long-term exposure to cigarette smoke leads to increased synthesis of ROS and decreased activity of anti-oxidative enzymes in LR-MSCs. CSE-induced oxidative stress increases the expression of inflammation-related genes and induces the generation of a pro-inflammatory phenotype in MSCs 48 . Pro-inflammatory MSCs exhibit increased production of inflammatory cytokines such as TNF-α, IL-1β, and IFN-γ, alongside diminished synthesis of immunosuppressive cytokines including IL-10, IL-35, and TGF-β 109 . Additionally, they show a reduced ability to suppress activated T cells. The immunomodulatory properties of LR-MSCs in COPD patients were found to be impaired exclusively in current smokers, whereas the immunoregulatory functions of LR-MSCs in former smokers were comparable to those in healthy controls 92 . The findings indicate that the adverse effects of cigarette smoke on the immunosuppressive function of LR-MSCs are not permanent, and the immunosuppressive potential of LR-MSCs can be nearly fully restored following smoking cessation 92 . At the same time, numerous studies demonstrate that CSE leads to the overproduction of ROS, mitochondrial dysfunction, and oxidative stress in MSCs46,47,111–113. Furthermore, CC-derived tar and carbon monoxide (CO) are known to induce oxidative stress, increase the release of ROS, and activate mitogen-activated protein kinase (MAPK) signaling cascades in immune cells exposed to combustible cigarettes, leading to increased production of inflammatory cytokines 114 (Table 3). Accordingly, most recent study by Kastratovic et al. 31 clarified the impact of various tobacco products on the intricate relationships between mesenchymal stem cells derived from periodontal ligament (PDL-MSCs) and T lymphocytes, essential for sustaining oral tissue homeostasis. Co-culturing T cells with PDL-MSCs exposed to combustible cigarette smoke resulted in an immune response marked by elevated levels of inflammatory IFN-γ and IL-17-producing CD4+ and CD8+ T cells 31 , as well as elevated synthesis of TNF-α and IFN-γ in PDL-MSCs (Table 2). This response may intensify bone resorption, thereby worsening damage to periodontal tissue. Moreover, the latest research by Park et al., 46 using transcriptomic profiling, revealed that 3R4F exposure elevates inflammation, cellular aging, wound repair, and ROS in human mesenchymal stem cells (hMSCs). Hence, RNA sequencing was performed to examine the alteration of the transcriptomic profile of 3R4F-treated hMSCs 46 . Gene set enrichment analysis (GSEA) using the gene ontology biological process (GO BP) gene sets identified four significantly upregulated categories in 3R4F-treated hMSCs: inflammatory response, aging, wound healing, and response to oxidative stress gene sets 46 . The inflammatory response involved enriched gene sets with upregulated expression of NLRP3, IL-1β, and IL-6. To confirm the RNA-seq results, Park et al. assessed the expression levels of the proinflammatory genes NLRP3, IL-1β, IL-6, IL-8, and TNF-α by qRT-PCR. The 3R4F treatment enhanced the expression of proinflammatory genes in hMSCs 46 . Similarly, they identified that IL-10 and IDO, known immunosuppressive factors of MSCs, were both downregulated by 3R4F treatment 46 . Additionally, 3R4F inhibited the anti-proliferative effect of hMSCs on T lymphocytes. Collectively, they came to conclusion that 3R4F perturbs immune homeostasis by altering global gene expression patterns of hMSCs, with functional consequences demonstrated in vitro 46 . Correspondingly, it should be noted that the relationship between oxidative stress and inflammation in cigarette smoke-exposed MSCs is bidirectional, with ROS serving both as initiators and amplifiers of inflammatory responses. The oxidative environment facilitates the activation of redox-sensitive transcription factors, including NF-κB, thereby increasing proinflammatory gene expression97,115–117.

The impact of cigarette smoke on MSCs immunomodulatory and hepatoprotective potential. This figure illustrates how exposure to cigarette smoke abrogates immunomodulatory and hepatoprotective potential of MSCs. MSCs that were cultured in cigarette smoke-exposed medium acquired a pro-inflammatory phenotype, resulting in significantly higher secretion of inflammatory cytokines (IFN-γ, TNF-α, IL-17, IL-6) and suboptimal production of hepatoprotective and immunosuppressive cytokines (TGF-β, HGF, IL-10, NO, KYN). MSCs: mesenchymal stem cells; IFN-γ: interferon gamma; TNF-α: tumor necrosis factor alpha; TGF-β: transforming growth factor beta; HGF: hepatocyte growth factor; NO: nitric oxide; KYN: kynurenine; Created with CorelDraw.
Research gaps and future directions
There is a considerable gap in the field in terms of the lack of standardized protocols for CSE preparation 118 . Especially having in mind that various research groups utilize differing methodologies, concentrations, and exposure durations, which in turn makes direct comparisons difficult 119 . Some scientists have come up with the virtual tar concentration approach, which could be a solution, but not many of the researchers are adopting it yet 118 . Therefore, consensus guidelines for physiologically pertinent exposure concentrations grounded in the actual nicotine and toxicant levels found in human smokers as well as standardized biomarkers to assess MSCs dysfunction in smokers should be developed23,46,93,120–122. Moreover, the differing traits of MSCs isolated from various sources must be taken into consideration23,88,120. Notably, the vast majority of the research focuses on in vitro studies when examining the impact of cigarette smoke on MSCs. This demands incorporation of clinical studies using MSCs isolated from actual smokers versus non-smokers to validate in vitro findings 23 . Hence, it must be emphasized that in vivo studies using physiologically relevant exposure models can only offer definite conclusions regarding the impact of cigarette smoke on MSCs and mirror clinical scenarios26,46,123. Finally, alternative tobacco products must be reconsidered, keeping in mind their less detrimental effect on MSCs30,31,106, even though they are not risk-free 88 . Future research ought to examine potential interventions to alleviate the effects of cigarette smoke on MSCs, including antioxidant therapies or targeted pathway inhibitors46,124,125. Some studies suggest that certain compounds, such as vitamin C or specific inhibitors, can partially restore MSC function 126 .
The donor’s smoking history and the recipient’s smoking status must be considered when using MSCs in regenerative medicine. Both aspects significantly influence the efficacy of treatment, albeit in distinct manners, and pose peculiar obstacles to regenerative medicine. Thus, MSCs derived from smokers exhibit significantly reduced growth rates, demonstrating a significant decrease in proliferation compared to non-smoking controls 29 as well as impaired functional capabilities such as migratory, differentiation, and proliferative potential 48 . Moreover, studies comparing MSCs from diabetic smokers versus non-smokers reveal that smoking status significantly decreases glycolytic capacity and maximal mitochondrial respiration 127 . This metabolic dysfunction directly impacts the cells’ synthetic function and overall therapeutic potential. Conversely, when MSCs are administered to smokers, they must contend with a modified milieu throughout the body, which complicates their functionality and viability, regardless of the initial health of the donor cells27,29. In addition, it must be emphasized that defects originating from the donor occur singularly, whereas the recipient’s smoking habit continually exposes them to detrimental toxins26,46,48. Hence, this persistent assault implies that even healthy transplanted MSCs will be compromised by the toxins consistently present in the body, which may exacerbate the therapeutic impact over time. The tissue environment in which MSCs must engraft and function is significantly altered by the recipient’s smoking history. As it has been recently displayed by a study of Siggins et al., 26 exposure to cigarette smoke reduces the quantity of endogenous MSCs in the bone marrow niche, which makes the environment less supportive to transplanted cells. In a nutshell, only a thorough analysis of donors’ and recipients’ habits, particularly their smoking history, along with future and deeper research, can mitigate the obstacles regarding MSCs therapy.
Conclusion
As MSCs are an attractive regenerative medicine tool, and since their usage is increasing in preclinical and clinical studies, it is of paramount importance to assess all of the noxas that may impair their therapeutic efficacy. Yet, there is a significant gap in cigarette smoke exposure research, as varying methodologies, concentrations, and exposure durations complicate direct comparisons. It is essential to establish consensus guidelines for exposure concentrations relevant to physiological conditions and standardized biomarkers to evaluate MSC dysfunction in smokers. The inclusion of clinical studies utilizing MSCs from active smokers is essential for the validation of in vitro results.
Footnotes
Acknowledgements
Not applicable.
Ethical considerations and consent to participate
This manuscript is a review article and does not involve any ethical issues. All authors reviewed and approved the final version of the manuscript.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Author contributions
D.P., D.P., and D.N. contributed to Conceptualization; Visualization; Writing – original draft; Writing – review & editing. V.V. supervised the study and provided critical feedback and revisions. All authors contributed to the finalization of the manuscript and approved the final version
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All information in this review was obtained from previously published literature. Therefore, no new data were generated or analyzed, and all referenced data are publicly available.
