Abstract
The mechanisms underlying scarless versus fibrotic wound healing remain a critical challenge in regenerative medicine. To elucidate the mechanisms of scarless repair, the axolotl (Ambystoma mexicanum), a model organism with exceptional regenerative capacity, has gained increasing prominence. Although axolotls are capable of regenerating complex structures such as limbs and tails, whether their skin regeneration is uniformly scarless—especially across different anatomical sites—remains undefined. Here, we demonstrate that adult axolotl tail skin achieves scarless regeneration, while dorsal skin repair results in fibrotic scarring. Through comparative histological and transcriptomic analyses of full-thickness excisional wounds, we identify accelerated re-epithelialization and reduced collagen deposition in tail skin compared to dorsal wounds. Developmental trajectory studies reveal prolonged retention of a hypodifferentiated state in tail skin, contrasting with early stratification in dorsal tissue. Mechanistically, we find that the extracellular matrix (ECM) glycoprotein gene Tenascin-N (TnN) exhibits higher expression in tail skin versus dorsal skin. The reported TnN downstream PI3K-Akt signaling pathway, demonstrated by Western blotting of pAkt, is significantly activated in tail skin versus dorsal skin during homeostasis and regeneration. These findings establish the axolotl as a key model for dissecting how developmental priming and ECM dynamics orchestrate regenerative versus fibrotic repair, offering novel insights for therapeutic strategies targeting scarless healing.

Introduction
The skin, the largest organ of the human body and the primary barrier against external environmental insults 1 , is composed of multiple layers, including the epidermis, dermis, and hypodermis. This structure plays vital roles in physiological processes, including pathogen defense, thermoregulation, sensory transmission, and immune surveillance 2 . Moreover, the complex, dynamic interactions between epidermal keratinocytes and dermal fibroblasts, along with intact skin appendages, constitutes the structural foundation for the skin to achieve effective barrier function and maintain tissue homeostasis 3 . However, as the most exposed organ, the skin constantly encounters environmental insults. The healing of severe skin injuries often involves complications, most notably fibrotic scars that impair function or cause disfigurement, which significantly diminish patients’ quality of life and impose substantial medical and socioeconomic burdens. Therefore, gaining deeper insights into the mechanisms of skin wound repair—particularly those regulating fibrosis inhibition and scarless regeneration—is essential for advancing clinical outcomes. In light of these challenges, skin wound repair remains a central focus of regenerative medicine research.
Skin wound repair relies on the coordinated interactions between diverse cell types and extracellular matrix (ECM) and is orchestrated by a sophisticated regulatory network comprising diverse signaling molecules such as cytokines 3 , neuropeptides 4 , and growth factors 5 . Although significant progress has been made in elucidating the molecular and cellular mechanisms of mammalian tissue repair, the inherently complex cellular interactions, dynamic changes in signaling pathways during wound healing 6 , and the absence of ideal research models collectively hinder the achievement of perfect, scarless regeneration. To overcome these challenges and the ethical limitations associated with obtaining human skin samples, the development of suitable animal models is crucial for elucidating the mechanisms of skin regeneration. Currently, mammalian models—such as rodents (e.g., mice and rats) and pigs—are widely employed in studies of wound repair and scar formation. However, these models have limitations in elucidating the regulatory mechanisms of scarless regeneration. For instance, wound healing in mice is highly dependent on panniculus carnosus contraction, a process that markedly differs from the predominantly re-epithelialization-based healing observed in humans 7 . Although porcine skin is structurally similar to human skin, it is cumbersome to manipulate and incurs high costs 8 . More importantly, these mammalian models invariably develop fibrotic scars following injury, thereby limiting their utility in investigating strategies to achieve fully functional regeneration. The composition and dynamic remodeling of the ECM are considered key determinants of repair outcomes 9 . However, the scar-prone nature of these models hinders a comprehensive elucidation of the ECM regulatory network underlying perfect regeneration. Therefore, the development and utilization of alternative models with superior regenerative capacity are essential for elucidating the mechanisms of scarless healing.
The axolotl (Ambystoma mexicanum), a prominent model in regenerative biology, has received considerable attention for its exceptional regenerative abilities. Its skin shares a highly conserved evolutionary structure with that of mammals 10 , consisting of the epidermis, dermis, and hypodermis and containing core components such as keratinocytes, fibroblasts, and collagen. Unlike mammals—especially adults, whose deep skin injuries typically result in scarring—the axolotl’s skin exhibits extraordinary regenerative capacity. Upon injury, axolotl skin rapidly re-epithelializes and activates resident stem cells 11 , enabling the reconstruction of tissue with near-normal structure and function. Even under stimulation by pro-fibrotic agents (e.g., bleomycin), axolotl skin exhibits significantly lower levels of collagen deposition compared to mammals, rendering it a unique and valuable model for investigating the mechanisms of scar regulation 12 . Nevertheless, whether axolotl skin can achieve true “perfect regeneration” following injury remains controversial. Some studies have indicated that, although the overall tissue architecture may be restored, the fine organization of dermal collagen fibers may differ from that of uninjured skin 13 . Considering the axolotl’s unique regenerative capacity, further elucidation of its regenerative mechanisms—and the key factors enabling scarless healing—requires the development of more refined injury models.
Regenerative capacity is often closely linked to developmental stage and tissue differentiation status, with early developmental or less differentiated tissues generally displaying greater regenerative potential 14 . Most current studies of axolotl skin injury utilize full-tail or limb amputation models 15 or localized skin injury paradigms 13 . However, these approaches have intrinsic limitations in isolating skin-specific regenerative responses: the former focuses on multitissue/organ coordination, obscuring the precise contribution of isolated skin tissue; the latter, often involving superficial wounds or partial excisions on appendage skin, lacks systematic comparison of regenerative potential across intra-organismal anatomical regions. Consequently, whether the axolotl—renowned for its robust regenerative prowess—displays uniform skin regenerative ability across dorsal, tail, and other body regions, and whether such potential heterogeneity is governed by local developmental maturity or microenvironmental cues, remains unaddressed. To address this knowledge gap regarding regional differences regarding regional specificity in axolotl skin regeneration and its relationship with developmental trajectories, we performed standardized full-thickness excision (FTE) on both dorsal and tail skin of adult axolotls. Based on the theoretical framework suggesting a link between development and regeneration 16 , we hypothesize that tail skin may maintain a relatively lower state of differentiation during development which may underlie its enhanced regenerative potential. This study aims to systematically compare the differences in regenerative rates and fibrosis levels between dorsal and tail skin following injury, combined with transcriptomic analyses of uninjured tissue, to provide the first systematic evidence of regional heterogeneity in axolotl skin regenerative potential and to further explore the underlying molecular basis of this heterogeneity and its possible association with intrinsic developmental trajectories. The findings of this study will underscore the importance of the axolotl as a powerful comparative model for skin regeneration research and provide new insights into the intrinsic relationship between tissue developmental status and regenerative capacity.
Materials and Methods
Animals
The d/d strain Ambystoma mexicanum used in this study was obtained from Elly M. Tanaka laboratory (Research Institute of Molecular Pathology, Vienna Biocenter, Vienna, Austria), and bred and maintained locally in freshly dechlorinated tap water at 20°C, with daily feedings. Animals were raised to various developmental stages for histological analysis: 30, 40, and 48 days post-fertilization (dpf); and 4, 8, 11 (sexual maturity), and 24 months post-fertilization (mpf). Animals were anesthetized in 0.03% (w/v) benzocaine (Sigma, E1501-500G) prior to imaging, surgery, and sample collection. All animal procedures were conducted in compliance with Chinese animal welfare legislation, approved by the Biomedical Research Ethics Committee of Guangdong Provincial People’s Hospital (approval number KY2024-192-01).
FTE Wounding
Adult axolotls were deeply anesthetized using 0.03% benzocaine. Sterile 3 mm biopsy punches were used to create FTE wounds penetrating the full skin thickness on the dorsal back and tail regions. Specifically, two wounds were made on each side of the dorsal region (located at the sixth myotome anterior to the cloaca) and the tail region (at the fourth myotome posterior to the cloaca), totaling four wounds per animal. After wounding, the epidermis at the center of the 3-mm circle was carefully removed with fine tweezers before animals were returned to clean water. Tissue samples were harvested at designated time points post-injury for subsequent analyses.
Wound Closure Assessment
Wound re-epithelialization was assessed using a vital dye method based on fast green staining, as previously described in axolotl wound healing models (Lévesque et al.). Briefly, 0.1% Fast Green (Sigma, F7252) was applied to each wound for 1 minute and then rinsed with phosphate-buffered saline (PBS). Wounds retaining the dye were considered incompletely epithelialized. Images were captured under standardized lighting conditions. Wound area and stained regions were quantified using ImageJ. The relative stained area was calculated by normalizing to the initial stained area at 0 hpi.
Tissue Processing for Histology and Immunofluorescence
For comprehensive histological and immunofluorescence assessments, harvested tissue samples were briefly rinsed in 1× PBS. Subsequently, samples were immersed in 30% sucrose solution in 1× PBS overnight at 4°C for cryoprotection. The samples were then rapidly snap-frozen in Tissue-Tek (4583, Sakura, Torrance, CA, USA) using liquid nitrogen–cooled isopentane and stored directly at −80°C. Cryosections, typically 10 to 20 μm thick, were cut using a cryostat and stored at −20°C for subsequent analysis.
Masson’s Trichrome Staining
Masson’s trichrome staining was performed using the Solarbio Masson’s Trichrome Stain Kit (Cat# G1346) with modifications for cryosections. In brief, cryosections were air-dried, rehydrated, fixed in Bouin’s solution, and stained sequentially with Weigert’s iron hematoxylin, Biebrich scarlet-acid fuchsin, and aniline blue solutions. Quantitative analyses of Leydig cell density, stratum compactum thickness, and dermal gland density were performed using ImageJ.
Immunofluorescence Staining
Antigen retrieval was performed at 85°C for 10 min in Citrate Antigen Retrieval solution (Sangon, E673001-0100) as the samples were cryosections. After the antigen retrieval, the sections were washed with phosphate-buffered saline with Tween-20 (PBST) to remove the retrieval solution. Then, the sections were blocked in 5% donkey serum (Solarbio, ASL050) and incubated overnight at 4°C with primary antibodies diluted in antibody dilution buffer (Genefist, GF1600-1). The primary antibodies used in this study were KRT4 (Sino Biological, 200539-T42), mouse monoclonal Anti-Collagen Type I (COL1A1) Antibody (C2456, Sigma-Aldrich, COL-1), PCNA (D3H8P) XP Rabbit mAb, Alexa 488-donkey anti-mouse IgG (Jackson, 711-547-003), Alexa 555-donkey-anti-rat IgG (Invitrogen, SA5-10027), Alexa 647-donkey-anti-mouse IgG (Jackson, 715-607-003), and CY3-donkey-anti-mouse IgG (Jackson, 715-165-151). After a PBST wash, the sections were incubated for 2 h at room temperature with secondary antibodies and DAPI (Sigma, D9542). The secondary antibodies used were Cy3-AffiniPure donkey anti-rat IgG (Jackson, 712-165-153), Cy5-conjugated goat anti-rabbit IgG (Thermofisher, A10523) and Alexa Fluor 647-donkey anti-mouse IgG (Jackson, 715-606-150). Sections were mounted with anti-fade mounting mediums (Bio-rad, BUF058C) and imaged using a confocal microscope (LSM980) equipped with ×20 objectives. Signal quantification was done using Fiji.
RNA Extraction, cDNA Synthesis, Quantitative Real-Time PCR Analysis
Total RNA was isolated from uninjured dorsal and tail skin tissues of sexually mature (11-month-old) axolotls using TRIzol Reagent (Invitrogen, Cat. No. 15596018) following the manufacturer’s protocol. Complementary DNA (cDNA) was synthesized from the total RNA using the HiScript 1st Strand cDNA Synthesis Kit (Vazyme, Cat# R212), in accordance with the manufacturer’s instructions. Quantitative PCR was performed with specific primer sets on an ABI QS5 Real-Time PCR Detection System, utilizing the AceQ qPCR SYBR Green Master Mix (Vazyme, Cat# AQ101) as per the provided protocol. Complementary DNA concentrations were normalized to the beta-actin gene. The sequences of the primers used for qPCR are provided in Supplementary Tables S2.
Bioinformatics Analysis of RNA-seq
For RNA sequencing, total RNA was extracted from uninjured dorsal and tail skin tissues of 11-month-old axolotls as described above. The RNA samples were subsequently sent to Berry Genomics for next-generation sequencing (NGS) using the Illumina HiSeq platform. Sequencing was conducted at a high depth to ensure comprehensive transcriptomic coverage. The raw sequencing data in the form of FASTQ files were processed by trimming adapter sequences and removing low-quality bases using the Trimmomatic tool. The reads were then mapped against the current axolotl reference genome (https://www.axolotl-omics.org/, AmexG_v6.0-DD) using HISAT2 v2.2.1 with standard parameters. Transcript quantification was then conducted using StringTie with standard parameters and the option of assembling transcripts. Finally, the FPKM of each gene was calculated based on the length of the gene and the read count mapped to the gene. Prior to differential gene expression analysis, for each sequenced library, the raw read counts were adjusted with the DESeq2 program package using one normalized scaling factor, and genes were classified as significantly differentially expressed if P adj < 0.05 and|log2 fold change| ≥ 1. GO enrichment analysis and KEGG enrichment analysis of DEGs were performed with Cluster Profiler (3.4.4) software.
Western Blotting
Total protein extraction from dorsal and tail skin wound tissues harvested at 0, 3, and 7 dpi axolotl tissues was performed using a commercially available lysis buffer (Affinibody, AIWB-012). After centrifugation at 12,000 rpm at 4°C for 20 min, the supernatant was collected, and protein concentration was measured by the BCA protein quantification kit (Vazyme, E112-01) following the manufacturer’s protocol. A total of 30 μg of protein per group was loaded to 4% to 12% SDS-PAGE gel. Gel electrophoresis was conducted at 160V for 60 min. Proteins were transferred to PVDF membrane (Immobilon, IPVH00010) via semi-dry transfer at 20V for 15 min. The membrane was blocked in 5% bovine serum albumin (BSA; Sigma, V90933) for 1 h at room temperature. Primary antibodies used included rabbit anti-Phospho Akt (1:1000, Cell signaling # 4056S), rabbit anti-total AKT (1:1000, Cell Signaling #4691), and mouse-anti-Beta Actin Monoclonal antibody (1:25,000, Proteintech Group, 66009-1-1g), which were incubated at 4°C overnight. After thoroughly washing, membranes were incubated with secondary antibodies rat-anti-mouse-HRP (1:10,000, Jackson, 115-035-068) or donkey-anti-rabbit-HRP (1:10,000, Jackson, 711-035-152) for 1 h at room temperature. Then, after TBST (0.1% Tween) washing for three times. Signal was visualized using the NcmECL Ultra kit (Ncmbio, P10100), and images were acquired using the Touch Imager (E-BLOT).
Quantification of Immunofluorescence and Western Blot Signal Intensity
All image-based quantifications were performed using Fiji (ImageJ), following standardized workflows for histological and immunofluorescent signal analysis. Background fluorescence was removed using the “Subtract Background” function. PCNA positivity was calculated as the percentage of PCNA+ nuclei among total DAPI+ nuclei within re-epithelialized wound epidermis. α-SMA-positive myofibroblasts were identified as cells showing cytoplasmic α-SMA immunofluorescence, nuclear co-localization with DAPI, and elongated spindle-like morphology. Manual cell counting was performed in the central wound region, and results expressed as α-SMA+ cell density (cells/mm²). COL1A1 fluorescence intensity was quantified as mean fluorescence intensity (MFI) within the dermal regions of interest (ROIs) corresponding to the regenerating wound area. Background fluorescence was removed using the “Subtract Background” function in Fiji (ImageJ). For normalization, the MFI of each ROI was divided by the MFI of uninjured tissue from the same animal. The final intensity values are reported as normalized intensity (AU). Western blot band intensities for p-Akt, total Akt, and β-actin were quantified using Fiji by measuring integrated density (area × mean gray value) for each band after background subtraction. To correct for loading variation, p-Akt and total Akt values were normalized to β-actin. The p-Akt/total Akt ratio was then calculated using these normalized values for statistical comparison 17 .
Statistical Analysis
Statistical analyses were performed using GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA). Quantitative data are presented as mean ± standard error of the mean (SEM) or standard deviation (SD), as stated in figure legends. Comparisons between two groups were assessed using unpaired two-tailed Student’s t-tests. For multiple-group comparisons, one-way analysis of variance (ANOVA) was performed followed by Dunnett’s multiple comparisons test. Statistical significance was defined as P < 0.05. In all graphs, **P < 0.01, ***P < 0.001, and ****P < 0.0001.
Results
Distinct Morphological and Healing Differences Between Dorsal and Tail Skin in Axolotls Following FTE
To systematically compare the regenerative capacity of dorsal and tail skin in adult axolotls and to characterize region-specific morphological responses following injury, we established a standardized FTE injury model. A 3-mm skin biopsy punch was used to create full-thickness wounds on both dorsal and tail regions, carefully avoiding damage to the underlying muscle tissue (Fig. 1a). The healing process was longitudinally monitored at defined time points post-injury, and wound areas were quantitatively analyzed to identify spatiotemporal heterogeneity in tissue repair across anatomical regions.

Morphological differences in wound healing between axolotl dorsal and tail skin following full-thickness excision injury. (a). Creation of full-thickness excisional (FTE) skin injury models on axolotl dorsal and tail skin using 3-mm biopsy punches. (b) Representative gross images of dorsal and tail wounds at 0, 7, 35, and 70 days post-injury (dpi). The original injury sites were indicated by black circles. Tail wounds progressively closed and became visually indistinct by 35 dpi, whereas dorsal wounds retained a clear wound boundary at 70 dpi. (c) Quantitative analysis of wound area in dorsal and tail skin at 35 and 70 dpi. Tail skin exhibited a significantly faster healing rate at both time points. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. ****P < 0.0001; n = 3. Scale bar, 1 mm. (d) Macroscopic appearance of dorsal and tail skin at 200 dpi. Tail wounds showed complete regeneration without visible scarring, while dorsal wounds displayed a depressed scar phenotype.
Macroscopic observations revealed that tail skin exhibited a notable hyperemic response with visible red blood cell infiltration in the subcutaneous tissue at 7 days post-injury (dpi). By 35 dpi, the regenerated epidermis remained lightly pigmented but matched uninjured skin in thickness; by 75 dpi, the regenerated area was macroscopically indistinguishable from surrounding skin (Fig. 1b). In contrast, dorsal skin showed significantly less hyperemia during the inflammatory phase (0–7 dpi), suggesting slower vascular reconstruction. At 35 dpi, a distinct boundary persisted between injured and uninjured epidermis. Moreover, by 70 dpi, dorsal wounds exhibited an atrophic scar phenotype.
Quantitative analysis of wound areas revealed significantly faster healing in tail versus dorsal skin at both 35 and 70 dpi (Fig. 1c). At 200 dpi, dorsal wounds exhibited hallmarks of atrophic scarring, such as a depressed surface, whereas tail wounds showed no apparent scar formation (Fig. 1d).
In summary, macroscopic observations demonstrate significant regional heterogeneity in axolotl skin regeneration: tail skin exhibits accelerated regeneration and scarless healing, while dorsal skin shows delayed repair with a propensity for atrophic scar formation.
Re-Epithelialization Analysis Reveals Accelerated Wound Closure in Axolotl Tail Skin Compared to Dorsal Skin
Rapid re-epithelialization is recognized as a critical step in successful wound healing, as it swiftly seals the wound and effectively prevents infection and fluid loss 18 . To quantitatively compare potential regional differences in epithelial wound coverage between tail and dorsal skin, Fast Green staining was used to mark exposed wound surfaces as an indirect indicator of re-epithelialization progress. Fast Green selectively labels uncovered cells in the wound bed, providing a convenient method to monitor wound closure 10 . Theoretically, once the newly formed epidermis completely covers the wound bed, the stained area will markedly diminish or even disappear.
Early staining results demonstrated that Fast Green effectively labeled the entire wound area in both dorsal and tail skin injuries (Fig. 2a, f), indicating full exposure of the wound beds in both regions during the initial injury phase. However, at 3 hours post-injury (hpi), a markedly reduced stained area was observed in tail wounds compared to dorsal wounds (Fig. 2B), suggesting that epidermal cell migration and wound coverage were initiated earlier in the tail region. By 12 hpi, the Fast Green-stained area at the edges of tail wounds was no longer detectable, whereas dorsal wounds still exhibited extensive staining (Fig. 2b, g), clearly indicating that tail skin undergoes re-epithelialization at a significantly faster rate than dorsal skin. At 24 hpi, the Fast Green-stained area in tail wounds further diminished, becoming considerably smaller than that in dorsal wounds (Fig. 2c, h), indicating that re-epithelialization in tail skin was nearing completion. By 30 hpi, Fast Green staining was virtually undetectable in tail wounds, suggesting complete epidermal coverage of the tail wound surface. In contrast, the central region of dorsal wounds continued to display Fast Green staining (Fig. 2d, i), with complete clearance of the stain not occurring until 48 hpi (Fig. 2e, j). Quantitative analysis of the stained wound areas (Fig. 2B) corroborated these observations, confirming that the re-epithelialization rate in tail skin is significantly faster than that in dorsal skin.

Visualization of wound closure in axolotl excisional wounds. (A) Representative images of Fast Green staining in tail (a–e) and dorsal (f–j) skin wounds at multiple time points post-injury. (a, f) Immediately after injury (0 hours post-injury, hpi): entire wound beds are stained, indicating full exposure of the wound surface. (b, g) At 12 hpi: peripheral staining was absent in tail wounds, suggesting the initiation of re-epithelialization; dorsal wounds remain largely stained. (d, i) At 30 hpi: tail wounds showed no visible staining, indicating complete epidermal coverage, while dorsal wounds still retained central staining. (e, j) At 48 hpi: both wound types are fully re-epithelialized, with no staining remaining. (B) Quantitative analysis of the relative wound epithelialization area over time. Tail wounds demonstrated significantly faster re-epithelialization compared to dorsal wounds. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. *P < 0.05, **P < 0.01, ***P < 0.001; n = 3. Scale bars, 1.5 mm.
Both visual observations and statistical analyses confirmed that the re-epithelialization process in tail skin is markedly more rapid than in dorsal skin, enabling quicker wound closure and establishing a more favorable microenvironment for subsequent dermal regeneration and tissue remodeling.
Superior Epithelial Migration, Differentiation, and Dermal Reconstruction in Axolotl Tail Skin Post-Injury Compared to Dorsal Skin
To elucidate the cellular mechanisms underlying the observed regional differences in wound healing between axolotl tail and dorsal skin, we performed Masson’s trichrome staining on wound tissues at various time points after FTE (Fig. 3A). Histological analysis using Masson’s trichrome staining unequivocally revealed significant regional heterogeneity in the wound repair process. Notably, compared to dorsal skin, tail skin appears to have superior regenerative capacity, as suggested by three key features: a faster re-epithelialization rate, enhanced epidermal differentiation, and more complete dermal reconstruction—findings that are consistent with our prior macroscopic observations.

Histological assessment of regional differences in axolotl skin wound healing using Masson’s trichrome staining. (A) Skin sections collected at 0, 8, and 12 hours, and 14, 30, and 200 dpi were stained with Masson’s trichrome to assess re-epithelialization, Leydig cell development, and dermal organization. Panels (a–f) represent tail skin; panels (g–l) represent dorsal skin. Yellow arrowheads indicate wound margins. (a, g) At 0 hpi: only skin was removed without disturbing the underlying muscle. (b, h) At 8 hpi: complete re-epithelialization occurred in tail wounds, while epithelial migration had just initiated in dorsal wounds. (c) At 12 hpi: Leydig cells (green arrows) appeared in the tail wound epidermis. (i) At 12 hpi: dorsal wounds showed complete re-epithelialization but contained markedly fewer Leydig cells. (d, j) At 14 dpi: tail wounds exhibited abundant mature Leydig cells, whereas dorsal wounds showed only sparse and immature Leydig-like cells. Dermal cell aggregation (blue arrows) was observed beneath the wound epidermis. (e, k) At 30 dpi: both wound types restored the basement membrane (blue staining); however, only tail wounds formed distinct dermal layers including the stratum spongiosum (ss) and the stratum compactum (sc), while dorsal wounds lacked clear dermal stratification. (f, l) At 200 dpi: both regions showed fully stratified skin with glandular structures (green arrows); tail wounds exhibited collagen organization comparable to uninjured tissue, while dorsal wounds showed excessive collagen deposition (blue staining), indicative of fibrotic repair. (B) Quantification of Leydig cell density in the regenerated epidermis at hpi, comparing dorsal and tail wounds. (C) Measurement of stratum compactum thickness at 200 dpi using uninjured skin as a reference. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. ****P < 0.0001; n = 5. Scale bars, 200 μm.
By 8 hpi, histological analysis revealed complete re-epithelialization in tail wounds, evidenced by a continuous epidermal layer covering the entire wound bed (Fig. 3b). In contrast, dorsal skin at the same time point exhibited only the initial signs of re-epithelialization (Fig. 3h). Furthermore, as regeneration advanced, tail skin not only re-epithelialized faster but also exhibited accelerated cellular differentiation. At 12 hpi, Leydig cells—rod-shaped epidermal differentiation markers—had already emerged in tail wounds and were significantly more abundant than in dorsal skin (Fig. 3B). These cells were also observed in uninjured epidermis. Conversely, dorsal skin only achieved complete re-epithelialization by 12 hpi (Fig. 3i). By 14 dpi, the number of Leydig cells in tail skin approached levels observed in uninjured skin, and the cells exhibited mature morphology (Fig. 3d). In comparison, dorsal skin at the same time point exhibited only a few underdeveloped structures resembling Leydig cells (Fig. 3j). Collectively, these histological findings demonstrate that tail skin initiates an efficient epidermal differentiation program early after injury, while the epidermal differentiation process in dorsal skin is significantly delayed.
The temporal differences in dermal reconstruction further underscore the regional heterogeneity in regenerative capacity between dorsal and tail skin. Over time, the gap between the wound epidermis and the underlying muscle gradually narrowed. By 14 dpi, a large number of dermal cells had accumulated beneath the wound epidermis in tail skin (Fig. 3d, j; blue arrows), resembling the dermal cell migration patterns reported by Endo et al. 19 in limb regeneration models. By 30 dpi, tail wounds had developed an initially organized collagen fiber network (Fig. 3e), accompanied by the restoration of basement membrane integrity (indicated by blue staining for collagen). In contrast, the dermal architecture of dorsal wounds at the same time point remained incomplete, lacking the expected stratum compactum (Fig. 3k). Importantly, long-term observations at 200 dpi showed that both dorsal and tail wound sites achieved complete skin regeneration in axolotls. These regenerated tissues reconstructed functional, stratified structures—including epidermis, dermis, and embedded glands—that were functionally and histologically comparable to normal skin (Fig. 3f, l; green arrows indicate glands). However, dorsal wounds showed pronounced blue collagen deposition in the stratum compactum (Fig. 3l), which was significantly thicker than in tail skin (Fig. 3C), suggestive of excessive collagen accumulation and potential fibrosis. This observation further supports the presence of excessive collagen deposition and fibrosis in dorsal skin, a pattern resembling typical mammalian scar formation. Conversely, tail skin wounds at 200 dpi displayed collagen deposition comparable to normal tissue, with restored stratum compactum integrity and no evident fibrosis (Fig. 3f). The regenerated tail skin was virtually indistinguishable from uninjured tissue, indicative of complete regeneration—including restoration of the stratum compactum—through a scarless healing process that reinstated full structural and functional integrity.
In summary, compared to dorsal skin, which exhibits more pronounced collagen deposition closely associated with excessive fibrosis during regeneration, axolotl tail skin demonstrates a faster re-epithelialization rate, more efficient epidermal differentiation, and more rapid and complete dermal reconstruction. This is achieved by establishing a collagen fiber network that closely approximates the thickness of normal tissue, effectively preventing abnormal collagen accumulation and ultimately leading to near-complete skin regeneration. This regeneration is characterized by the restoration of structural and functional integrity, rendering the regenerated tissue indistinguishable from uninjured skin.
Enhanced Proliferation and Keratin 4 (KRT4) Expression in Axolotl Tail Skin Compared to Dorsal Skin Post-Injury Suggest a Role for KRT4 in Early Dermal Reconstruction
Previous studies have documented significant regional differences in re-epithelialization rates between axolotl dorsal and tail skin following injury. To further elucidate the cellular mechanisms underlying this disparity and characterize the potential role of keratinization, we utilized immunofluorescence staining to examine epidermal cell proliferation and KRT4 expression dynamics. Specifically, we performed DAPI/PCNA/KRT4 immunofluorescence staining on dorsal and tail wound tissues at various time points following FTE injury (Fig. 4a, b). These analyses aimed to systematically evaluate KRT4 expression patterns and regional differences in cellular proliferation.
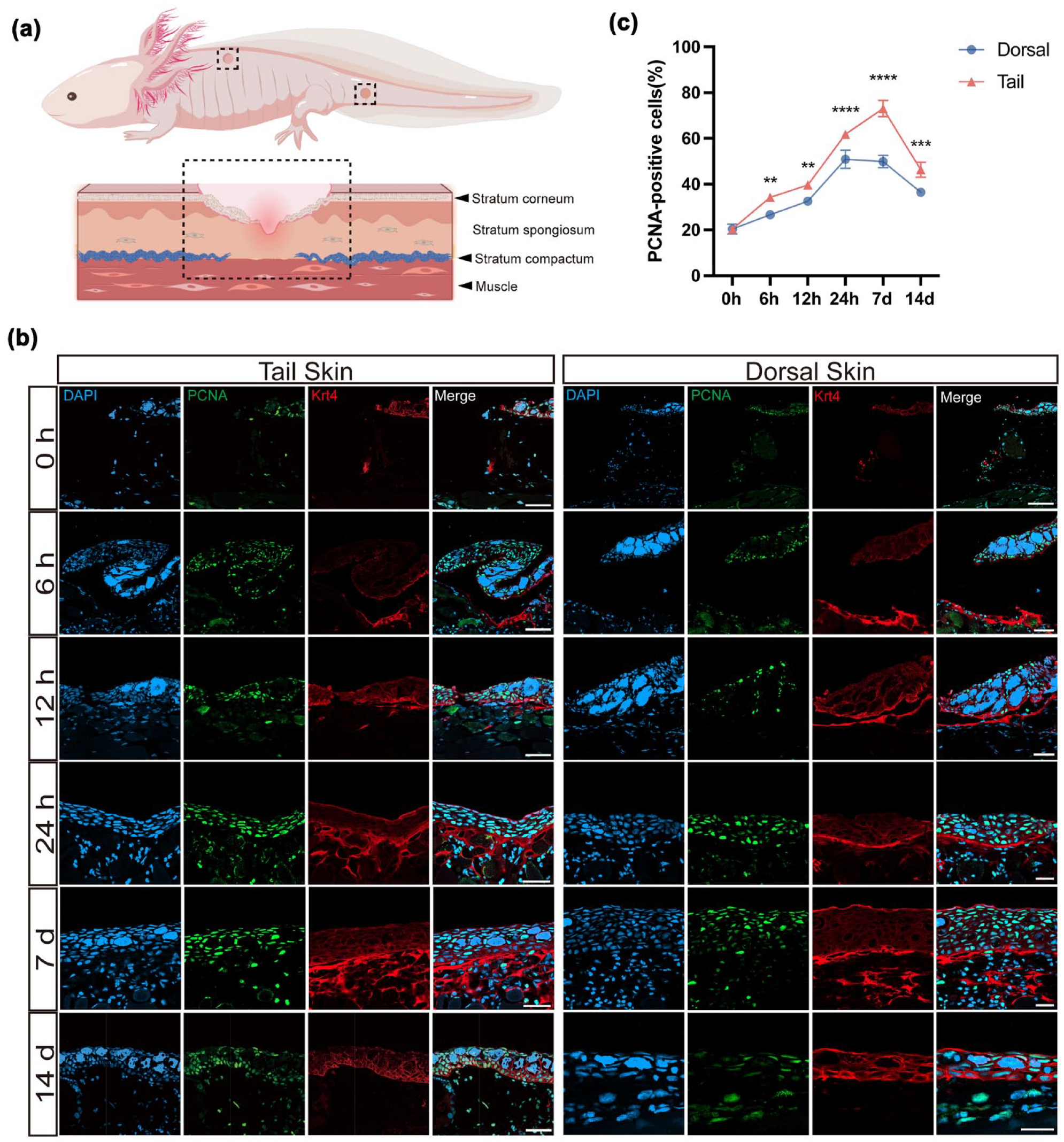
Immunofluorescence analysis revealed regional differences in epidermal proliferation and KRT4 expression during axolotl skin wound healing. (a) Schematic illustration of the wound sampling regions (dashed boxes) from dorsal and tail skin. (b) Representative immunofluorescence images of dorsal and tail wounds stained for DAPI (blue), PCNA (green), and KRT4 (red) at multiple time points post-FTE (from 6 hpi to 14 dpi). In uninjured skin, KRT4 expression was confined to the epidermis. Between 6 hpi and 7 dpi, KRT4 expression transiently extended into subepidermal regions, suggesting injury-induced ectopic expression. By 14 dpi, KRT4 signal was again restricted to the epidermis. Notably, at 24 hpi and 7 dpi, tail wounds exhibited markedly higher levels of subepidermal KRT4 accumulation compared to dorsal wounds. Scale bars, 100 μm. (c) Quantification of PCNA-positive cell density in dorsal and tail skin wounds at indicated time points. Tail wounds consistently showed significantly higher proliferative activity across all stages of wound healing. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. **P < 0.01, ***P < 0.001, ****P < 0.0001; n = 5. Scale bars, 100 μm.
Immunofluorescence staining revealed that KRT4 expression in uninjured normal skin was strictly confined to the epidermal layer (Fig. 4b), consistent with its established role as a marker of differentiated epidermal cells 20 . However, following FTE injury, both dorsal and tail wounds exhibited marked alterations in KRT4 expression dynamics. During the early post-injury phase (6 hpi to 7 dpi, Fig. 4b), KRT4-positive signals extended beyond the epidermis into subepidermal tissues, likely due to injury-induced transient ectopic expression in non-epithelial cell types 21 . Notably, this ectopic expression exhibited clear spatiotemporal specificity: by 14 dpi, KRT4 localization returned to the epidermis (Fig. 4b), suggesting that keratin gene expression is dynamically regulated across distinct phases of repair and may serve functional roles during early wound healing 22 .
To characterize differences in cell proliferation and KRT4 expression, we observed that KRT4 accumulation in subepidermal regions was significantly higher in tail wounds than in dorsal wounds at 24 hpi and 7 dpi. These observations support the hypothesis that epidermis-derived cells contribute to dermal reconstruction in tail skin, consistent with the findings of Ayaka Ohashi et al. 15 on keratinocyte-driven collagen deposition in axolotls. Analysis of PCNA immunostaining revealed consistently higher densities of PCNA-positive nuclei in tail wounds compared to dorsal regions (Fig. 4c). This elevated proliferative response likely provides a cellular foundation for the enhanced regenerative capacity observed in tail skin.
In summary, our findings demonstrate that tail skin exhibits a markedly superior regenerative capacity compared to dorsal skin. This advantage is driven by sustained cell proliferation and tightly regulated, phase-specific keratin gene expression. Together, these processes promote rapid and orderly skin regeneration with minimal scarring, ultimately restoring full tissue architecture. These coordinated mechanisms likely underpin the enhanced regenerative prowess observed in tail skin.
Dorsal Skin Wound Healing Exhibits Region-Specific Fibrosis Driven by Aberrant ECM Remodeling and Protracted Myofibroblast Activity
In mammalian wound healing, myofibroblasts secrete ECM components, including collagen fibers, and their excessive activation is closely linked to fibrosis23,24. Thus, the differentiation and proliferation of myofibroblasts must be tightly regulated to prevent pathological scarring 25 . Axolotl tail skin demonstrates near-scarless regeneration, while dorsal skin healing often exhibits signs of fibrosis, offering a compelling model to study regional differences in ECM remodeling. To characterize the spatiotemporal differences in ECM dynamics during wound repair, we performed immunofluorescence staining on dorsal and tail wound tissues at 7 and 30 dpi, evaluating the expression of type I collagen (Col1a1), the predominant collagen in the dermal matrix, and α-smooth muscle actin (α-SMA), a widely recognized marker of myofibroblasts (Fig. 5a). Pathological over-deposition of Col1a1, the most abundant collagen in the dermal matrix, is a key hallmark of fibrotic diseases 26 . α-SMA-positive myofibroblasts play a central role in wound contraction and ECM remodeling, and their excessive accumulation is associated with scar formation in fibrotic conditions 23 .
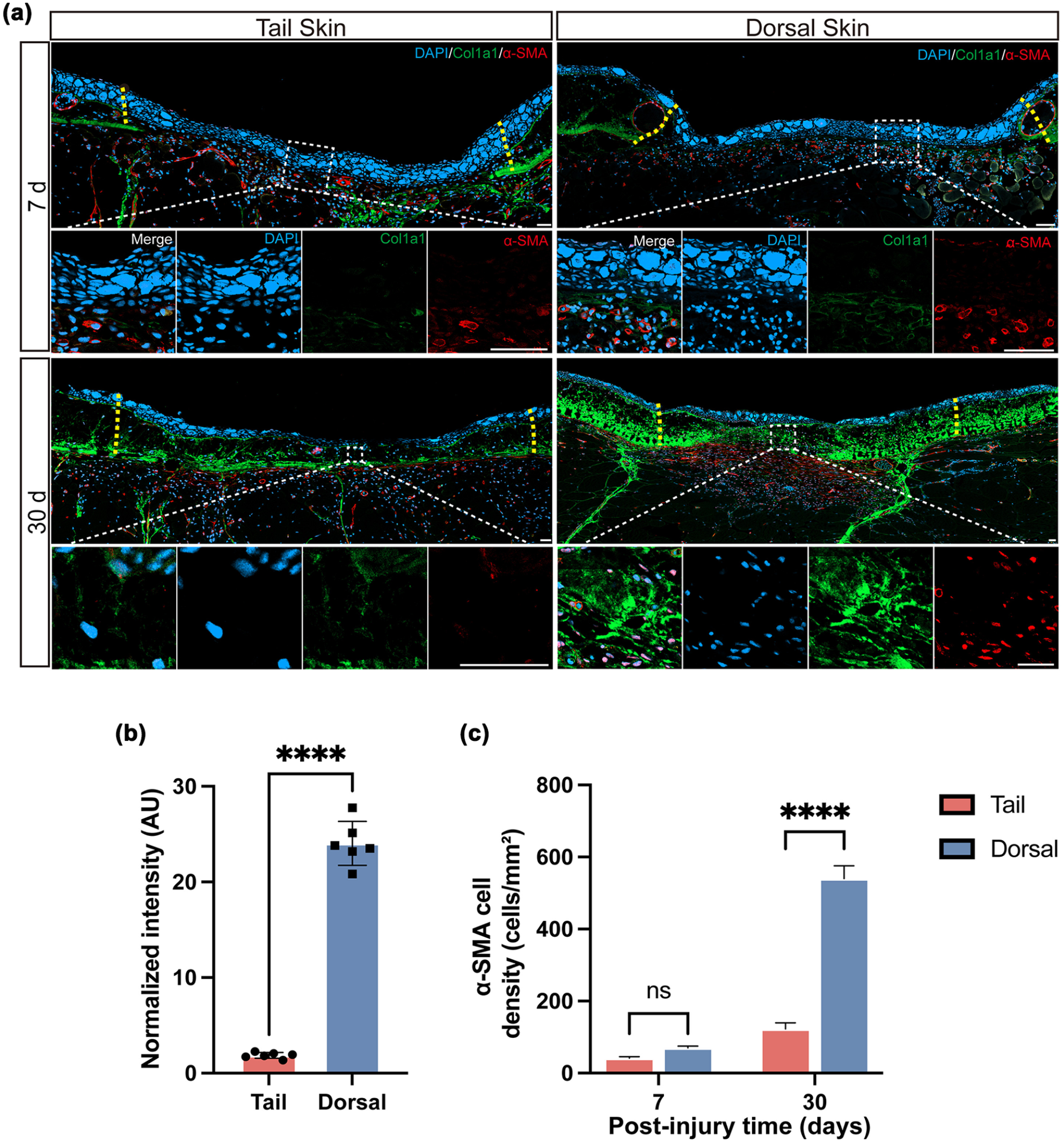
Regional differences in collagen deposition and myofibroblast accumulation during axolotl skin wound healing. (a) Representative immunofluorescence staining of type I collagen (Col1a1, green) and α-smooth muscle actin (α-SMA, red) in dorsal and tail wounds at 7 and 30 dpi. Col1a1 labels dermal collagen fibers, while α-SMA marks activated myofibroblasts involved in wound contraction and ECM remodeling. Tail wounds showed visibly reduced signal intensity for both markers compared to dorsal wounds, particularly at 30 dpi. (b) Quantification of Col1a1 fluorescence intensity in dorsal and tail skin wounds at 30 dpi, measured in arbitrary units (AU) using ImageJ. Tail wounds exhibited significantly lower collagen deposition. (c) Quantification of α-SMA-positive cell density (cells/mm²) in dorsal and tail skin wounds at 7 and 30 dpi. Tail wounds displayed significantly fewer α-SMA-positive cells at 30 dpi, indicating reduced myofibroblast accumulation. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. ****P < 0.0001; n = 5. Scale bars, 100 μm.
Immunofluorescence analysis revealed region-specific differences in fibrosis markers: at 30 dpi, Col1a1 signal intensity was significantly lower in tail wounds than in dorsal wounds, indicating reduced collagen deposition in the tail region (Fig. 5a, b). Quantitative analysis of α-SMA-positive myofibroblast density (Fig. 5c) showed no significant difference between dorsal and tail wounds at 7 dpi. By contrast, at 30 dpi, dorsal wounds showed a significantly higher density of α-SMA-positive cells than tail wounds, implying prolonged myofibroblast activity or delayed resolution, which may contribute to fibrotic outcomes. This observation aligns with mammalian scarring models, where sustained α-SMA expression correlates with fibrotic outcomes 27 .
Consistent with these findings, both Col1a1 and α-SMA immunostaining revealed significantly elevated levels in dorsal compared to tail wounds (Fig. 5b, c), further confirming the presence of fibrosis-associated collagen overaccumulation in dorsal skin. In contrast, tail skin exhibited no evidence of disorganized collagen architecture or myofibroblast accumulation. These results mirror the dense-layer fibrosis observed in dorsal wounds via Masson trichrome staining (Fig. 3), offering additional evidence of region-specific differences in ECM remodeling. Taken together, our data suggest that intrinsic developmental differences between dorsal and tail skin contribute to distinct collagen remodeling capacities during wound healing. These disparities ultimately give rise to two divergent regenerative outcomes: scarless repair in the tail and fibrosis-associated healing in the dorsal skin.
Axolotl Dorsal and Tail Skin Exhibit Significant Morphological Heterogeneity During Development
For decades, wound repair has been hypothesized to recapitulate developmental programs, implying that the intrinsic developmental trajectory of tissues significantly influences their regenerative capacity. Prior findings have unequivocally demonstrated that, following injury, dorsal skin undergoes fibrotic scarring, whereas tail skin executes rapid, near-scarless repair. To uncover the developmental roots of this anatomical heterogeneity in regenerative competence, we systematically tracked spatiotemporal divergence in morphogenetic programs between axolotl dorsal and caudal skin from embryonic stages (30 days post-fertilization, dpf) through sexual maturation (2 years post-fertilization, ypf).
Histological analysis revealed that during early developmental stages (30–40 dpf), both dorsal and tail skin were composed of a single-layered embryonic ectodermal epithelium (Fig. 6a, b, h, i). By 48 dpf, early Leydig cells (green arrows) and a nascent basement membrane (BM; green arrows) emerged in both regions, consistent with previously reported features 28 , with no significant differences observed. Marked regional divergence in developmental tempo and architecture became apparent from 4 months post-fertilization (mpf). At 4 mpf, dorsal skin had established a canonical epidermal–dermal bilayer, characterized by a thickened epidermis with a dense Leydig cell population and a dermis differentiated into a gland-rich stratum spongiosum and acellular stratum compactum (Fig. 6k). In stark contrast, tail skin remained at a low differentiation state with indistinct stratification, exhibiting a 4-month delay in dermal layer formation relative to dorsal counterparts (Fig. 6d). Dermal stratification in tail skin commenced at 8 mpf, coinciding with the gradual integration of glandular structures into the dermis, as seen in other highly regenerative tissues and near-complete glandular differentiation was achieved by 11 mpf (Fig. 6f). Notably, the overall architecture of 11 mpf tail skin mirrored that of 4 mpf dorsal skin, highlighting a substantial developmental lag in tail skin. By sexual maturity (11 mpf), both dorsal and tail skin had developed a tri-layered structure—comprising epidermis, dermis, and hypodermis—with histological features analogous to mammalian skin (Fig. 6o, p). However, regional heterogeneities persisted: dorsal skin exhibited greater thickness (Fig. 6C), whereas tail skin harbored a significantly higher density of dermal glands (Fig. 6B). In fully mature adults (24 mpf), both skin types displayed epidermal thinning and enhanced dermal collagen deposition, indicative of continuous adaptive remodeling throughout ontogeny.

Developmental progression and regional heterogeneity in axolotl dorsal and tail skin. (A) Masson’s trichrome staining of tail (a–g) and dorsal (h–n) skin at multiple developmental stages: 30, 40, and 48 days post-fertilization (dpf); 4, 8, 11, and 24 months post-fertilization (mpf). (a, h) At 30 dpf and (b, i) at 40 dpf, both skin regions consisted of a single-layer ectodermal epithelium (green dashed lines indicate epidermal boundary). (c, j) At 48 dpf, early Leydig cells and a nascent basement membrane (BM) appeared (green arrows). (d, k) At 4 mpf, dorsal skin exhibited complete stratification with abundant Leydig cells and dermal layers, including the stratum spongiosum (ss) and stratum compactum (sc); tail skin remained poorly stratified. (e, l) At 8 mpf, dermal stratification initiated in tail skin. (f, m) At 11 mpf, both dorsal and tail skin exhibited mature tri-layered organization with developed dermal glands (green arrows). (g, n) At 24 mpf, both skin regions showed epidermal thinning and increased dermal collagen deposition. (o, p) High-magnification views of dorsal and tail skin at 11 mpf illustrate detailed layered structures, including stratum corneum (SC), Leydig cells, BM, ss, sc, hypodermis, and underlying muscle. (B) Quantification of dermal gland density (glands/cm²) in dorsal and tail skin at 11 mpf. Tail skin exhibited significantly higher gland density. (C) Quantification of total skin thickness (μm) in dorsal and tail skin at 11 mpf. Dorsal skin was significantly thicker than tail skin. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. ****P < 0.0001; n = 5. Scale bars, 20 μm for (a, b, h, i); 200 μm for all other panels.
In summary, axolotl skin exhibits pronounced regional heterogeneity in developmental trajectories: dorsal skin undergoes early epidermal and dermal stratification, potentially enabling the rapid establishment of an effective physical barrier, whereas tail skin maintains a hypodifferentiated state for an extended period, a trait that may endow it with enhanced tissue plasticity and regenerative potential.
Transcriptomic Analysis Reveals Significant Region-Specific Gene Expression Differences Between Dorsal and Tail Skin in Mature Axolotls
Having identified substantial developmental differences between the dorsal and tail skin of axolotls, we employed transcriptome sequencing to characterize their molecular profiles, investigating whether these differences are inherently programmed and might influence injury responses. Transcriptomic analyses were performed on dorsal and tail skin from sexually mature axolotls (11 mpf), with the aim of identifying intrinsic molecular signatures underlying their distinct regenerative capacities.
Transcriptome sequencing of dorsal and tail skin from sexually mature axolotls was performed to identify intrinsic molecular signatures that may underlie their disparate regenerative capacities. Differential expression analysis identified 110 differentially expressed genes (DEGs) between dorsal and tail skin, with 21 upregulated in the dorsal skin and 89 in the tail skin, demonstrating significant transcriptional divergence at sexual maturity (Fig. 7a).

Transcriptomic profiling reveals distinct gene expression signatures between dorsal and tail skin in mature axolotls. (a) Volcano plot illustrating differentially expressed genes (DEGs) between dorsal and tail skin. Genes were classified as significantly differentially expressed using thresholds of adjusted Padj < 0.05 and|log2(fold change)| ≥ 1. (b) GO enrichment analysis of the identified DEGs, highlighting significantly enriched biological processes and molecular functions (FDR < 0.05) in dorsal versus tail skin. BP significantly enriched in DEGs include regulation of cell morphogenesis, ATPase activity modulation, and negative regulation of T-cell activation. (c) The top 10 significantly enriched pathways from the KEGG database among the DEGs (P < 0.05), illustrating key signaling and metabolic pathways that are differentially regulated between dorsal and tail skin, such as motor proteins, focal adhesion, and leukocyte transendothelial migration pathways. (d) Heatmap visualizing the expression patterns of a selection of genes associated with significantly enriched GO terms and KEGG pathways, providing an overview of the distinct molecular signatures characterizing dorsal and tail skin. (e) Quantitative real-time PCR (qPCR) validation of representative DEGs (Il20rb, Pla2g4c, Tagln, and TnN). Expression trends were consistent with RNA-seq results, confirming upregulation of TnN in tail skin and elevated expression of Il20rb, Tagln, and Pla2g4c in dorsal skin. Data are presented as mean ± SEM. Statistical significance for qPCR was assessed using unpaired two-tailed Student’s t-test. *P < 0.05, **P < 0.01, ***P < 0.001; n = 3.
Gene Ontology (GO) enrichment analysis revealed that these DEGs are significantly associated with regulation of cell morphogenesis, ATPase activity modulation, and negative regulation of T-cell activation (Fig. 7b), highlighting intrinsic differences in morphogenetic, metabolic, and immune-regulatory pathways. Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis indicated significant enrichment in motor proteins, focal adhesion, and leukocyte transendothelial migration pathways (Fig. 7c), underscoring divergent molecular programs for cell motility and immune surveillance.
Integration of GO and KEGG data identified 16 candidate regulators (see Supplementary Table S1 for gene functions and references), including Pla2g4c, a critical enzyme in inflammatory cascades, and Tagln, a muscle-fiber structural protein—both implicated in fibrotic and immune responses. In tail skin, TnN (Tenascin-N) and Myl2 were specifically upregulated; TnN is an ECM glycoprotein that modulates cell adhesion and matrix remodeling 29 , engaging ECM–receptor interactions and the PI3K-Akt cascade (Fig. 7d) 30 .
qPCR validation confirmed that Il20rb, Pla2g4c, and Tagln are significantly elevated in dorsal skin, while TnN shows marked upregulation in tail skin (Fig. 7e). Il20rb has been shown to drive keratinocyte hyperproliferation via STAT3 activation, consistent with the thicker dorsal epidermis observed 31 . Elevated TnN may modulate cell-matrix adhesion 32 and influence cell migration and ECM dynamics 33 , thereby contributing to the accelerated regenerative response in tail skin.
Collectively, our study uncovers significant differential gene expression profiles between uninjured dorsal and caudal skin in axolotls. These DEGs are functionally enriched in epidermal barrier reconstitution, wound resolution, cell motility, and immune modulation. This suggests that dorsal and caudal skin may exhibit significant differences in healing speed following injury. Furthermore, these findings provide crucial molecular insights for a deeper understanding of the region-specific regeneration mechanisms of axolotl skin.
PI3K-AKT Signaling Pathway Activation Is Significantly Higher in Tail Skin Than in Dorsal Skin During the Early Stages of Wound Healing
Transcriptome analysis (Fig. 7 and Supplemental Table S1) revealed elevated expression of the ECM glycoprotein TnN in tail skin under homeostatic conditions. Given the involvement of TnN in signaling pathways relevant to the skin dermis, ECM-receptor interactions, and the PI3K-Akt signaling cascade 30 , and considering the critical role of the PI3K-Akt pathway in wound healing-related processes such as cell survival, proliferation, inflammation, and fibrosis 34 , we hypothesized that this pathway is differentially activated during the early phases of wound healing in dorsal and tail skin. This hypothesis was subsequently investigated using Western blot analysis (Fig. 8a).

Western blot showing differential activation of the PI3K-Akt pathway in dorsal and tail skin wounds. (a) Representative Western blot images of phosphorylated Akt (p-Akt), total Akt, and β-actin from dorsal and tail skin wound tissues collected at 0, 3, and 7 dpi. (b) Quantification of p-Akt/total Akt ratios at each time point, normalized to β-actin and measured using ImageJ. Tail wounds exhibited significantly higher PI3K-AKT pathway activation compared to dorsal wounds at all time points. Data are presented as mean ± SEM. Statistical significance was determined using unpaired two-tailed Student’s t-test. *P < 0.05, **P < 0.01, ***P < 0.001; n = 3.
Western blot analysis demonstrated a significantly higher p-Akt/Total-Akt ratio in the wound area of tail skin compared to the dorsal skin wound area at 0, 3, and 7 dpi (Fig. 8b). This indicates a markedly enhanced activation of the PI3K-Akt pathway in tail skin wound sites relative to dorsal skin during the early stages of wound repair. This earlier and more pronounced activation may contribute to the accelerated initiation of regeneration observed in tail skin. Activation of the Akt signaling pathway promotes cell survival, proliferation, and modulation of early inflammatory responses, which are essential for the prompt commencement of tissue repair.
In summary, the activation kinetics of the PI3K-Akt pathway during the initial stages of wound healing exhibit significant regional disparities between dorsal and tail skin. These findings potentially offer critical early molecular mechanistic insights into the divergent wound healing outcomes characteristic of these two anatomical locations.
Discussion
In this study, we systematically established full-thickness skin wound models in axolotls and revealed previously uncharacterized regional heterogeneity in regenerative capacity between dorsal and tail skin. Tail skin exhibited accelerated re-epithelialization, enhanced epidermal differentiation, and near-complete dermal architectural restoration, collectively reflecting a highly efficient and scarless regenerative response, as supported by both histological and molecular evidence. Conversely, dorsal skin healing follows a fibrotic trajectory, characterized by sustained myofibroblast accumulation and excessive collagen deposition, ultimately culminating in visible scar formation during the chronic remodeling phase. Together, these observations suggest that tail skin possesses intrinsic pro-regenerative properties enabling near-perfect repair, while dorsal skin is predisposed to fibrosis. These findings highlight the anatomical specificity and context-dependent nature of skin regeneration in axolotls.
Developmental Regulation of Regional Heterogeneity in Axolotl Skin Regeneration
It has long been recognized that wound repair partially recapitulates developmental processes35,36, suggesting that a tissue’s initial developmental state may significantly influence its regenerative capacity. Our developmental analysis revealed that axolotl tail skin persists in a hypodifferentiated state during development, whereas dorsal skin undergoes more rapid differentiation, thereby forming distinct epidermal-dermal layers and completing the development of appendages such as skin glands at an earlier stage.
This sustained hypodifferentiation in tail skin may serve as a mechanism for preserving cellular plasticity and regenerative potential, a phenomenon observed in other highly regenerative species. For instance, studies on reindeer have shown that antler velvet skin, capable of scarless healing, contains fibroblasts resembling fetal fibroblasts, which contrast with the pro-fibrotic fibroblasts found in adult dorsal skin 37 . The developmental delay observed in axolotl tail skin may confer enhanced regenerative abilities by retaining a higher abundance or activity of stem/progenitor cells into adulthood. This is consistent with findings that stem cell abundance and activity are typically higher during early development and decline with tissue maturation 38 .
In essence, the “developmental delay” of tail skin likely allows it to retain relatively high stem/progenitor cell numbers or activity in adulthood, thereby facilitating more effective initiation of cell proliferation, migration, and differentiation after injury—processes that promoting faster, more complete regeneration while minimizing fibrogenic outcomes. These developmental disparities underscore the role of intrinsic tissue maturity in shaping regional regenerative responses, potentially accounting for the observed differences in regenerative outcomes.
Molecular Mechanisms Underlying Regional Differences in Axolotl Skin Regeneration
Transcriptomic profiling of uninjured axolotl skin revealed distinct gene expression signatures between dorsal and tail regions, with tail skin enriched for genes governing cell migration, differentiation, and ECM remodeling—including TnN—whereas dorsal skin preferentially expressed inflammatory and fibrogenic genes such as Pla2g4c and Tagln. As an ECM glycoprotein, TnN is highly expressed in tissues requiring dynamic matrix turnover 29 and plays a critical role in orchestrating ECM-receptor interactions critical for tissue remodeling 30 . This molecular feature likely endows tail skin with enhanced cellular motility and matrix plasticity, thereby facilitating accelerated re-epithelialization following injury—a process central to scarless regeneration. Conversely, Tagln—a marker of fibrotic processes—exhibits conserved roles in promoting collagen deposition and excessive myofibroblast activation, as observed in human keloids and other pathological scars39,40. The upregulation of Tagln in dorsal skin suggests evolutionary conservation of fibrogenic pathways between axolotls and mammals, whereby excessive ECM cross-linking and inflammatory signaling may drive scar formation. These divergent molecular profiles provide a mechanistic framework for the observed differences in regenerative outcomes. Collectively, the differential expression of TnN, Pla2g4c, and Tagln forms a molecular basis for regional regenerative disparities, whereby tail skin leverages ECM remodeling capacity to facilitate tissue restoration, while dorsal skin activates conserved fibrogenic programs leading to scarring. These findings bridge developmental heterochrony and molecular signaling, underscoring the interplay between tissue ontogeny and regenerative mechanisms.
A critical finding of this study is the marked regional specificity in injury-induced signaling dynamics, with tail skin showing higher TnN expression and more robust early activation of the PI3K-Akt pathway (0–7 dpi). Early activation of this pathway promotes keratinocyte and dermal cell proliferation/migration, facilitating wound bed establishment and granulation tissue formation, which suggests that the PI3K-Akt signaling cascade may play a pivotal pro-repair role in the early regenerative response of tail skin.
As shown in studies on porcine skin by Zou et al. 30 , TnN—an ECM glycoprotein—mediates ECM-receptor interactions in the dermis and activates the PI3K-Akt signaling cascade. This suggesting that high TnN expression in axolotl tail skin may similarly regulate this pathway during regenerative repair. This mechanistic homology is further supported by zebrafish heart regeneration, where the homologous protein Tenascin-C engages integrin αvβ3 to trigger focal adhesion kinase (FAK)-dependent PI3K-Akt activation, thereby promoting cardiomyocyte migration 41 . An analogous mechanism likely operates in axolotl tail skin: TnN binding to integrin receptors could phosphorylate FAK, which in turn activates PI3K via p85 subunit phosphorylation—an established pathway in cell signaling. This signaling cascade may enhance keratinocyte and dermal cell proliferation/migration, suppress apoptosis, and modulate inflammatory responses, thereby inhibiting excessive ECM deposition and fibrosis 34 . In contrast, dorsal skin exhibits suboptimal PI3K-Akt activation, coinciding with elevated expression of the fibrogenic marker Tagln and reduced ECM remodeling capacity. The resulting deficiency in early regenerative signaling permits uncontrolled collagen deposition and myofibroblast accumulation, driving scar formation. Thus, we propose that the high levels of TnN in tail skin—compared with suboptimal PI3K-Akt activation in dorsal skin—may facilitate early upregulation of this signaling pathway, thereby promoting rapid re-epithelialization, efficient epidermal differentiation, and complete dermal architecture reconstruction after injury to achieve scarless skin regeneration.
Regional Specificity of Regeneration: Insights From Axolotls and Other Model Organisms
This study clearly demonstrates striking regional specificity in regenerative capacity—even in axolotls, renowned for their robust regenerative abilities—where dorsal and tail skin exhibit distinct repair outcomes. These findings underscore the critical role of local anatomical location and microenvironmental heterogeneity in shaping regenerative responses, a phenomenon documented across diverse evolutionary lineages, including mammals. For example, similar regional disparities in wound healing have been reported in porcine burn models 42 , indicating that such specificity is not exclusive to axolotls.
Notably, the active proliferation of tail epithelial cells and dynamic expression of KRT4 observed in our study align closely with Ohashi et al.’s 15 finding that keratinocytes play a critical role in driving dermal matrix remodeling in axolotls—providing cellular-level evidence for the superior regenerative capacity of tail skin. This phenomenon is not unique to axolotls: in mammals, although cardiac regenerative capacity is limited, specific regions like the cardiac apex show modest regenerative potential after injury 43 . Even in highly regenerative planarians, regeneration of different body fragments is strictly regulated by positional information 44 . These cross-species observations suggest that intrinsic positional information, together with local microenvironmental factors such as vascular distribution, innervation, and immune cell composition, is pivotal in determining regenerative capacity and patterns. Interestingly, macroscopic observation revealed more visible vasculature in tail skin compared to dorsal skin. Given that vascular supply is known to facilitate regeneration, this anatomical difference may partially contribute to the superior healing observed in tail skin.
Thus, the dorsal-tail regenerative differences observed in axolotl skin provide further evidence for the universality of regional specificity in regenerative biology, transcending taxonomic boundaries.
Significance, Limitations, and Future Directions
This study is the first to systematically compare the regenerative capacity of adult axolotl dorsal versus tail skin, revealing significant differences in their wound-healing outcomes.
Unlike prior work focusing on juvenile stages or limb amputation sites in axolotls 15 , our investigation fills a critical gap by directly comparing skin regeneration across distinct anatomical regions within the same organism. Axolotl tail skin thus serves as a unique in vivo model directly juxtaposed with the fibrosis-prone dorsal region, offering an ideal platform to dissect endogenous pro-regenerative and anti-fibrotic mechanisms with potential relevance to human pathological scarring and organ fibrosis. Moreover, the highly efficient, scarless regeneration observed in tail skin suggests the feasibility of isolating regenerative cell populations for autologous transplantation in future skin-repair therapies. Finally, targeting the regulation of TnN expression or activity and the precise temporal activation of the PI3K-Akt pathway during tail regeneration holds promise for novel anti-fibrotic and pro-scarless therapeutic strategies.
Future studies will employ gene-editing approaches to dissect the roles of TnN and the PI3K-Akt pathway in regional regeneration, and will leverage single-cell and spatial transcriptomics to resolve the cellular and molecular landscapes driving dorsal-tail heterogeneity.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251348730 – Supplemental material for Comparative analysis of dorsal and tail skin reveals region-dependent heterogeneity in axolotl skin regeneration
Supplemental material, sj-docx-1-cll-10.1177_09636897251348730 for Comparative analysis of dorsal and tail skin reveals region-dependent heterogeneity in axolotl skin regeneration by Lieke Yang, Qian Chen, Jinman Li, Yan Hu, Yaoxin Liu, Binbin Lu, Shuaibin Pei, Tingyi Huang, Yanmei Liu and Ji-Feng Fei in Cell Transplantation
Footnotes
Ethical Considerations
Ethical approval to report this case was obtained from the Ethics Review Committee of Guangdong Provincial People’s Hospital (approval no: KY Q 2022 395 03).
Author Contributions
J-FF and YL conceived and planned the experiments. LY, QC, BL, SP, and YH performed the experiments. KY, HT, and JL analyzed the data and prepared the original figures. J-FF, LY, YL, BL, and QC wrote the manuscript. All authors revised the manuscript and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant nos. 31970782; 92268114; and 32070819), the National Key R&D Program of China (grant nos. 2021YFA0805000 and 2019YFE0106700), the High-level Hospital Construction Project of Guangdong Provincial People’s Hospital (grant no. DFJHBF202103 1and KJ012021012).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the animal welfare legislation in China. All animal experiments were approved by the Biomedical Research Ethics Committee of Guangdong Provincial People’s Hospital and adhered to the institution’s approved protocols.
Statement of Informed Consent
This article does not involve human subjects, and informed consent is therefore not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
