Abstract
Selective depletion of TCRαβ+ and CD45RA+ subsets of apheresed hematopoietic progenitor cells, HPC(A), enables haploidentical hematopoietic stem cell transplant (haplo-HSCT) by circumventing risks of graft-versus-host disease. Here, we analyze our institution’s large series of ex vivo T-cell depletion processes to review procedure performance and explore factors that affect depletion efficiency and graft composition. Over 6 years, 91 haploidentical donors underwent peripheral blood CD34+ stem cell mobilization with granulocyte-colony stimulating factor, with 12 (13%) receiving additional pre-emptive plerixafor. HPC(A) was split into two fractions for TCRαβ and CD45RA depletion with the CliniMACS PLUS device. TCRαβ depletion resulted in a median 4.3 (interquartile range, 4.1–4.5) log reduction, with CD34 recovery at 98% (94%–103%) and TCRγδ+ cell recovery at 89% (74%–98%). CD45RA depletion resulted in a median 4.8 (4.3–5.2) log reduction, with CD3+/CD45RO+ cell recovery at 41% (34%–47%) and CD34 recovery at 58% (51%–68%). TCRαβ depletion efficiency was maintained even when total nucleated cell counts exceeded the maximal specified number, provided the target fraction was within capacity of the depletion kit. Platelet contamination did not affect depletion efficacy or CD34 recovery. Age increases the proportion of CD45RO+ memory cells and TCRαβ subset in HPC(A), while plerixafor increases the latter. Although statistically significant correlation exists between pre-depletion cell composition and depletion performance for some cell subsets, the post-depletion product still met pre-specified threshold without being affected to a clinically relevant extent, over a wide range of input cell numbers. Such robustness of the depletion systems is critical for successful performance of haplo-HSCT.
Figure partially created in BioRender, https://BioRender.com/3n12d59
Keywords
Introduction
For patients with high-risk hematological malignancies who lack a suitable human leukocyte antigen (HLA)-matched donor, haploidentical hematopoietic stem cell transplantation (haplo-HSCT) presents itself as a promising treatment option. However, one major challenge to circumvent in the HLA-mismatched setting is the inherent risk of graft-versus-host disease (GvHD). One of the well-established strategies for GvHD prophylaxis in haplo-HSCT is that of ex vivo T-cell depletion1–3, which involves graft manipulation to remove T-cell subsets that mediate GvHD.
Since alloreactivity is restricted to TCRαβ+ T cells 4 , TCRαβ depletion allows for preservation of TCRγδ+ T cells and NK cells which not only mediate graft-versus-leukemia effect, but also possess anti-infective properties5,6. Likewise, the benefits of CD45RA+ T-cell depletion have gained increasing recognition as it removes these naïve T cells that can cause GvHD, while preserving CD45RO+ memory T cells that promote immune reconstitution 7 . This approach is derived from animal studies that have demonstrated the lack of ability of memory T cells from unprimed donors in inducing GvHD 8 .
In our institution, we combine the merits of both processes and selectively deplete TCRαβ+ and CD45RA+ T-cell subsets of apheresed hematopoietic progenitor cells, HPC(A), to facilitate our haplo-HSCT protocol using a TCRαβ-depleted graft with concurrent CD45RA-depleted T-cell add-back. In this report, we examine our 6 years of experience with these two selective ex vivo T-cell depletion processes to review depletion efficiency and cell recovery rates. We take this opportunity further to explore factors, including donor characteristics and mobilization method, that may influence leukapheresis product composition and in turn affect the performance of depletion processes.
Materials and methods
Fig. 1 provides a schematic overview of the methodology of this study.

Schematic overview of study methodology.
Data source
Donors’ data were derived from our institution’s transplant database and electronic records with approval from the SingHealth Centralised Institutional Review Board (ECOS 2015-2419). All donors have provided written informed consent for use of their clinical and laboratory results.
Leukapheresis
Haploidentical donors received 5 days of subcutaneous granulocyte colony-stimulating factor (G-CSF) administered daily at 10–16 µg/kg of body weight (rounding off to nearest vial size of 600, 900, or 1200 µg) to mobilize CD34+ hematopoietic stem cells. The target CD34 yield is set at 4 × 106 CD34/kg. Donors with suboptimal day 4 CD34 counts received pre-emptive plerixafor (Mozobil®; Sanofi, Ridgefield, NJ) on the evening of day 4 before planned harvesting on day 5. Leukapheresis procedures were performed with a continuous-flow blood cell separator (Spectra Optia®; Terumo BCT, Lakewood, CO) using a continuous mononuclear cells program following manufacturer’s instructions. Standard protocol involved processing three times of donors’ total blood volume with anticoagulant citrate dextrose solution over 5 to 6 h.
T-cell depletion procedure
HPC(A) was divided into two fractions after quantification of CD34+ cell counts. The priority was to allocate a fraction of the product with at least 4 × 106 CD34/kg for TCRαβ depletion, while the remaining product undergoes CD45RA depletion for concurrent memory lymphocyte add-back and future donor lymphocyte infusion (DLI). Both fractions were subjected to immunomagnetic depletion following instructions of the depletion kits and operation manual of CliniMACS PLUS device (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). Each TCRαβ and CD45RA depletion procedure was accomplished with a single set of depletion kit and CliniMACS separation column. In brief, for TCRαβ depletion, the two-step process involved first incubating with TCRαβ antibody conjugated to Biotin, followed by washing to remove excess Biotin conjugates. The Biotin-labeled cells were then secondarily labeled with anti-Biotin conjugated microbeads and loaded on to the semi-automated CliniMACS PLUS device to collect TCRαβ-negative fraction in the eluate. For CD45RA depletion, labeling was performed directly with anti-CD45RA conjugated beads. Program DEPLETION 3.1 was used for both depletion processes.
Immunophenotyping
On days 4 and 5 of mobilization, donor peripheral blood CD34+ counts were determined by flow cytometry using BD FACSCanto flow cytometer and BD FACSCanto Clinical Software (Becton Dickinson, San Jose, CA), following the International Society for Hematotherapy and Graft Engineering (ISHAGE) guidelines 9 for CD34 enumeration. Immunophenotypic analysis was performed on aliquots of apheresis products before and after labeling and depletion. CD34+, CD45RO+, CD45RA+, TCRαβ+, and TCRγδ+ cells were quantified in viable leukocytes identified by fluorochrome-conjugated monoclonal antibodies and 7-aminoactinomycin D staining. The list of monoclonal antibodies used is detailed in Supplemental Table 1. A minimum of 5 × 105 events was analyzed for each run.
Statistical analysis
Results are expressed as median values with ranges comprising minimum and maximum values, or the use of interquartile ranges (IQR) wherever appropriate. Mann–Whitney U test was used to establish differences between sets of data. Pearson correlation coefficient was applied to determine the strength of correlation between sets of paired data. P values of ≤0.05 denoted statistical significance. All figures were created with GraphPad Prism version 10.
Results
Donor and product characteristics
A total of 91 haploidentical donors underwent 101 apheresis sessions for stem cell collection from February 2017 to January 2023. Eighty-one donors (89%) underwent a single leukapheresis session, whereas the remaining required a second session. A total of 87 TCRαβ depletion processes and 90 CD45RA depletion processes were performed on these leukapheresis products. Median donor age was 33 years (range, 10–69), including 56 males and 35 females.
Twelve donors had median peripheral blood CD34+ counts of 19 × 106/l (IQR 12–28 × 106/l) on day 4 of G-CSF and received one dose of plerixafor due to concerns of inadequate collection. This resulted in comparable peripheral blood CD34+ counts on the day of apheresis for donors who received plerixafor and those who did not (Fig. 2).

Box and whisker plots of peripheral blood CD34+ hematopoietic stem cell counts in donors on day 4 of mobilization and day of apheresis (day 5). Donors are divided into those who received plerixafor and those who did not. Lower and upper fences represent the 25th and 75th percentiles, with the median in between. Whiskers extend to 1.5 times of the interquartile range (Tukey method). Dots represent outlier values.
The harvesting process yielded a median total nucleated cell (TNC) count of 57.1 × 109 (IQR 43.8–73.9 × 109) in the product. After quantification of CD34+ cell count, the main bulk of HPC(A), comprising a median of 77% (IQR 72–87%) of product volume, was allocated as stem cell graft after TCRαβ depletion and the remaining was subjected to CD45RA depletion for use as memory T-cell add-back.
Depletion efficiency
Depletion efficiency is defined as log10 of the ratio between residual viable cell counts in the post-depletion product and original cell counts of interest in HPC(A) prior to depletion.
For TCRαβ depletion, the median TCRαβ cell count in HPC(A) was 12.2 × 109 (IQR 9.9–17.7 × 109), which was reduced to 0.6 × 106 (IQR 0.4–1.1 × 106) after depletion, giving a median log depletion of 4.3 (IQR 4.1–4.5) (Fig. 3). This translated to a median of 0.018 × 106 TCRαβ/kg recipient weight (range, 0.002–0.147 × 106) in the infused product, of which 2 cases marginally exceeded the pre-specified threshold of 0.1 × 106 TCRαβ/kg recipient weight. Both cases involved pediatric recipients with significant weight disparity from their respective parent donors, accounting for a relatively higher TCRαβ proportion.

Scatter dot plot illustrating the efficiency of TCRαβ+ and CD45RA+ T-cell depletion processes. Depletion efficiency is represented as log depletion, with each dot representing a single depletion process, and the horizontal bars indicating median values.
For CD45RA depletion, the median CD45RA cell count was 2.6 × 109 (IQR 1.3–3.3 × 109) and 0.03 × 106 (IQR 0.01–0.08 × 106) pre- and post-depletion respectively, giving a median log depletion of 4.8 (IQR 4.3–5.2) (Fig. 3). For the protocol-specified dose of 1 × 106 CD3+CD45RA-/kg recipient weight for lymphocyte add-back, the median residual CD45RA count was 0.07 × 103 CD45RA/kg (range, 0–3.9 × 103), with no cases exceeding the threshold of 0.05 × 106 CD45RA/kg recipient weight.
Cell recovery
To evaluate cell recovery, a comparison was made between viable CD34+, TCRγδ+, and CD3+/CD45RO+ cell counts in the pre-depletion apheresed product versus respective cell counts after depletion.
Table 1 shows that for TCRαβ depletion, recovery of TCRγδ+ cells was high at 89%, with similarly excellent CD34+ cell recovery rates of 98% (Fig. 4a.i). The minimal loss of CD34 subset enabled infusion of a good stem cell dose at a median of 5.7 × 106/kg recipient weight (IQR 2.1 – 11.7 × 106/kg) for the 57 adult patients who received the TCRαβ-depleted stem cell product.
Recovery rates of cell subsets of interest with TCRαβ and CD45RA depletion processes.

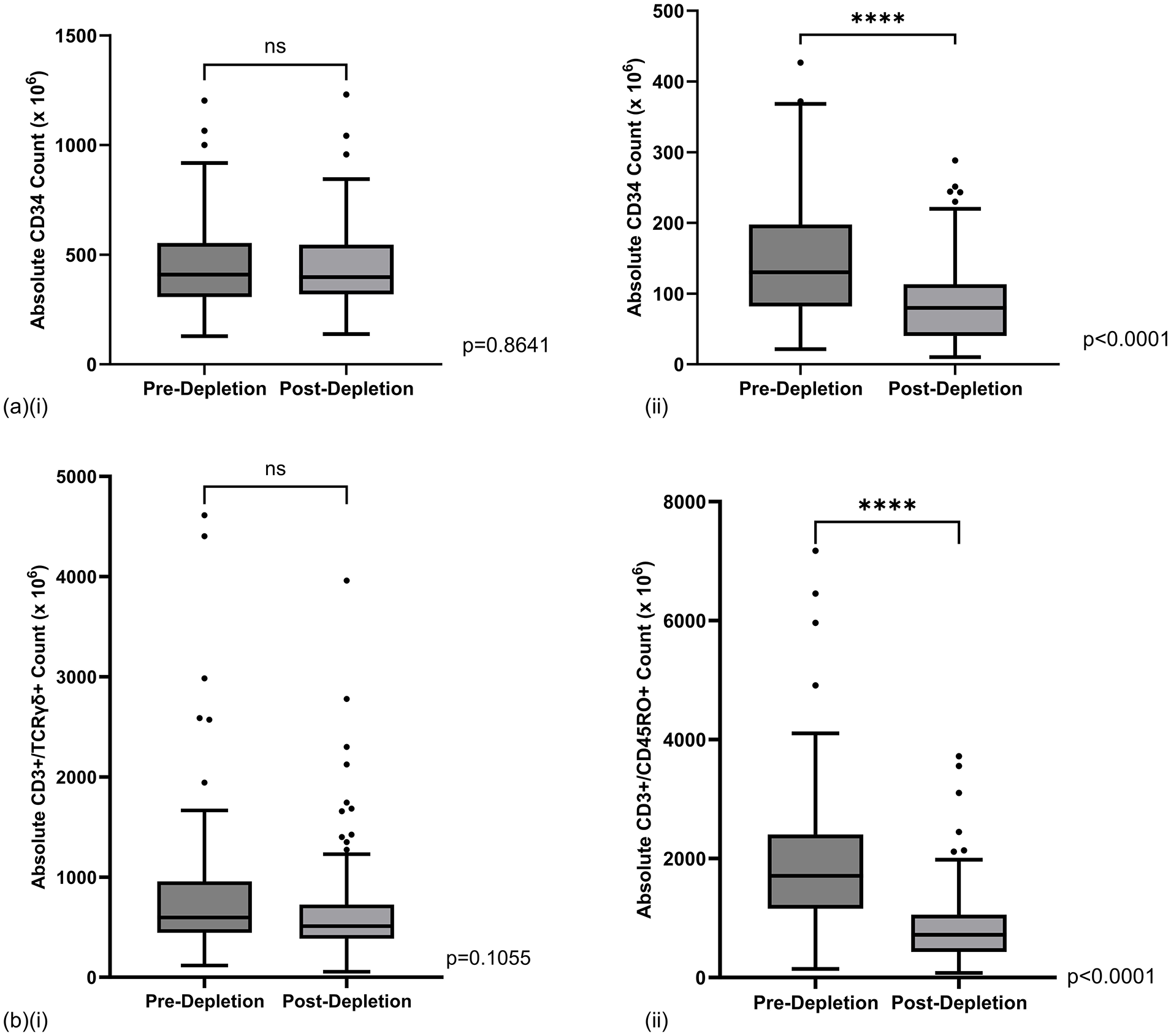
Box and whisker plots of (a) absolute CD34+ counts and (b) respective CD3+ target cell counts in apheresed products before and after (i) TCRαβ depletion and (ii) CD45RA depletion. Lower and upper fences represent the 25th and 75th percentiles, with the median in between. Whiskers extend to 1.5 times of the interquartile range (Tukey method). Dots represent outlier values. ns: not statistically significant, ****P value ≤ 0.0001 (Mann–Whitney U test)
On the other hand, CD45RA depletion resulted in a significantly greater loss with a lower median recovery rate of 41% (IQR 34%–47%) for the CD3+/CD45RO+ subset (Table 1). Similarly, this depletion process also lost a significant proportion of CD34+ cells, with a median recovery rate of 58% (IQR 51%–68%) (Fig. 4a.ii), which, however, did not have any clinical impact as the main bulk of CD34+ stem cells for infusion was contributed by the larger fraction allocated for TCRαβ depletion.
In both depletion processes, cell viability was preserved, with median post-depletion CD3+ T-cell viability of 99.0% (IQR 98.5%–99.4%) and 99.9% (IQR 99.9%–99.9%), respectively, for the TCRαβ and CD45RA depletion processes.
Factors that affect performance of depletion processes
TNC number
We show that there was a weak negative correlation between pre-depletion TNC count over a wide range (21.6 to 90.8 × 109 cells) and TCRαβ log depletion (Fig. 5a), but TNC count did not affect CD34+ and TCRγδ+ cell recovery rates.

Effect of total nucleated cell (TNC) number of apheresed product on the performance of depletion processes. (a) Scatterplot of the relationship between pre-depletion TNC count and TCRαβ log depletion. r: Pearson’s correlation coefficient (b) Box and whisker plots of (i) TCRαβ log depletion, (ii) TCRγδ+ cell recovery rates, and (iii) CD34+ cell recovery rates, stratified by whether maximum TNC count of 60 × 109 was exceeded. Lower and upper fences represent the 25th and 75th percentiles, with the median in between. Whiskers extend to 1.5 times of the interquartile range (Tukey method). Dots represent outlier values. ns: not statistically significant, *P ≤ 0.05 (Mann–Whitney U test)
By manufacturer’s protocol, one vial of CliniMACS TCRαβ/Biotin is sufficient for labeling up to 24 × 109 TCRαβ+ cells out of a maximum TNC count of 60 × 109 cells. We encountered 19 cases which exceeded the maximum stipulated TNC count (median of 71 × 109, range 60.8 to 90.8 × 109), without exceeding the upper limit of 24 × 109 in the target TCRαβ+ fraction. This resulted in a significantly lower depletion efficiency in comparison to the group that did not exceed the 60 × 109 TNC limit, but remained highly efficient with a median of 4.1 (IQR 3.9–4.3) log reduction (Fig. 5b.i). As a result, none of the 19 cases exceeded the threshold of 1 × 105 TCRαβ T cells/kg of recipient weight post-depletion. Furthermore, TCRγδ+ T cell and CD34 recovery rates were not significantly compromised (Fig. 5b.ii and 5b.iii).
Pre-depletion cell subset composition
For TCRαβ depletion, there was no association between pre-depletion CD34+ counts (range, 128.5–1065.5 ×109) and CD34 recovery rates (Supplemental Fig. 1a.i). However, for CD45RA depletion, a higher pre-depletion CD34+ count (range, 21.6 to 426.8 ×109) was found to be moderately associated with higher CD34 recovery (Supplemental Fig. 1b.i).
We observed a weak negative correlation between pre-depletion TCRαβ:TCRγδ ratio of HPC(A) (range, 4.6–92.5) and depletion efficiency (Supplemental Fig. 1a.ii). On the other hand, a weak positive correlation was demonstrated between pre-depletion CD45RA: RO ratio of HPC(A) (range, 0.45–4.33) and depletion efficiency (Supplemental Fig. 1b.ii). However, such effect on depletion efficiency did not result in products exceeding allowed thresholds, over a wide range of pre-depletion cell composition.
Platelet contamination
The median platelet concentration of HPC(A) was 1396 × 109/l (IQR 1054–1705 × 109/l) with an absolute count of 379 × 109 (IQR 288–501 × 109). The median residual platelet count was 37 × 109 (IQR 28–53 × 109) and 26 × 109 (IQR 19–42 × 109), respectively, after TCRαβ and CD45RA depletion processes. Pre-depletion platelet concentration (range, 516–4034 × 109/l) did not affect depletion efficiency for both depletion processes (Supplemental Fig. 2a), but we found a weak negative correlation with TCRγδ+ cell recovery rates (Supplemental Fig. 2b). CD34 recovery was not affected by pre-depletion platelet count. No association was demonstrated between pre-depletion platelet concentration of HPC(A) and CD45RO recovery.
Factors that affect composition of HPC(A)
As we have demonstrated above that cell composition of the mobilized leukapheresis product can impact depletion performance, we went one step upstream to explore factors that may influence the composition of HPC(A).
Donor age
We observed a moderate positive correlation between age and TCRαβ:TCRγδ composition of HPC(A) (Fig. 6a.i), which corroborates with existing knowledge on age-related changes of the immune system10,11.

Effect of donor age and the use of plerixafor as mobilization agent on the composition of apheresed products. (a) Scatterplots of the relationship between donor age and HPC(A) composition in terms of (i) TCRαβ:TCRγδ ratio and (ii) CD45RA: CD45RO ratio. r: Pearson’s correlation coefficient (b) Box and whisker plots of (i) total nucleated cell count of HPC(A), (ii) TCRαβ:TCRγδ ratio of HPC(A), and (iii) post-depletion TCRγδ+ counts, stratified by whether donors received plerixafor. Lower and upper fences represent the 25th and 75th percentiles, with the median in between. Whiskers extend to 1.5 times of the interquartile range (Tukey method). Dots represent outlier values. ns: not statistically significant, *P ≤ 0.05; **P ≤ 0.01 (Mann–Whitney U test).
We also demonstrated a negative correlation between age and CD45RA:CD45RO composition of HPC(A) (Fig. 6a.ii). This supports the finding that the expression of CD45 isoforms shifts from naïve T-cell phenotype to memory T-cell phenotype with increasing age 12 .
Plerixafor use
HPC(A) from the 12 plerixafor-mobilized donors had a significantly higher median TNC count of 84.6 × 109(IQR 63.4–97.7 × 109), compared with 54.7 × 109(IQR 43.3–70.4 × 109) in HPC(A) from donors who were mobilized with G-CSF alone (Fig. 6b.i).
Plerixafor use also led to a significantly higher TCRαβ:TCRγδ ratio of HPC(A) (Fig. 6b.ii) but post-depletion TCRγδ+ cell counts were not compromised (Fig. 6b.iii). There was no difference observed in terms of CD45RA:CD45RO ratio of HPC(A) between donors who did or did not receive plerixafor.
Other variables
We also examined the effect of variables, such as donor gender (Supplemental Fig. 3a) and weight (Supplemental Fig. 3b), but did not find any significant correlation with HPC(A) composition.
Discussion
This analysis shows that within our large series of T-cell depletion processes, we have achieved highly efficient and consistent T-cell subset depletion which were comparable to those reported in other studies utilizing the CliniMACS device for immunomagnetic depletion13–15. This is a fundamental factor that underlies the successful operation of our haplo-HSCT program. In a nationwide multi-center study reviewing the outcomes of patients who underwent haplo-HSCT using ex vivo T-cell depletion methods as outlined in this paper, we observed an acceptable cumulative incidence of grade III to IV acute GVHD of 12% and a very low chronic GVHD of 5% at 2 years, resulting in favorable clinical outcomes of GvHD-free and relapse-free survival (GRFS) of 57.3% at 2 years 16 . A retrospective comparison of clinical outcomes with a contemporary cohort of propensity score-matched patients who underwent haplo-HSCT with an unmanipulated graft followed by post-transplant cyclophosphamide (PTCy) showed a lower risk of chronic GVHD (hazard ratio (HR): 0.08; 95% confidence interval (CI): 1–62; P = 0.016), resulting in a more favorable 5-year GRFS (50% vs 30%; P = 0.03) in the recipients of ex vivo T-cell depleted graft, as compared to haplo-PTCy recipients 17 .
Leukapheresis and cell processing are expensive and labor-intensive procedures. To accommodate the protocol of infusing a stem cell graft depleted of TCRαβ+ T cells, followed by add-back of memory T-cell subset, optimal proportioning of the leukapheresis product is necessary to maximize the utilization of both the harvested product and the depletion kit. Information on the expected recovery of stem cells and target cells of interest has facilitated decisions on the allocation of aliquot sizes for both depletion processes. This allows us to adopt flexibility in fractionating HPC(A) for downstream processing, depending on our desired CD34 stem cell yield for the TCRαβ-depleted graft, and optimization of the remaining mobilized product for CD45RA depletion, taking into consideration an individualized assessment of the recipient’s future needs for further DLI. In this series, based on the accumulated experience that is continuously being refined, a single leukapheresis session could provide enough cells for both depletion processes for all except 10 donors who required a second leukapheresis session. We observe that while the TCRαβ depletion process yielded excellent recovery of CD34+ stem cells (Table 1, Fig. 4a.i), the CD45RA depletion process led to loss of more than half of the target CD45RO+ cells (Table 1, Fig. 4b.ii). This results from the heterogeneous nature of CD45 isoform expression, leading to the loss of a proportion of CD45RO+ cells with weak CD45RO expression. CD34+ cell recovery rate was also compromised in the process of CD45RA depletion, as a substantial proportion of CD34+ hematopoietic stem cells express CD45RA18,19. Notably, in the 89 CD45RA depletion processes, we did not encounter any case with variant pattern of CD45RA:CD45RO expression that could result in failure of recovery of CD45RO+ cells 19 .
The success of depletion processes depends on a good harvest that contains adequate numbers of cells of interest in the leukapheresis product, which in turn may be affected by factors inherent in the donors or mobilization method. The proportion of memory T cells increases with age, as demonstrated in our series of 91 cases with an inverse correlation between age and CD45RA: CD45RO ratio (Fig. 6a.ii). This information is relevant in the planning of apheresis sessions. In situations where repeated DLI with memory T cells is planned, a larger blood volume will need to be processed from younger donors with the aim of collecting more TNC, or a larger fraction of the leukapheresis product will need to be allocated for CD45RA depletion, since the leukapheresis product is expected to contain a lower proportion of CD45RO+ cells in younger donors.
Our findings pertaining to the impact of donor age and use of plerixafor on the composition of HPC(A), although irrelevant for transplant protocols that involve depletion of these cells, may have potential immunological impact for HSCT using unmanipulated grafts, and is worth further investigation.
We made one worthwhile observation from this analysis. Nineteen cases, including five that were plerixafor-mobilized, challenged the capacity of the TCRαβ depletion kit by exceeding the 60 × 109 TNC limit but without exceeding the target fraction capacity of 24 × 109. Despite this, CD34 fraction and TCRγδ subset recovery was not compromised (Fig. 5b.ii and 5b.iii). Although TCRαβ depletion efficiency was indeed lower compared to the other 68 cases (Fig. 5b.i), the process remained highly efficient with a median of 4.1 (IQR 3.9–4.3) log reduction and threshold dose was not exceeded as a result. This is a valuable observation which supports continuation of such practice to obviate unnecessary wastage.
Another significant observation we made was that platelet number did not affect depletion efficiency for both TCRαβ and CD45RA depletion processes (Supplemental Fig. 2). Platelet contamination has been recognized as a potential factor that may exert a negative impact on CD34+ cell recovery through interference on antibody binding in immunomagnetic CD34+ cell selection processes20,21. The potential adverse effects of platelet contamination may have been partially abrogated by the thorough manual washing steps in our cell preparation processes, as demonstrated by Dykes et al 22 . Not unexpectedly, with the extensive ex vivo manipulation, the post-depletion product contains minimal platelets, and therefore unlike unmanipulated HPC(A), these selective T-cell-depleted stem cells cannot be expected to result in any increase in platelet count post-infusion.
We have shown in further correlative analysis that donor characteristics and mobilization method can affect leukapheresis product composition, which in turn has some impact on depletion performance, with several correlations demonstrating statistical significance. Nevertheless, both depletion systems can accommodate variations in leukapheresis product composition over a broad range of up to 20-fold difference without compromising depletion efficiency and cell recovery to a clinically meaningful degree, such that pre-specified doses could be achieved in the graft for infusion and dose limits were not exceeded. This testifies to the robustness of both depletion systems that can handle products of a wide range of composition for depletion and recovery of cell subsets of interest. Although CliniMACS Prodigy®, a new-generation fully automated system for both depletion processes 23 , is now available, CliniMACS PLUS is expected to remain in use by many laboratories, therefore our findings remain relevant.
Conclusion
In this report, we have summarized our results and experience with ex vivo TCRαβ+ and CD45RA+ T-cell depletion over a span of 6 years. Our results show that the depletion systems are robust and reproducible in the hands of our highly competent laboratory personnel. This approach has enabled haplo-HSCT to be conducted safely with low risks of GVHD.
We have also highlighted interesting observations on a variety of donor-related and mobilization-related factors that influence the immune cell subsets and composition of the leukapheresis product, which are worth further exploration in future studies.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251336965 – Supplemental material for Selective TCRαβ+ and CD45RA+ T-cell depletion of hematopoietic stem cell graft: An analysis on factors that affect depletion performance
Supplemental material, sj-docx-1-cll-10.1177_09636897251336965 for Selective TCRαβ+ and CD45RA+ T-cell depletion of hematopoietic stem cell graft: An analysis on factors that affect depletion performance by Chieh Hwee Ang, Gina Gan, Ren How Ho, Kee Khiang Heng and Yeh Ching Linn in Cell Transplantation
Footnotes
Ethical Considerations
The study received ethical approval from the SingHealth Centralised Institutional Review Board (ECOS 2015-2419).
Author Contributions
C.H.A. analyzed the data and wrote the manuscript. G.G., R.H.H., and K.K.H. performed the cell depletion processes. G.G. collected the data from cell depletion processes. Y.C.L. conceptualized the study, edited and reviewed the manuscript. All authors agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
All donors have provided prior written informed consent to allow access to their electronic records and usage of their clinical and laboratory results for this study.
Data Availability Statement
Data collected and analyzed in this study is available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
