Abstract
Donor-specific antibodies (DSAs) are essential causes of graft rejection in haploidentical hematopoietic stem cell transplantation (haplo-HSCT). DSAs are unavoidable for some patients who have no alternative donor. Effective interventions to reduce DSAs are still needed, and the cost of the current therapies is relatively high. In this study, we retrospectively analyzed the data of 11 DSA-positive patients who received haplo-HSCT at our center and evaluated the therapeutic efficacy of the combination of intravenous immunoglobulin (IVIG), dexamethasone and high dose of transfused mononuclear cells (MNCs) for DSA desensitization. The kinetics of DSAs at different times and the engraftment and transplantation outcomes were also observed. We found that all patients had successful donor-cell engraftment and that no patient developed poor graft function. The median engraftment times of neutrophils and platelets were 14 days (range, 11–24 days) and 13 days (range, 11–123 days), respectively. The DSA levels of all patients became negative or dropped under 2000 within 22 days after HSCT. A total of 36.4% of patients developed grade II–IV acute graft-versus-host disease (aGVHD), and 9.1% of patients died of severe gastrointestinal aGVHD. Of the 7 surviving patients, four were diagnosed with chronic GVHD. After a median follow-up of 28.9 months (2.0–52.1 months), four patients died: of relapse (two), aGVHD (one), and multiple-organ failure (one). The 2-year OS, DFS, and NRM were 63.6%, 45.4%, and 18.2%, respectively. Combination therapy with IVIG, dexamethasone, and a high dose of MNCs transfusion, a simple and efficient procedure, was safe and effective for DSA desensitization and peripheral blood stem cell (PBSC) engraftment.

Keywords
Introduction
Haploidentical hematopoietic stem cell transplantation (haplo-HSCT) has become a viable option for transplant candidates without human leukocyte antigen (HLA)-matched donors. Severe complications, including primary graft failure, acute graft-versus-host disease (aGVHD), and infection, are major obstacles to this approach. Donor-specific anti-HLA antibodies (DSAs) were first identified in solid organ transplantation and are considered a cause of hyperacute graft rejection 1 . In HSCT, DSAs have been associated with primary graft failure, delayed engraftment, and poor survival2,3,4. Heavy transfusion, multiparity, and offspring recipients are high-risk factors for developing DSAs5,6,7.
DSAs are found in approximately 10% to 21% of haploidentical donors 8 . Therefore, the European Society for Blood and Marrow Transplantation consensus guidelines recommend routine testing for DSAs for optimal donor selection 9 . Since the number of donors is limited, DSAs are likely to exist in the donors who are available. This makes DSA desensitization before transplantation particularly urgent. Although some strategies have been empirically applied, such as rituximab or bortezomib to inhibit antibody production8,10,11,12, intravenous immunoglobulin (IVIG) or platelet transfusion to neutralize antibody-mediated reactions10,12,13, and plasma exchange for antibody depletion8,14,15, the procedures are complex, and the efficacies vary between individuals.
In this study, we investigated the prevalence of DSAs in haplo-HSCT patients from our center. Based on immune thrombocytopenia (ITP) therapy, we explored the efficacy and safety of the use of IVIG, dexamethasone, and high dose in mononuclear cells (MNCs) transfusions for efficient DSA desensitization intervention.
Materials and Methods
Patients and the Corresponding Donors
From April 2019 to October 2021, 11 consecutive DSA-positive patients (two weakly positive, three positive, and six strongly positive) who received haplo-HSCT at our hospital were retrospectively analyzed. The HLA class I and II antibodies were sequenced in both directions before HSCT. The results were analyzed based on the patients’ clinical characteristics. The study was approved by the ethics committee of the 920th Hospital of Joint Logistics Support Force and registered with ClinicalTrials.gov (NCT06471478). All procedures were performed according to the standards of the Declaration of Helsinki, and all patients enrolled signed informed consent forms.
DSA Testing
Blood samples from patients were collected at the following 5 time points: 2 weeks before HSCT, 0 day [before hematopoietic stem cells (HSCs) transfusion], +8 days, +15 days, and +22 days after HSCT. DSA information was collected with a LABScreen Kit. Serum anti-HLA antibodies were tested using single antigen beads according to the manufacturer’s instructions. Anti-HLA antibody profiles were collected using LABScanTM100 (Template setting based on the instructions of the kit). The mean channel fluorescence intensity (MFI) was used to measure the DSA level based on the strength of fluorescence from each antigen bead. MFI > 10,000 is defined as “strongly positive”; 5,000 < MFI ≤ 10,000 is defined as “positive”; 500 < MFI ≤ 5000 is defined as “weakly positive”; and an MFI less than or equal to 500 is defined as “negative” 9 .
HSC Mobilization and Collection
Donors were given with subcutaneous injection of 10 µg/kg/day granulocytecolony-stimulating factor (G-CSF) on -4 to -1 day before transplantation, blood routine and peripheral CD34+ cell count were measured on -1 day and 0 day. If the peripheral CD34+cell count <20/µl on -1 day, the donor was subcutaneously injected with an additional 12 mg plerixafor 12 h before HSCs collection.
Leukapheresis was performed on 0 day with the cobe spectra blood cell separator (Fresenius Medical Care, Bad Homburg v. d. Höhe, Germany) through the bilateral peripheral venous accesses of donors. According to the recommendation of the American Society for Blood and Marrow Transplantation, the optimal mobilization was recommended as CD34+cell count ≥4 × 106/kg. Mononuclear cell count, which was defined as the concentration of the sum of monocytes and lymphocytes in the grafts, was also used to describe the dose of grafts. Mononuclear cells of grafts were measured to ensure a minimal count ≥8 × 108/kg MNC count. If the MNC count did not reach the standard after a single leukapheresis, a repeat leukapheresis could be performed on day 1.
DSA Reduction Treatment
All patients received 1 g/kg IVIG on -1 day and 25 mg/m2/d dexamethasone on -4 to -1 day before transplantation. On the basis of the conventional transfusion amount of HSCs mentioned above, additional MNCs were transfused based on the DSA level of each patient: 3 ± 2 × 108/kg, 6 ± 2 × 108/kg, and 9 ± 2 × 108/kg mononuclear cells for patients whose DSAs were weakly positive, positive, and strongly positive, respectively.
Transplantation Procedure
All patients received a reductive toxicity conditioning regimen. Patients diagnosed with acute myeloid leukemia (AML), acute lymphocytic leukemia (ALL), or thalassemia received 180 mg/m2 fludarabine, 100–200 mg/m2 busulfan, or 120 mg/m2 melphalan. Patients diagnosed with severe aplastic anemia (SAA) or paroxysmal nocturnal hemoglobinuria (PNH) were given 100 mg/kg cyclophosphamide, 180 mg/m2 fludarabine and 100 mg/m2 melphalan. aGVHD prophylaxis consisted of cyclosporin A (CsA, CsA was administered at a serum concentration of 250 ± 50 ng/ml from +1 to +90 days and gradually tapered up to +180 day after HSCT), 30 mg/kg posttransplant cyclophosphamide (+3 day and +4 day), 5 mg/m2 short-term methotrexate (+3 day and +6 day) and 15 mg/kg mycophenolate mofetil q8h (+5 day to +35 day). The first-line aGVHD treatment was 2 mg/kg methylprednisolone, and the second-line aGVHD treatment included etanercept, budesonide, basiliximab, and sirolimus, according to the condition of each individual. Ganciclovir and acyclovir could be given to prevent cytomegalovirus infection, and sulfamethoxazole was given to prevent Pneumocystis carinii pneumonia 16 .
Follow-up
Full-blood chimerism was monitored on +14 day, +28 day, +30 day, +60 day, and +90 day and later monitored twice a month. During transplantation, routine blood tests were performed daily before engraftment. Neutrophil engraftment was defined as >0.5 × 109/L for three consecutive days, and platelet engraftment was defined as >20×109/L for three consecutive days. Primary graft failure was defined as no appearance or complete loss of donor-derived neutrophils by +28 day according to short tandem repeat (STR) chimerism analysis. Poor graft function was defined as the presence of at least two cytopenic counts beyond +28 day with a transfusion requirement associated with hypoplastic-aplastic bone marrow.
The primary endpoint was the incidence of primary graft failure. The secondary endpoints were the incidence of poor graft function, acute and chronic GVHD, nonrelapse mortality (NRM), overall survival (OS), and disease-free survival (DFS).
Statistics
Graft-versus-host disease incidence, OS, DFS, and NRM were statistically analyzed with SPSS version 26.0 (IBM, Armonk, NY, USA). Continuous variables that did not conform to the normal distribution were described by medians (range). OS and DFS survival curves were plotted using Kaplan‒Meier analysis. DSA variation curves were drawn using GraphPad Prism 6.07 (GraphPad Software Inc., San Diego, CA, USA) according to the detailed DSA MFI on different time points.
Results
Patient Characteristics
Information on the 11 enrolled patients and their corresponding donors is listed in Table 1. Three patients were male and eight were female. The median age was 35 years (range, 4–59 years). Patient 1 had received autologous HSCT 1 year earlier. Two (patients 1 and 5) of the six patients with hematological malignancies were not remission and received the salvage HSCT. Almost all patients had multiple blood transfusions before HSCT (more than 30 units). Of the eight female patients, six were multiparous (more than twice) and two were diagnosed with transfusion-dependent thalassemia. A total of 45.5% of the patients were in the “child-to-mother” transplantation model, and serum iron overload was present in seven (63.6%) patients. All patients had transfusion history, and the median number of units was 22 units (ranged for 4–62 units).
Patient and Donor Information.

Abbreviation: M/F: male/female, HLA: human leukocyte antigen, ALL: acute lymphocytic leukemia, AML: acute myeloid leukemia, SAA: severe aplastic anemia, PNH: paroxysmal nocturnal hemoglobinuria.
Kinetics of DSA After Desensitization
The number of HSCs required was calculated according to the disease type, DSA level and individual conditions before transplantation. The median numbers of mononuclear cells and CD34+ cells were 26.5×108/kg (16.9–28.6 × 108/kg) and 17.6×106/kg (15.4–24.8 × 106/kg), respectively.
The most common positive DSA was anti-HLA DQ. Seven patients had more than one kind of DSA against this locus. Six patients had strongly positive DSAs with MFI greater than 10,000, and five had weakly positive DSAs with an MFI less than 5,000. All weakly positive and positive DSAs directly became negative or decreased to less than 1,000 within +8 day. Most strongly positive DSAs decreased more slowly than did the other DSAs but gradually dropped under 2,000 within +22 day (Fig. 1 and Table 1).

Dynamic variation of DSA MFI. DSA: donor-specific antibody, MFI: mean channel fluorescence intensity, pre-HSCT: pre-hematopoetic stem cell transplantation.
Hematopoietic Reconstitution After Haplo-HSCT
All patients had successful donor-cell engraftment, and no patients developed primary graft failure or poor graft function. The median engraftment times of neutrophils and platelets were 14 days (ranging from 11 days to 24 days) and 13 days (ranging from 11 days to 123 days), respectively, which are comparable with our previous results 14 . Patient 5 experienced delayed platelet reconstitution and needed platelet transfusions until +123 day, which may have been caused by infection and iron overload. Chimerism analysis was routinely performed after neutrophil engraftment, and 10 patients had persistent complete donor chimerism during the follow-up. Patient 5 acquired complete remission and complete donor chimerism after HSCT, but the chimerism rapidly decreased to 89.4% 3 months after HSCT due to leukemia relapse (Table 2).
Transplantation Information and Outcomes.
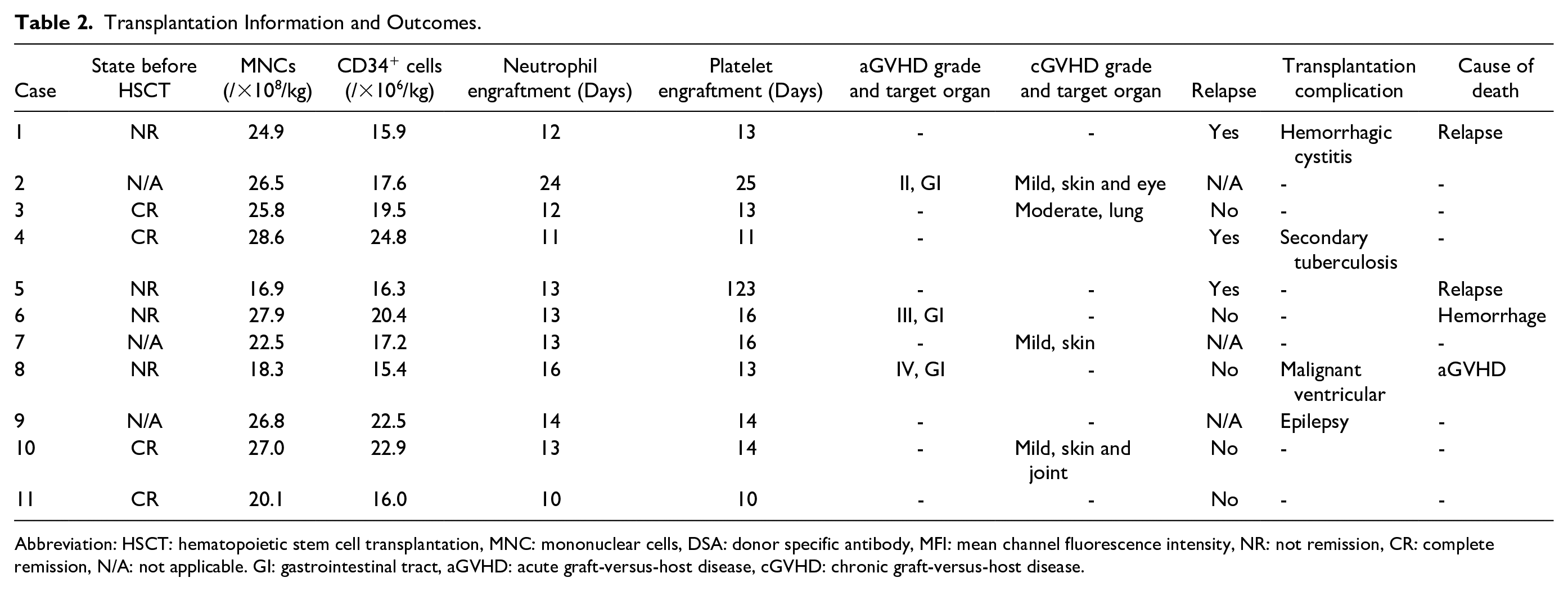
Abbreviation: HSCT: hematopoietic stem cell transplantation, MNC: mononuclear cells, DSA: donor specific antibody, MFI: mean channel fluorescence intensity, NR: not remission, CR: complete remission, N/A: not applicable. GI: gastrointestinal tract, aGVHD: acute graft-versus-host disease, cGVHD: chronic graft-versus-host disease.
Transplantation Outcomes
After a median follow-up of 28.9 months (2.0–52.1 months), six patients (patients 2, 3, 7, 9, 10, and 11) achieved long-term survival, with a donor chimera rate greater than 99%. Patient 4 relapsed 26.6 months after haplo-HSCT, at which point she stopped treatment. She is still alive. Four patients died: Patient 1 and Patient 5, who were in the NR state before HSCT, died of relapse within half a year after transplantation. Patient 6 died of cerebral hemorrhage 76 days after HSCT. Patient 8, who was diagnosed with PNH, had a long blood transfusion history of more than 10 years and was in poor physical condition before HSCT. He died of GVHD and multiple-organ failure on day 55 (Table 2).
Three (27.3%) patients developed grade II-IV aGVHD, and 1 patient died of severe gastrointestinal GVHD. Of the 7 surviving patients, 4 (36.4%) were diagnosed with cGVHD. The skin, eye, and joints were the main tissues involved. Patient 3 developed lung cGVHD for which he needed home oxygen therapy. None of the patients developed hepatic veno-occlusive disease, thrombotic microangiopathy or posttransplant lymphoproliferative disorders. Other transplantation-related complications are listed in Table 3. The 2-year OS and 2-year DFS were 63.6 and 45.4%, respectively (Figs 2 and 3), and the NRM was 18.2% during the observation time.
Transplantation Outcomes.

Abbreviation: GI: gastrointestinal tract, N/A: not applicable; aGVHD: acute graft-versus-host disease, cGVHD: chronic graft-versus-host disease.

Overall survival.

Disease-free survival.
Discussion
DSA has been strongly associated with primary graft failure, poor graft function and delayed engraftment after haplo-HSCT, which may result in increased transplantation-related mortality and poor prognosis2,3,4,17. In this study, we analyzed the demographic and the clinical characteristics of DSA-positive patients and applied a novel DSA desensitization strategy involving the combination of IVIG, dexamethasone, and high dose for peripheral blood stem cell (PBSC) transfusion. Our results demonstrated that this strategy can significantly decrease DSAs, ensure hematopoietic reconstitution, and yield satisfactory transplantation outcomes.
Previous strategies for DSA desensitization are mainly based on the following mechanisms: the first and most common method is plasmapheresis. Over 10–50 units of fresh frozen plasma were used for antibody removal. Generally, plasma exchange is performed more than 10 days before HSCT to avoid influencing the conditioning regimen18-20. Yoshihara et al. 21 reported a patient with a DSA level greater than 10,000 who underwent plasma exchange, the DSA level decreased significantly before transplantation but rebounded to baseline within 1 week because of newly produced antibodies by host residual antibody-producing cells. Plasmapheresis eliminates both anti-HLA class I and II DSAs; it is accessible to most patients with significant and temporary efficacy but is limited by insufficient blood product supplementation and the requirement for patient platelet levels. The second strategy is to block DSA with IVIG or infusion of the donor’s platelet. HLA class I antigens are highly expressed on platelets; therefore, if only DSAs against HLA class I antigens exist, a large volume of the donor’s platelet could be infused on day -1 for DSA neutralization13,21. One study used platelet infusion as a single treatment and showed an inspiring effect with a rapid and remarkable reduction of DSAs, but it is useless to absorb DSAs specific to HLA class II antigens, as HLA class II antigens are not expressed on platelets 13 . IVIG shares similar mechanisms and can quickly eliminate existing antibodies. However, the time to response is dependent on the previous antibody level, and the DSA MFI may rebound after stem cell transfusion because of de novo DSAs 12 . The third strategy is to inhibit the production and function of new DSAs with CD20 antibodies or proteasome inhibitors. Rituximab can target CD20+ B cells, so it has been used to deal with DSA-mediated transplantation rejection in the setting of solid organs11,22,23. Chang et al. 11 used a single dose of 375 mg/m2 rituximab 3 days before transplantation, they found that it effectively reduced DSA and prevented the onset of primary graft failure in DSA-positive patients with 2,000 < MFI < 10,000 but did not in those with significantly increased antibody levels. Bortezomib is a proteasome inhibitor that can stimulate plasma cell apoptosis and suppress antibody secretion 21 . Choe et al. 12 tested several doses of 1.3 mg/m2 bortezomib and 2 g/kg IVIG before HSCT for 14 patients, and only two patients achieved a reduction of MFI to <2,000. This approach was barely satisfactory because of its long onset time, additional interventions are needed for most patients. Inhibition of the complement cascade represents a novel direction for DSA desensitization, but this approach has not been well defined due to the expensive price and limitations in sample size 24 . Ma et al. 25 found that the circulating T-follicular helper (Tfh) cells were positively related to the production of anti-HLA antibodies, and targeting Tfh cells is another strategy for DSA desensitization; further studies are required to identify potential agents for inhibiting cTfh cells. Each intervention has its own strengths and disadvantages, and most clinical studies tend to combine several treatments to decrease DSAs.
We referred to ITP therapy and proposed a novel DSA desensitization strategy. Dexamethasone is a preferred corticosteroid in first-line therapy for adult patients with ITP. A high dose of dexamethasone can inhibit inflammation and suppress the immune system, reducing tissue damage after conditioning chemotherapy as well as autoantibody generation 26 . During ITP therapy, IVIG can competitively bind to FcN receptors on mononuclear macrophages and block antibody-dependent cell-mediated cytotoxic effects 27 . Dexamethasone/IVIG seemed able to rapidly eliminate the existing DSAs, but were useless for the clearance of the de novo DSAs and high-level DSAs in HSCT. The grafts products contained enough HSCs and MNCs, which ensured a successful hematopoietic reconstitution. Moreover, large amounts of monocytes, lymphocytes, and platelets could persistently neutralize the de novo anti-HLA class I and II DSAs produced after HSCT to a large extent. We found that the time frame of DSA-MFI reduction in most patients was 0 day to +8 day, and some DSAs even rose at 0 day, which may have been caused by de novo DSAs. Considering the short-term efficacy of both IVIG and dexamethasone, we speculated that large amounts of MNCs transfusions play a predominant role in sustained DSA clearance and ensure the engraftment of HSCs.
The procedure is not only simple and works for any type of DSA but also fast-acting and not limited to any blood product. High dose in PBSCs contains more T cells and often results in a greater incidence of aGVHD. In our study, the incidences of aGVHD and cGVHD were 27.3 and 36.4%, respectively, which nearly coincided with our previous data 28 . No patient developed primary graft failure or poor graft function. The 2-year OS and DFS were relatively lower, as half of the patients were in poor condition before HSCT. HSC collection may be an obstacle to this strategy, especially for patients whose donors were old or underweight. However, in this study, most grafts reached the standard with the used of G-CSF, only one low-weight donor received G-CSF plus plerixafor. Due to the limited sample size, prospective studies with larger samples and subgroup analyses for different types of DSAs are needed to validate the efficacy and safety of this therapy and to further explore the response to this therapy.
In conclusion, the presence of DSAs is not an absolute barrier to HSCT. The combination of IVIG with dexamethasone and high dose of PBSCs transfusion has a certain effect on DSA reduction and is an efficient strategy for decreasing primary graft failure with a satisfactory transplantation outcome.
Footnotes
Acknowledgements
We thank the patients enrolled, their families, and our research team.
Author Contributions
Conception and design: SW.
Manuscript writing: XL.
Collection and assembly of data: YL, ZY, LL, SL, YD, and YC.
Data analysis: DZ and XH.
Final approval of manuscript: All authors.
Availability of Data and Material
The authors confirm that the data supporting the findings of this study are available within the article.
Ethical Approval
The study was approved by the ethics committee of the 920th Hospital of Joint Logistics Support Force and registered with ClinicalTrials.gov (NCT06471478).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
All patients enrolled signed informed consent forms.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint and Special Project of Kunming medical university (No. 202301AY070001-226), the Special Project for the Fundamental Research of Yunnan Province (No. 202301AU070024), the Biomedical Major Project of Yunnan Province (No. 202102AA100011), Yunnan Applied Basic Research Projects-Union Foundation (No. 202201AY070001-280).
