Abstract
Recent advances in biomedical technologies have extended the boundaries of previously established regulatory guidelines pertaining to stem cell research. These guidelines constrained the study of human pluripotent stem cells (hPSCs) and their derivatives from use under various conditions, including the introduction of hPSCs into the brains of host animals because of concerns of humanizing the brains of animal species. Other guidelines constrained the use of hPSCs in creating human-animal chimeras because of the potential contribution of human stem cells not only to the brain but also to the germline. Some regulatory guidelines forbid the growing of human embryos ex vivo beyond the stage of primitive streak development because of concerns regarding the creation of human forms of life ex vivo. At the subcellular level, there are guidelines regulating the transfer of mitochondria within human embryos. At the molecular level, there are guidelines regulating genome editing to prevent permanent genetic alterations in germline cells. These and other issues related to stem cells have been reviewed, and new research guidelines established by the International Society for Stem Cell Research (ISSCR) for its membership. Because many of the recommended changes by the ISSCR impact research being conducted by members of the American Society for Neural Therapy and Repair (ASNTR), the ASNTR established a task force to review relevant recommendations by the ISSCR to determine which new guidelines to adopt for research conducted by the ASNTR society membership. The final ASNTR recommendations are presented in this document.
Keywords
Introduction
Stem cells offer the possibility of replacing dysfunctional cells and organs in order to repair the human body. Since the characterization of human embryonic stem cells (ESCs) by Thomson et al. 1 and the generation of induced pluripotent stem cells (iPSCs) by Takahashi and Yamanaka 2 , tremendous strides have been made in characterizing somatic cells differentiated from ESCs and iPSCs. Recent advances further push the boundaries of stem cell research to the limits of current research guidelines established by certain countries, scientific institutions, and scientific societies 3 . These boundaries were initially established to limit the manipulation of pluripotent human stem cells into animal hosts that would result in human-like neurological capabilities, produce human–animal chimeras where human cells would contribute to the germ line, and grow human embryos beyond the development of the primitive streak. Advances in technology also raised concerns about the use of gene-editing approaches that would introduce permanent changes in the human germline, as well as concerns regarding human mitochondrial transfer and exchange.
These and other issues were assessed by a panel convened by the International Society for Stem Cell Research (ISSCR). As a result of their deliberations, this panel has provided recommendations for new guidelines for the conduct of stem cell research by members of its society. Several of those recommended guidelines impact the nervous system research conducted by members of the American Society for Neural Therapy and Repair (ASNTR). As a consequence, a task force was convened by the ASNTR to review the new guidelines recommended by the ISSCR for stem cell research to determine whether the ASNTR should adopt these recommendations as well. This report provides recommendations for incorporating new guidelines for research on stem cells by the ASNTR based on those recommended by the ISSCR.
Topic 1: Studies Transplanting Human Stem Cells or Their Direct Derivatives Into the Central Nervous System of Animals
ISSCR recommendation 2.2.1.1
Research involving the transfer of human stem cells or their direct neural or glial derivatives into the central nervous system (CNS) of postnatal animal hosts requires review by institutional animal research oversight committees supplemented by reviewer expertise in stem cell or developmental biology. Such oversight should weigh the potential benefits of the research and utilize available baseline non-human animal data grounded in rigorous scientific evidence or reasonable inferences and involve diligent application of animal welfare principles.
Background and rationale for ISSCR recommendation 2.2.1.1
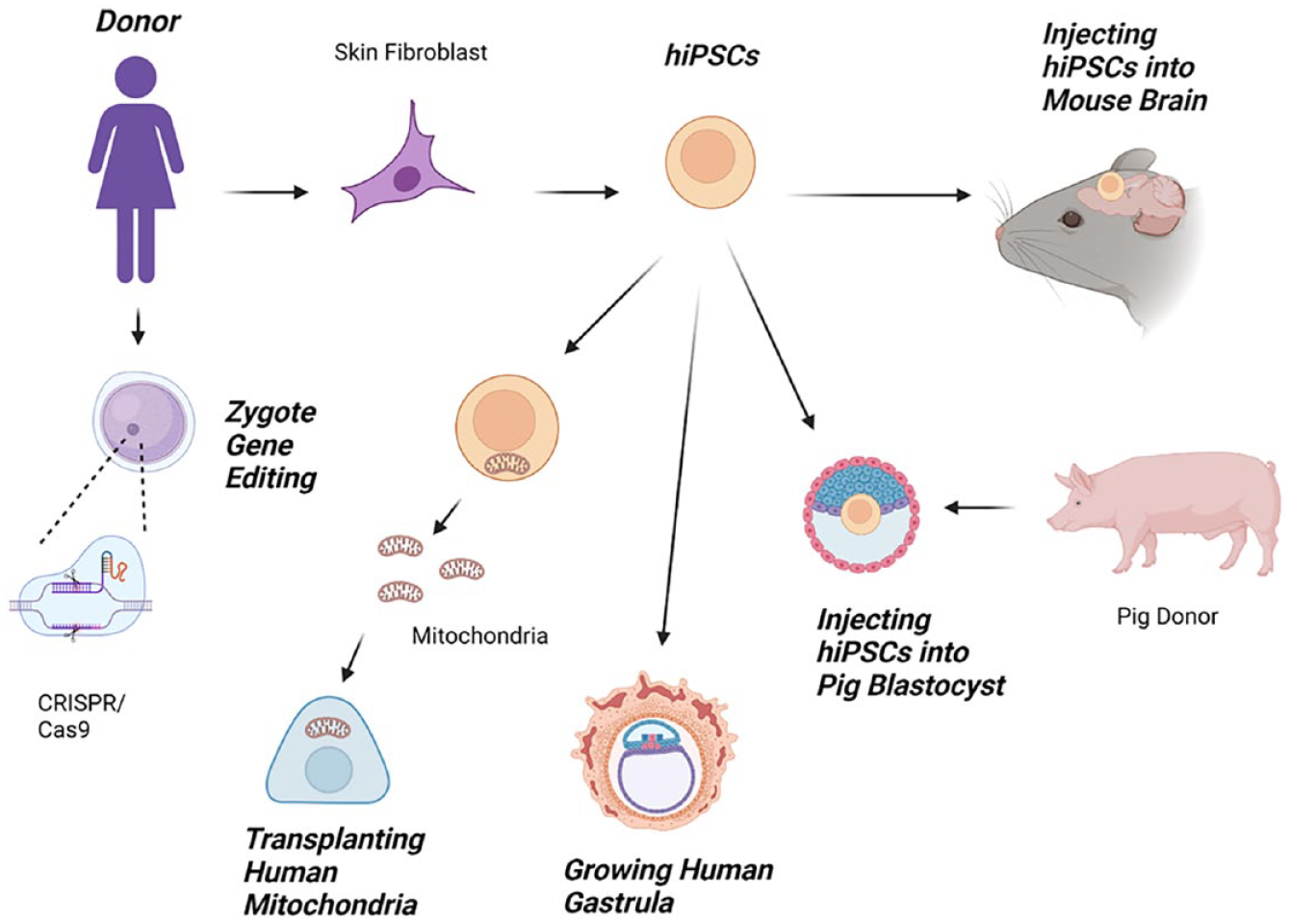
The use of human pluripotent stem cells (hPSCs) has become increasingly relevant in biomedical research exploring stem cell biology. As a result of this sustained interest, along with advancements in science and technology and successful experimentation, transplanting hPSCs into animal hosts has created another avenue for producing animal models of human disease, generating human organs for transplantation, and evaluating stem cell therapies4,5. While these advancements may be beneficial, it is necessary to recognize the potential concerns of safety and efficacy associated with transferring hPSCs and their derivatives into animal hosts while ensuring proper animal welfare.
Pluripotent stem cells are a category of undifferentiated stem cells with the potential to generate all human cell types during development 4 . There are two types of hPSCs: ESCs and human iPSCs. ESCs are pluripotent stem cells derived from the inner cell mass of early developing embryos4,6. Human iPSCs are pluripotent stem cells generated from adult somatic cells by induction with select transcription factors2,4. As a result of their pluripotency, ESCs and hiPSCs are ideal candidates for generating a wide range of cells through targeted differentiation protocols and have been used to generate models of human neurological disease.
Grafting hPSCs or their direct derivatives into the animal CNS has been performed to understand stem cell integration and survival post-transplantation (Fig. 1). In particular, studies demonstrated that human iPSC-derived neuronal cells grafted into rodent brains survive up to 2 years after transplantation 7 , integrate and form synapses with endogenous neurons 8 , and extend their axons toward relevant targets8,9. Researchers have translated these transplantation protocols into animal models of CNS disease and injury, including Parkinson’s disease 10 , stroke 11 , traumatic brain injury (TBI) 12 , and spinal cord injury (SCI) 8 , to assess therapeutic efficacy and understand post-transplant cell biology for potential clinical translation. In many of these studies, transplanted cells survived and improved overall animal health and function, paving the way for clinical trials, including the ongoing human iPSC transplantation clinical trials for SCI 13 and amyotrophic lateral sclerosis (ALS) 14 in Japan. While preclinical studies may provide promising results that suggest therapeutic benefits, this does not always translate to clinical trials, as animal models do not capture the full complexity of human disease and injury; therefore, researchers opted to generate updated animal models using human stem cells. Espuny-Camacho et al. described grafting human iPSC-derived neurons into postnatal mice to generate an Alzheimer’s mouse model that followed relevant Alzheimer’s pathology 15 .

Grafting human stem cells or their direct derivatives into the central nervous system of host animals is a successful model for understanding stem cell integration and survival after transplantation.
While these advancements in modeling and treating CNS diseases and injuries in animals with hPSCs have been fruitful for understanding neurological diseases and the therapeutic value of the neural and glial derivatives, it is important to highlight the accompanying public and scientific concerns. In a survey studying US public readiness for human–animal chimeric embryo research, one primary concern participants raised was the incorporation of human cells into the brain of animal hosts and the potential for neurological humanization of animal hosts 16 . Based on the current state of stem cell transplantation research, the ISSCR subcommittee guidelines described that biological humanization is not scientifically consistent with the idea of cognitive humanization of animal hosts 17 . A literature review of preclinical peer-reviewed publications revealed no evidence to suggest cognitive humanization of animals in response to human–animal cell integration 18 . Nevertheless, this concern remains influential in outlining guidelines for the use of human stem cells in the CNS of postnatal animal hosts.
Legal and institutional boundaries exist to regulate human–animal stem cell transplantation. These policies can vary from the local to the national level, as well as between institutions. Generally, any research involving animals requires institutional approval before animal use to ensure animal safety and welfare through every protocol involving the use of animals. To use hPSCs for transplantation into animals, additional oversight often is needed from committees 19 . Approval from these oversight committees, along with compliance with other additional institutional, local, and national regulations, should be the primary concern for this research. However, conducting human–animal stem cell transplantation research in adherence to regional regulation is complicated by the lack of both national and international consensus. Within the US alone, states such as Arizona and Louisiana restrict human-derived cell contribution to animal brain tissue to less than 51%, in contrast to California, which has specific restrictions for non-human primates but generally allows for a wider range of research with Stem Cell Research Oversight approval 3 . On an international level, guidelines for human stem cell transplantation in animals remain largely unspecified. Many countries, including Canada, Australia, and Germany prohibit the transplantation of animal cells into human embryos but have no specific guidelines for the inverse—introducing human cells into animal embryos3,20. There have been discussions to overcome this ambiguity in regulation, including the United Kingdom’s Guidelines for Human Embryonic Stem Cell Research 3 . In 2019, Japan’s Ministry of Education, Culture, Sports, Science, and Technology relaxed regulations on human–animal chimeric embryo research 21 . These new guidelines have eased restrictions and currently allow for research on animal brains containing human cells from human–animal stem cell transplantation 22 . Considering the variety of international recommendations and regulations, the guidelines recommended by the ISSCR are meant as considerations to supplement existing legal restrictions and aim to generate international consensus on human–animal stem cell transplantation.
The ISSCR recommendation on the transfer of hPSCs or their neural or glial derivatives into the CNS of postnatal animal hosts includes enhancing behavioral monitoring and data-collection procedures, considering limited study design, and ensuring proper expertise in animal research review committees 17 . For behavioral monitoring and data collection, additional considerations should be made on assessing cognitive and developmental trajectories after introducing human stem cells or their derivatives into the animal CNS, particularly the brain. This includes assessing behavioral baselines, regular monitoring for changes and enhancements of animal cognition, and appropriately adjusting research protocols in response to these data. When designing these studies, it is recommended to begin by limiting pilot studies to obtain the necessary information on the developmental progression of modified animals, and then adjusting protocols to provide relevant findings to the review committees. In addition, the animal review committees should include members with sufficient and relevant scientific and clinical expertise to assess and make judgments on human–animal chimera protocols. These recommendations are intended to ensure the maintenance of animal welfare while addressing public and scientific concerns.
ASNTR recommendation
The ASNTR Task Force found reasons to adopt ISSCR recommendation 2.2.1.1 with additional considerations for implementation. The ASNTR Task Force agreed with the overall approach of building on existing regulatory structures to allow for widespread adoption. Responsible progression of human stem cell research in animals through behavioral monitoring and flexibility in study design should be maintained. While not every institution has the access and resources necessary to create animal review committees with the specific member composition, the ASNTR Task Force recommended that the exact composition of the committee does not need to be consistent across institutions, so long as an array of perspectives is considered. Furthermore, it was suggested that the animal review committees should increase the diversity of perspectives by including biosafety and bioethics as considerations for approving human–animal stem cell chimera protocols. When adhering to these guidelines, human–animal chimera research can progress safely and responsibly.
Topic 2: Human–Animal Chimeric Embryo Research
ISSCR recommendation 2.2.2.2
Chimeric embryo and in utero research should proceed for the minimum time necessary to achieve the scientific aim. This research must proceed incrementally, stopping at well-defined time points to assess the degree and scope of chimerism during development before proceeding to full gestation, if full gestation is among the well-justified goals of the research. To avoid unpredictable and widespread chimerism, researchers should endeavor to use targeted chimerism strategies to limit chimerism to a particular organ system or region of the gestating chimeric animal.
Background and rationale for ISSCR recommendation 2.2.2.2
There is an ever-growing organ demand for transplantation therapy, and the severe shortage of donors cannot meet this demand. The fields of interspecies chimerism and blastocyst complementation demonstrate potential as an alternative method for generating whole transplantable organs and cells. Major efforts in the chimerism field are aimed at developing functional human organs in host animals like pigs. These functional organs can then be extracted and used for organ transplantation in a clinical setting. Furthermore, when using blastocyst complementation, the host embryo is genetically modified to enhance donor cell contribution to specific organs of interest (Fig. 2).

Exogenic human organs and cells are generated in host animal chimeras via blastocyst complementation in gene-edited embryos. The host embryo is genetically modified to enhance donor cell contribution to specific organs of interest, and functional organs are then extracted and used for organ transplantation.
Several studies have evaluated interspecies chimerism using different combinations of donor–host species, such as rat–mouse, marmoset–mouse, human–mouse, and rhesus monkey–pig, among others. Evolutionarily close species such as mice and rats successfully formed interspecies chimeras with multiple chimeric organ systems. The Nakauchi group at Stanford generated rat pancreas in PDX1-/- mice hosts 23 as well as mouse islets in PDX1-/- rats 24 . The mouse islets that developed in the mouse–rat chimera were functional and maintained normal host blood glucose levels when implanted into a diabetes-induced mouse model. Blastocyst complementation also has been successfully implemented in the generation of other organs, such as rat thymus in an athymic mouse model 25 and functional mouse kidney in the SAL1-/- rat model 26 .
The ultimate goal of the interspecies chimerism field is to develop human organs in host animals for transplantation therapy. More recent studies have focused on using human or non-human primate cells as donor cells for interspecies chimerism and blastocyst complementation experiments; however, the chimeric efficiency of these evolutionarily distant species has been limited. Multiple studies are beginning to tackle the barriers to chimerism 27 by using different strategies, such as stage-matching donor and host cells28,29, overexpressing anti-apoptotic genes in the donor cells 30 , and gene editing host embryos.
In a human–mouse chimerism study, human iPSC-derived neural crest cells injected in utero into embryonic-day-8.5 (E8.5) mouse blastocysts successfully formed human–mouse interspecies chimeras (~30% efficiency). The level of chimerism from human cell contribution was much lower than that in mouse–mouse chimeric controls 31 . Human PSCs injected into in vitro gastrulating mouse embryos (E6–E7.5) also formed chimeras, with high human contribution in the developing chimeric fetus with ~70% of chimeric embryos with human cells 32 .
Human iPSCs overexpressing anti-apoptotic genes, such as BCL230,33 and BMI1 34 , successfully integrated into mouse blastocysts and formed human–mouse chimeras with an increased efficiency until E10.5 in vitro. BCL2 overexpressing human iPSCs also complemented ETV2-null pig embryos and differentiated into the endothelial lineages in the E18 human–pig chimera 35 . Although successful chimerism was observed between human and non-human hosts, the chimeric efficiency and human donor cell contribution have been extremely low in most studies.
Significant progress in human–pig interspecies chimerism was recently reported by Wang et al. 36 . Human cell contribution as high as 50%-65% was observed in pig kidneys. The authors demonstrated that transient expression of BCL2 in the human donor stem cells greatly increased the efficiency of the chimeric contribution of the human cells to the mesonephros in the kidneys of SIX1-/- SALL1-/- pig embryos assessed at E28 that provided a nephric-defective niche for the human cells 36 . The study also showed a limited contribution of human cells to the CNS and genitalia. These findings suggest that the blastocyst complementation strategy could be used to prevent widespread chimerism in other organs, such as the brain and genitalia, which would raise ethical concerns in the field. This study, for the first time, clearly demonstrated that high contribution of human cells to organs in host organisms like pigs is possible, while also avoiding contribution to off-target organs.
ASNTR recommendation
It will be possible to achieve a higher level of human cell contribution in specific organs of host animals in future studies, which would help to move the field closer to the primary goal of developing whole human organs in host animals for transplantation therapy. Furthermore, complementation strategies have demonstrated the ability to target specific organs for chimerism and to avoid widespread chimerism. Adopting the ISSCR Recommendation 2.2.2.2. will ensure that the field of chimerism continues to progress toward its fundamental goal, carefully and incrementally in an organ-specific manner.
Some concerns regarding the recommendation include the “minimum time necessary” for in utero chimeric research and having “well-justified” research goals are both subjective and not well-defined in the recommendation. As observed in multiple chimerism studies, even with complementation strategies in place, widespread chimerism is sometimes unavoidable. Both these concerns require careful consideration by the researchers. In addition, an oversight committee or consultation with bioethicists would be ideal to ensure that the research progress is not hampered in the long term. The ASNTR Task Force supported the adoption of ISSCR Recommendation 2.2.2.2.
Topic 3: Culture of Human Embryos Beyond the Formation of the Primitive Streak or 14 Days
ISSCR recommendation 2.2.2.1
Given the advancements in human embryo culture and the potential for such research to yield beneficial knowledge that promotes human health and well-being, the ISSCR calls for national academies of science, academic societies, funders, and regulators to lead public conversations touching on the scientific as well as the societal and ethical issues raised by this research. Should broad public support be achieved within a jurisdiction, and if local policies and regulations permit, a specialized scientific and ethical oversight process could weigh whether the scientific objectives necessitate and justify the time in culture beyond 14 days, ensuring that only a minimal number of embryos are used to achieve the research objectives.
Background and rationale for ISSCR recommendation 2.2.2.1
The ISSCR recommendation 2.2.2.1 suggests that culturing human embryos beyond the current 14-day limit would require broad public support, local policies and regulatory permits, and a specialized scientific and ethical oversight process. The current limitation for in vitro human embryo culture up to 14 days was proposed by the Warnock Committee in 1984 37 , and since, there has been an international consensus regarding this time point. The justification for this time point is controversial and was set to ease public concern regarding human embryo research. Fourteen days occur before the formation of the primitive streak, which begins on day 15. Fourteen days is also the last developmental time point when identical twins can be formed and, therefore, is often considered by some organizations as the time at which individual identity is formed. In 1984, methods to culture human embryos beyond 14 days did not exist.
Today, techniques exist to culture human embryos beyond 14 days (Fig. 3). Magdalena Zernicka-Goetz’s group developed mouse in vitro systems for studying mouse implantation 38 and demonstrated that the system can be used to study human embryo post-implantation stages until day 14 39 . The group was forced to conclude their studies on day 14 due to the 14-day limit on the culture of human embryos. Jacob Hanna’s group successfully cultured mouse embryos for up to 6 days in culture from the pre-gastrulation stages until advanced organogenesis up to day 11 (mouse gestation period is 18–20 days) 40 . If ethical concerns are addressed, research groups hope to study human development and disease using human embryos in the same system. Studying human embryo development beyond day 14 will be beneficial for understanding human embryo gastrulation and specification of the three germ layer lineages. Extending the 14-day limit also will be important to study and minimize miscarriage and aid with in vitro fertilization (IVF) techniques.
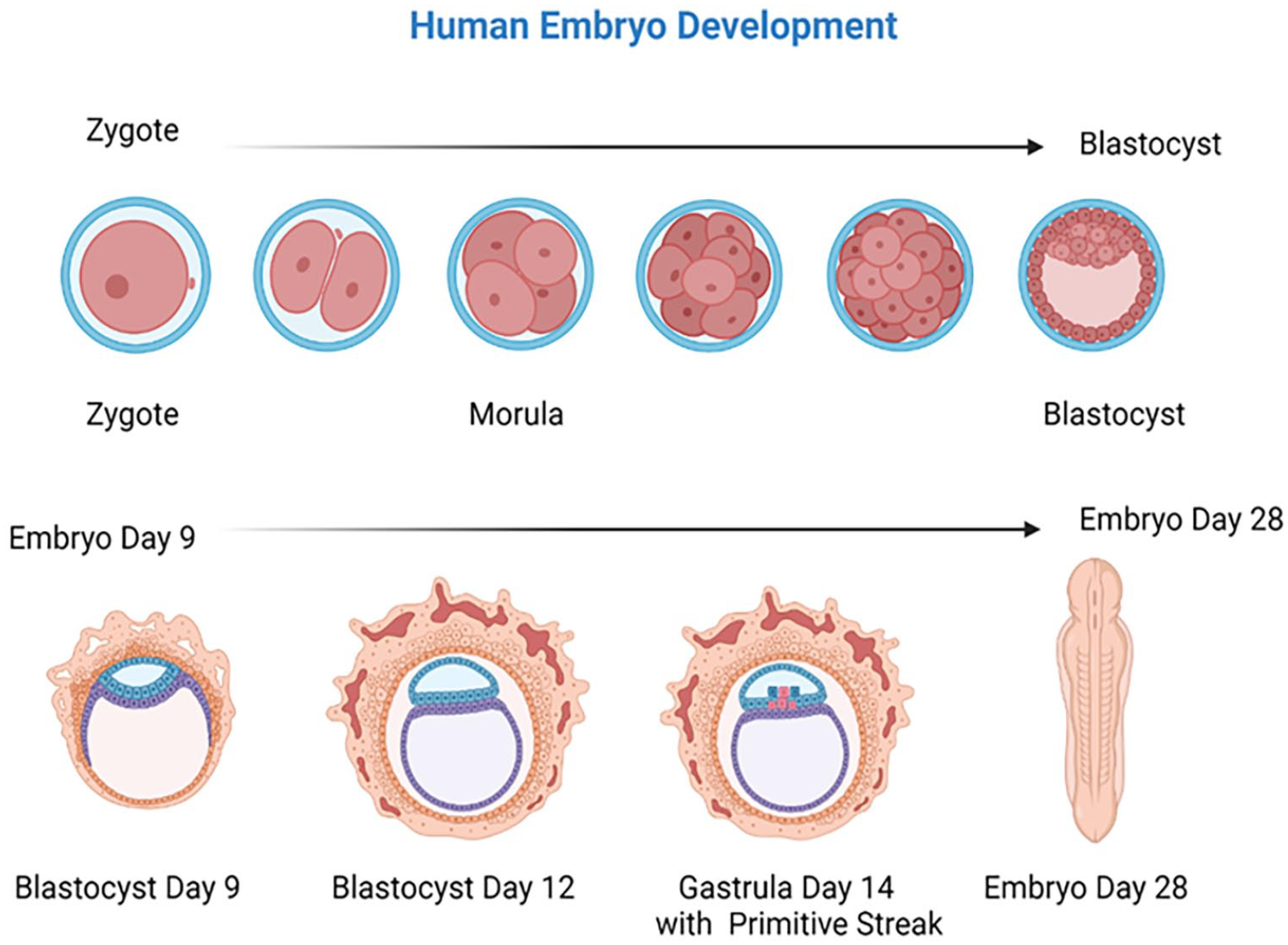
Human embryo development from zygote to day-28 embryo. Current restrictions on growing human embryos in tissue culture limit growth to day 14 before the formation of the primitive streak at day 15. Extending the 14-day limit to days 15–28 would be beneficial to understanding human embryo gastrulation and to understand why miscarriages occur and aid the development of in vitro fertilization techniques.
ASNTR recommendation
Adopting the ISSCR Recommendation 2.2.2.1 guideline will be an important step toward the progression of human embryo studies. Research groups would be responsible for adhering to local policies and consulting with ethical oversight committees to proceed incrementally and ethically. In addition to adopting the guideline, a new in vitro limit should be set, as a new limit has not been proposed in the guideline and remains a drawback to the recommendation. The new in vitro limit for culturing human embryos should address public concerns regarding human embryo research. The use of alternative model systems to study human embryo development, such as human ESC- and iPSC-derived embryo models, should be encouraged whenever possible. These models have been effective in closely replicating human embryo development until day 14 41 , can be used to study further stages of development, and consider the current ethical concerns with direct usage of human embryos. The ASNTR Task Force supported the adoption of ISSCR Recommendation 2.2.2.1.
Topic 4: Mitochondrial Replacement Techniques
ISSCR recommendation 2.2.3
Further research is needed to refine and assess the safety and efficacy of mitochondrial replacement techniques (MRT), including minimizing the risk of mitochondrial carryover and disruptions to the interaction between mitochondrial and nuclear genomes. In addition, research on polar body transfer techniques and the use of mitophagy or genome editing is needed to reduce or eliminate pathogenic mitochondrial DNA (mtDNA). Such research should be subjected to review by a special oversight process.
Background and rationale for ISSCR recommendation 2.2.3
The mitochondrial proteins that are necessary to perform functions for producing energy for the cell are transcribed from both nuclear DNA and mtDNA. mtDNA only contains 37 genes and is a closed circular molecule. The mitochondria found in the embryo originate from the oocyte cytoplasm 42 ; therefore, these mtDNA diseases are not sex-specific but rather inherited by all offspring. Mutations within the mtDNA can be detrimental to various metabolic pathways. In recent decades, over 300 ribosomal RNA (rRNA) and transfer RNA (tRNA) genes, as well as over 300 points in coding and control regions, have been identified 43 . High levels of mutant mtDNA can lead to heritable and often fatal diseases for which there are no cures 44 , leading prospective parents who are afflicted and wish to conceive to pass these mutant mtDNA onto their children. Laws and regulations for gene editing vary between regions and countries.
The United Kingdom (UK) was the first to approve and pass legislation in 2015 that allowed MRT 45 . Within the UK legislation, there are policies on compensation for oocyte donors and for an egg donor registry system, secure contact information for children born using donor gametes, and regulation for preventing genetic diseases on a case-by-case basis46,47. Only licensed clinics can perform MRT, and it is only allowed in limited types of cases 48 . Within the US, oocyte donation is allowed, but regulations differ between states. US legislation prohibits the Food and Drug Administration (FDA) form federally funding applications that include a human embryo that was intentionally created or modified to include heritable genetic modification 31 . Despite the availability of donated eggs, current legislation limits the use of MRT in both research and clinical applications. In addition, there are professional societies like the American Society of Reproductive Medicine that have guidelines on financial compensation for oocyte donors or best practices, but these are non-binding policies 47 . Mexico, on the other hand, has no national regulations for oocyte donation or regulation of human genetic information 49 . The lack of explicit federal regulations allowed for a controversial MRT performed in Mexico by a US-based physician 50 . Thus, differing national regulations may impact the ability to collaborate in stem cell research and provide patient care in the same manner across nations.
Types of MRTs
A possible prevention strategy for mtDNA disease is to transfer nuclei of at-risk zygotes and oocytes to unaffected and enucleated eggs or zygotes from donors (Fig. 4). Currently, the two major techniques at the forefront of assisted reproductive technology (ART) are pronuclear transfer (PNT) and maternal spindle transfer (MST), requiring micromanipulation.
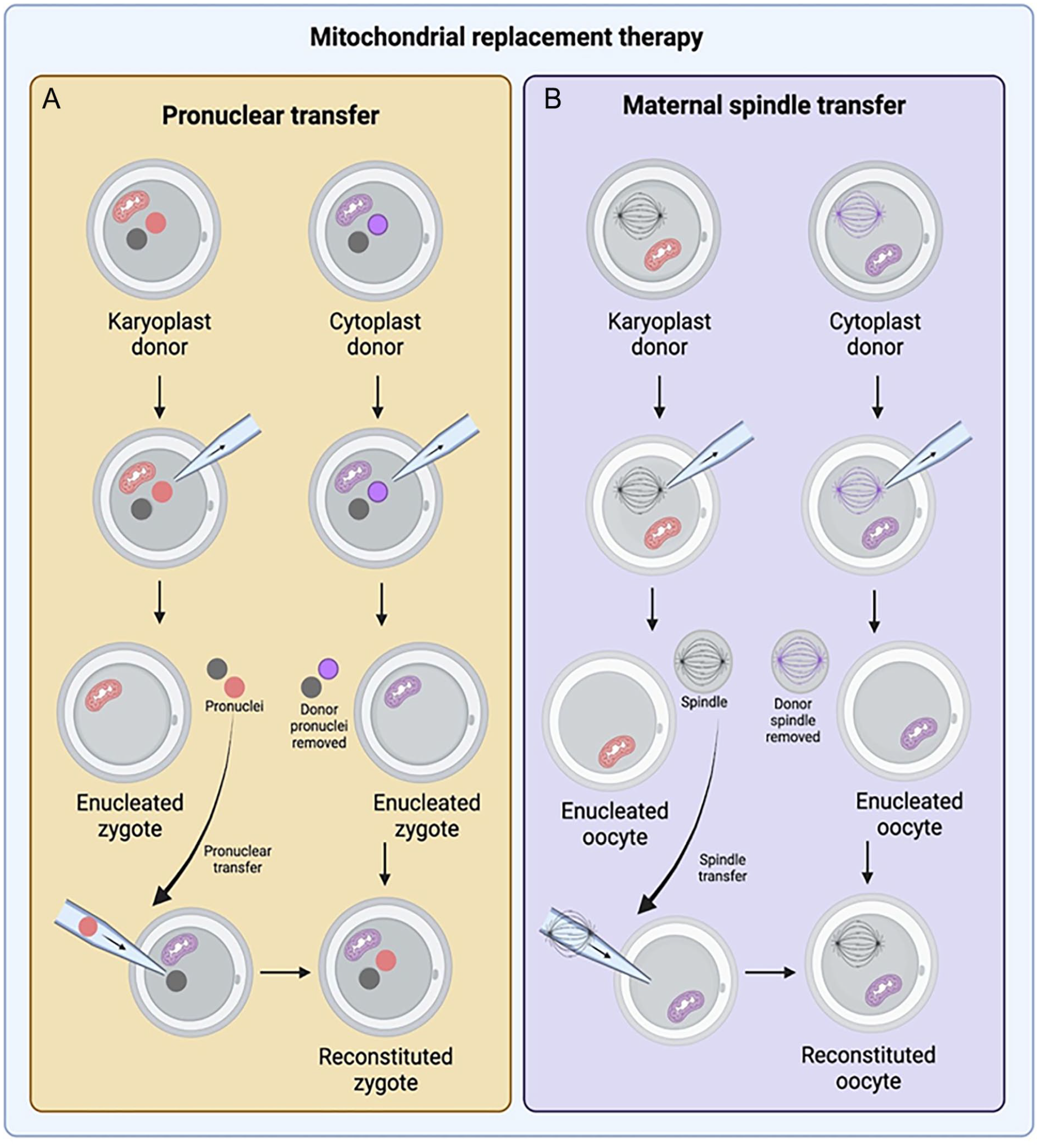
Mitochondrial transfer by (A) pronuclear transfer (PNT) and (B) maternal spindle transfer (MST). For PNT, pronuclei from the at-risk zygote (pink) are removed and transferred to the disease-free donor’s enucleated zygote (purple). For MST, the metaphase II spindle complex of the at-risk oocyte (black/pink) is removed and transferred to a disease-free donor’s enucleated oocyte (purple).
PNT requires the isolation and transfer of pronuclei of at-risk zygotes to another enucleated disease-free donor zygote 51 . The resulting zygote will contain healthy donor mtDNA and DNA from both parents and are potentially transferable 43 . The advantage of PNT is that both maternal and paternal genomes are within the pronuclei. During the first mitosis, the assembly of a normal bipolar spindle will make transmission of chromosomes to daughter cells more likely during the division to the two-cell stage 52 . Optimization of PNT leads to enhanced development of blastocysts without incidence of aneuploidy or gene expression patterns. In addition, the carryover of mutant maternal mtDNA is less than 2.0%43,53. Disadvantages of PNT include decreased blastocyst development rate compared to typical fertilized embryos, a small risk of aneuploidy, and ethical concerns related to utilizing this technique 52 . In China, an infertile woman underwent ART using PNT, and it led to an initial triplet pregnancy. Ultimately, the pregnancy resulted in no live births due to selective fetal reduction, respiratory distress in one of the two remaining fetuses, and cord prolapse in the last fetus 54 .
MST operates by isolating and transferring the metaphase II spindle complex of an at-risk oocyte to an enucleated donor egg43,51. The reconstituted and fertilized oocyte is a potentially transferable embryo. Non-human primate studies have been successful with viable offspring. Comparable postnatal growth and development appear unaffected in non-human primates55,56. These zygotes have normal fertilization rates, blastocyst development, and ESC isolation rates 53 , and the carryover of mutant maternal mtDNA is less than 1.0% 43 . As aforementioned, a controversial MRT case in Mexico led by a US-based physician was performed using MST. The woman previously had four miscarriages, and two children died of MTATP6 mutation in mtDNA. The use of MST resulted in a son, who was healthy at 7 months of age. At 7 months old, the boy’s mtDNA mutation load in his tissues was 2.36%–9.23%, which was much lower than that in her previous two deceased children. No further information on the boy’s health has been reported 57 .
Long-term impacts
Currently, there are no consistent long-term follow-ups after MRT; therefore, it is difficult to assess the long-term implications that arise from ARTs. Due to the lack of knowledge surrounding children born from MRT and the mitochondrial diseases being prevented, a long-term follow-up plan would help MRT research better address any long-term impacts following the procedure. These long-term impacts include the presence of the mitochondrial disease and sequelae. Long-term follow-ups should be specific to the mitochondrial disease at hand and coordinated between the physician and parents during childhood. Once the child is legally competent and there is no evidence of disease, follow-ups may be refused 58 .
ASNTR recommendation
The ASNTR Task Force found that the ISSCR Recommendation 2.2.3 on MRT offered several advantages and disadvantages. The advantages discussed included 1) the ability to treat heritable and possibly fatal mtDNA diseases, 2) the opportunity to explore and refine the ART techniques discussed earlier to make them safe and efficacious before clinical application, 3) the requirement of a special oversight process, and 4) the encouragement for further research on MRTs in humans to better understand the techniques and clinical implications. The disadvantages discussed included 1) the risk of mitochondrial carryover still being possible; 2) the disruption of mitochondrial and nuclear genomes; 3) the ethical implications of accessibility of resources for the ARTs, genetic modification of germline, and genetic link to parents and donor; and 4) the current laws and regulations restricting research in certain countries. Despite these disadvantages, the task force agreed to endorse this recommendation due to the need to address and support additional research to better understand MRT as a potential therapeutic for treating mtDNA disease. Further research would help ensure the safety and efficacy of MRT before clinical application of the ART. The ASNTR Task Force ultimately endorsed the ISSCR Recommendation 2.2.3 on MRT.
Topic 5: Emerging Categories of Embryo Research That Merit Close Review—Heritable Genome Editing
ISSCR recommendation 2.2.3.1
Until there is further scientific clarity on how to achieve desired genetic alterations, additional evidence for safety, and wider discussion and consensus on ethics (ie, whether it should be done and, if so, under which circumstances), any attempts to edit the mitochondrial genome or modify the nuclear genome of human embryos for the purpose of human reproduction is premature and should not be permitted at this time.
Background and rationale for ISSCR recommendation 2.2.3.1
With the emergence of genome editing, its potential uses in human embryos have sparked an important debate surrounding its ethics. Currently, there are many diseases, such as cystic fibrosis, alpha-1 antitrypsin, hemophilia, beta thalassemia, sickle cell, and infections, that potentially could be prevented through gene therapies 59 . For example, cystic fibrosis, a genetic disease that affects the lungs and pancreas, among other organs, causes difficulty breathing and increased susceptibility to pneumonia. This recessive disease is caused by mutations in both copies of the cystic fibrosis transmembrane conductance regulator (CFTR) gene. There is potential for this disease to be prevented with gene therapy prior to or during pregnancy by targeting the CFTR 60 . In vitro research has successfully utilized clustered regularly interspaced palindromic repeats (CRISPR)/Cas9 to inactivate CFTR in human epithelial cells 61 , thus creating the possibility for human translation.
Various methods for generating genetic alterations are currently in development. Of the more prevalent methods is the CRISPR/Cas9 system, which functions by attaching itself to the target DNA sequence with the help of a guide RNA and then excising the target DNA at that location 62 (Fig. 5). The targeted DNA can undergo a variety of processes to disable, alter, replace, or remove the target sequence depending on the tools in place. While CRISPR has demonstrated precise targeting abilities, it is still a developing biotechnology with potential off-target effects 63 . These off-target effects may occur when CRISPR/Cas9 cleaves a non-targeted genomic site, and the effects of these off-target events can be detrimental.

There are various methods for generating genetic alterations. One of these methods is the CRISPR/Cas9 system, in which gene editing of the human zygote and germline transfers edited genes to subsequent generations.
In 2015, Liang et al. performed the first set of experiments using CRISPR-associated systems in human tripronuclear zygotes and showed that CRISPR cleaved specific genes in embryonic cells 64 . The embryos experienced mosaicism, development of two or more genetically unique sets of cells in the body, and off-target cleavage in some cells. However, subsequent studies minimized mosaicism by modulating the cell cycle stage during editing 65 . Together, these experiments highlight the potential for gene editing in humans at early cell stages and the associated risks.
In 2018, a researcher crossed an ethical line by allowing two genetically altered embryos to develop to term, making them the first genetically altered humans 66 . The researcher used a CRISPR system designed to target and delete 32 base pairs from the CCR5, the gene responsible for coding a protein necessary for human immunodeficiency virus (HIV) to infect T-cells. Some humans, particularly a select few in northern Europe, are born without this gene and have been shown to have HIV infection resistance with no known detrimental effects 67 . The goal of this research was to produce humans that would be resistant or immune to the variants of HIV that require the CCR5 protein. At the time, the CRISPR technology was unable to make this precise change without errors. Reportedly one of the children was a heterozygote, and both children experienced mosaicism. The CRISPR system did not delete 32 base pairs but instead made numerous changes to CCR5. Those changes would theoretically disable the CCR5 protein, but these genomic edits have not previously been recorded in humans, and the true effect of these edits is unknown. This research prompted outrage from numerous communities.
ASNTR recommendation
Risks such as off-target effects, improper DNA repair, and downstream genetic effects of genome editing remain to be addressed thoroughly. Besides the safety concerns, there are ethical concerns such as consent, human rights, religious views, availability, and equality. With all these concerns in mind, the ISSCR recommendation 2.2.3.1 was accepted by the ASNTR Task Force. While potentially preventable diseases will remain a risk under this recommendation, this does ensure that unintended and off-target effects are not caused in humans. The recommendation also ensures that sanctioned research can be done safely and that no ethical concerns will be violated. When considering all the potential risks, a clear consensus in favor of this recommendation was reached.
Topic 6: Classifying Stem Cell-, Cell-, and Tissue-Based Interventions
ISSCR recommendation 3.1.1
Stem cells, cells, and tissues that are substantially manipulated or used in a non-homologous manner must be proven safe and effective for the intended use before being marketed to patients or incorporated into standard clinical care.
Background and rationale for recommendation 3.1.1
The therapeutic use of manipulated stem cells, cells, or tissues for non-homologous treatments is complex, speculative, and has potential health, ethical, and regulatory risks to recipients68,69. There is a need for products to be thoroughly tested in preclinical and clinical studies and evaluated by regulators before these products are distributed as drugs, biologics, and advanced-therapy medicinal products 70 . Manipulated stem cells, cells, or tissues are classified based on the degree of manipulation: minimally manipulated, substantially manipulated, or for non-homologous use71,72.
Minimally manipulated stem cells, cells, and tissues generally are non-expanded cells that have been isolated from biological material, where processing does not alter the relevant biological characteristics of the tissue’s utility for reconstruction or repair 73 . Minimally manipulated cells and tissues are subjected to fewer regulatory requirements. Despite available literature containing varied protocols and studies performed in vastly different clinical settings, minimally manipulated stem cells have been shown to be safe and beneficial in the short term74–76. For example, several studies have examined autologous minimally manipulated adipose-derived mesenchymal stem cells (MSCs) for treating osteoarthritis and tissue preparation used for structural support 77 and have concluded the treatment to be safe and successful in patients78–80. These adipose-derived MSCs commonly are enzymatically extracted from fat lipoaspirates as a stromal vascular fraction, followed by ex vivo expansion, which often lead to significant cellular senescence and decreased multipotential and regulatory challenges 81 . Novel methods have been developed for harvesting and processing adipose tissue without the need for enzymatic treatment or expansion. These methods lead to clinical utility for autologous minimally manipulated adipose-derived MSCs suitable for direct injection in the clinic or cryopreservation for future use 82 .
Challenges of using minimally manipulated cells include optimizing cell isolation techniques, developing advanced devices for delivery systems, and defining standardized protocols to ensure long-term safety and efficacy. In situ tissue engineering proposes a new approach for the use of minimally manipulated cells in the operating room75,83,84. Unlike other therapeutic products containing cultured cells, minimally manipulated stem cells, cells, and tissues do not require authorization in order to be marketed, making their safety profile an attractive therapeutic option. With these types of cells and tissue, the responsibility rests on the clinician to invite independent scrutiny of the manipulation process, such that scientific and regulatory experts can determine the proper level of regulatory oversight. Since it is advisable to contact legally authorized regulatory bodies if there is uncertainty or disagreement about the regulatory status of particular therapeutic interventions, this lack of oversight is dependent on the ethics of the clinician.
In contrast, substantially manipulated stem cells, cells, and tissues are subjected to processing steps that alter their original structural or biological characteristics (eg, isolation and purification processes, tissue culture and expansion of the cells, genetic manipulation, etc) 71 . For example, the enzymatic digestion of tissue is considered a substantial manipulation and must therefore be subject to regulatory oversight as an advanced-therapy medicinal product85–87. The safety and efficacy of substantially manipulated cells and tissues cannot be assumed because the composition of the intervention may differ from the original tissue source. Demonstration of safety and effectiveness will depend on the particular intervention and the specific condition targeted, and therefore subject to approval by the US FDA.
Cells and tissue used in a homologous manner, whether substantially manipulated or not, means that they are used to maintain the original function in the same anatomical environment. Cells and tissue used in a non-homologous manner, whether substantially manipulated or not, means that they are repurposed to perform a different function in the recipient prior to being removed, processed, and transplanted or otherwise delivered from the donor. While there are potential benefits to non-homologous use of cells and tissue, these also can pose serious risks.
Adipose-derived cell therapy has demonstrated a favorable safety profile given the underreporting of adverse events88,89. There is a need for future research to focus on higher-quality reporting of adverse events and adequately controlled trials with rigorous and comprehensive adverse event reporting. Only adverse events that are looked for will be found. For example, there are well-documented reports of vision loss with the use of adipose-derived stromal cells to treat macular degeneration 90 . A meta-analysis of the safety profile and efficacy of MSC therapy for acute myocardial infarction and ischemic heart failure also found that the clinical trials reported highly variable definitions and descriptions of adverse events 91 .
ASNTR recommendation
The ISSCR recommendation 3.1.1 is as follows and depends on the degree of manipulation of the stem cells, cells, and tissues (Fig. 6). For minimally manipulated stem cells, cells, and tissues, the ISSCR recommends that the FDA and other international regulators release detailed standards to delineate when manipulation of cell-based products can no longer be considered minimal or their use homologous and must be subject to regulatory oversight as an advanced-therapy product. For substantially manipulated stem cells, cells, and tissues, the ISSCR recommends that they be evaluated by national regulators as drugs, biologics, and advanced-therapy medicinal products. For non-homologous use of stem cells, cells, and tissues, the ISSCR recommends evaluation by regulators following the completion of well-designed and carefully controlled preclinical and clinical studies. The ASNTR Task Force supported this recommendation.

Recommendations for FDA approval or no FDA approval before a therapeutic can be marketed depend on the degree of manipulation of the stem cells and derivative cells and tissues. The reprogramming of cells for cellular therapy is considered to be a substantial manipulation of stem cells, cells, and tissues and requires FDA approval, while minimally manipulated stem cells, cells, and tissues do not require authorization to be marketed.
Discussion
Advances in biomedical technologies have extended the boundaries of previously established regulatory guidelines pertaining to stem cell research. Such guidelines included limiting the study of hPSCs and their derivatives from use under various conditions. Other guidelines constrained the use of hPSCs in creating human–animal chimeras because of the potential contribution of human stem cells to the brain and germline. Advances in technology raised concerns about the use of gene-editing approaches that would introduce permanent changes in the human germline and concerns regarding human mitochondrial transfer and exchange.
These and other issues were assessed by the ASNTR Task Force to review the new guidelines recommended by the ISSCR for stem cell research and determine whether these guidelines should also be adopted by the ASNTR. After the ASNTR Task Force endorsed the support of the ISSCR guidelines, these recommendations were each put to a vote by the ASNTR membership. The majority of those voting were in favor of accepting the ISSCR recommendations.
Topic 1: Studies Transplanting Human Stem Cells or Their Direct Derivatives Into the CNS of Animals
The ASNTR Task Force found reasons to adopt ISSCR guideline 2.2.1.1. We recommend that the exact composition of the committee does not need to be consistent across institutions but should focus on ensuring that a comprehensive array of perspectives is considered. Furthermore, it was suggested that the animal review committees should increase the diversity of perspectives by including biosafety and bioethics as considerations for approving human–animal stem cell chimera protocols.
Topic 2: Human–Animal Chimeric Embryo Research
The ASNTR Task Force found reasons to adopt ISSCR guideline 2.2.2.2. Adopting this recommendation will ensure that chimeric embryo and in utero research should proceed for the minimum time necessary to achieve the scientific aim.
Topic 3: Culture of Human Embryos Beyond Formation of the Primitive Streak or 14 Days
The ASNTR Task Force found reasons to adopt ISSCR guideline 2.2.2.1. In addition to adopting the guideline, we recommend that a new in vitro limit should be set, despite no proposal for such a new limit in the guideline, a drawback to the recommendation. This discussion on a new limit should address public concerns regarding human embryo research.
Topic 4: MRTs
The ASNTR Task Force found reasons to adopt ISSCR guideline 2.2.3. There is a need for additional research to better understand MRT as a potential therapeutic to treat mtDNA disease. Further research would help ensure the safety and efficacy of MRT before clinical application of the ART.
Topic 5: Emerging Categories of Embryo Research That Merit Close Review—Heritable Genome Editing
The ASNTR Task Force found reasons to adopt ISSCR guideline 2.2.3.1. Until there is further scientific clarity, any attempts to edit the mitochondrial genome or modify the nuclear genome of human embryos for the purpose of human reproduction are premature and should not be permitted at this time. The recommendation also ensures that sanctioned research can be done safely, and no ethical concerns will be violated.
Topic 6: Classifying Stem Cell-, Cell-, and Tissue-Based Interventions
The ASNTR Task Force found reasons to adopt ISSCR guideline 3.1.1. For minimally manipulated stem cells, cells, and tissues, we recommend following FDA guidelines for oversight as an advanced therapy product. For substantially manipulated stem cells, cells, and tissues, we recommend that the investigational product should be evaluated by national regulators as drugs, biologics, and advanced-therapy medicinal products. For non-homologous use of stem cells, cells, and tissues, we recommend it be evaluated by regulators following the completion of well-designed and carefully controlled preclinical and clinical studies.
This concludes the recommendations for incorporating new guidelines for research on stem cells by the ASNTR based on those recommended by the ISSCR.
Footnotes
Acknowledgements
The authors acknowledge the work of the ISSCR Task Force in developing new research guidelines for their membership, which provided the basis for our discussions.
Author Contributions
All authors were members of the American Society for Neural Therapy and Repair Task Force to review and evaluate new research guidelines by the International Society for Stem Cell Research that pertained to research by members of the ASNTR. Based on discussions by the task force, authors AR, AS, IHC, PS, and SRV wrote sub-sections of the manuscript, which was then edited by ATC, GLD, KF, AWG, AMP, JR, PRS, L-RZ, LVZ, and WCL.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paul Sanberg is the editor of the journal Cell Transplantation but did not participate in the journal’s editorial review process for acceptance of this manuscript for publication. The authors declare no other conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
