Abstract
Cerebral ischemic stroke is a major cerebrovascular disease and the leading cause of adult disability. We and others previously demonstrated that transplantation of human Wharton’s jelly mesenchymal stromal cells (WJ-MSCs) attenuated neuronal damage and promoted functional improvement in stroke animals. This study aimed to investigate the protective effects of human WJ-MSC exosome (Exo) transplant in cellular and rat models of cerebral stroke. Administration of Exo significantly antagonized glutamate-mediated neuronal loss and terminal deoxynucleotidyl transferase (TdT)-mediated dUTP-X nick end labeling (TUNEL) in rat primary cortical neuronal cultures. Adult male rats underwent a 60-min middle cerebral artery occlusion (MCAo); Exo or vehicle was injected through the tail vein 5–10 min after the MCAo. Two days later, the rats underwent a series of behavioral tests. Stroke rats receiving Exo developed a significant improvement in locomotor function and forelimb strength while reductions in body asymmetry and Bederson’s neurological score. After the behavioral test, brain tissues were harvested for histological and quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) analyses. Animals receiving Exo had less infarction volume, measured by 2,3,5-triphenyl tetrazolium chloride (TTC) staining. Transplantation of Exo increased the expression of protective neurotrophic factors (BMP7, GDNF) and anti-apoptotic factors (Bcl2, Bcl-xL) in the ischemic brain. These findings suggest that early post-treatment with WJ-MSC Exo, given non-invasively through the vein, improved functional recovery and reduced brain damage in the stroke brain.
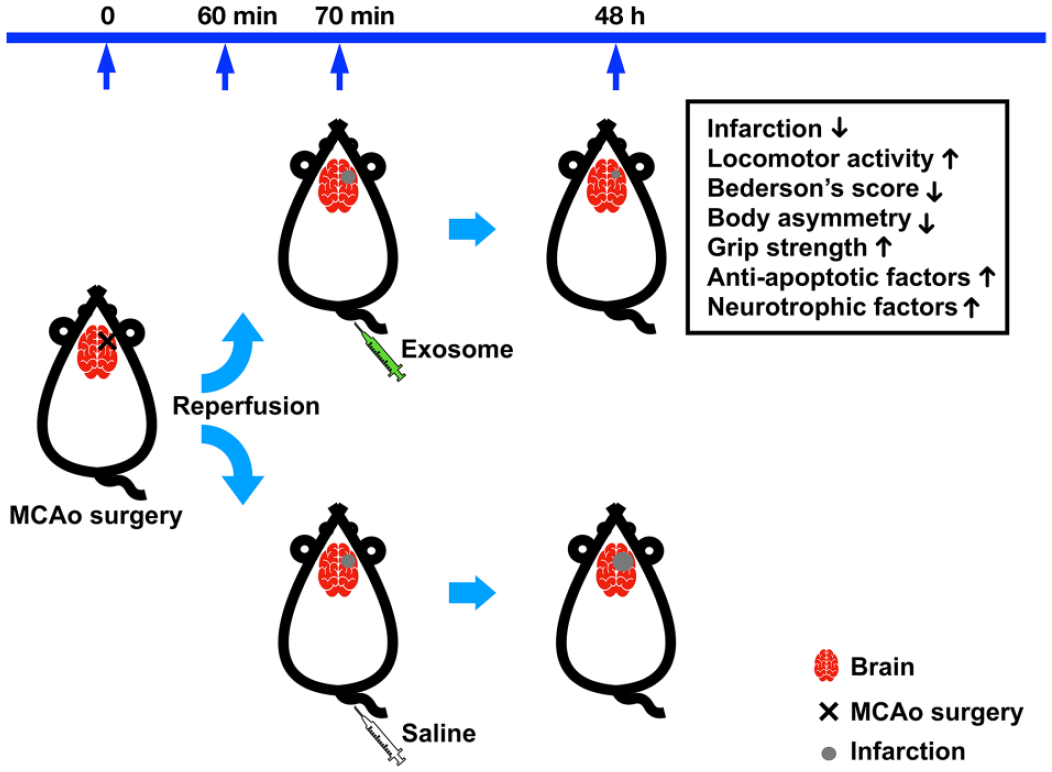
Introduction
Cerebral stroke is the second most fatal disease worldwide, with approximately 75% of its survivors experiencing permanent disabilities. It imposes heavy economic burdens on patients and society1,2. Cerebral ischemic stroke, usually caused by a blood clot that blocks blood circulation in the brain, accounts for 87% of the stroke population 3 . Cerebral ischemia leads to pathological reactions such as mitochondrial dysfunction, glutamate excitotoxicity, inflammation, and apoptosis, eventually causing neuronal damage in the brain4,5. Currently, the treatment for cerebral ischemic stroke is limited to rapid recanalization by endovascular thrombectomy and intravenous thrombolysis by injection of tissue plasminogen activator 6 . Medications to ameliorate cerebral ischemia-mediated neuronal degeneration are still limited. Developing adjunctive therapies to facilitate neuronal restoration in post-cerebral ischemic stroke is crucial and necessary.
Increasing evidence supports that mesenchymal stromal cell (MSC) transplant promotes neurogenesis, angiogenesis, and functional recovery in experimental stroke models7,8. We previously reported that intracerebral transplantation of human placenta–derived MSCs reduced activation of microglia and brain infarction in the stroke brain 9 . These transplants released trophic factors or cytokines, leading to protection, anti-inflammation, and repair10 –14. MSCs can self-renew and have the potential to differentiate into various cell lineages of three germ layers: endoderm (endothelial cells and hepatocytes), mesoderm (adipocytes, chondrocytes, and osteoblasts), and ectoderm (neurons and glia cells)15,16. MSCs can be obtained from various tissues such as adipose tissue, bone marrow, cord blood, placenta, or tooth buds.
The human umbilical cord (hUC) has been increasingly considered a source of MSCs due to its abundant supply from discarded tissues and less ethical concerns17,18. We previously demonstrated that transplantation of MSCs from the human Wharton’s jelly of the umbilical cord [Wharton’s jelly mesenchymal stromal cell (WJ-MSCs)] attenuated neuronal damage and promoted functional improvement in stroke animals 19 . We also reported that the protective effect of these cells was indirectly mediated through the production of protective factors from the transplants 19 .
The protective effects of MSCs may also be attributed to the exosomes (Exos) in the graft cells. A growing body of studies has suggested that MSC-derived Exos hold promise as a treatment for cerebral ischemic stroke20,21. Exo treatment reduces neuroinflammation22,23, oxidative stress 24 , and apoptosis 25 while promoting angiogenesis 26 and neuroregeneration 27 in the damaged brain. Further research is essential for a complete understanding of the underlying mechanism and to translate these findings into effective treatments for ischemic stroke patients.
Exos are lipid bilayer vesicles, typically 50–150 nm in diameter, produced by almost all types of cells, including MSCs, immune cells, and cancer cells 28 . Their surface is decorated with transmembrane proteins such as CD9, CD63, and CD81. The interior of Exos contains a diverse array of proteins, DNA, RNA, metabolites, and other bioactive substances, which can vary depending on the type of parental cells 29 . Exos are formed as intraluminal vesicles (ILVs) in multivesicular bodies (MVBs) through the inward budding of endosomal membranes 30 . When MVBs fuse with the cell membrane, Exos are released to the extracellular space as vesicles, through which they can transfer their cargo to other cells by membrane fusion or vesicle internalization 30 . In addition, Exos can cross the blood–brain barrier (BBB), as demonstrated in the rodent models that received intranasal or intravenous delivery of Exos loaded with catalase, recombinase, siRNA, or curcumin31 –34. Due to their stability, biocompatibility, low immunogenicity, functional contents, and ability to cross BBB, Exos have the potential to be used as cell-free therapeutic agents for cerebral ischemic stroke.
This study aimed to investigate the protective effects of Exos derived from human WJ-MSCs in a rat model of cerebral ischemic stroke. We found that intravenous administration of WJ-MSC Exos after transient middle cerebral artery occlusion (MCAo) improved neurological function recovery and reduced brain damage in stroke rats. Our findings support that human WJ-MSC-derived Exo is potentially useful for the early post-treatment of ischemic stroke.
Materials and Methods
Human WJ-MSCs
Newborn umbilical cord tissues were collected from term deliveries of healthy donors who had signed the written informed consent. This study was approved by the Institutional Review Board (IRB) of Fu Jen Catholic University (approval no. C111157) and conducted in accordance with the Declaration of Helsinki. Donors’ peripheral blood samples were tested negative for hepatitis B virus (HBV) antigens and antibodies against HIV1, HIV2, hepatitis C virus (HCV), and syphilis. The isolation and expansion of WJ-MSCs was undertaken within a Good Manufacturing Practice (GMP)-grade cell processing unit (#EZCPi ONE, YJ Biotechnology, New Taipei City, Taiwan) approved by the Taiwan Food and Drug Administration (TFDA) under the regulations of Good Tissue Practice (GTP). The umbilical cord specimen was collected in a sterile bottle filled with Hank’s balanced salt solution (HBSS) supplemented with antibiotics (streptomycin 100 μg/ml, penicillin 100 IU/ml) and processed for culturing WJ-MSCs within 24 h. After removing the umbilical arteries, veins, and adventitia, we sliced Wharton’s jelly tissues into pieces smaller than 1 mm3. The slices were treated with collagenase type II at 37°C in a humidified incubator with 5% CO2 for 18 h. Cells were harvested by centrifugation and grown in Iscove’s Modified Dulbecco’s Medium (IMDM) supplemented with 5% human platelet lysate (hPL) at 37°C in a humidified incubator with 5% CO2. Flow cytometry analysis confirmed the expanded cells expressing high levels of MSC surface markers (CD73, CD90, and CD105) but no hematopoietic stem cell marker (CD34).
Exosome Purification
The conditioned media of human WJ-MSCs cultures was harvested and centrifuged at 4,000 × g for 10 min, followed by filtration through a 0.22-μm filter to remove cell debris and larger particles. The clarified conditioned media was circulated through a tangential flow filtration (TFF) device (0.05 μm pore size) by a peristaltic pump to reduce the volume by 10 folds. The ultrafiltrated media was subjected to exosome (Exo) purification using the exoEasy Maxi kit (#76064; Qiagen; Hilden, Germany) per the manufacturer’s written instructions. Briefly, the ultrafiltrated media was mixed with an equal volume of binding buffer and applied into a membrane-based spin column. After centrifugation at 4,000 × g for 1 min, the column was washed once with 10 ml of washing buffer, followed by centrifugation at 4,000 × g for 5 min. The bound Exos were eluted from the column with 2 ml of elution buffer by centrifugation at 4,000 × g for 5 min. The eluates were concentrated in saline to 0.5 ml using an ultrafiltration spin column (100 kDa MWCO; Cytiva; Uppsala, Sweden). The purified Exos were aliquoted and stored at −80°C until use.
Western Blot Analysis
Lysates of human WJ-MSCs and Exos were prepared for Western blot analysis as previously described35,36. Briefly, WJ-MSCs (8 × 105 cells) or Exos (2 × 1010 ptc) were resuspended in 100 µl of phosphate-buffered saline (PBS) and mixed with 100 µl of 2× Laemmli buffer (125 mM Tris-HCl pH 6.8, 20% glycerol, 10% β-mercaptoethanol, 4% sodium dodecyl sulfate (SDS), and 0.004% bromophenol blue in distilled water), followed by passing through a 25-gauge needle 20 times to reduce viscosity. After being heated at 90°C for 10 min, the Laemmli buffer–treated samples were subjected to electrophoresis separation on 12% SDS-polyacrylamide gels. The separated proteins were transferred from the gel to a polyvinylidene fluoride (PVDF) membrane (#RPN303F; GE HealthCare; Buckinghamshire, UK) by electrophoresis. The membrane was then incubated with 10-ml blocking buffer (5% skim milk and 0.1% Tween-20 in PBS) for 1 h and then incubated with appropriate primary antibodies in 3-ml blocking buffer (in a 50-ml centrifuge tube) at 4°C overnight with gentle rotation. Six rabbit monoclonal antibodies against five exosome markers CD9 (1:2,000, #ab263091; Abcam), CD63 (1:2,000, #ab134045; Abcam), CD81 (1:2,000, #ab109201; Abcam), Hsp70 (1:2,000, #ab181606; Abcam), and TSG101 (1:2,000, #ab125011; Abcam) and one endoplasmic reticulum marker Calnexin (1:2,000, #ab133615; Abcam), and one mouse monoclonal antibody against a cellular housekeeping protein β-actin (1:3,000, #GTX629630; GeneTex) were used as primary antibodies. After washing with 0.1% Tween-20 (in PBS) three times for 10 min each, the membrane was incubated with horseradish peroxidase (HRP)-conjugated goat polyclonal antibodies against mouse IgG (1:2,500; #GTX213111-01; GeneTex) or rabbit IgG (1:2,500; #GTX213110-01; GeneTex) in 3-ml blocking buffer for 1 h, followed by the washing procedure described above. The target proteins on the membrane were detected using an enhanced chemiluminescence reagent (##GTX14698; GeneTex), and the light emission signals were captured by the CCD camera-based image system MultiGel-21 (TOPBIO, New Taipei City, Taiwan). The intensity of the detected images was quantified by ImageJ software (http://imagej.net).
Exosome Particle Analysis
The purified Exos were analyzed by nano-flow cytometry (nFCM) to determine particle size, concentration, and surface markers. Samples were diluted in PBS to the concentration, achieving a flow rate within an optimal range of 2,000–12,000 counts per minute. Based on the calibration curve, the particle size and concentration were determined by converting the flow rate and side scattering intensity using NanoFCM software (NanoFCM, Profession V2.315). For the detection of Exo surface markers, samples were stained with fluorescent dye–conjugated monoclonal antibodies against CD9, CD63, or CD81 at 37°C for 1 h. After centrifugation at 100,000 × g at 4°C, Exo pellets were washed once with PBS and resuspended in PBS for phenotype analysis by nFCM. In addition, the particle size, concentration, and surface markers of exosomes from 5% hPL-supplemented fresh medium and WJ-MSC conditioned medium were examined using the ZetaView MONO system (Particle Metrix; Ammersee, Germany).
Transmission Electron Microscopy
The Exo resuspension (10 μl, in PBS) was added to a carbon-coated grid for 60 s. The grid was blotted with filter papers to remove the excessive solution, followed by conventional negative staining with 1% uranyl acetate (10 μl) for 90 s. After blotting with filter papers, the grid was washed once with distilled water (20 μl) and left to air-dry for 20 min. Exo images were recorded using a transmission electron microscope (JEM-1400, JEOL) operated at an acceleration voltage of 120 kV.
Animals
Adult male Sprague-Dawley rats were purchased from BioLASCO Co., Ltd. (Taipei, Taiwan) and housed in an environment with controlled conditions: light on at 7:00 and off at 19:00, constant room temperature at 24 ± 2°C, and relative humidity at 50 ± 10%. All animals habituated for 2 weeks after their arrival and had free access to drinking water and autoclaved food. Animal experiments were conducted under the protocol approved by the Animal Research Ethics Board at the National Health Research Institutes in Taiwan (approval no. NHRI-IACUC-112034-A). We have carefully read and adhered to the ARRIVE guidelines.
In this exploratory study, we aimed to investigate whether human WJ-MSC Exos can attenuate neuronal damage caused by cerebral ischemic stroke. The sample size of each group in a two-sided t-test comparison was chosen based on assuming an alpha level of 0.05, a power of 90% chance of detecting the effect, and an effect size of 1.6 standard deviation units 37 . Given the attrition and unexpected loss of animals or data during the experiment, we increased the sample size by 20%. Therefore, a total of 12 rats per group was considered necessary. The random numbers were generated using the RAND function of Mac’s Numbers application to allocate rats to experimental groups. A total of 29 rats (male, 3 months old) were randomly separated into three groups: naive (n = 5), stroke surgery followed by the treatment with saline (n = 12), or Exos (n = 12). One animal of the saline-treated group died during the stroke surgery. The researchers who conducted the animal experiments were aware of the group allocation, but the animal care staff and the researchers who analyzed the data were unaware of it.
Primary Cortical Neuronal Cultures
The brain cortex tissues were aseptically collected from fetuses (at embryonic days 14–15) of timed-pregnant rats (Sprague-Dawley). After removing the meninges and blood vessels, the dissected cortices were pooled and treated with 0.05% trypsin at room temperature for 20 min. Cells were harvested by low-speed centrifugation and rinsed with pre-warmed Dulbecco’s modified Eagle’s medium (DMEM) to remove trypsin. Cell pellets were dissociated in the neuron culture medium by repeatedly pipetting and then grown in a 96-well plate, which was precoated with poly-
Immunocytochemistry
PBS was used in the washing step, and antibodies were diluted in PBS. The primary cortical neuron (PCN) cells grown in a 96-well plate were fixed with 4% PFA. After washing once, cells were permeabilized by incubating with 100 μl of blocking solution (composed of 5% bovine serum albumin, 0.1% Triton X-100, and 1× PBS) for 1 h. After washing once, cells were incubated with a mouse monoclonal antibody against MAP2 (1:500; #MAB3418; Millipore; Darmstadt, Germany) at 4°C overnight. After washing three times, cells were incubated with an Alexa Fluor 488-conjugated goat antibody against mouse IgG (1:1,000; Invitrogen) for 2 h. After washing three times, cells were examined by an inverted fluorescence microscope Nikon Eclipse Ti1 (Nikon) using an excitation wavelength of 450–500 nm and a detection wavelength of 515–565 nm (green). Images (10× magnification) were acquired from four fields per well by a camera DS-Qi2 (Nikon) and analyzed using NIS Elements AR 5.11 Software (Nikon).
Terminal Deoxynucleotidyl Transferase (TdT)-Mediated dUTP-X Nick End Labeling
The PCN cells grown in a 96-well plate were analyzed for DNA fragmentation using the In Situ Cell Death Detection Kit (#11684795910; Roche; Indianapolis IN, USA) per the manufacturer’s instructions. Briefly, cells were fixed with 4% PFA and, after washing once, incubated with 0.1% Triton-X 100 (in 0.1% sodium citrate) on ice for 2 min to permeabilize the cell membrane. After washing twice, cells were incubated with 50 μl of TUNEL reaction mixture for 60 min at 37°C in a humidified chamber. After washing three times, cells were examined by an inverted fluorescence microscope Nikon Eclipse Ti1 (Nikon) using an excitation wavelength of 450–500 nm and a detection wavelength of 515–565 nm (green). Images (20× magnification) were acquired from four fields per well by a camera DS-Qi2 (Nikon), and TUNEL+ cells were manually counted.
Transient MCAo
Rats (male, 3 months old; n = 24) were anesthetized by intraperitoneal (i.p.) injection with sodium pentobarbital (35 mg/kg). A burr hole of about 2–4 mm in diameter was made using a dental drill in the right squamosal bone. Afterward, common carotids were clamped bilaterally by arterial clips, and the right distal middle cerebral artery (MCA) was ligated with a 10-0 nylon suture. After 60-min ischemia, the suture and arterial clips were removed to restore blood supply and generate cerebral ischemia-reperfusion injuries. One animal died on the table during the surgery. Rats were administered with 0.5 ml of saline (n = 11) or human WJ-MSC Exos (1011 ptc/ml; n = 12) via the tail-vein injection 5–10 min after reperfusion. Rats were kept in an incubator at 37°C until recovering from anesthesia.
Locomotor Activity Test
Rats (naive, n = 5; MCAo/saline, n = 11, MCAo/Exo, n = 12) were individually placed in transparent acrylic chambers (42 cm × 42 cm × 30 cm; length × width × height), equipped with infrared sensors aligned in the horizontal and vertical directions (AccuScan Instruments, Inc.). After rats habituated for 30 min, the locomotor activity data were collected by recording the events of beam interruption for 2 h during the light cycle (2 p.m.–4 p.m.). Six parameters were measured, including horizontal activity (HACTV; the number of beam interruptions detected by the horizontal sensors), total distance traveled (TOTDIST; distance traveled by the animals), movement numbers (MOVNO; the counts of horizontal movements followed by a break lasting over one second), movement time (MOVTIME; the total time when the animal was detected moving), vertical activity (VACTV; the number of beam interruptions detected by the vertical sensors), vertical movement number (VMOVNO; the number of vertical movements followed by a period during which the animal must go below the level of the vertical sensor for at least one second), and vertical movement time (VTIME; the total time when the animal was detected moving vertically).
Body Asymmetry Test
Rats (naive, n = 5; MCAo/saline, n = 11, MCAo/Exo, n = 12) were examined for lateral movement using the elevated body swing test38,39 when suspended 20–30 cm above the table by lifting their tails. The frequency of the turning of the head or upper body opposite to the lesion brain side was counted in 20 consecutive trials. The stroke rats presented asymmetric lateral movement with almost 20 turns per 20 trials, while the naive rats showed almost equal frequency in each direction.
Modified Bederson’s Neurological Test
Rats (naive, n = 5; MCAo/saline, n = 11, MCAo/Exo, n = 12) were assessed for the degree of neurological deficits, graded on a scale of 0–3 when suspended 20–30 cm above the table by lifting their tails39,40. If rats stretch both forelimbs toward the table, grade 0 (no observable deficit) is assigned. If rats draw one forelimb back to the breast and extend the other forelimb straight, grade 1 is assigned. In addition to the behavior in grade 1, if rats exhibit decreased resistance to lateral push and display no circling, grade 2 is assigned. In addition to the behavior in grade 2, if rats twist the upper half of their body, grade 3 is assigned.
Grip Strength Test
Forelimb strength was assessed by a grip strength meter (#BIO-GS3; Bioseb). Rats (naive, n = 5; MCAo/saline, n = 7, MCAo/Exo, n = 6) were positioned horizontally over the grid of the grip strength meter to allow their right or left forepaws to attach to the top portion of the grid stably. When the rat grasped the grid, it was pulled by the root of the tail away from the grid. The maximal grip strength value displayed in grams was recorded by the grip strength meter. After three consecutive trials, the mean value of each forelimb of each rat was calculated.
2,3,5-Triphenyl Tetrazolium Chloride Staining
Two days after MCAo, rats were decapitated. The collected brains (MCAo/saline, n = 7, MCAo/Exo, n = 6) were sliced into 2-mm-thick sections, which were immersed in PBS supplemented with 2% 2,3,5-triphenyl tetrazolium chloride (TTC, #T8877; Sigma) at room temperature for 15 min and then fixed in 4% PFA for 20 min. The images of stained brain slides were taken with a digital scanner, and infarction areas were measured using the ImageJ software. The infarction volume was obtained by multiplying the sum of infarction areas and the average thickness (2 mm) of brain slides.
RNA Extraction
Cortical tissues of the infarcted or non-infarcted brain hemisphere (MCAo/saline, n = 11, MCAo/Exo, n = 12) were subjected to RNA extraction by Trizol Reagent (#15596-018; Ambion) according to the manufacturer’s instruction 41 . Briefly, each cortical tissue was lysed with 0.4 ml of Trizol reagent, and the lysate was homogenized by pipetting up and down several times. After incubation on ice for 5 min, the lysate was mixed with 80 µl of chloroform, followed by incubation on ice for 3 min. After centrifugation at 12,000 × g at 4°C for 15 min, the upper aqueous phase containing the RNA was transferred to a 1.5-ml microcentrifuge tube and mixed with 0.2 ml of isopropanol. After incubation on ice for 10 min, the sample was centrifuged at 12,000 × g at 4°C for 10 min. The precipitated RNA pellet was washed once with 0.4 ml of 75% ethanol, air-dried for 10 min, and resuspended in 30 µl of RNase-free water (#10977-015; Invitrogen). To degrade potentially contaminated DNA fragments, we treat the resuspended RNA with two units of DNase I (#M0303S, NEB) in 100 µl of 1× DNase I reaction buffer at 37°C for 10 min. RNA was then re-extracted by chloroform, re-precipitated by isopropanol, and washed by ethanol, as described above. Finally, the purified RNA was quantified by a NanoDrop spectrophotometer and stored at −80°C until use. One animal’s cortical samples of the MCAo/Exo group were excluded due to the failure of RNA extraction.
qRT-PCR Analysis
The mRNA levels of selected genes, listed in Table 1, were determined by quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) performed on an ABI StepOnePlus system. First, the purified total RNA of each sample (MCAo/saline, n = 11, MCAo/Exo, n = 11) was transcribed into cDNA using the RevertAid First Stand cDNA Synthesis Kit (#K1622; ThermoFisher Scientific). Briefly, the RNA (1 µg) was mixed with 1 µl of random hexamer primers (100 µM) in distilled water to a volume of 12 µl and then incubated at 65°C for 5 min, followed by chilling on ice. Next, 4 µl of 5× reaction buffer, 2 µl of dNTP (10 mM), 1 µl of RNase inhibitor (20 U/µl), and 1 µl of M-MuLV reverse transcriptase (200 U/µl) were added to the chilled mixtures. The total mixture (20 µl) was incubated at 25°C for 5 min, followed by 42°C for 60 min. Finally, the reaction was terminated by heating at 70°C for 5 min. A 40-fold dilution of the resulting cDNA was made in distilled water. The mRNA levels of most of the selected genes were determined by SYBR Green-based qPCR. Briefly, 2 µl of diluted cDNA, 10 µl of 2× SYBR Green PCR master mix (#K0371; ThermoFisher Scientific), and 2 µl of each primer (5 µM) were mixed in distilled water to a final volume of 20 µl. The PCR cycling program was set as follows: 50°C for 2 min and 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 15 s and annealing/extension at 60°C for 1 min. The primer sequences used for the PCR were designed by the Primer-3 program and listed in Table 1. The specificity of PCR products was verified by performing a melting-curve analysis (95°C for 15 s, 60°C for 1 min, and 95°C for 15 s) at the end of the PCR. The mRNA levels of GAPDH and BMP7 genes were determined using commercially available primers and probes per the manufacturer’s instructions. Briefly, a mixture containing TaqMan Fast Advanced Master Mix (5 µl), TaqMan probe (0.5 µl), distilled water (2.5 µl), and diluted cDNA (2 µl) was prepared. The PCR cycling program was set as follows: 95°C for 20 s, followed by 40 cycles of denaturation at 95°C for 1 s and annealing/extension at 60°C for 20 s. The mRNA levels of examined genes were normalized to that of the reference gene GAPDH and presented as fold changes using the 2-ΔΔCt method 42 .
Oligonucleotide Primers and Probes Used for qRT-PCR.
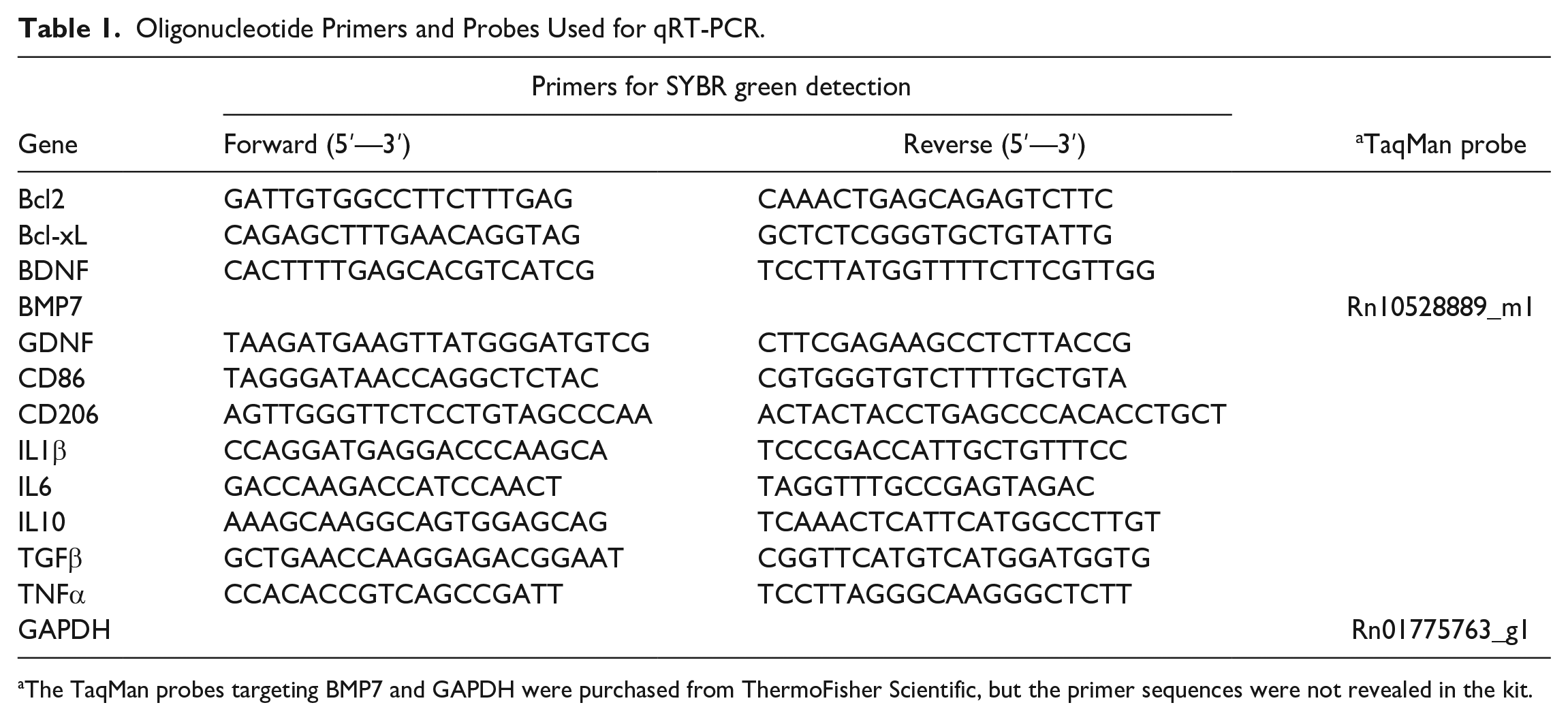
The TaqMan probes targeting BMP7 and GAPDH were purchased from ThermoFisher Scientific, but the primer sequences were not revealed in the kit.
MicroRNA Sequencing
Total RNA was isolated from WJ-MSC Exos using the miRNAeasy kit (Qiagen), followed by the construction of microRNA (miRNA) libraries utilizing the QIAseq miRNA Library Kit (Qiagen). Next-generation sequencing was performed on the Illumina NovaSeq 6000 platform with a sequence depth of 20 million raw reads per sample to assess the quantity and sequence profiles of the miRNA molecules.
Protein Array
Total proteins were extracted from WJ-MSC Exos using radioimmunoprecipitation assay (RIPA) buffer, and the levels of 40 cytokines and 40 growth factors were quantified using multiplexed sandwich enzyme-linked immunosorbent assay (ELISA)-based arrays. Specifically, the Quantibody Human Inflammation Array 3 (#QAH-IFN-3; RayBiotech; Norcross GA, USA) and Quantibody Human Growth Factor Array 1 (#QAH-GF-1; RayBiotech; Norcross GA, USA) were employed. Protein signals labeled with Cy3 dye were scanned using an Innopsys InnoScan laser scanner. The concentrations of the target proteins were determined by comparing the signals from the samples against a standard curve.
Statistical Analysis
All data were presented as mean values ± standard error (SEM) and analyzed using GraphPad Prism 9.2.0 statistical software (GraphPad Software, Inc.). The infarction volume and locomotor activity between the Exo-treated and vehicle-treated groups were compared using the unpaired, two-tailed Student’s t-test. In the qRT-PCR analysis, the difference in examined genes among groups was determined by ordinary one-way analysis of variance (ANOVA), followed by Tukey’s multiple comparison test. The percentages of MAP2+ counts and TUNEL+ counts among groups treated with different doses of Exos and glutamate were compared using one-way ANOVA, followed by post hoc Dunnett’s multiple comparison test. The assessments of neurological deficits, including Bederson’s score, body asymmetry, and forelimb grip strength, were compared among groups using one-way ANOVA, followed by post hoc Dunnett’s test. Statistical significance is considered when the P value is less than 0.05.
Results
Characterization of Human WJ-MSC Exos
Cells isolated from Wharton’s jelly of hUCs were grown as monolayer cells attaching to the surface of culture vessels. They exhibited a liner and stretch morphology similar to that seen in fibroblasts (Fig. 1A). After trypsinization, cells were examined by flow cytometry and confirmed to express MSC surface markers (CD73, CD90, CD105) at high percentages (>95%) (Fig. 1B–D) but almost no hematopoietic stem cell marker (CD34; <2%) (Fig. 1E). This finding is consistent with previous studies on the characterization of human WJ-MSCs43 –45. Vesicles were purified from the conditioned medium of human WJ-MSC cultures by combining ultrafiltration and affinity adsorption methods, as previously described46,47. Transmission electron microscopy showed that the purified vesicles have a typical spherical shape (Fig. 2A). By nFCM, vesicle size was detected in the range of 45–180 nm with an average diameter of 73.0 ± 24.5 nm (Fig. 2B), and typical Exo surface markers (CD9, CD63, CD81) were found on the vesicle surface (Fig. 2C–F). Western blot analysis revealed that these vesicles contained Exo markers (CD9, CD63, CD81, HSP70, TSG101) but no endoplasmic reticulum marker (Calnexin) (Fig. 2G). These results indicated that human WJ-MSCs were successfully isolated from hUCs, and WJ-MSC-derived Exos were purified without contamination of endoplasmic reticulum components.
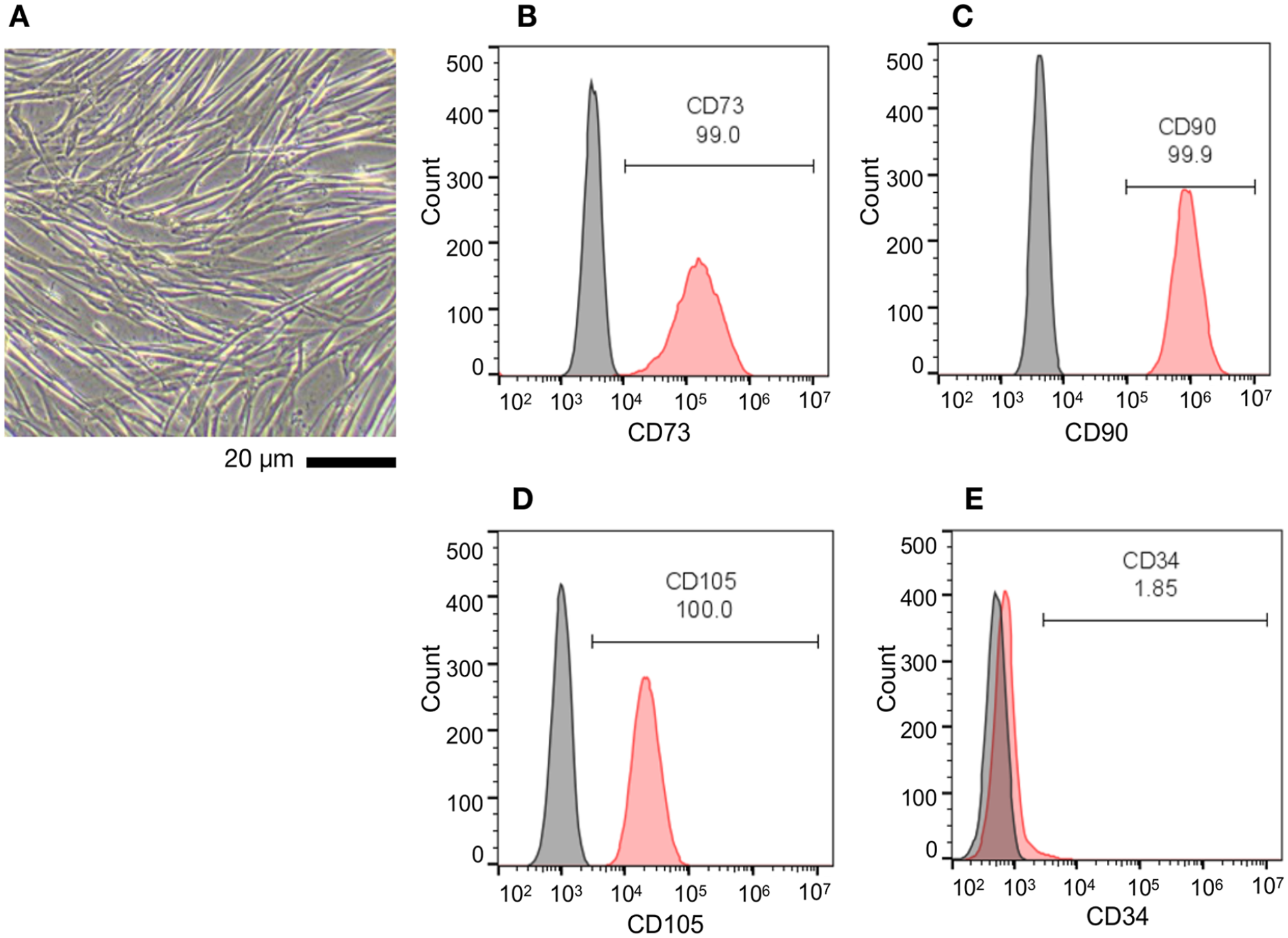
Characterization of human WJ-MSCs. (A) Phase-contrast image depicting the morphology of WJ-MSCs. Cells are observed as an adherent monolayer on the surface of a culture dish and display fibroblast-like morphology. Scale bar: 20 μm. (B—E) Cells were analyzed for specific surface markers by flow cytometry using fluorescein-conjugated antibodies targeting MSC surface markers (CD73, CD90, CD105) and a hematopoietic stem cell marker (CD34).
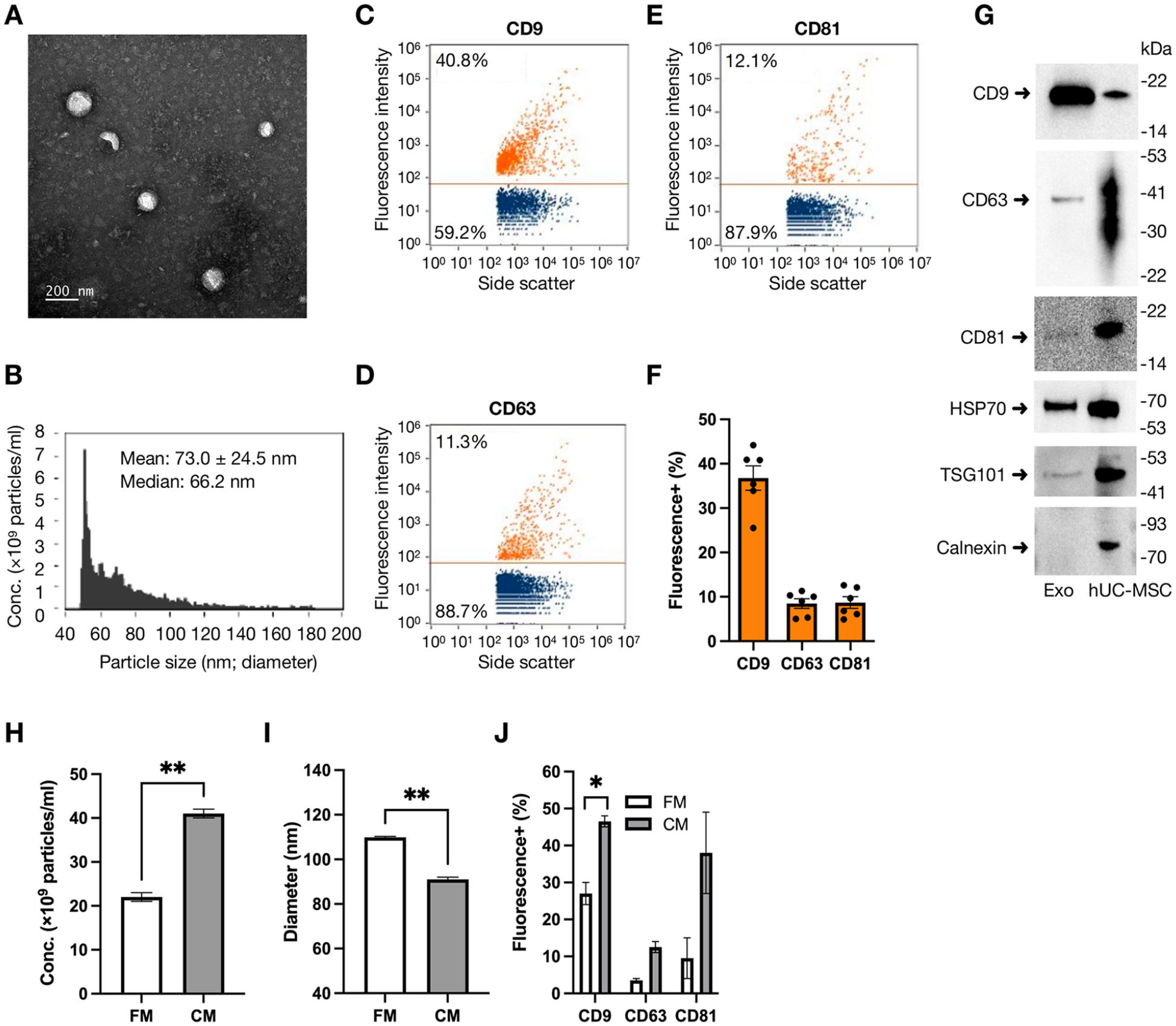
Characterization of Exos. Exos were purified from the culture medium of WJ-MSCs. (A) Exos were photographed under transmission electron microscopy. Scale bar: 200 nm. (B–F) Exos were further analyzed by nano-flow cytometry for particle concentration, size distribution (B), and surface markers. A set of representative dot plots revealed the percentage of Exos positive for the surface markers probed by fluorescein-labeled antibodies against CD9 (C), CD63 (D), and CD81 (E). (F) A statistical graph presented the percentage counts of indicated markers obtained from six batches of purified exosomes. Data were expressed as mean ± SEM. (G) Exo and WJ-MSC lysates were examined by Western blot analysis to detect protein markers of Exo (CD9, CD63, CD81, HSP70, TSG101) and endoplasmic reticulum membrane (Calnexin). The molecular weight (kDa) and migration location of protein markers were indicated. The hPL-supplemented fresh medium (FM) and conditioned medium (CM) collected from WJ-MSC cultures were examined by the ZetaView particle analyzer to determine the concentration (H), particle size (I), and surface markers (J) of exosomes in the medium. Duplicated results are presented as mean ± SEM, and significant differences between groups are indicated (*P < 0.05; **P < 0.01; Student’s t-test).
We recognize the concern about potential contamination from exosomes present in the 5% hPL-supplemented medium. To address this issue, we carefully monitored the exosome yield and characteristics to differentiate between exosomes from WJ-MSCs and those originating from hPL. After 6 days of WJ-MSC culture, the exosome concentration in the conditioned medium increased from 2.2 × 1010 ptc/ml (present in the fresh medium) to 4.1 × 1010 ptc/ml (P = 0.0055; Fig. 2H), with a reduction in mean particle size from 110 ± 0.4 nm to 91 ± 0.9 nm (P = 0.003) (determined by ZetaView, Fig. 2I). Over time, WJ-MSCs likely absorb or degrade exosomes from their culture medium, resulting in a higher portion of MSC-derived exosomes in the conditioned medium. In addition, we observed a significant increase in the percentage of exosomes expressing the surface marker CD9, from 27 ± 3% in the fresh medium to 46.5 ± 1.5% in the conditioned medium (P = 0.0283) (determined by ZetaView, Fig. 2J). Moreover, the miRNA profiles of exosomes purified from the conditioned medium (Fig. 7A) were markedly different from those of hPL-derived exosomes. Specifically, the seven most abundant miRNAs in the WJ-MSC-derived exosomes are distinct from those typically found in hPL exosomes48,49. These findings suggest that exosomes in the conditioned medium are primarily derived from WJ-MSCs rather than hPL.
WJ-MSC-Derived Exos Attenuated Glutamate-Induced Neurodegeneration and Apoptosis in PNC Culture
Cerebral ischemia leads to neuronal death in the brain, which is partly attributed to the release of excitatory amino acids50 –52. The protective effects of human WJ-MSC-derived Exos against glutamate-mediated neurotoxicity were examined in the PNCs (Fig. 3). Cells were treated with saline (as control), glutamate (Glu), or Glu + WJ-MSC Exos (Exo) at different concentrations for 48 h. Similar to previous reports53,54, Glu (100 μM) significantly reduced Microtubule-Associated Protein 2 (MAP2) immunoreactivity (100 ± 5.4% vs 53.3 ± 2.9%, P < 0.0001) and increased TUNEL-positive cell counts (100 ± 8.8% vs 307.5 ± 6.0%, P < 0.0001), compared with the control. Administration of Exos significantly and dose-dependently increased MAP2 immunoreactivity (Fig. 3A, C; ×1 Exo: 74.6 ± 3.2%, P = 0.0029; ×10 Exo: 85 ± 3.7%, P < 0.0001) and antagonized Glu-mediated TUNEL (×1 Exo: 126.6 ± 6.0%, P < 0.0001; ×10 Exo: 124.1 ± 6.5%, P < 0.0001; Fig. 3B, D). These results suggest that WJ-MSC Exo protects PNCs from glutamate-mediated neurodegeneration and apoptosis.
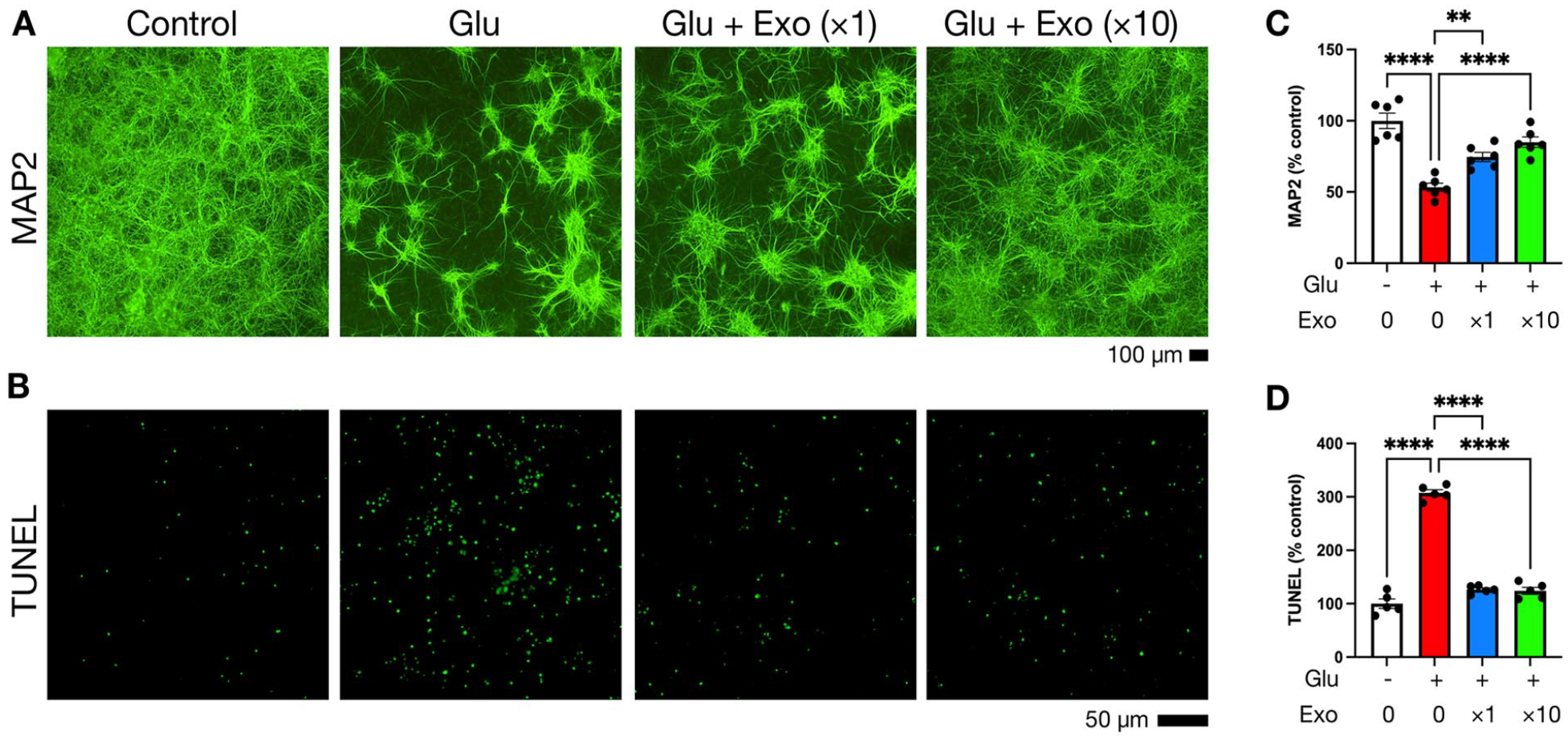
Exos protect cortical neurons from excitotoxicity. Rat primary cortical neurons were grown in a 96-well plate for 10 days and then treated with saline (as control), glutamate (Glu; 100 μM), or Glu + WJ-MSC Exos at different concentrations (×1: 5×109 ptc/ml; ×10: 5×1010 ptc/ml) for 48 h. After fixation, cells were subjected to immunofluorescence staining to probe MAP2 neuronal marker (A) and TUNEL assay to examine DNA fragmentation (B). In each group, images were taken from 5 to 6 wells to measure fluorescence intensity (for MAP2 staining) and fluorescent dot counts (for TUNEL assay). (C, D) Data were normalized to the control group and expressed as mean percentage values ± SEM. Significant differences between groups are indicated (**P < 0.01; ****P < 0.0001; one-way ANOVA followed by Tukey’s multiple comparison test).
WJ-MSC Exos Improved Motor Activity and Reduced Neurological Deficits in Stroke Rats
Stroke rats were transplanted with WJ-MSC Exos (Exo) or saline (Veh) via the tail vein 5–10 min after MCAo. Two days later, rats were individually placed in the activity chamber to record locomotor activity for 2 h (Fig. 4A, B). The stroke rats receiving saline (n = 11) or Exos (n = 12; 5 × 1010 ptc of exosomes per animal) exhibited lower locomotor activity than naive rats (n = 5) (Fig. 4B). However, Exos significantly increased locomotor activity in stroke animals (Exo vs Veh; HACTV: 9,614 ± 689 vs 7,607 ± 589, P = 0.0395; TODIST: 3,655 ± 315 vs 2,811 ± 205, P = 0.0391; MOVNO: 354 ± 23 vs 288 ± 24, P = 0.0635; MOVTIME: 370 ± 23 vs 315 ± 24, P = 0.1199; VACTV: 1,127 ± 100 vs 812 ± 54, P = 0.0133; VMOVNO: 146 ± 10 vs 117 ± 8, P = 0.0339; VTIME: 397 ± 39 vs 262 ± 21, P = 0.007) (Fig. 4B). Two neurological tests were conducted. The stroke rats receiving saline (n = 11) or Exos (n = 12) had higher Bederson’s neurological scores and body asymmetry than the naive rats (n = 5) (Fig. 4C, D). Exos significantly reduced Bederson’s scores (Exo vs Veh; 1.75 ± 0.18 vs 2.46 ± 0.16, P = 0.0073) and body asymmetry (Exo vs Veh; 16.58 ± 0.61 vs 18.82 ± 0.44, P = 0.0074) in the stroke rats (Fig. 4C, D). Forearm strength was examined by the grip test. The stroke rats receiving saline (n = 7) or Exos (n = 6) had weaker grip strength than the naive rats (n = 5, Fig. 4E). Compared with saline, Exos improved the grip strength (411 ± 24 vs. 305 ± 21, P = 0.005, Fig. 4E). These results suggest that intravenous WJ-MSC Exos improved functional recovery in stroke animals.
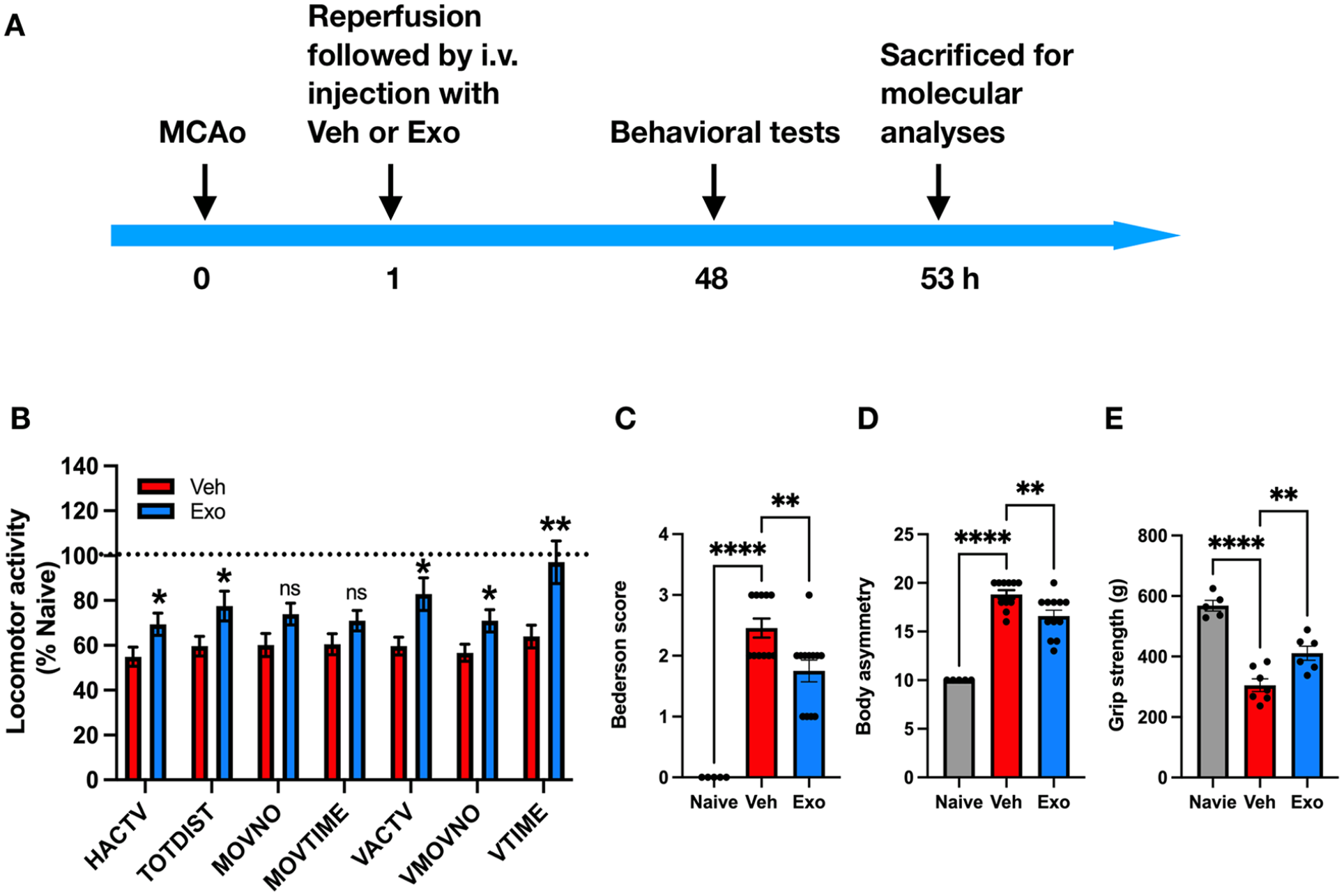
Exos improve locomotor activity and attenuate neurological deficits in ischemic stroke rats. (A) Experiment timeline. Adult rats underwent transient MCAo surgery by ligating the right distal MCA for 60 min. After reperfusion, rats were injected with saline (Veh; n = 11) or WJ-MSC exosomes (Exo; n = 12) via the tail vein. Two days later, naive rats (n = 5) and MCAo rats were examined for (B) locomotor activity for 2 h, (C) Bederson’s neurological deficit score, (D) body asymmetry in 20 trials, and (E) grip strength of the left forelimb. Finally, animals were sacrificed, and brain tissues were collected for molecular analyses. Data were expressed as mean values ± SEM. Significant differences between groups are indicated (*P < 0.05; **P < 0.01; ****P < 0.0001, ns: not significant; B: Student’s t-test, Exo vs Veh; C, D, E: one-way ANOVA followed by Dunnett’s multiple comparison test).
Transplantation of WJ-MSC Exos Mitigated Brain Infarction
A total of 13 stroke rats were used in this study. Human WJ-MSC Exos (Exo; n = 6) or saline (Veh; n = 7) was given through the tail vein 5–10 min after MCAo. Brain tissues were collected for TTC staining 2 days after MCAo. As seen in Fig. 5, infarct volume was significantly reduced in animals receiving Exos (64.1 ± 16.8 mm3 vs 112.5 ± 12.5 mm3, P = 0.0382; Fig. 5A, B), suggesting that intravenous administration of WJ-MSC Exos reduces brain infarction.

Exos reduce cerebral infarction in ischemic stroke rats. Adult rats underwent transient MCAo surgery by ligating the right distal MCA for 60 min. After reperfusion, rats were injected with saline (Veh; n = 7) or WJ-MSC exosomes (Exo; n = 6) via the tail vein. (A) Two days after MCAo, rats were sacrificed, and brains were sliced for TTC staining to examine the volume of infarct areas (white patch). Scale bar: 10 mm. (B) The integrated infarction volume was measured from scanned images of brain slides. Data were expressed as mean values ± SEM. Significant differences between groups are indicated (*P < 0.05; Student’s t-test).
WJ-MSC Exo Transplants Increased the Expression of Anti-Apoptotic Factors (Bcl2, Bcl-xL) in the Ischemic Brain Cortices
Adult rats were subjected to the MCAo and administered either saline (Veh; n = 11) or WJ-MSC Exos (Exo; n = 11) via the tail vein. Two days after MCAo, the cortical tissues of the lesioned side and contralateral (non-lesioned) brain hemispheres were collected for qRT-PCR analysis. Exo treatment significantly upregulated the expression of anti-apoptotic factors Bcl2 (Exo: 1.40 ± 0.07 vs Veh: 1.10 ± 0.06, P = 0.0471; Fig. 6A1) and Bcl-xL (Exo: 1.51 ± 0.07 vs Veh: 1.19 ± 0.06, P = 0.0001; Fig. 6A2) in the lesioned cortex. In contrast, Bcl2 (Exo: 1.275 ± 0.09 vs Veh: 1.261 ± 0.08, P = 0.9991; Fig. 6A1) and Bcl-xL (Exo: 1.08 ± 0.05 vs Veh: 1.11 ± 0.05, P = 0.981; Fig. 6A2) were not altered by Exos in the non-lesioned cortex.
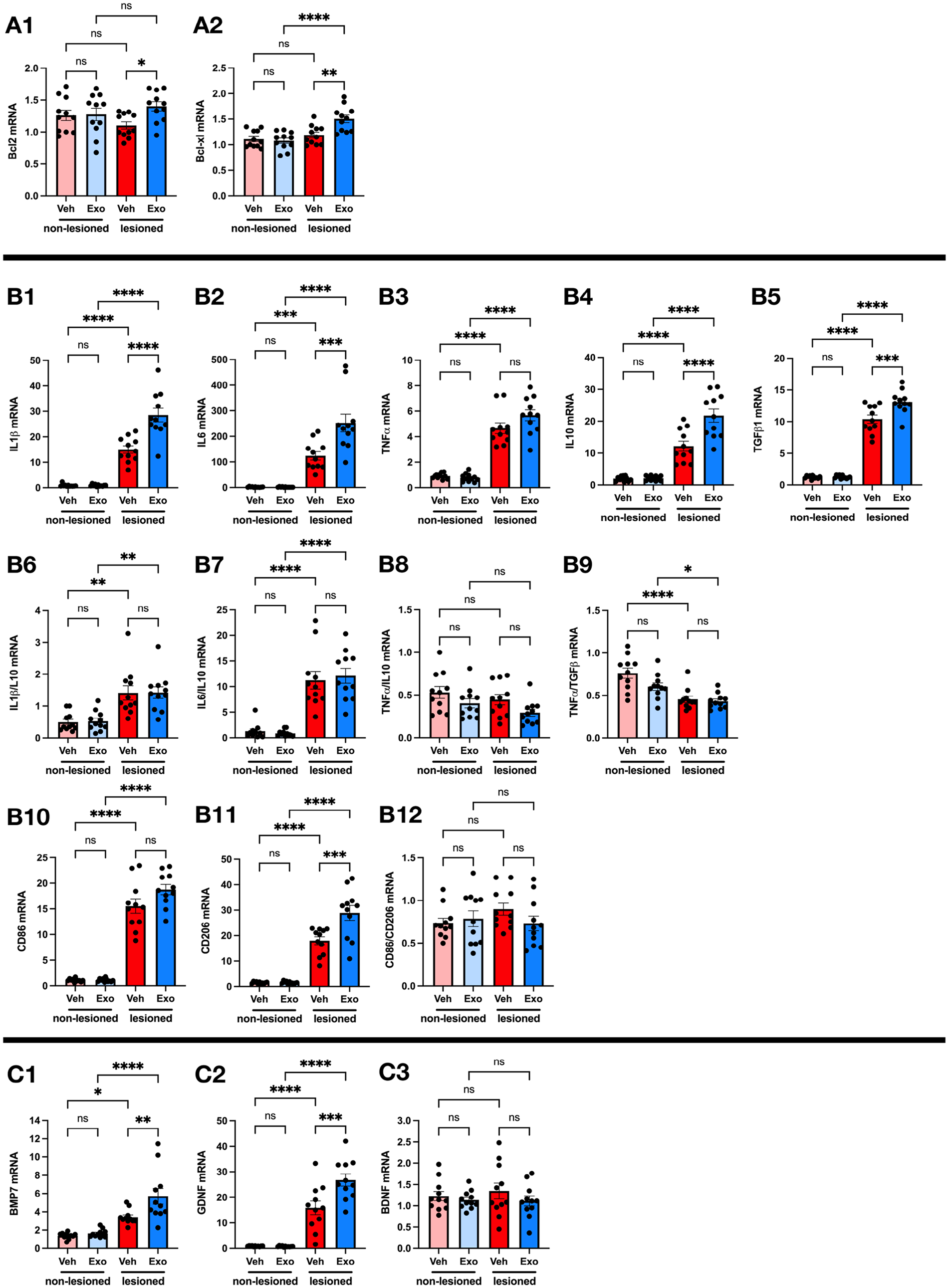
Effects of Exo treatment on the expression of apoptotic, inflammatory, and neurotrophic factors in the brain of ischemic stroke rats. Adult rats were subjected to transient MCAo surgery by ligating the right distal MCA for 60 min. After reperfusion, rats were injected with saline (Veh; n = 11) or WJ-MSC exosomes (Exo; n = 11) via the tail vein. Two days after MCAo, the cortex tissues of lesioned and non-lesioned brain sides were collected for qRT-PCR to examine the mRNA levels of apoptotic (Bcl2, Bcl-xl; A1, A2), inflammatory (CD86, CD206, IL1β, IL6, IL10, TGFβ1, and TNFα; B1–B12), and neurotrophic factors (BMP7, GDNF, and BDNF; C1, C2, C3). The mRNA level of examined genes was normalized to that of GAPDH and measured as fold or ratio changes. Data are expressed as mean values ± SEM, and significant differences between groups are indicated (*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, ns: not significant; one-way ANOVA followed by Tukey’s multiple comparison test).
WJ-MSC Exos Altered the Expression of Inflammatory Markers in the Stroke Brain
To investigate the effect of WJ-MSC-derived Exos on brain inflammation after cerebral ischemia, we examined the mRNA levels of pro-inflammatory cytokines (IL1β, IL6, TNFα), anti-inflammatory cytokines (IL10, TGFβ), and markers for M1 (CD86) and M2 (CD206) microglia in lesioned cortices. Exos significantly increased IL1β (28.53 ± 2.70 vs 15.00 ± 1.45, P < 0.0001; Fig. 6B1), IL6 (252.20 ± 34.50 vs 123.80 ± 16.55, P = 0.0002; Fig. 6B2), IL10 (21.73 ± 2.07 vs 12.18 ± 1.55, P < 0.0001; Fig. 6B4), TGFβ1 (13.12 ± 0.55 vs 10.38 ± 0.66, P = 0.0004; Fig. 6B5), and CD206 (28.91 ± 2.94 vs 17.96 ± 1.57, P = 0.0002; Fig. 6B11) in the lesioned cortex. There was no significant difference (Exo vs Veh) in the level of TNFα (5.68 ± 0.43 vs 4.66 ± 0.42, P = 0.102; Fig. 6B3) and CD86 (18.76 ± 0.99 vs 15.54 ± 1.41, P = 0.0567; Fig. 6B10) and the ratios of IL1β/IL10 (1.42 ± 0.18 vs 1.42 ± 0.22, P > 0.9999; Fig. 6B6), IL6/IL10 (12.11 ± 1.45 vs 11.25 ± 1.73, P = 0.9515; Fig. 6B7), TNFα/IL10 (0.29 ± 0.04 vs 0.45 ± 0.06, P = 0.2498; Fig. 6B8), TNFα/TGFβ (0.43 ± 0.03 vs 0.45 ± 0.04, P = 0.9807; Fig. 6B9), and CD86/CD206 (0.73 ± 0.09 vs 0.89 ± 0.07, P = 0.426; Fig. 6B12) in the lesioned cortices between groups. These findings suggest that Exo transplants upregulated pro- and anti-inflammatory genes in the stroke brain.
WJ-MSC Exo Transplants Upregulated Protective Neurotrophic Factors in the Stroke Brain
We further investigated whether WJ-MSC Exos promoted the expression of neurotrophic factors after cerebral ischemia by analyzing the mRNA levels of three neurotrophic factors, BMP7, GDNF, and BDNF, in the cortex tissues of lesioned and non-lesioned brain hemispheres of stroke rats treated with Exos or saline (Fig. 6C1–C3). In the Veh group, the mRNA levels of BMP7 (3.42 ± 0.24 vs 1.39 ± 0.10, P = 0.0141; Fig. 6C1) and GDNF (15.90 ± 2.68 vs 0.85 ± 0.05, P < 0.0001; Fig. 6C2), but not BDNF (1.35 ± 0.19 vs 1.23 ± 0.11, P = 0.9072; Fig. 6C3) were significantly higher in the lesioned cortices than in the non-lesioned cortices, indicating that stroke increased the expression of BMP7 and GDNF. In the lesioned cortices, transplantation of Exos significantly increased BMP7 (5.71 ± 0.85 vs 3.42 ± 0.24, P = 0.0049; Fig. 6C1), GDNF (26.82 ± 2.37 vs 15.90± 2.68, P = 0.0006; Fig. 6C2), but not BDNF(Exo: 1.12 ± 0.12 vs Veh: 1.35 ± 0.19, P = 0.5723; Fig. 6C3). In contrast, GDNF, BDNF, BMP7 were not affected by Exo transplantation in the non-lesioned cortices. These findings suggest that the administration of WJ-MSC Exos further upregulated the expression of BMP7 and GDNF in the ischemic brain.
WJ-MSC Exos Contain Numerous Bioactive Molecules That May Contribute to the Beneficial Effects Observed in Stroke Rats Treated With Exos
It remains unclear whether these therapeutic outcomes arise from the direct action of the Exos or their paracrine effects. To investigate this, we further analyzed the molecular contents of WJ-MSC Exos, focusing on miRNAs, growth factors, and cytokines, which may be involved in neuroprotection, anti-inflammatory, or anti-apoptotic mechanisms. The miRNA profiles, determined by next-generation sequencing, were ranked based on total read counts (Fig. 7A). The top seven miRNAs accounted for 71.71% of the total 384 miRNAs identified in the Exos (Fig. 7B). Growth factors and cytokines were quantified using multiplexed sandwich ELISA arrays. Among the 40 growth factors examined, 30 were detected and ranked by their abundance (Fig. 7C), with the top five comprising 89.72% of the total detected growth factors (Fig. 7D). Similarly, 38 of the 40 cytokines were identified and ranked (Fig. 7E), with the top seven representing 93.36% of the total cytokines in the Exos (Fig. 7F). The most prominent bioactive molecules—miRNAs (miR-16-5p, miR-126-3p, let-7f-5p, miR-125b-5p, let-7a-5p, let-7i-5p, miR-93-5p), growth factors (IGBP-4, BMP-4, BMP-7, IGFBP-2, FGF-4), and cytokines (TIMP-2, TIMP-1, ICAM-1, RANTES, G-CSF, IL-6sR, TNFRII)—have been previously reported to play neuroprotective roles in stroke models (as detailed in the Discussion section). Conversely, the minor components are unlikely to exert significant biological effects due to their low abundance. These findings suggest that the therapeutic effects of WJ-MSC Exos may, in part, be due to the major bioactive molecules they encapsulate.
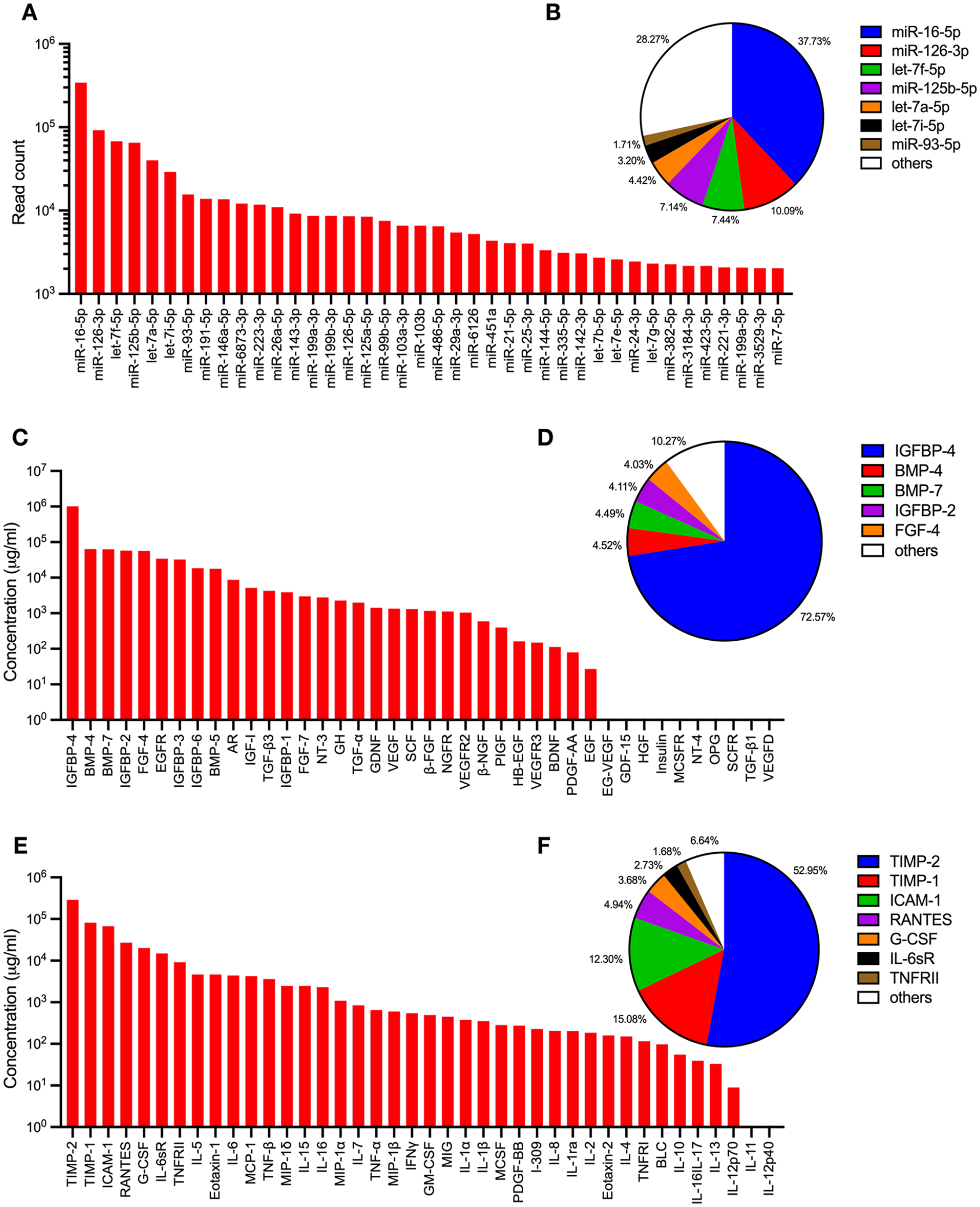
Bioactive molecule profiles in Exos. The miRNA content of WJ-MSC Exos was analyzed via next-generation sequencing. The total read counts of the top 40 miRNAs are displayed (A), with the top seven miRNAs accounting for 71.71% of the total 384 miRNAs identified in the Exos (B). Growth factors and cytokines present in the Exos were quantified using multiplexed sandwich ELISA-based arrays. Of the 40 growth factors analyzed, 30 were detected (C), with the top five comprising 89.72% of the total detected growth factors (D). In addition, 38 of the 40 cytokines were identified (E), with the top seven representing 93.36% of the total cytokines detected in the Exos (F).
Discussion
In this study, we characterized Exos isolated from human WJ-MSCs and investigated their potential in the treatment of cerebral ischemic stroke. We found that systemic transplantation of Exos from human WJ-MSCs significantly reduced brain infarction, lowered Bederson’s neurological scores and body asymmetry, and increased locomotor activity and forearm grip strength in stroke rats. Exo treatment also led to increased expression of neurotrophic factors (BMP7, GDNF) and anti-apoptotic factors (Bcl2, Bcl-xL) in the brain tissue damaged by MCAo. We also revealed that Exo administration significantly reduces glutamate-mediated neuronal loss and apoptosis in PNC cultures. Our data support the idea that early post-treatment with WJ-MSC Exos protects against neuronal damage and improves functional recovery in the brain affected by stroke.
Cells isolated from Wharton’s jelly of hUCs exhibit morphology similar to that of fibroblasts and express MSC surface markers (CD73, CD90, CD105), but not the hematopoietic stem cell marker (CD34). This observation aligns with previous studies on the characterization of human WJ-MSCs43 –45. Vesicles purified from the conditioned medium of human WJ-MSC cultures are spherical, ranging in size from 45 to 180 nm, and display typical Exo markers (CD9, CD63, CD81, HSP70, TSG101) without the presence of endoplasmic reticulum marker (Calnexin), consistent with Exo characterization standards30,55. These findings confirm that human WJ-MSCs were successfully isolated from hUCs, and the Exos collected for this study were purified from WJ-MSCs without contamination of endoplasmic reticulum components.
The protective effect of WJ-MSC Exos was first examined in PNC cultures. A high dose (100 µM) of glutamate was used to generate neurodegeneration as previously described56,57 and to simulate overflow of concentrated glutamate during cerebral ischemia 58 . Glutamate significantly reduced MAP2 immunoreactivity and increased TUNEL activity. Both responses were significantly mitigated by WJ-MSC Exos, indicating that these Exos attenuated glutamate-induced apoptosis and neuronal loss. This neuroprotective effect is further supported by in vivo experiments, where systemic administration of WJ-MSC Exos increased the expression of anti-apoptotic factors Bcl-2 and Bcl-xL in the lesioned brain tissues. These data suggest that WJ-MSC Exos induced protection through the suppression of apoptosis.
Comparatively, Exos derived from other sources also processed anti-apoptotic properties in stroke brains. For example, Exos from rat bone marrow MSCs inhibit apoptosis by reducing KDM6B expression and reduce neuronal damage in stroke rats 59 . Exos from mouse bone marrow MSCs inhibit apoptosis by suppressing CDK6 in stoke mice 60 . Exos derived from rat bone marrow MSCs downregulated caspase-8-dependent apoptosis in oxygen-glucose-deprived cultures of oligodendrocytes 25 . While these findings highlight the therapeutic potential of MSC-derived Exos through anti-apoptosis in mitigating neuronal damage and promoting recovery in ischemic stroke, further investigation is required to determine the anti-apoptotic mechanisms of WJ-MSC Exos.
Numerous studies have demonstrated that the use of MSCs can enhance neurological function recovery in stroke animals. This benefit is accomplished by promoting the growth of new blood vessels and nerve cells while also reducing inflammation and cell death61 –65. Notably, evidence suggests that the beneficial effects of MSCs are partly mediated by the secretion of Exos rather than direct cell replacement12,66,67. Recent investigations utilizing Exos derived from MSCs obtained from various tissues, such as rat bone marrow, rat adipose, human bone marrow, and hUC blood, have shown promising results in improving neurological function recovery in animal models of ischemic stroke12,22,68,69. Furthermore, studies have demonstrated that direct injection of hUC-MSCs into the brain or systemic administration can reduce brain injury and neurological deficits in rodent stroke models9,70 –72. Therefore, Exos derived from hUC-MSCs may have similar effects in protecting neurons and enhancing functional recovery in the rat model of ischemic stroke.
The beneficial effects of Exo treatment on protection and repair in cerebral ischemic stroke rats are highlighted by its ability to enhance the expression of essential neurotrophic factors, such as BMP7 and GDNF. BMP7 possesses neurogenic and neuroprotective properties, promoting neuronal differentiation and dendritic growth, while GDNF exerts potent trophic effects on various types of neurons, facilitating cell survival and axonal outgrowth73 –77. After stroke-induced injury, these neurotrophic factors are upregulated, contributing substantially to promoting neuronal survival, regeneration, and functional recovery78 –80. By enhancing the upregulation of BMP7 and GDNF, WJ-MSC-derived Exos may create a suitable microenvironment for neurogenesis and synaptic plasticity within the injured brain tissue, ultimately restoring neurological function. However, the expression of BDNF was not altered in the stroke rats after Exo treatment. Although BDNF is a well-known neurotrophic factor implicated in neuronal survival, neurogenesis, and synaptic plasticity81 –84, its unchanged expression suggests that the beneficial effects of Exo treatment in ischemic stroke rats may not be attributed to the action of BDNF.
MSC-derived Exos may prevent or reverse neuronal damage by delivering bioactive molecules such as miRNAs and proteins that regulate critical signaling pathways involved in neuronal repair and functional recovery85 –87. Notably, seven miRNAs—miR-16-5p, miR-126-3p, let-7f-5p, miR-125b-5p, let-7a-5p, let-7i-5p, and miR-93-5p—make up 71.71% of the 384 miRNAs identified in WJ-MSC Exos, each potentially contributing to stroke recovery. miR-16-5p, the most abundant, has been shown to promote the survival of cortical neurons and reduce apoptosis by targeting AKT3 in an oxygen-glucose deprivation model of cerebral ischemia 88 . Members of the let-7 miRNA family offer neuroprotection in cerebral ischemic stroke by regulating proteins involved in inflammatory pathways, including the SMAD3/TGFβ axis within the central nervous system 89 . In addition, overexpression of miR-126-5p has been shown to protect the BBB and reduce pro-inflammatory cytokines in a mouse model of MCAo 90 . miR-125b-5p and miR-93-5p are involved in modulating excitotoxicity and oxidative stress, respectively, after ischemic stroke91,92.
Among the growth factors present in WJ-MSC Exos, five (IGFBP-4, BMP-4, BMP-7, IGFBP-2, and FGF-4) account for 89.72% of the total identified and may contribute to stroke recovery. IGFBP-4, the most abundant growth factor, has been shown to promote neuronal survival and neurite outgrowth following cerebral ischemia 93 . BMP-7 and FGF-4 have been linked to neuroregeneration by stimulating the proliferation of neuronal precursors94,95, while BMP-4 has been shown to support neuronal stem cell proliferation and cell survival in an in vitro model of oxygen-glucose deprivation 96 .
In addition, seven cytokines—TIMP-2, TIMP-1, ICAM-1, RANTES, G-CSF, IL-6sR, and TNFRII—representing 93.36% of the total cytokines in WJ-MSC Exos may also contribute to recovery after stroke. TIMP-2 and TIMP-1, the most abundant, reduce ischemic damage by modulating matrix metalloproteinase (MMP) activity and activating intracellular kinase-dependent pathways97,98. ICAM-1 in Exos may help decrease infarct size by preventing leukocyte migration into the brain parenchyma 99 . While RANTES is typically pro-inflammatory, it may promote cell survival and neurotrophic factor upregulation by activating Akt and Erk1/2 pathways in peri-infarct areas 100 . G-CSF has been shown to reduce lesion size, improve motor function, and lower mortality in transient ischemic stroke, likely by acting on its receptors in the ischemic penumbra 101 . Last, IL-6sR and TNFRII serve as buffers to prevent overstimulation from IL6 and TNFα, two pro-inflammatory cytokines involved in ischemic brain injury102,103. These improvements highlight the potential of Exo therapy as a multifaceted and effective strategy for reducing neurological deficits caused by ischemic stroke.
We previously demonstrated the intracerebral transplantation of human WJ-MSCs reduced brain infarction and improved motor functions in stroke rats 19 . The protective effect was associated with a strong anti-inflammatory effect in the lesioned cortex. In contrast, in this study, we found that human WJ-MSC Exos given intravenously did not process strong anti-inflammatory function. Exo treatment resulted in an increased expression of anti-inflammatory cytokines (IL10, TGFβ) and an M2 microglia marker (CD206) in the lesioned cortices. At the same time, there was an upregulation of pro-inflammatory cytokines (IL1β, IL6, TNFα) and an M1 microglia marker (CD86). These results indicate that Exos triggered pro- and anti-inflammatory pathways but did not skew to either pathway, as the ratios of pro/anti-inflammation cytokines and microglia polarization (M1/M2) were not altered. The differential inflammatory responses between WJ-MSCs and WJ-MSC Exos may be attributed to the route, dose, or timing of transplantation, highlighting the intricate nature of our research. Further investigation is necessary to understand the specific mechanisms by which Exos orchestrate this delicate balance.
Exo treatment for ischemic stroke offers several advantages over cell-based therapies that use MSCs. One major advantage is avoiding potential safety concerns associated with cell transplantation, such as immune rejection, tumorigenesis, and embolism formation104 –106. Exos can be administered via various non-invasive routes, such as intravenous injection, intranasal delivery, and intra-arterial infusion12,107,108. Therefore, Exo therapy is conducted through more accessible administration routes than invasive procedures required for cell transplantation, reducing procedural risks and costs and enhancing patient compliance. In addition, Exos possess unique properties that allow them to traverse biological barriers, including the BBB, enabling targeted delivery to the site of injury in the brain109,110. Exos are extracellular vesicles secreted by MSCs, containing bioactive molecules, including proteins, nucleic acids, and lipids, that modulate various cellular processes involved in neuroprotection, angiogenesis, neurogenesis, and anti-inflammatory responses. Exo treatment could be a promising and versatile therapeutic strategy for ischemic stroke, with the potential to overcome many limitations associated with cell-based therapies.
To bridge our present findings to clinical applications, we need to address several critical aspects. (1) Delivery Routes: In our study, we administered Exos systemically via intravenous injection, which is feasible for clinical use. However, alternative routes such as intra-arterial delivery or local administration to the ischemic brain region may offer more precise targeting, minimizing off-target effects and potentially improving outcomes111,112. A detailed evaluation of the pros and cons of these methods is necessary to determine the most effective delivery strategy for human applications. (2) Dosage: Establishing an optimal dose for clinical application is crucial 113 . Our study demonstrated efficacy with a specific dose in rats, but human trials would need to consider dose scaling based on body size and stroke severity. Dose-response studies will help refine therapeutic regimens to ensure both efficacy and safety for patients 114 . (3) Timing of Administration: The timing of Exo treatment is another critical aspect. Our data suggest that early post-stroke intervention is beneficial, but in clinical settings, stroke patients may present at different times after onset. Exploring how treatment windows affect outcomes and understanding the balance between early and delayed interventions are essential for maximizing recovery in diverse clinical scenarios 115 . (4) Patient Selection: Not all stroke patients will respond equally to Exo therapy. Potential selection criteria such as stroke type, severity, and patient-specific factors (e.g., age, comorbidities, genetic predispositions) could influence treatment efficacy116,117. Identifying reliable biomarkers to predict responsiveness could help patient selection and improve treatment outcomes 118 . (5) Safety and Regulatory Considerations: Safety issues such as immune responses, potential off-target effects, and long-term safety in patients must be carefully evaluated. Current clinical guidelines for cell-based therapies may be adapted to address the regulatory challenges regarding the use of MSC-derived Exos as therapeutic agents 119 . By addressing these considerations in the clinical translation of WJ-MSC Exo therapy, we aim to provide a clearer roadmap for future clinical trials and potential therapeutic use in stroke patients.
Despite the promising findings of this study, further investigation is needed to fully understand the therapeutic potential of Exos derived from WJ-MSCs in treating cerebral ischemic stroke. First, this study did not assess whether the administered Exos reached the damaged brain region, which is crucial for validating their direct therapeutic effects. Second, the impact of different Exo dosages on treatment outcomes was not explored, making it uncertain whether there is an optimal dose or whether the observed benefits are dose-dependent. Third, the long-term effects of Exo treatment were not evaluated, leaving it unclear whether the improvements in brain damage and neurological outcomes are sustained over time or whether additional treatments are required for lasting therapeutic benefits. In addition, this study lacked a detailed immunohistochemistry analysis of neuronal pathology in the penumbra region, the peri-infarct area critical for behavioral recovery after stroke120 –122. Examining these aspects could provide detailed insights into the cellular and molecular changes induced by Exo treatment. These limitations need to be addressed in future research to establish the efficacy, optimal administration protocols, and long-term sustainability of WJ-MSC-derived Exos as a treatment for cerebral ischemic stroke.
In conclusion, we demonstrate that early intravenous administration of Exos from human WJ-MSCs can significantly enhance functional recovery and mitigate brain damage in the stroke animal model. Given the non-invasive nature of this therapeutic approach, our study not only offers promising insights but also opens up exciting possibilities for potential clinical applications in stroke treatment.
Footnotes
Acknowledgements
We thank Mr Yen-Shen Wu (Electron Microscope Laboratory of Tzong Jwo Jang, College of Medicine, Fu Jen Catholic University) for TEM technical assistance and Riverfront Women’s Hospital for collecting human umbilical cords.
Author Contributions
Conceptualization: YHC
Data curation: YSC, KJW, SJY, KLW, CYH, YSC, KYC, YSW, EKB, TWH, SHL, CHL, SCH, YHC
Formal analysis: YSC, KJW, YSW, YHC
Funding acquisition: YHC
Investigation: YSC, KJW, SJY, KLW, CYH, YSC, KYC, YSW, EKB, TWH, SHL, CHL, SCH
Methodology: YSC, KJW, SJY, CYH, YSC, KYC, YSW, EKB, YHC
Project administration: YHC
Resources: SJY, KLW, SCH, YW, YHC
Supervision: YW, YHC
Validation: YSC, KJW, CYH, YSW, EKB, YHC
Visualization: YHC
Writing-original draft: YHC
Writing-review & editing: YW, YHC
All authors read and approved the final manuscript.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (IRB) of Fu Jen Catholic University (approval no. C111157) on July 18, 2023. The Institutional Animal Care and Use Committee of the National Health Research Institutes in Taiwan approved the experimental procedures used in this study (approval no. NHRI-IACUC-112034-A) on March 21, 2023.
Statement of Human and Animal Rights
All animal housing and experiments were conducted in strict accordance with the institutional Guidelines for the Care and Use of Laboratory Animals at the National Health Research Institutes.
Statement of Informed Consent
Written informed consent for donating discarded umbilical cords in this study was obtained from the donors before participating.
Data Availability
All relevant data included in the paper are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Y-SC, K-LW, C-YH, Y-SC, and K-YC are employees of YJ Biotechnology Co. Ltd. YW is a member of the editorial board of the journal of Cell Transplantation. The authors, including K-JW, S-JY, Y-SW, E-KB, T-WH, S-HL, C-HL, S-CH, and Y-HC, declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The funding agencies were not involved in the writing of this manuscript or the decision to submit it for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by YJ Biotechnology Co. Ltd., Taiwan [FJCU-7100491, C12-018], and the National Science and Technology Council, Taiwan [NSTC 112-2314-B-030-009, MOST 111-2314-B-030-007].
