Abstract
Many studies support the idea that long noncoding RNAs (lncRNAs) are significantly involved in the process of cardiomyocyte (CM) regeneration following a myocardial infarction (MI). This study aimed to systematically review the emerging role of lncRNAs in cardiac regeneration by promoting CM proliferation after MI. Furthermore, the review summarized potential targets and the underlying mechanisms of lncRNAs to induce heart regeneration, suggesting utilizing lncRNAs as innovative therapeutic targets for mitigating MI injuries. We searched the PubMed, Scopus, and Web of Science databases for studies on lncRNAs that play a role in heart regeneration after MI. We used search terms that included MI, lncRNAs, CM, and proliferation. Relevant English articles published until June 11, 2023, were systematically reviewed based on inclusion and exclusion criteria. A total of 361 publications were initially identified, and after applying the inclusion and exclusion criteria, nine articles were included in this systematic review. These studies investigated the role of critical lncRNAs in cardiac regeneration after MI, including five upregulated and four downregulated lncRNAs. Acting as a competitive endogenous RNA is one of the main roles of lncRNAs in regulating genes involved in CM proliferation through binding to target microRNAs. The main molecular processes that greatly increase CM proliferation are those that turn on the Hippo/YAP1, PI3K/Akt, JAK2-STAT3, and E2F1-ECRAR-ERK1/2 signaling pathways. This systematic review highlights the significant role of lncRNAs in heart regeneration after MI and their impact on CM proliferation. The findings suggest that lncRNAs could serve as potential targets for therapeutic interventions aiming to enhance cardiac function.
Keywords
Introduction
Cardiovascular diseases are one of the main causes of death worldwide. Myocardial infarction (MI) is a common cardiovascular accident with high morbidity and mortality. Although reperfusion interventions have improved MI-related mortality, acute MI remains a life-threatening disease worldwide1,2. One of the main ways to overcome the long-term complications caused by heart infarction is to use therapeutic methods such as reducing the loss of cardiomyocytes (CMs) or increasing the number of CMs. Studies have shown that adult CMs can undergo proliferation under certain conditions, which highlights the potential for cardiac repair after infarction by enhancing endogenous cardiac regeneration3,4.
Long noncoding ribonucleic acids (lncRNAs) are RNA molecules longer than 200 nucleotides that control gene expression and protein function through various molecular processes during transcription or posttranscription. LncRNAs regulate gene expression at both transcriptional and translational levels in the cytoplasm through multiple pathways. They have been proven to operate as attractors, binders, and scaffolding for other regulatory factors involved in epigenetic alterations 5 . They are also engaged in organizing multiple components of the transcription and splicing process as well as subnuclear structures 6 . Evidence has shown that lncRNAs play an important role in pathologies such as cancer, cardiovascular and neurodegenerative disease, and could serve as innovative targets for treating these disorders7–9.
LncRNAs have recently been identified as vital factors regulating cardiac development, the heart’s physiological function, and the pathological processes of cardiovascular diseases. For example, Klattenhoff et al. revealed the essential role of Braveheart in the CM differentiation of pluripotent stem cells. Ounzain et al. 10 showed that CARMEN crucially regulates the differentiation of cardiac precursor cells. Furthermore, the expression of CARMEN was upregulated in patients with hypertension 11 . Fendrr, a lateral mesoderm-specific lncRNA, has been demonstrated to play a crucial role in regulating heart and body wall development in mice 12 . Wang et al. 13 demonstrated the lncRNA Chear as an essential epigenetic checkpoint for stress-induced cardiac hypertrophy. This study aimed to systematically review the literature about lncRNAs involved in heart regeneration after MI and summarize the molecular mechanism by which lncRNA contributes to CM proliferation. These findings may provide new insights into a novel targeted therapy after MI in the future.
Material and Methods
Search Strategy
The study was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A thorough search was conducted in the PubMed, Scopus, and Web of Science databases for English literature on lncRNAs related to heart regeneration following MI up to June 11, 2023. The search terms are outlined in Table 1.
The Search Terms Used for Database Searching.

Inclusion and Exclusion Criteria
We considered articles qualified for inclusion if they were about lncRNAs involved in cardiac regeneration and CM proliferation after MI in humans, animals, and cell cultures. We excluded articles if they were about: (1) other accompanying underlying diseases such as heart failure and cardiomyopathies, etc.; (2) the role of stem cells, circRNA, and microRNA; (3) letters, reviews, and other nonoriginal articles; and (4) other languages except English.
Data Extraction and Quality Assessment (Q.A.)
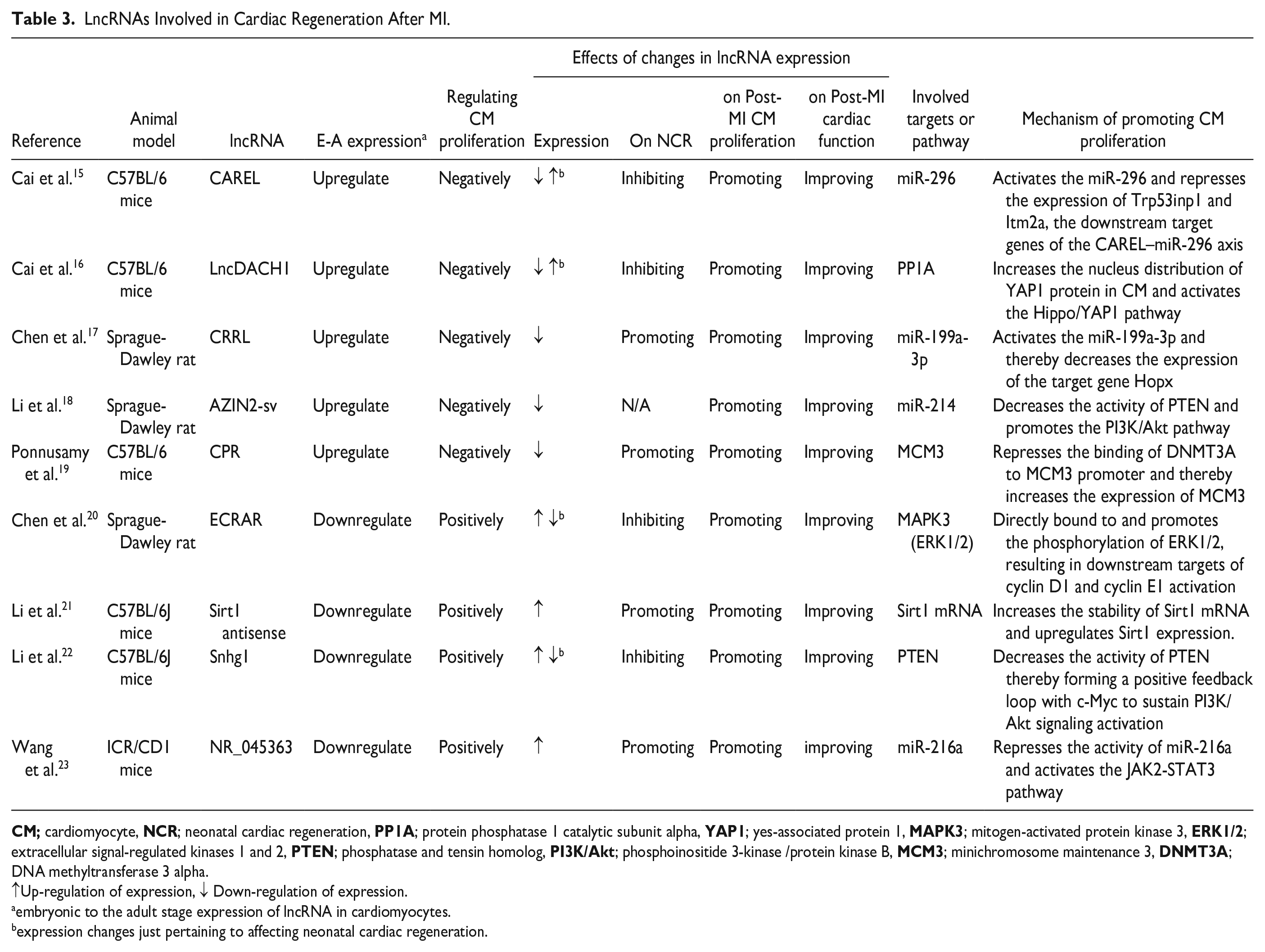
We extracted these details from each study: authors, journal, year of publication, experimental model, involved lncRNA name, embryonic to adult stage expression of lncRNA in CMs, how regulating CM proliferation, effects of changes in lncRNA expression, involved targets or pathways, molecular mechanisms. Then we grouped the extracted data according to the type of expression of lncRNA in embryonic to adult stages. The studies were assessed for quality using the Würzburg Methodological Quality Score 14 . The W-MeQS has 12 elements designed for evaluating the quality of a laboratory procedure. These 12 items check for selection bias, performance bias, attrition bias, detection bias, reporting bias, reagent status, chart status, and measurement status. These are all things that are common in both in vitro and in vivo studies. The maximum score that can be obtained from this tool is 12, so one point is given for each fulfilled item, and the final score will be the sum of the total points. A higher W-MeQS score indicates a greater study quality (Table 2). Two authors conducted the Q.A. separately, and any discrepancies were settled by discussion with a third author.
Quality Assessment of Included Studies Based on Würzburg Methodological Quality Score.
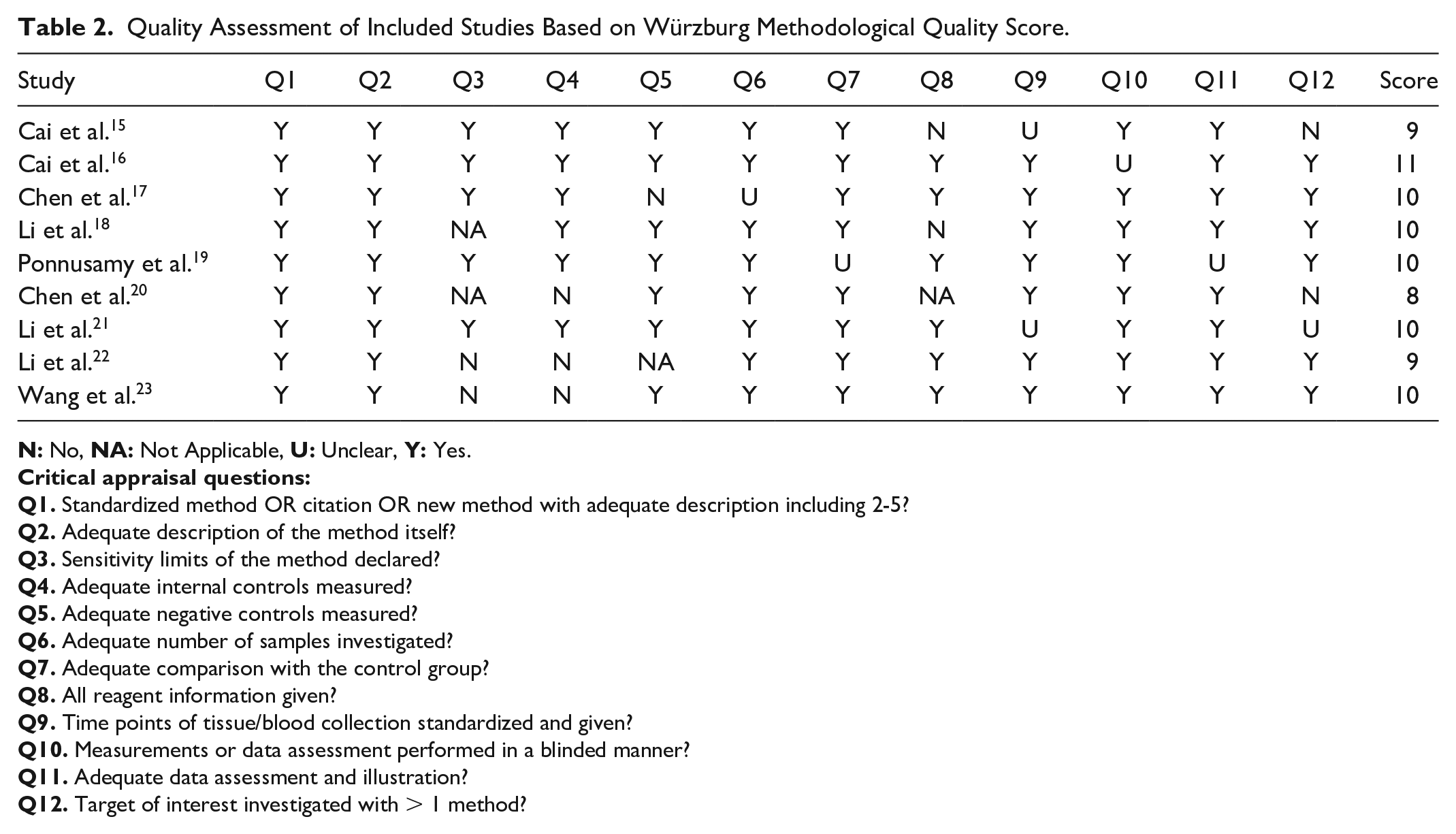
LncRNAs Involved in Cardiac Regeneration After MI.
↑Up-regulation of expression, ↓ Down-regulation of expression.
embryonic to the adult stage expression of lncRNA in cardiomyocytes.
expression changes just pertaining to affecting neonatal cardiac regeneration.
Results
A total of 361 publication titles were downloaded from PubMed, Scopus, and Web of Science after using the relevant search terms of which 203 publications were excluded; 134 titles were duplicated, 68 titles were not original articles, and one of them was not English. Furthermore, 106 articles were excluded after screening based on their titles and abstracts. Among the remaining publications, 51 full-text articles were retrieved. Subsequently, 42 of these articles were excluded for various reasons: including 17 about apoptosis, 20 about regulating the MI process, and five not about hypoxia-induced myocardial injury.
Overall, nine publications were incorporated into this systematic review (Figure 1). Each of the nine included studies investigated the role of a critical lncRNA in cardiac regeneration after MI, including five upregulated and four downregulated lncRNAs during embryonic to adult stage (Table 2). The studies included in this review had W-MeQS scores ranging from eight to 12, with a mean score of 9.67, suggesting that they were of good quality (Table 2). The Q.A. scores were distributed as follows: one item scored 11, five articles scored 10, two pieces scored 9, and one article scored 8. Figure 2 demonstrates the molecular mechanisms by which involved lncRNAs promote the CM proliferation.
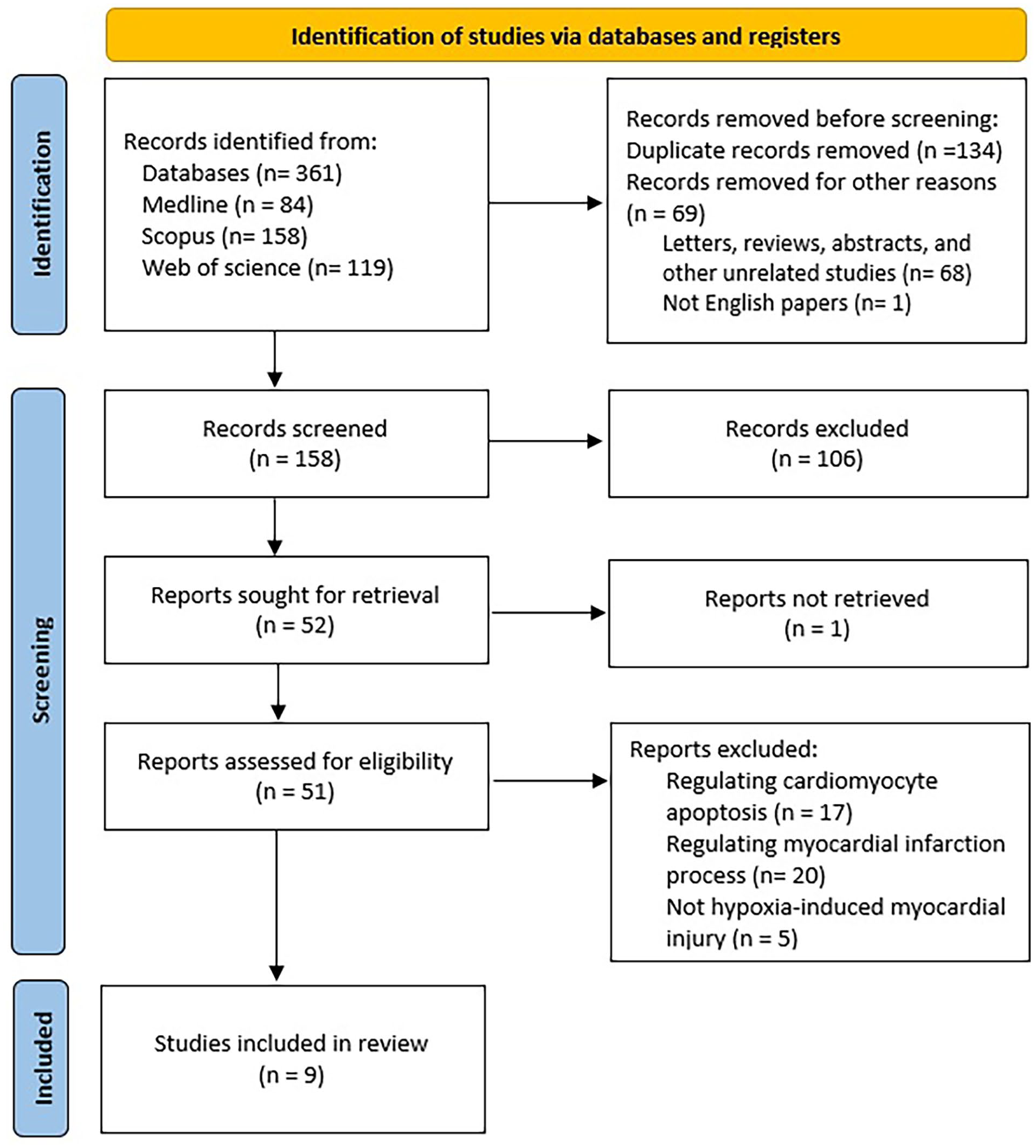
Flow chart of the systematic review process based on the PRISMA guidelines.
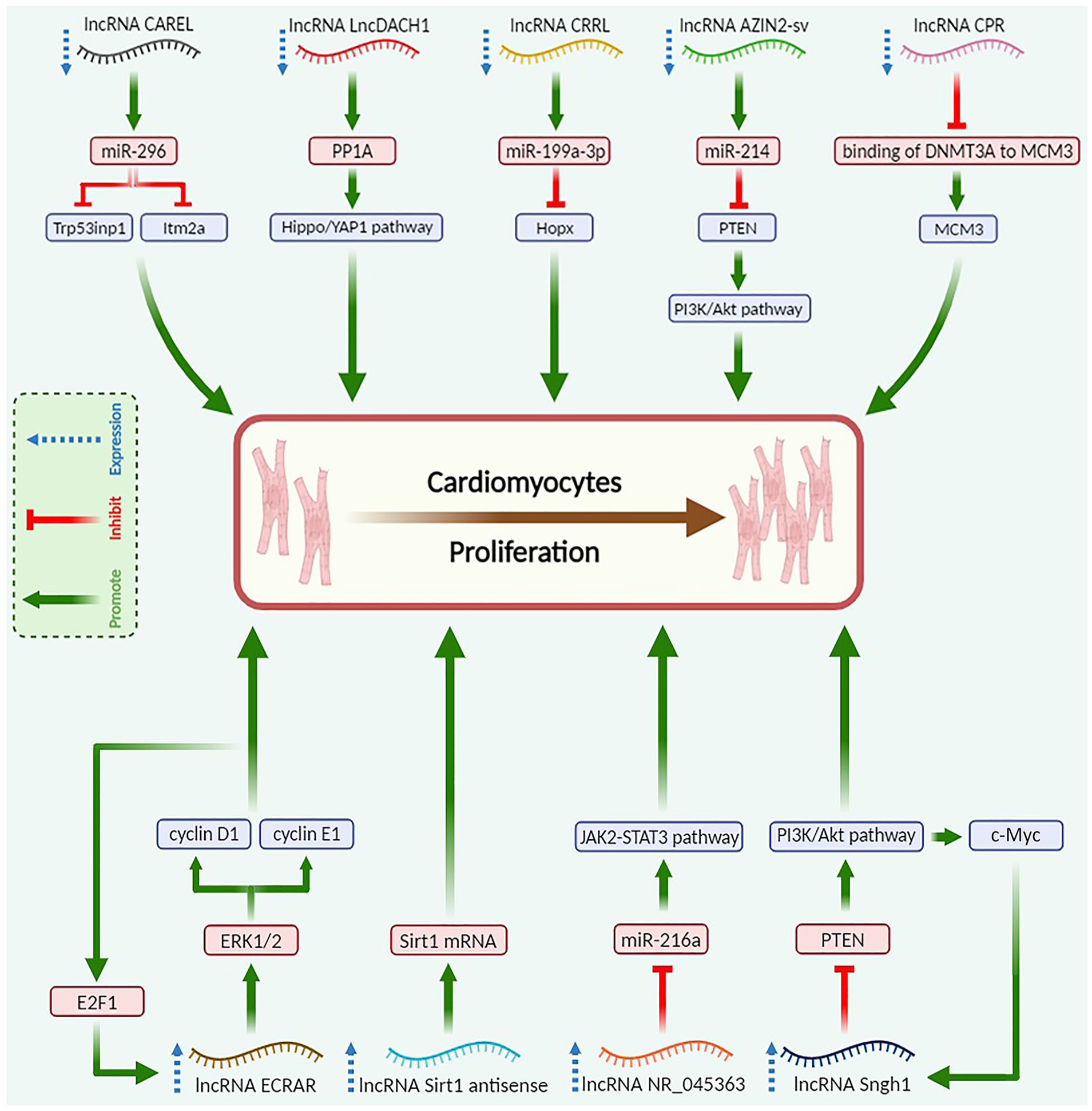
Molecular mechanisms of lncRNAs to promote the CM proliferation. Created with BioRender.com.
LncRNAs With Upregulated Expression in Embryonic to the Adult Stage
Increased expression of lncRNA CAREL in the newborn heart leads to the impairment of the heart’s regeneration capacity. CAREL deficiency increases the ability of cardiac cells to replicate after ischemic damage. Myocardial scar size has significantly decreased, and ejection fraction (EF) has improved along with increased mitotic activity, supporting these findings. CAREL is an endogenous RNA that competes with miR-296 and helps control the growth of CM. miR-296 is interested in specific genes called TRP53INP1 and ITM2A 15 .
LncDACH1 is the other lncRNA identified as a key player in cardiac regeneration via negatively regulating CM proliferation. The overexpression of LncDACH1 suppresses neonatal cardiac regeneration (NCR). Knockout of LncDACH1 enhances cardiomyogenesis following an ischemic injury. LncDACH1 directly binds to the protein phosphatase 1A (PP1A), limiting its activity within CMs. By antagonizing PP1A, LncDACH1 modulates the Yes-associated protein 1 (YAP1) phosphorylation levels. By starting the transcription of certain genes, YAP1 is involved in many normal and diseased heart processes, such as cell death, differentiation, and proliferation 16 .
LncRNA CRRL (Cardiac Regeneration-Related LncRNA) regulates cardiac function and remodeling following MI. Reducing CRRL enhances adult cardiac performance and decreases left ventricular (LV) remodeling after an MI. Notably, CRRL knockdown promotes CM proliferation without inducing cardiac hypertrophy, potentially offering a promising avenue for enhancing cardiac repair. CRRL overexpression inhibits post-MI healing in rat hearts. CRRL functions as a competitive endogenous RNA (ceRNA) for miR-199a-3p to promote CM replication by targeting Hopx, a gene involved in cardiac repair 17 .
LncRNA AZIN2-sv suppresses CM reproduction both in vitro and in vivo. Loss of AZIN2-sv after an MI, on the contrary, seems to protect cardiac function by possibly increasing endogenous CM proliferation and angiogenesis. Notably, the specific impact of this lncRNA on neonatal CM proliferation remains unexplored. In the cytoplasm, AZIN2-sv binds directly to miR-214 at certain spots. This controls CM proliferation by changing the miR-214/PTEN/Akt pathway 18 .
Inhibition of Cardiomyocyte Proliferation Regulator (CPR) lncRNA effectively promotes karyokinesis and cell division in both adult and postnatal CMs. This suggests that CPR is involved in stopping CM proliferation. CPR overexpression inhibits cardiomyogenesis and impairs NCR. DNA methylation silences the MCM3 gene, and CPR helps with this by bringing DNMT3A to the promoter CpG sites 19 .
LncRNAs With Downregulated Expression in Embryonic to Adult Stage
The in vivo and in vitro experiments have shown that the overexpression of lncRNA ECRAR may be involved in NCR by promoting postnatal CM proliferation through the ERK1/2-mediated pathway. The E2F1-ECRAR-ERK1/2 signaling mechanism irreversibly helps the G1/S transition, leading to cell cycle re-entry and inducing proliferation of CM. Knockdown of ECRAR suppresses post-MI cardiomyogenesis and heart recovery. Echocardiography (Echo) and morphological evaluations demonstrate that ECRAR overexpression improves post-MI cardiac function due to remarkably inducing myocardial regeneration after MI 20 .
LncRNA Sirt1 antisense has a potential regulatory effect on CM regeneration and cardiac repair. The overexpression of Sirt1 antisense lncRNA is necessary for proliferation of neonatal CM in vivo and in vitro. Sirt1 antisense lncRNA enhances Sirt1 mRNA stability by extending its half-life, upregulating Sirt1 protein level in Sirt1 antisense lncRNA overexpressed CM. The expression of Sirt1 in vivo is stimulated by lncRNA Sirt1 antisense to increase post-MI CM proliferation and prevent CM apoptosis after MI. The two-dimensional Echo showed that overexpression of Sirt1 antisense lncRNA improves cardiac function after MI by reducing ventricular chamber enlargement, both in diastole and systole 21 .
Lack of lncRNA Snhg1 decreases the proliferative potential of CMs and inhibits NCR. Snhg1 reduces phosphatase and tensin homolog (PTEN) protein levels, activating PI3K-AKT phosphorylation to promote post-MI cardiac repair. C-Myc, regulated by the PTEN/PI3K/Akt pathway, acts as an activator for Snhg1. It forms a positive feedback loop with Snhg1 to enhance CM proliferation. In addition, Snhg1 controls angiogenesis by increasing vascular endothelial growth factor (VEGF). Echo results show improvement in heart function after Snhg1 overexpression 22 .
Analyzing the heart of neonatal mice after apex resection revealed that the upregulation of lncRNA NR_045363 plays an important role in the NCR. The downregulation of lncRNA NR_045363 was found to have adverse effects on DNA synthesis, cardiac cell cytokinesis, and CM mitotic activity. The overexpression of NR_045363 downregulates miR-216a levels, which leads to an increase in the protein levels of phosphorylated JAK2-STAT3, suggesting that NR_045363 may regulate the JAK2-STAT3 axis by binding to miR-216a, thus promoting CM proliferation. NR_045363 overexpression improves heart function after MI including enhancing LVEF and LV fractional shortening, smaller LV end-systolic and diastolic diameters, and reduced infarct size 23 .
Discussion
The capacity of the new CM generation during MI is too restricted in the adult human heart. Thus, restoring the lost CMs and preserving the cardiac function by relying on the endogenous regeneration rate of CMs is indeed impossible. Accordingly, regenerative techniques are receiving extensive interest. Studies have shown that neonatal CMs or even adult ones can undergo proliferation and remodeling under certain conditions, highlighting the ability for cardiac repair after infarction through improving endogenous cardiac regeneration3,4,24. It might be possible to produce new CMs from embryonic or induced pluripotent stem cells and give them to the heart either as a cell suspension or upon ex vivo generation of contractile myocardial tissue. Either way, the generation of a particular gene or a more successful selection of miRNAs can stimulate CMs’ innate capacity for proliferation 25 .
Recently, lncRNAs have gained significant attention due to their crucial role in regulating gene expression and various biological processes. Five times exceeding the number of protein-coding genes, 96,308 lncRNA transcripts are known from human genome 26 . Unlike protein-coding genes and miRNAs, the majority of lncRNAs have poor interspecies sequence conservation 27 , although it is speculated that conservation of their secondary structure might exist 28 . LncRNAs are classified into six major categories based on their genomic location and orientation relative to protein-coding genes: (1) sense (2) sense intronic (3) antisense (4) bidirectional (5) enhancer (6) intergenic. In addition, they are divided into signal, decoy, guide, scaffold, enhancer, or sponge lncRNA based on their cellular functions 7 .
A growing body of evidence has revealed the vital roles that lncRNAs play in various processes, including immunological response, disease development, pluripotency and differentiation circuit control, and chromosomal dynamics 29 . Since lncRNAs are widely expressed in the cardiovascular system, it is thought that they comprise a sophisticated regulatory network that controls both normal and abnormal aspects of heart function. LncRNAs have a crucial role in the development of MI and in preventing complications following a heart attack by controlling angiogenesis, cell proliferation, and other biological processes30,31. Research indicates that lncRNAs can be identified in plasma or urine and exhibit dynamic changes with the onset and progression of heart failure. Recent studies have found that lncRNAs such as LIPCAR, SENCR, and MIAT can be detected in plasma, being potential biomarkers for heart failure 32 .
Moreover, studies have found different lncRNAs involved in the pathogenesis of other heart diseases. Heat2 is an immunogenic lncRNA that increases in the blood of patients with heart failure and regulates cell function 33 . A study by Wang et al. 34 discovered that lncRNA CHRF, which is an endogenous miR-489 sponge, binds directly to miR-489 and controls the production of Myd88 and cardiac enlargement. Another study indicated that increased levels of lncRNA CCRR reversed adverse effects, while reducing CCRR destroyed the intercalated disks and gap junctions, decreasing longitudinal heart conduction 35 .
There is significant potential for discovering new lncRNAs involved in the regulation of cardiac regeneration. However, the identification process is hampered by the limited availability of libraries for systematic screenings, as all known lncRNAs associated with cardiac regeneration have been discovered in recent years. In this systematic review, we found nine lncRNAs with a crucial role in cardiac regeneration after MI which have been investigated since 2018. Our review showed that lncRNAs that affect cardiac regeneration could be divided into positive and negative regulators. The CAREL, DACH1, CRRL, AZIN2-sv, and CPR lncRNAs negatively regulate CM proliferation by various mechanisms: CAREL and CRRL act as sponge lncRNAs; DACH1 is a decoy lncRNA that directly binds to a specific protein phosphatase; AZIN2-sv is a sponge and protein stabilizer lncRNA; and CPR acts as a guide lncRNA. Compared to other negative regulators, CPR has effects on CM number and post-MI CM apoptosis, making it a favorable target.
In contrast, ECRAR, Sirt1 antisense, Snhg1, and NR_045363 are the positive regulators of CM proliferation. ECRAR is a signal lncRNA, Sirt1 antisense stabilizes mRNA and increases protein level, Snhg1 is a signal lncRNA, and NR_045363 acts as a sponge lncRNA. Among positive regulators, Sirt1 antisense and ECRAR might be more potent in cardiac regeneration due to additional effects on CMs compared to others including decreasing CMs apoptosis and CMs cross-sectional area by Sirt1 antisense and decreasing post-MI fibrosis and increasing capillary and arteriole density by ECRAR.
There has been extensive research into the therapeutic potential of lncRNAs due to their vital role in regulating CM proliferation. The first step is finding a way to target lncRNA to the intended destination to enact their biological functions. Over the past two decades, viral particles have emerged as an effective way for delivering therapeutic noncoding RNAs, packaged within plasmids. Adenoviral-based gene delivery, in particular, has been widely used in scientific research due to its high transfection efficiency and robust transgene expression. For instance, adenovirus vectors carrying shAZIN2-sv, when injected into the myocardium, have been shown to preserve cardiac function in adult rats, reduce infarct area, and promote angiogenesis from 14 to 60 days post-MI injury 18 . However, there are some obstacles: (1) their transient expression profile limits their utility in treatment, and (2) they can trigger host immune responses.
Furthermore, overcoming the gap between basic and clinical research requires considerable effort. A significant hurdle in developing new therapeutic approaches is that most of the present findings have not yet been investigated, or have failed in large animal models. The poor sequence conservation of lncRNAs between different species makes animal translational studies challenging36,37. As a result, only lncRNAs that are well conserved across species hold promise as viable targets for new therapies, yet the number of such conserved lncRNAs is still quite limited12,38,39.
So far, only two studies involving lncRNAs have been conducted in large animals. Hennessy et al. 40 identified lncRNA CHROME, which was upregulated in nonhuman primates with atherosclerotic vascular disease. Similarly, Li et al. 41 found that the lncRNA H19 was overexpressed in two mouse models of abdominal aortic aneurysm and in a low-density lipoprotein receptor (LDLR) knockout mini-pig aneurysm model. There have not been any clinical trials conducted to look into the possibility of lncRNAs proliferating in the heart yet. Ultimately, since no lncRNA treatment strategy has been used in large animals to date, more and more therapeutic research in large animal models is required.
The novel treatments based on targeting the expression of lncRNAs about CM proliferation may potentially prevent life-threatening complications after MI and improve cardiac function by inducing cardiac regeneration. Furthermore, the combination of lncRNA-based targeted therapy with conventional treatments for MI and post-MI complications may be a promising option to improve cardiac function after MI, leading to prolonged survival and a good prognosis in patients with MI. However, there are many limitations and challenges. One of the remaining issues is the development of techniques that can adjust CM proliferation rate as a means of both increasing and limiting this process. Unintended proliferation can lead to serious pathologies including uncontrolled hypertrophy and myocardial enlargement. Several investigations on both rats and pigs have documented severe consequences in response to extended treatment with different miRNAs; these included cardiomegaly, impaired heart function, arrhythmia, and sudden death42,43.
The real role that increased CM proliferation can play when considering the heart as a whole, is another issue that has to be confirmed. Several research works have documented noteworthy enhancements in both heart structure and function, which have been linked to this procedure. A little increase in CM cell cycle activity was, nonetheless, occasionally seen; however, this was insufficient to support the overall positive benefits16,23,44. It is important to take into account that additional factors like increased angiogenesis, hypertrophy, decreased apoptosis, or changed metabolic activity can also be important.
In addition, humans possess unique genetic and physiological features that are not fully simulated by research models, complicating the translation of findings into human applications. This problem is especially noticeable with lncRNAs, which are not well conserved across species; hence, in order to apply findings to human biology, humanized models or organoid cultures are frequently required 21 . In addition, the myocardium is made up of several cell types that interact and communicate in complex ways 43 . It is essential to comprehend how these various cell types interact with regeneration processes. Gaining further insight into these multicellular interactions may help regulate and promote the growth of CMs.
Although the development of lncRNA therapy for cardiac dysfunction is still in its early stages, lncRNAs present a promising new avenue for future treatment. Since more lncRNAs are likely to be involved in the control of cardiac regeneration in the near future, lncRNA-based therapy is a very promising strategy for the treatment of heart failure. However, to create more effective, safe, and efficient therapeutic approaches for the treatment of patients with cardiac diseases, a deep and comprehensive understanding of the molecular targets and regulatory mechanisms of lncRNAs is required. The safety and effectiveness of new lncRNA treatments will need to be assessed in big animals before human clinical trials are started since small rodent models are the main in vivo tools used in heart regeneration research. In addition, to analyze the many biological and molecular processes that underlie the activity of lncRNAs, models using rodents and cell cultures will be required. A single lncRNA can bind hundreds or even thousands of mRNAs, influencing gene expression networks. Moreover, many lncRNAs might target a single mRNA, illustrating the intricate web of connections.
Therefore, more research is required to gain a deeper knowledge of the precise roles and molecular processes of lncRNAs in cardiac regeneration. Furthermore, to overcome the numerous intrinsic barriers to heart regeneration, combinatorial treatment methods may be necessary for the repair of the failing human heart. The development of new MI treatments hence requires consideration of a combinatorial lncRNA targeting method.
Conclusion
Taken together, this systematic review provides fundamental evidence about critical lncRNAs, which play a significant role in promoting or inhibiting the post-MI proliferation of CMs, leading to cardiac regeneration. In the future, advancements in sequencing and interfering technologies will uncover other lncRNAs related to heart regeneration, necessitating more significant translational investigations. However, recent studies have documented important findings that could be fundamental to future research on the role of lncRNAs in cardiac regeneration after life-threatening cardiovascular diseases, especially MI. Thus, lncRNAs whose expression is up- or downregulated in embryonic CM may be good targets for new medicines that can help MI patients overcome the problem caused by CM death.
Footnotes
Author Contributions
D.D. has contributed to the study concept and design and compiled the database search results. D.D. and AH.B. designed the inclusion and exclusion criteria and determined the steps of data collection. AH.B. and M.R. reviewed the literature and extracted data. D.D. and AH.V. prepared the figures. AH.B. and AM.A. generated the tables. All authors wrote the original draft. D.D. and M.R. critically revised the manuscript. D.D. supervised this project. All authors read and approved the final manuscript.
Availability of Data and Materials
Not applicable (the current study was performed based on published literature and no datasets were generated).
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
