Abstract
Islets have a high demand for oxygen and most of them will die of hypoxia injury before and after transplantation. Hypoxic damage is one of the key factors associated with islet graft dysfunction. Mesenchymal stem cells (MSCs) have multiple functions and can enhance the therapeutic effect of islet transplantation. In this study, islets were cultured together with or without MSCs derived from umbilical cord (hUC-MSCs) under normal and hypoxic conditions. The effect of hUC-MSCs on the survival and function of isolated islets was detected by immunofluorescence and ELISA. Hypoxia-inducible factor 1 alpha (HIF-1α) and PFKFB3 mRNA and protein expression in different conditions were tested by real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) and Western Blot. The islets co-cultured with hUC-MSCs have improved viability and function compared with islets cultured alone. The mRNA transcription of HIF-1α in the co-cultured group increased. The protein expression of PFKFB3 increased with the increase of HIF-1α. This study found that hUC-MSCs could protect islets from dysfunction caused by hypoxia, and HIF-1α/PFKFB3 played an important role in hypoxic resistance, suggesting a potential strategy to improve the outcome of islet transplantation.
Introduction
Diabetes has become a rising public health problem worldwide. It is estimated that more than 500 million people worldwide suffer from diabetes 1 . Allogenic islet transplantation is one of the most potential treatment options for restoring normal blood glucose in patients with type 1 diabetes 2 . However, despite some progress in recent years, pancreatic islet transplantation still has some shortcomings, such as the shortage of donor organs, the need for lifelong immunosuppression after surgery, and the low long-term survival rate. The survival of transplanted islets is closely related to the microenvironment of the transplantation site. Recent studies have shown that whether the islets are transplanted into the liver or other sites, the role of oxygen (O2) is important for the survival of the islets 3 . Islet hypoxia is the main cause contributing to early graft loss. Loss of graft function caused by immune injury and hypoxia are the main obstacles that limit the widespread application of islet transplantation 4 . Unfortunately, the mechanism of hypoxia and hypoxic injury in islet transplantation is not fully understood. In addition, pancreatic islets are cell clusters with an average diameter of about 150 μm. Isolated islets cultured in conventional conditions are usually exposed to a dynamic change of oxygen partial pressure from normoxic, through hypoxic, to anoxic levels.
Our previous research found that co-culture of mesenchymal stem cell (MSCs) and islets in vitro can prolong the survival time of islets and enhance the tolerance of islets to ischemia and hypoxia5,6. Human umbilical cord-derived MSCs (hUC-MSCs) have several advantages than the adult MSCs 7 , easier to obtain, no damage to mother and child, and no significant changes in the biological characteristics of cells after cryopreservation and resuscitation, which make their applications in experiments more convenient and reliable. Therefore, it is very important to explore the protective effect of hUC-MSCs on islet transplantation and to clarify the mechanism of hUC-MSCs against the ischemia and hypoxic injury in islet transplantation.
The major regulator of cellular adaptation to low oxygen stress is hypoxia-inducible factor 1 alpha (HIF-1α). Under hypoxia conditions, HIF-1α activates a series of target genes involved in cell energy metabolism, cell survival and apoptosis, and angiogenesis8–10. According to reports, HIF-1α is an important molecule that regulates pancreatic islets cell function regardless of normoxia or hypoxia3,11. It is well known that the glycolytic activator 6-phosphofructo-2-kinase/fructose 2,6-bisphosphatase 3 (PFKFB3) is an important signal enzyme for glucose metabolism and a downstream substrate of HIF-1α signaling pathway12–14. In this study, we hypothesize that hUC-MSCs may have a cytoprotective effect on pancreatic islets during hypoxia by regulating HIF-1α/PFKFB3. To verify this hypothesis, we performed co-culture of the purified mouse pancreatic islets with a certain number of hUC-MSCs under hypoxia in vitro. We use this experimental model to test whether MSCs would enable pancreatic islets to survive under conditions of hypoxic stress and whether this effect is associated with HIF-1α/PFKFB3.
Materials and Methods
Isolation and Expansion of hUC-MSCs
All protocols and animal procedures detailed below were approved by the Ethics Committees of Lu He Hospital, Capital Medical University, and were performed following the principles of Management Rules of the Ministry of Health of China. The isolation and passage of hUC-MSCs was done following the methods of Bharti et al 15 . The umbilical cord was washed with phosphate buffer saline (PBS) at 4°C, cut into 1 to 2 cm small pieces, and the umbilical cord arteries and veins were removed (Fig. 1A). After removing 1 umbilical vein and 2 umbilical arteries, carefully bluntly separated the Wharton’s jelly, and then mechanically cut them into 1 mm3 fragments with ophthalmic scissors. The pellets obtained were seeded in 10-cm culture dish supplemented with α-MEM medium (Biological Industries Israel Beit Haemek Ltd, Kibbutz Beit-Haemek, Israel) supplemented with 10% fetal bovine serum, penicillin (100 U/mL), and streptomycin (100 μg/mL). The cells were cultured to 80% to 90% confluence in incubator at 37°C and 5% CO2. In the same culture media, the same procedure was continued until they attained the fifth passage.
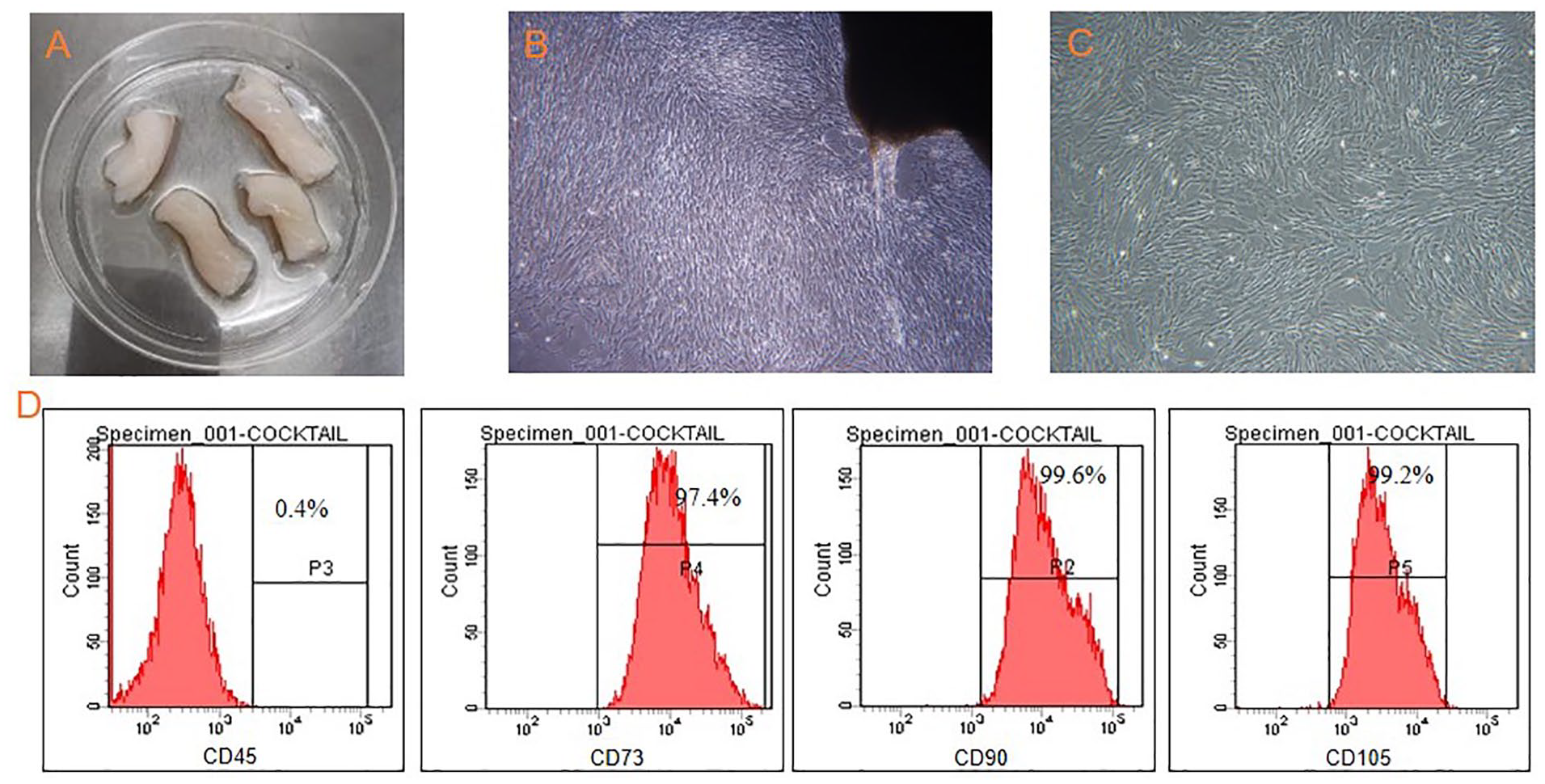
Morphology and imunophenotyping of hUC-MSCs. (A): Cutting of cord pieces. (B): The primary hUC-MSCs were grown from the edge of tissue explants (4×). (C): Homogeneous population of fibroblast-like MSCs at passage 3 (4×). (D): Flow cytometry analysis of hUC-MSCs at passage 3. hUC-MSCs: human umbilical cord-derived MSCs; MSCs: mesenchymal stem cells.
Flow Cytometry Test and Characterization of hUC-MSCs
The primary, third and fifth passage hUC-MSCs were collected, respectively, and characterized for the expression of unique cell surface markers. The hUC-MSCs were digested with 0.25% trypsin and washed with PBS containing 0.5% bovine serum albumin to prepare a cell suspension. Mouse anti-human monoclonal antibodies, CD73 (APC Tagged), CD90 (FITC Tagged), CD105 (PerCP-cy5.5Tagged), and CD45 (PE Tagged) (BD Biosciences) were added, mixed well, and incubated for 30 min on ice. Appropriate isotype controls were run to determine nonspecific fluorescence. We followed the methods of Ali et al 16 . After completion of incubation, the cells were washed and resuspended in 300 μl of PBS. Then were transferred into polystyrene round-bottom tubes and detected on BD FACS CantoII (BD, Franklin Lakes, NU, USA). The data was analyzed using Flowjo software (FlowJo LLC., Ashland, OR, USA).
Mouse Islets Isolation and Co-Culture
Male C57BL/6 mice were used at 8–12 weeks of age for all experiments. The isolation and purification of mouse islets were performed by collagenase P and Ficoll as we previously described 17 . Eighty to 100 mouse islets were used per experimental well. For co-culture experiment, 1 × 105 hUC-MSCs were seeded into the lower chamber of a 24-well transwell plate (CorningBioCoat, Corning, NY, USA) containing co-culture medium. After hUC-MSCs became attached, the medium was changed immediately and the freshly isolated islets were placed into the upper chamber (indirect contact co-culture). To test the direct contact co-culture, islets were seeded into wells having hUC-MSCs without transwell. Before the hypoxia operation, the specific inhibitor 2-methoxyestradiol 18 (2-MeOE2 or 2ME2, 100 μM, S1233, Selleck, Shanghai, China) and the activator dimethyloxalylglycine 19 (DMOG, 100 μM, S7483, Selleck, Shanghai, China) were, respectively, added to the different conditions of islets culture medium to suppress or activate HIF-1α. At the same time, in the presence of hUC-MSCs and without hUC-MSCs, the islets of different groups were placed in normoxia (20% O2, 37°C, 5% CO2) and hypoxia (1% O2, 37°C, 5% CO2) co-cultured for 48 h. The following functional-related parameters were evaluated.
Islet viability determination
To detect the viability of the isolated islets, the cell permeable esterase substrate fluorescein diacetate (FDA, Sigma Aldrich) and cell impermeant nucleic acid stain propidium iodide (PI, Sigma Aldrich, Inc., St.Louis, MO, USA) were used to perform viable cell and dead cell staining. This experiment was carried out by 3 independent researchers, and the FDA/PI staining followed the procedures of the NIH Clinical Islet Transplantation Consortium 20 . The 24 μM (9.9μg/mL) FDA stock solution was made in acetone while 750 μM (0.5 mg/mL) stock of PI was made in Dulbecco’s phosphate-buffered saline (DPBS). Islets were incubated with 0.46 μM of FDA in DPBS for 2 min followed by 14.34 μM of PI for 30 s. Fluorescent microscope was used immediately to observe and access pictures. After staining with FDA, viable cells showed bright fluorescent green. Dead cells appeared bright fluorescent red/orange. The ratio of green cells to red cells represented the percentage of islet viability. The viability of islets treated with and without hUC-MSCs under normoxic and hypoxic conditions was evaluated.
Islet functionality-glucose-stimulated insulin secretion (GSIS) assay
To evaluate the GSIS level of pancreatic islets in different experimental groups, we conducted experiments according to the protocol published in Somesh et al 21 . A subset of islets (n = 10) from each experimental condition were carefully selected and added to 24-well plate containing PBS. First, we washed islets with 300 μL of PBS twice, then added 350 μl of Kreb’s Ringer Hepes Buffer (KRBH) without glucose to each well, and incubated for 1 h at 37°C in CO2 incubator. After incubating for an hour, islets were washed with PBS twice, and sequentially KRBH buffer containing low glucose (2 mmol/L) and high glucose (20 mmol/L) were added to stimulate islets. After stimulation with high-glucose and low-glucose solutions for 1 h, the supernatant was collected and stored at −20°C. A commercial mouse insulin ELISA kit (Millipore, #EZRMI-13K, USA) was used to detect insulin levels in all samples. The ratio of insulin secreted in high glucose to that secreted in low glucose was expressed as the stimulated index (SI).
Quantitative RT-PCR
According to the total RNA extraction kit (TRIZOL, Invitrogen, Carlsbad, CA, USA), the total RNA was extracted using Trizol reagent from approximately 400 islets under each condition, and quantified with Nanodrop 2000 (Thermo Fisher Scientific, Madison, WI, USA). One to 2 μg total RNA was reverse-transcribed into cDNA using the iScript cDNA Synthesis Kit (Cat# 1708891; Bio-Rad, Hercules, CA, USA). After cDNA was arranged in a certain order, quantitative RT-PCR reactions were carried out with SYBR Green PCR Master Mix (Bio-Rad) system. The reference standard was the housekeeping gene 18s. The 2−ΔΔCT method was used as described previously by Schmittgen and Livak 22 . We recorded the Ct value when the housekeeping gene and the target gene of the sample reach the upper inflection point during PCR amplification and converted it to −ΔΔCT to analyze the relative expression of the target gene.
Western Blot Analysis
Thirty islets under different conditions were collected, centrifuged, and washed twice with pre-cooled PBS. Appropriate amount of cell lysate radio-immunoprecipitation assay (RIPA) containing protease inhibitors was added to the cells. The pellets were resuspended and lysed on ice for 15 min, then centrifuged at 4°C, 12,000 × g for 10 min, and the supernatant obtained was the total protein. The protein concentration was measured by bicinchoninic acid (BCA) method (Thermo Fisher Scientific, Madison, WI, USA). Then they were boiled at 95°C for 5 min, separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membrane, blocked in 5% nonfat milk for 1 h, washed with 1×Tris-buffered saline with Tween-20 (TBST) and incubated with the primary antibody HIF-1α (1:1000), PFKFB3 (1:500), and GAPDH (1:1000) overnight. The membrane was washed with 1× TBST and incubated for 1 h at room temperature with the corresponding horseradish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody (1:3000) or goat anti-mouse secondary antibody (1:3000) for 1 h at room temperature. After washing in 1× TBST again, ECL luminescent reagents were added and displayed according to the manufacturer’s instructions on Bio-Rad’s ChemiDoc XRS+ system.
Results
Characterization of hUC-MSCs
The hUC-MSCs were grown from the edge of tissue explants (Fig. 1B) and created a homogeneous monolayer of adherent spindle-shaped cells at passage 3 (×40) (Fig. 1C). The immunophenotypes of hUC-MSCs were identified by flow cytometry. hUC-MSCs at passage 3 were used in the subsequent experiment. Fig. 1D showed the representative pictures of the characteristics of the MSCs at passage 3. Fig. 1D showed that the percentage of CD90+, CD105+, CD73+, and CD45+positive cells were 99.6%, 99.2%,97.4%, and 0.4% respectively, which met the MSCs criteria defined by the International Society for Cellular Therapy 23 . It showed that the hUC-MSCs used in this study have good MSCs biological characteristics.
hUC-MSCs Enhance Survival of Islets Exposed to Hypoxia
Fig. 2 showed a micrograph of islets co-cultured with hUC-MSCs. The islets were slightly attached to the surface of hUC-MSCs at the bottom of the culture plate. After 48 h, pancreatic islets in normoxia were still intact, preserving their clustered structure better in co-culture with hUC-MSCs (Fig. 2. C1, D1) than islet alone (Fig. 2. B1). And there is no difference between the direct contact co-culture and indirect contact co-culture (C1 and D1). The envelope of islet became incomplete in hypoxia (B2), the majority cells inside the islet were apoptosis to show black appearance under the inverted microscope. In the co-culture group, the morphology of islet showed a clustered structure has no change in hypoxia (C2 and D2) compared with in normoxia. No cells inside the islet were apoptosis to show black appearance under the inverted microscope. The morphology of hUC-MSCs alone in normoxia (A1) and hypoxia (A2) showed no obvious change.

hUC-MSCs, murine islets alone, and islets co-cultured with hUC-MSCs at passage 3. hUC-MSCs were precultured for 2 days before the co-culture with islets. hUC-MSCs cultured alone in normoxia (A1) and hypoxia (A2); islets cultured alone in normoxia (B1) and hypoxia (B2); co-culture of pancreas islets with hUC-MSCs direct contact (C1, C2) and indirect contact (D1, D2) in normoxia (C1, D1) and hypoxia (C2, D2) in vitro. PI/FDA staining for determination of cell viability in pancreatic islets alone in normoxia (E1) and hypoxia (E2), islets cultured with hUC-MSCs in normoxia (F1) and hypoxia (F2). The small bright spots correspond to the intense red staining of PI-positive (dead) cells. The diffusely stained regions correspond to the FDA-stained (live) cells. In vitro assessment of experimental groups post 48-h co-culture period. Original magnifications: ×40. hUC-MSCs: human umbilical cord-derived mesenchymal stem cells; PI: propidium iodide; FDA: fluorescein diacetate.
Islet viability
The co-cultured islets were stained by FDA/PI. More disintegrated islets were observed in islet alone in the normoxia (E1) and hypoxia (E2) than the co-cultured group (F1, F2). For the co-culture group, whether under normoxia or hypoxia, most of the cells in the islets are green, only a few are red, and the survival rate of islets was >90%. The survival rate of the islet in the group of islet culture alone was only less than 50% (normoxic) and less than 30% (hypoxic).
Islet functionality
Basal insulin secretion under static incubation with 2.0 mM glucose tended to be lower but not significant compared with that in co-cultured islets in normoxia (Fig. 3A). The secretion significantly increased with an increase of glucose concentration (20.0 mM) (Fig. 3B). The stimulation index (SI) was significantly different between the co-cultured group and islet alone group both in normoxia and hypoxia conditions (Fig. 3C) (P < 0.05). GSIS was impaired in islets alone in hypoxia. Compared with the islets cultured alone, the co-cultured islets with hUC-MSCs maintained higher sensitivity to glucose stimulation. hUC-MSCs maintain the functionality of the islets under hypoxic conditions.

Results of the glucose-stimulated test. To assess the function of different conditions, insulin secretion induced by glucose was measured. Duplicate batches of 10 islets each were incubated for 1 h in the presence of 2.0 mM or 20.0 mM glucose. Insulin content secreted during basal (A) and stimulated (B) phase are expressed as ng/mL. Insulin expressed as the stimulation index (SI) (mean ± SD) for 3 independent experiments. hUC-MSCs: human umbilical cord-derived mesenchymal stem cells. *P < 0.05, compared with islets alone group.
Regulation of HIF1α and PFKFB3 in Normoxia and Hypoxia
We conducted the qRT-PCR amplification of HIF-1α by using the specific primers after the reverse transcription. The results suggested that in normoxia, the expression of HIF-1α was weak. After hypoxia, the levels of HIF-1αincreased in both groups, especially the islets and hUC-MSCs co-cultured group (Fig. 4).
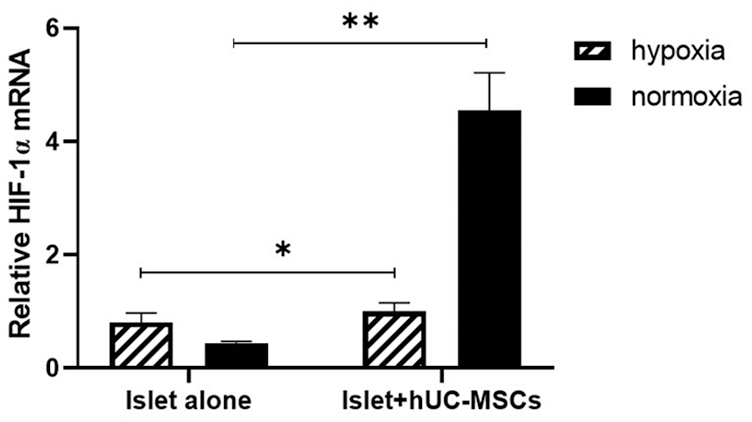
real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis of HIF-1α gene expression in islets. HIF-1α mRNA expression of islets was detected by quantitative real-time PCR (qPCR) assay after islets were cultured with or without hUC-MSCs for 48 h in normoxia and hypoxia. HIF-1α: hypoxia-inducible factor 1 alpha; hUC-MSCs: human umbilical cord-derived mesenchymal stem cells. *P < 0.05; **P < 0.001, n = 3 per group, statistical significance was determined by the unpaired t test.
We manipulated the levels of HIF-1α and PFKFB3 in the islets of different conditions in the presence and absence of hUC-MSCs. After related interventions, the expression and content of glycolysis-related pathway proteins in each group are shown in Fig. 5. The results of Western blot showed that HIF-1α protein expression was significantly up-regulated under hypoxia culture. HIF-1α and PFKFB3 protein expression in the co-culture group were significantly higher than that in the control group under hypoxic conditions. After intervention with HIF-1α inhibitor and agonist, the expression of each group of proteins was significantly higher and lower than that of the islet alone group, respectively.

Western blot of HIF1α and PFKFB3 levels in protein extracts from islets cultured in different conditions.
Discussion
The death and apoptosis of islets were caused by damage during islet isolation and purification, long time of isolation, and no microvascular regeneration at the early stage of transplantation. MSCs can secrete many biologically active substances, reduce the adverse effects of damage factors on other cells, and maintain the growth and vitality of cells. Many studies have used it as a seed cell to repair transplanted islet damage24,25. However, the specific mechanism by which MSCs play an important role in enhancing the ability of islet cells to resist hypoxic injury is currently still unclear. HIF-1α is a nuclear transcription factor that adapts to intracellular oxygen concentration. It consists of a regulatory HIF-1 subunit and a structural HIF-1 cellular subunit. Under normal physiological conditions, HIF-1α is in a dynamic balance of expression and degradation 26 . Hypoxia quickly activates the expression of HIF, especially HIF-1α, while the hydroxylation of prolyl hydroxylase is inhibited, and one of the main changes in the promoted cell metabolism is from aerobic to anaerobic metabolism 27 . The disruption of the hypoxia signaling pathway caused by HIF-1α knockout can also lead to islet dysfunction 28 . Therefore, it is inferred that the biologically active substances secreted by MSCs may regulate the HIF1-α pathway to prevent its binding to the β subunit into the nucleus under hypoxic conditions, thereby maintaining the mitochondrial oxygen consumption and ensuring the function of pancreatic islets in response to glucose stimulation.
In the present study, the isolated hUC-MSCs confirmed the unique characteristics of MSCs and displayed positive expression of classical MSCs cell surface markers (Fig. 1). It was showed that hUC-MSCs could maintain islet function through co-culture in vitro and effectively reduce islet cell death, improve cell activity (Figs. 2 and 3), which may be a joint effect of stem cell secretions on anti-inflammatory, pro-cell repair, and anti-apoptosis.
We further investigated the expression of HIF-1α at the transcriptional and protein levels. Because HIF-1α is a major regulator of cellular and developmental responses to hypoxia. Similar to the results of published studies, we found that HIF-1α expression is low under normoxic conditions, but is relatively high under hypoxia. HIF1α regulates the expression of various genes and mediates adaptive responses to hypoxia (Fig. 4).
Therefore, we developed the current work based on our interest in using intracellular HIF-1α activity as an indicator of the activation of these downstream pathways. According to previous studies, HIF-1α is expressed in different tissues and regulates target genes involved in angiogenesis, cell proliferation, and inflammation, and its expression is associated with different disease states 29 . Studies have shown that HIF-1α is a key transcription factor driving PFKFB3 expression in macrophages 30 . Montemurro et al showed that early cell calcineurin of type 2 diabetes patients increase glycolysis by activating HIF1α/PFKFB3, that is, the fission of mitochondria and their perinuclear distribution induce the conversion from oxidative phosphorylation to glycolysis. It can resist the increase of cytosolic Ca2+ caused by toxic oligomer stress, and the continuous death of β cells delaying beta-cell loss at the expense of beta-cell function through activating HIF1α/PFKFB313. Similarly, HIF1α/PFKFB3 is activated in the β cells of patients with type 1 diabetes, and likely contributes to the relatively slow rate of beta cell loss at the expense of early defective glucose-induced insulin secretion 14 . In this study, we demonstrated that there is an interaction between PFKFB3 and HIF-1A using an agonist and inhibitor of HIF-1α (Fig. 5). Specifically, hUC-MSCs promote islet hypoxia tolerance through the HIF-1A/PFKFB3 pathway, highlighting a new mechanism by which MSCs protect islets.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Lu He Hospital, Capital Medical University and Capital Medical University Institutional Animal Care and Use Committee (IACUC), Beijing, China.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Ethics Committee of Lu He Hospital, Capital Medical University approved protocols (2021-LHKY-020-02). All of the experimental procedures involving animals were conducted in accordance with the guidelines and approved by the IACUC of Capital Medical University (AEEI-2021-031), Beijing, China.
Statement of Informed Consent
Written informed consent was obtained from the patients, and their personal information were not involved in this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the following grants: Capital Medical University Funding (PYZ19085), Discipline construction project of Peking Union Medical College, and Lu He Hospital Funding (LHYY2019-JC19).
