Abstract
Keywords
Introduction
Cervical esophageal defection is caused by esophageal cancer, malignancies in the head and neck region and the upper gastrointestinal tract, structural congenital anomalies, massive burns, infections, and autoimmune diseases. 1 In recent years, the incidence of esophageal cancer has increased worldwide. 2 Surgical replacement therapy requires the stomach, jejunum, colon, and other autologous tissues as a substitute for esophageal tissue. Unfortunately, replacement may lead to high morbidity and mortality. Furthermore, damage to normal tissue seriously impacts the quality of life.3–5 The human esophagus has limited regeneration capacity. Unlike other organs, the esophageal cannot be transplanted. Therefore, esophageal tissue engineering has been developed to construct bionic esophageal stents, which avoids obtaining material from the patient and reduces the risk of high-risk surgery and high mortality and morbidity driven by postoperative surgery. 6 The success of esophageal tissue regeneration will ultimately depend on the ability of the scaffold to mimic the properties of natural tissue. In addition, it depends on providing an environment in which normal regeneration can occur. One of the major challenges and difficulties in esophageal tissue engineering is the optimization of the source and quality of seed cells.
Mesenchymal stem cells (MSCs) are a heterogeneous subset of stromal stem cells found in various fetal and adult tissues, which can differentiate into multiple mesodermal cells. 7 MSCs are promising tissue regenerative cells with multidirectional differentiation, high expansion capability, and immunomodulatory capacity. Multiple studies have demonstrated that human umbilical cord mesenchymal stem cells (UCMSCs) retain the differentiation potential of adult stem cells both in vitro and in vivo. UCMSCs are capable of differentiating not only into adipocytes, osteoblasts, and chondrocytes.8,9 but also into endothelial cells, epithelial cells, neural stem cells, hepatocytes, cardiomyocytes, germ cells, and islet cells under specific induction conditions. 10 Studies have shown that UCMSCs have a greater proliferative capacity than MSCs derived from bone marrow and adipose tissue. Their basic biological properties are not preceded by significant differences upon culture expansion. 11 Therefore, UCMSCs may be a more ideal seed cell for tissue engineering research and applications.
Studies in recent years have shown that the microenvironment in which stem cells reside has an induction of differentiation.12,13 MSCs are expanded in the microenvironment and receive growth signals determining their cell fate. These signals include cell-cell, cell-matrix interactions, and transcriptional programs that activate or suppress MSCs genes. 14 The latest pharmacological studies demonstrated that some natural herbs or extracts can effectively induce directed differentiation of stem cells into target cells without toxic side effects. 15 The herbal medicine that acts on stem cells in a way similar to that of the channel ushering drug, which can guide the homing and differentiation of stem cells and promote directed migration. 16 Resveratrol is a polyphenolic compound named 3, 5, 4-trihydroxypentadiene, widely found in plants such as polygonum cuspidatum, peanuts, grapes, and veratrum. 17 The broad biological effects of resveratrol include anti-aging, anti-inflammatory, antioxidant, cardiovascular protective, hypolipidemic, immunomodulatory, and antitumor effects. 18 The anti-inflammatory and antioxidant properties of resveratrol leading to a reduction in the expression of pro-inflammatory factors are well recognized. 19 Furthermore, resveratrol has been implicated in promoting osteogenesis, adipogenesis, and neurogenesis of MSCs in vitro. 20 To date, no studies have been reported on the effect of resveratrol on the specific differentiation of hUCMSCs into esophageal fibroblasts.
Fibroblasts have long been known as important matrix components, providing structural integrity to tissues and contributing to healing after injury or damage by producing proteins such as collagen. 21 However, orientated differentiation of MSCs into target cells is critical to injury healing. Therefore, in the present study, our objective was to elucidate the pharmacological effect and the underlying mechanism of resveratrol on the differentiation of hUCMSCs into HEFs under co-culture conditions. The findings from the study may provide important clues for generating high-quality seed cells for the tissue-engineered esophagus and its clinical application.
Materials and methods
Materials
Purified Resveratrol (MF: C14H12O3, MW: 228.24, purity > 98 %) was purchased from Shanghai Yuan Ye Biotechnology Co. Ltd (Shanghai, China). A stock solution (10 μM) of resveratrol was dissolved in DMSO and frozen at - 20°C until use. The PI3K inhibitor LY294002 was purchased from MCE (New Jersey, USA). Dimethyl sulfoxide (DMSO) was obtained from Sigma-Aldrich (St Louis, MO, USA). Details of the antibodies used are provided in sections 2.4, 2.5, 2.8, and 2.11.
Cell culture
Isolation and culture of primary esophageal fibroblasts
Primary human esophageal fibroblast lines were established as previously described. 22 Isolation and culture of HEFs were approved by the ethics committee of the First Affiliated Hospital of Guangdong Pharmaceutical University (approval number: 2021-IEC-153) and with the written informed consent of the patients; normal esophageal tissues greater than 5 cm away from esophageal cancer were collected from patients undergoing esophagectomy. Exclusion criteria included that all patients who underwent esophagectomy were positive for infectious agents such as HIV, hepatitis B virus, hepatitis C virus, and infectious syphilis. Under aseptic conditions, the submucosa was removed from normal esophageal tissue and cut into 1 mm3 tissue pieces. The tissue pellets were placed into cell culture flasks. Dulbecco’s modified Eagle’s medium/Ham’s nutrient mixture F12 (DMEM/F12) containing 100 U/ml penicillin, 100 µmol/ml streptomycin, and 10% fetal bovine serum was added to the culture flask (all from Gibco, Grand Island, NY, USA). Flasks were placed in a constant temperature incubator containing 5% CO2 at 37°C for incubation. The medium was changed every 3 days. Cells were passaged when they reached 90% confluence. To ensure cell viability, only cells from the 3rd to 5th generation were used for this experiment.
Isolation and culture of primary human umbilical cord mesenchymal stem cells
The isolation and culture of hUCMSCs were approved by the Ethics Committee of the First Affiliated Hospital of Guangdong Pharmaceutical University (Ethical Approval number: 2021-IEC-153); fresh human umbilical cords were collected from healthy term parturients. Written informed consent from the patient was obtained prior to the study. Exclusion criteria included all deliveries that were positive for infectious agents such as HIV, hepatitis B virus, hepatitis C virus, and infectious syphilis, and those with pre-diagnosed genetic abnormalities. Human umbilical cord mesenchymal stem cells were isolated as previously described. 23 Under aseptic conditions, Wharton’s jelly was peeled off from the umbilical cord and cut into 1 mm3 tissue chunk size. The tissue chunks were placed in cell culture flasks. Dulbecco’s modified Eagle medium (DMEM/F12) containing 100 U/ml penicillin, 100 µmol/ml streptomycin, and 10% fetal bovine serum was added to the culture flask. Flasks were placed into a constant temperature incubator containing 5% CO2 at 37°C. The culture medium was changed every 3 days. Cells were passaged when they reached 90% confluence. To ensure cell viability, only cells from the 3rd to 5th generation were used for this experiment.
Multilineage differentiation
To establish osteogenic lipogenesis of hUCMSCs in monolayer culture, 3rd generation cells were inoculated into 6-well plates using lipogenic induction medium (HUXUC-90,031, Cyagen Biosciences, and osteogenic induction medium (HUXUC-90,021, Cyagen Biosciences,) to culture hUCMSCs according to the manufacturer’s instructions. 21 days later, oil red O staining and alizarin red (all from Cyagen Biosciences, Suzhou, China) staining were performed to assess the ability of hUCMSCs to differentiate into adipocytes and osteoblasts, respectively.
Immunofluorescence staining
For immunofluorescence staining, HEFs were fixed with 4% paraformaldehyde (Biosharp, Guangzhou, China) for 30 min at room temperature (RT), closed with 10% sheep serum (ZSGB-BIO, Beijing, China), and left overnight with Vimentin antibody (1:200, AF7013, Affinity Biosciences, OH, USA) at 4°C. The next day, the samples were incubated for 1 h with the CY3-conjugated goat anti-rabbit IgG (H+L) antibody (Bioss, Beijing, China). Finally, the nuclei were stained with DAPI (Beyotime, Shanghai, China), and the samples were analyzed with an inverted fluorescence microscope (Olympus, Japan).
Phenotypic characterization by flow cytometry
To identify the cell phenotype, 3rd generation cells were subjected to flow cytometric identification. The expression of surface markers of hUCMSCs was detected by flow cytometry. Cultured cells were resuspended in pre-chilled phosphate-buffered saline (PBS) at a concentration of 106 cells/ml and incubated with CD34-FITC Clone 4H11, CD45-PerCP-Cy5.5 Clone 30-F11 (all from EBioscience, Thermo Fisher Scientific, Waltham, MA, USA), HLA-DR-FITC Clone G46-6, CD105-APC Clone 266, CD90-FITC Clone 5E10 and CD73- PerCP-Cy™5.5 (all from BD Pharmingen, Franklin Lakes, NJ, USA), respectively, for 30 min at 4°C. Subsequently, the cells were washed twice with PBS and then flow cytometry (BD Biosciences, BD FACSCalibur) was used to detect surface markers in hUCMSC. Data were processed with FlowJo10 (FlowJo, LLC).
Cell proliferation assay
To evaluate the effect of resveratrol on cell proliferation, hUCMSCs were seeded in 96-well plates (6000 cells/well) and then treated with resveratrol at various concentrations (0 μM, 0.1 μM, 1 μM, 2.5 μM, 5 μM, 10 μM, 20 μM, 50 μM, 100 μM) for 1, 3, 5, 7, and 14 days. Cell proliferation was determined using the Cell Counting Kit-8 (Dojindo Molecular Technologies, Japan) following the manufacturer’s instructions. Absorbance at 450 nm was measured using a spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA).
Co-culture of hUCMSCs and HEFs cells
The differentiation of hUCMSCs was induced using a co-culture technique. Cells were cultured in 0.4 µm pore size Transwell® system inserts, which ensured no direct cell-to-cell contact (corning, Inc. USA) at a density of 1.3 × 105 cells per insert. hUCMSCs were inoculated into six-well plates, and HEFs were inoculated into the insert. The cells were then expanded separately and hUCMSCs and HEFs were inoculated in a 2:1 ratio in the lower and upper chambers using Transwell chambers, respectively. Resveratrol (0.1 μM) was added to the culture medium. Meanwhile, resveratrol (0.1 μM) and LY2904200 (10 μM) were added to the other culture medium. The culture was maintained for 14 days, and the medium was changed every 2 to 3 days.
The experiment consisted of five different groups: HUCMSCs cultured alone, HEFs cultured alone, HUCMSCs and HEFs co-cultured, HUCMSCs and HEFs co-cultured with resveratrol added for 14 days of induction, and HUCMSCs and HEFs co-cultured with resveratrol added for 7 days of induction followed by addition of the PI3K inhibitor LY294002. The Sirius red staining, immunocytochemistry, Western Blot, and qRT-PCR to examine the expression of genes and proteins related to each group of hUCMSCs.
Immunocytochemical staining
hUCMSCs were washed 3 times with PBS for 5 min and fixed with 4% paraformaldehyde for 30 min. The hUCMSCs were then washed 3 times with PBS for 5 min and incubated with 0.25% Triton X-100 (Beyotime, Shanghai, China) for 10 min at RT. hUCMSCs were sequentially washed 3 times with PBS for 5 min and blocked with 10% sheep serum for 30 min at RT. The hUCMSCs were then shaken from serum, collagen I (1:200; AF7001), collagen III (1:200, AF5457) antibodies were added, and cells were incubated overnight at 4° C. Beta actin (1:5000; AF7018) antibody (all from Affinity Biosciences, OH, USA) was subsequently added for 1 h at RT. Finally, hUCMSCs were washed with PBS 3 times for 5 min at RT. DAB (Solarbi, China) was used for color development and hematoxylin for re-staining.
Sirius red staining
After 14 days in culture, each group of cells was washed 3 times with PBS solution, fixed with 4% paraformaldehyde for 30 min in RT, and washed 3 times with PBS buffer. Cells were stained with Sirius red staining solution (Solarbi, China) for 10 min, and total collagen secretion of each group of cells was observed under an inverted microscope.
Quantitative real-time polymerase chain reaction
Primers used in quantitative real-time fluorescent quantitative PCR.

Western blot analysis
Cells were lysed in protein extraction reagent RIPA lysis buffer containing 10 mM phenylmethylsulfonyl fluoride (PMSF) and 1% phosphatase inhibitor mixture for 30 min at 4°C. The lysates were then centrifuged at 12,000 g for 15 min at 4°C, and the supernatant was retained. Protein concentration was determined using the BCA Protein Assay Kit. Equal amounts of protein from each extract were added to the protein sample buffer and heated at 5°C for 95 min, then run on a 10% SDS-polyacrylamide gel. These reagents were purchased from Beyotime Company (Shanghai, China). The separated proteins were electrically transferred to polyvinylidene difluoride nitrocellulose membranes (Millipore, Billerica, MA). Membranes were blocked with 5 % (w/v) bovine serum albumin (BSA/TBST), and specific primary antibodies were incubated on a shaker at 4°C overnight. The membranes were washed with TBST and then incubated for 1 h at RT with a secondary antibody (Beyotime, Shanghai, China). After being washed three times, the signal bands were detected by chemiluminescence western blot detection reagent (ECL kit) and autoradiographic film. The following antibodies were used: p-AKT (1:1000; AF0016), AKT (1:1000; AF6261), p-NF-κB (1:1000; AF2006), NF-κB (1:1000; AF5006), IL-6, TGF-β, collagen I (1:500; AF7001), collagen III (1:1000; AF5457), beta Actin (1:5000; AF7018), and GAPDH (1:5000; AF7021) antibody (all from Affinity Biosciences, OH, USA).
Statistical analysis
All data are expressed as mean ± standard error of the mean (SEM). Statistical significance was determined by nonparametric Kruskal–Wallis one-way ANOVA test with Bonfferoni’s correction and one-way ANOVA with Fisher LSD post-hoc test or Dunnett’s test. A computer program SPSS (version 22.0, Chicago, Illinois, USA) was used. The results were considered statistically significant when p < .05.
Results
Phenotypic characterization and lipogenic osteogenic differentiation ability of hUCMSCs
The hUCMSCs were characterized for their adhesion ability, typical mesenchymal stem cell marker expression, and lipogenic osteogenic capacity. The 3rd generation hUCMSCs grew in an appressed form and showed a characteristic spindle-shaped fibroblast morphology with vigorous cell growth (see Figure 1A). After 21 days of culture in an osteogenesis-inducing medium, alizarin red staining showed a large amount of calcium precipitation and a bright red color inside the cells, demonstrating the osteogenic differentiation ability of hUCMSCs (see Figure 1B). After 21 days of culture in a lipogenesis-inducing medium, oil red O staining showed a large amount of lipid precipitation in the cells and bright red granules in the cytoplasm, indicating that hUCMSCs had the ability of lipogenic differentiation in vitro (see Figure 1C). The immunophenotype of the cells was detected by flow cytometry. The high expression of CD73, CD90, and CD105 in human umbilical cord MSCs with positive rates of 99.4%, 97.5%, and 98.6%, respectively, and the low expression of the hematopoietic cell markers CD34, CD45, and HLA-DR, with positive rates of 0.29%, 4.62%, 0.2%, respectively, are shown in Figure 1D. Morphology of hUCMSCs, adipogenic and osteogenic differentiation ability, and flow cytometry analysis of hUCMSCs. (A) Morphology of hUCMSCs in P3 generation under an optical microscope (X100). (B) Osteogenic differentiation of hUCMSCs stained with alizarin red (X200). (C) HUCMSCs differentiated into adipocytes and stained with oil red O (X200). (D) Flow cytometry showed the expression of CD73, CD90, and CD105 in third-generation umbilical cord mesenchymal stem cells, with positive rates of 99.4%, 97.5%, and 98.6%, respectively. The expression of CD45, HLA-DR, and CD34 in third-generation umbilical cord mesenchymal stem cells, with positive rates of 4.62%, 0.2%, and 0.29%, respectively.
Phenotypic identification of HEFs
Inverted fluorescence microscopy showed the expression of vimentin in HEFs. After DAPI staining, the nuclei of HEFs showed blue fluorescence (see Figure 2-DAPI). Vimentin was expressed in the cytoplasm and surrounding area, showing red fluorescence (see Figure 2-vimentin). The merge results graph shows that each expression is in line with the characteristics of HEF (see Figure 2-merge). The identification results confirm that the cells are human esophageal fibroblasts. Expression of vimentin in HEFs. Typical images of immunofluorescent staining for vimentin in HEFs (X200).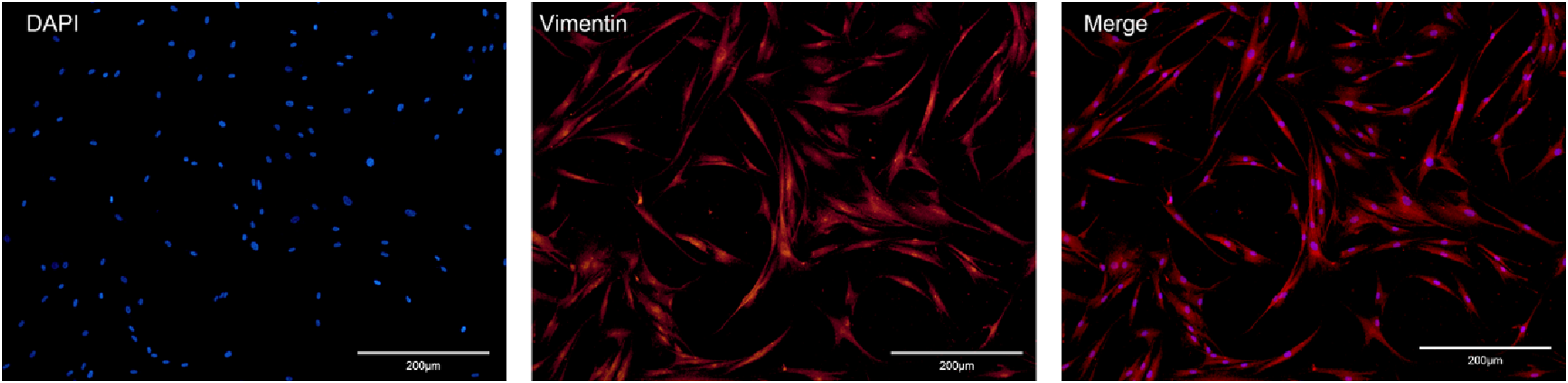
Resveratrol promotes the cell viability of hUCMSCs
To observe the effect of resveratrol on the cell viability of hUCMSCs, cells were cultured using a complete medium with different concentrations of resveratrol, and then cell viability was detected by CCK-8 on days 1, 3, 5, 7, and 14 after dosing treatment (see Figure 3). The results showed that compared to the blank control group, no significant differences in absorbance values were observed in the 0.1 μM, 1 μM, 2.5 μM, 5 μM, and 10 μM resveratrol-treated groups on days 1, 3, 5, and 7; the proliferation rate of hUCMSCs on day 7 decreased significantly at concentrations of 20 μM, 50 μM and 100 μM, indicating that low resveratrol concentrations of resveratrol had no toxic effects on cells. With the extension of the drug action time, the proliferation rate of hUCMSCs on day 14 increased significantly at resveratrol concentrations of 0.1 μM and 1 μM, and the differences were statistically significant. However, with the increased drug concentration of 2.5-100uM, the proliferation of hUCMSCs on day 14 began to be inhibited, and the differences were all statistically significant. The effect of resveratrol on the proliferation rate of hUCMSCs cultured separately for 1, 3, 5, 7, and 14 days. The effect of resveratrol on the cell viability of hUCMSCs through CCK8 (n = 5), the values are expressed as mean ± SEM, with *p < .05, **p < .01, ***p < .001.
Based on the above results, 0.1 μM of resveratrol culture solution was selected as the effective concentration for the subsequent co-culture experiment to observe its synergistic effect on the differentiation of hUCMSCs.
Resveratrol promotes the differentiation of hUCMSCs into fibroblasts in an indirect contact co-culture system
To establish a feasible and effective method for inducing differentiation of MSCs into HEFs, we used Transwell chambers and co-cultured MSCs with HEFs for 14 days to compare the expression of collagen genes in hUCMSCs in the co-culture group, the resveratrol-induced co-culture group, and hUCMSCs monoculture. As shown in Figure 4A, collagen I and collagen III of hUCMSCs increased significantly in the co-culture of resveratrol compared to the co-culture group and the monoculture of hUCMSCs. Resveratrol promotes fibroblast differentiation of hUCMSCs in an indirect co-culture system. Resveratrol at 0.1 μM and LY2904200 at 10 μM were used in the experiments (conducted in triplicate, n = 3). (A) Immune images showed the expression of collagen I, collagen III, and p-AKT/AKT in each group of hUCMSCs. (B) Treatment with LY294002 reduced the expression of collagen I, collagen III, and p-AKT/AKT in each group of hUCMSCs. The values are expressed as mean ± SEM, *p < .05, **p < .01, ***p < .001 by one-way ANOVA with Fisher’s LSD post hoc test (collagen I, p-AKT/AKT of Figure 4A; collagen III, p-AKT/AKT of Figure 4B), or one-way ANOVA with Dunnett’s test (collagen III, p-AKT/AKT of Figure 4A; collagen I, p-AKT/AKT of Figure 4B).
Phosphorylated AKT was decreased in hUCMSCs co-cultured with HEFs compared with hUCMSCs monoculture, which indicated that the phosphorylated AKT level of hUCMSCs was down-regulated after co-culture with HEFs. The results suggest that inhibited differentiation of hUCMSCs to HEFs may be associated with down-regulation of phosphorylated AKT expression. Phosphorylated AKT is significantly enhanced in the resveratrol-induced co-culture group, suggesting that resveratrol may promote the differentiation of hUCMSCs into HEFs through the AKT pathway. Therefore, in vitro, we explored whether resveratrol regulates the differentiation of hUCMSCs through the AKT pathway (see Figure 4B). LY294002 intervened in the resveratrol-induced co-culture group and was confirmed by Western blot experiments that LY294002 inhibited the regulation of the AKT pathway by resveratrol in hUCMSCs in the indirect contact co-culture system, and the expression levels of collagen I and collagen III were reduced, indicating that resveratrol may promote the differentiation of co-cultured hUCMSCs with HEFs into fibroblast by activating the AKT pathway.
Resveratrol promotes the differentiation of hUCMSCs into fibroblasts through the regulation of the AKT pathway in an indirect co-culture system
In an indirect co-culture system, we further supported that resveratrol may promote collagen synthesis and secretion in hUCMSCs via the AKT pathway. The results of Sirius scarlet staining showed that total collagen was secreted and deposited around the nucleus. Resveratrol promoted the enhanced secretion of total collagen (both collagens I and III) from hUCMSCs in co-culture with HEFs compared to the co-culture group and the UCMSCs monoculture group. However, the intervention with LY294002 significantly inhibited the effect of resveratrol on promoting the synthesis and secretion of total collagen in hUCMSC. This was confirmed by the immunocytochemical results of collagen I and collagen III, in which the staining of collagen I and III was strongly positive in the co-culture group treated with resveratrol, indicating resveratrol-promoted fibroblastic differentiation of hUCMSC (Figure 5A–C). Furthermore, the results of the qPCR also showed that LY294002 had an inhibitory effect on resveratrol. LY294002 reduced the expression of collagen I, collagen III, and AKT in hUCMSC co-cultured with HEFs (see Figure 5C). Together, these data suggest that resveratrol is important in promoting the differentiation of hUCMSCs into fibroblasts in the co-culture system, and that resveratrol promotes the differentiation of hUCMSCs through activation of the AKT signal pathway. Effect of resveratrol on hUCMSC differentiation into fibroblasts depends on the transduction of the AKT signal. Resveratrol at 0.1 μM and LY2904200 at 10 μM were used in the experiments (conducted in triplicate, n = 3). (A) After 2 weeks of culture, cultured umbilical cord mesenchymal stem cells were stained with Sirius red to observe the effect of collagen deposition in each group. (B) Collagen I expression in each group of hUCMSCs was detected by immunocytochemistry. (C) Collagen III expression detected in each group of hUCMSCs was detected by immunocytochemistry. (D) Quantitative real-time fluorescence detection of collagen I, collagen III, and AKT gene expression in each group of hUCMSCs. The values are expressed as mean ± SEM, *p < .05, **p < .01, ***p < .001, by one-way ANOVA with Dunnett’s test (D).
Resveratrol-mediated AKT pathway reduces the expression of IL-6, TGF-β, Caspase-9, and Bax, and inhibits the NF-κB pathway in hUCMSCs co-cultured with HEFs
Protein expression levels of NF-κB, IL-6, and TGF-β in each group of hUCMSCs were examined by Western Blot (see Figure 6A). The expression levels of NF-κB, IL-6, and TGF-β in the resveratrol co-culture group were significantly lower than in the co-culture group. After co-treatment with resveratrol and LY294002, the expression levels of NF-κB did not increase significantly, but the expression of IL-6 and TGF-β was significantly increased. The qRT-PCR results showed that resveratrol significantly decreased the expression levels of IL-6, TGF-β, NF-κB, caspase-9, and Bax, in co-cultured hUCMSC with HEF. However, after co-treatment with resveratrol and LY294002, the expression levels of N-κB and IL-6 were not significant but significantly increased the expression levels of TGF-β, caspase-9, and Bax (see Figure 6B). The results suggest that resveratrol may inhibit apoptosis and down-regulate the expression of IL-6 and TGF-β by activating the AKT signaling pathway. Furthermore, resveratrol can inhibit the NF-κB signaling pathway, thus promoting the differentiation of hUCMSCs into HEFs. Effects of resveratrol and LY294002 on the NF-κB signaling pathway and expression of mRNA in the hUCMSCs of each group (n = 3). Resveratrol at 0.1 μM and LY2904200 at 10 μM were used in the experiments. (A) Using Western blot to analyze the expression of IL-6, TGF-β, NF-κB, and p-NF-κB. (B) qRT-PCR analysis of the expression of IL-6, TGF-β, NF-κB, caspase-9, and Bax. Values are expressed as mean ± SEM, *p < .05, **p < .01, ***p < .001 by one-way ANOVA with Fisher LSD post-hoc test (TGF-β, IL-6, p-NF-κB/NF-κB of Figure 6A; TGF-β of Figure 6B), or nonparametric Kruskal–Wallis one-way ANOVA test with Bonfferoni’s correction (IL-6, NF-κB, Bax of Figure 6B), or one-way ANOVA with Dunnett’s test (caspase-9 of Figure 6B).
Discussion
In the present study, we presented data suggesting for the first time that under a co-culture condition in which hUCMSCs and esophageal fibroblasts are cultured together, resveratrol at a concentration of 0.1 μM promoted the differentiation of hUCMSCs into HEFs. We further showed that the underlying mechanism is associated with down-regulation of IL-6 and TGF-β via the AKT pathway and inhibition of the NF-κB pathway. The results of this study provided a promising strategy for constructing esophageal tissues engineered to find suitable seed cells and optimize their source and quality.
Previous studies have shown that resveratrol has a number of biological functions, such as anti-aging, anti-inflammation, and antioxidant stress. 18 Although multiple studies have indicated that resveratrol has a protective effect on MSCs, it is cytotoxic to MSCs in vitro. Resveratrol enhances the expression of Sirt1 and inhibits the expression of p53 and p16 to promote proliferation and decrease MSC aging at concentrations of 0.1 μM, 1 μM, and 2.5 μM. Resveratrol at concentrations of 5 μM and 10 μM promotes cellular senescence and apoptosis by inhibiting SIRT1 and PCNA and stimulating the expression of p53 and p16. 24 Our cytotoxicity experiments demonstrated that resveratrol promoted hUCMSC cell viability after 14 days in cultures at 0.1 μM and 1 μM concentrations. In contrast, with increasing resveratrol concentration, the cell viability of hUCMSC decreased significantly (Figure 3). Therefore, 0.1 μM concentration of resveratrol was chosen for the experiments in the current study.
Co-culture is a strategy for culturing multiple cell types in the same environment and controlling cell behavior through direct or indirect interactions between cells to drive tissue formation. 25 Cell co-culture technology can mimic the in vivo environment to a large extent and is an ideal culture modality for highly active cultivation and eventual promotion. In this study, we co-cultured hUCMSCs with HEFs in Transwell chambers to investigate the effects of resveratrol and fibroblast paracrine secretion on the cell biological behavior of MSCs. Although the indirect co-culture system does not fully simulate the spatial relationship between the two cell types, they are allowed to interact via paracrine secretion, thus excluding the possibility of cell fusion and endocytosis, among other phenomena. 26 In the present study, we found that the addition of resveratrol to the hUCMSC and HEF co-culture system significantly increased the expression of collagen I and collagen III in hUCMSC, and promoted the differentiation of hUCMSC into HEFs. Collagen I and collagen III are non-rigid extracellular matrices secreted by fibroblasts. They are also employed as broad molecular markers of the differentiation of fibroblasts.27,28
MSCs influence their microenvironment in different ways, such as immune regulation, hematopoietic, and tissue regeneration. The microenvironment also regulates the differentiation, proliferation, and function of MSCs. 24 The increased number of isolated culture conditions and passages is detrimental to MSCs and makes them more susceptible to microenvironmental influences. 29 Furthermore, differences in peripheral oxygen concentration will reduce the proliferation, differentiation, and anti-inflammatory effects of MSCs. 30 Previous studies have shown that resveratrol protects MSCs from inflammation and senescence. 31 Therefore, the effect of resveratrol in promoting the differentiation of hUCMSC in the co-culture system was further investigated in the following studies.
We observed that the expression of phosphorylated AKT decreased after the co-culture of hUCMSCs and HEFs, and that the expression of phosphorylated AKT was elevated after treatment with resveratrol. These results suggest that the activation of AKT is involved in the promotion of the differentiation of hUCMSCs by resveratrol. The PI3K/Akt pathway has been shown to play a crucial role in the regulation of the proliferation, survival, apoptosis, and differentiation of MSCs/progenitor cells. 32 PI3K is an essential upstream initiator of the PI3K/AKT/mTOR signaling pathway. AKT is the upstream hub of the PI3K/AKT/mTOR signaling pathway, controlling multiple aspects of cell growth, proliferation, survival, substance metabolism, and energy metabolism. 33 Brito et al. 34 showed that resveratrol increases cell viability and promotes DNA synthesis and proliferation in smooth muscle cells via the PI3K/Akt/mTOR/P70S6K pathway. In keeping with these observations, we demonstrated that resveratrol specifically activated the AKT pathway in hUCMSCs. In contrast, co-treatment with resveratrol and LY294002, an inhibitor of the PI3K/AKT signaling pathway, attenuated the resveratrol-induced differentiation of hUCMSCs into fibroblasts. This indicated that resveratrol promoted the differentiation of hUCMSCs into fibroblasts by activating the AKT signaling pathway.
An important finding of this study is that resveratrol activated the AKT pathway to down-regulate the expression of TGF-β and IL-6 and inhibited apoptosis, thus promoting the differentiation of hUCMSCs toward HEFs. As shown in Figure 6A, the decreased protein expression of TGF-β and IL-6 in hUCMSCs was reversed by the addition of LY294002. Our qRT-PCR results demonstrated that LY294002 significantly altered the reduction of mRNA expression of TGF-β, caspase-9, and Bax in hUCMSCs induced by resveratrol (Figure 6B). In agreement with our data, previous studies demonstrated that resveratrol altered the TGF-β-mediated downstream cell functions by inhibiting the production of TGF-β in an antitumor experimental study. 35 Resveratrol also modulated numerous cellular signaling pathways in a sirtuin-dependent manner, such as increasing phosphorylation of AKT and decreased activation of NF-κB. 36 Mechanically, it is believed that induced activation of Akt may activate or inhibit its downstream target proteins Bcl-x1/Bcl-2 related death factor (BAD), nuclear factor kappa B (NF-κB), aspartate specific cysteine protease 9 (caspase-9), etc., through phosphorylation, thus mediating cell growth, adhesion, migration, promoting cell survival, and anti-apoptosis. 37
In this study, we found that resveratrol significantly decreased the expression of genes and proteins of caspase-9, IL-6, NF-κB, and Bax in co-culture with hUCMSC. This may be because hUCMSCs inhibited their differentiation by upregulating the expression of IL-6 and NF-κB and promoting apoptosis after co-culture with HEFs. While resveratrol promoted the differentiation of hUCMSCs into HEFs by decreasing the expression of IL-6 and NF-κB and inhibiting apoptosis. Our findings are supported by previous observations. 20 Studies have shown that several stimuli and inhibitors play an essential role in the initial commitment and subsequent stages of differentiation of MSCs into a specific cell type. 38 A recent study showed that IL-6 gene expression was significantly upregulated in undifferentiated MSCs compared to differentiated derivatives of MSCs. 39 Studies have shown that IL-6 can impair the ability of MSCs to generate chondrocytes and maintain them undifferentiated by activating ERK1/2. 40 IL-1 and TNF-а inhibit osteogenesis and adipogenesis in MSCs by activating the canonical NF-κB signaling pathway and the IL-1R1/MyD88 signaling pathway. 41 Resveratrol decreases IL-6 and IL-8 by inhibiting NF-κB and AP-1, hindering the synthesis of nitric oxide synthase and cyclooxygenase-2 to improve membrane stability and reduce the release of inflammatory factors. 42
Studies have shown that TGF-β signaling plays an essential role in adipocyte differentiation in adipose tissue-derived stromal cells and bone marrow-derived MSCs. 43 In general, the TGF-β/activin-Smad2/3 pathway promotes the proliferation of preadipocytes while inhibiting their differentiation into adipocytes. Transforming growth factor-β inhibits myogenic cell proliferation, differentiation, and myotube formation. 44 Resveratrol acts on the TGF-β-mediated downstream cell functions by suppressing the production and down-regulating the activity of TGF-β. Consistent with previous studies, in this study, we demonstrated that resveratrol decreased the expression levels of TGF-β genes and proteins after co-culture with hUCMSC. It may be that after co-culture of hUCMSCs with HEFs, hUCMSCs inhibited their differentiation by upregulating TGF expression levels of TGF-β, while resveratrol promoted differentiation of hUCMSCs to HEFs by decreasing the expression of TGF-β. Since it is not possible to determine whether upregulation of TGF-β by hUCMSC acts on HEF to promote their fibrosis, resveratrol combined with hUCMSC co-culture with HEF facilitates the microenvironment of the hUCMSC culture, thus promoting its differentiation in the right direction.
Collectively, these results indicate the critical role of resveratrol in the induction of hUCMSC differentiation. Further studies are needed to explore the detailed molecular mechanisms by which HEFs mediate the differentiation of hUCMSCs and to elucidate the exact role of resveratrol in promoting hUCMSC differentiation. This forms a limitation of the study. Another limitation is that only limited biomarkers for fibroblastic differentiation of hUCMSCs are used in the study. Further study using more validation approaches might be needed in the future. Furthermore, the mechanism of action of resveratrol in promoting the differentiation of HUCMSC into esophageal fibroblast was investigated only under in vitro conditions. No animal experiments were conducted to study whether HUCMSCs differentiate into esophageal fibroblasts and maintain their ability and drive esophageal regeneration upon transplantation in vivo.
Conclusions
In conclusion, the results of this study suggest that resveratrol induces and promotes the differentiation of hUCMSCs into esophageal fibroblasts in vitro. The AKT pathway may be critical in promoting the differentiation of hUCMSCs into fibroblasts by resveratrol. Resveratrol down-regulates the expression of TGF-β and IL-6 via the AKT pathway. Simultaneously, resveratrol inhibits the NF-κB pathway, thereby promoting the differentiation of hUCMSCs into fibroblasts. Resveratrol can create a co-culture microenvironment that is conducive to differentiating hUCMSC into HEFs. It may be used to enhance the effect of hUCMSC on esophageal fibrogenic repair for esophageal tissue engineering and clinical rehabilitation of esophageal defects.45–49
Data availability statement
The data used to support the findings of this study are included within the article.
Footnotes
Acknowledgements
The authors thanks Dr Eileen McGowan of the University of Technology Sydney for her critical reading and editing.
Author contributions
Conceptualization, XC, SC, and YL; methodology, software, validation, and formal analysis, XC, ZS, QW, LS, JB, HC, and SC; investigation and data curation, XC, QW, HC, and ZS; resources, SC and HC.; writing—original draft preparation, XC.; writing—review and editing, XC, SC, HC, and YL; visualization, XC and YL; supervision, SC and YL.; project administration, SC, HC, and YL; funding acquisition, SC. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Guangdong Science and Technology Department (2020A0505100058), Department of Education of Guangdong Province (2019KZDXM024), and Guangdong Province Medical Products Administration (2022TDZ19).
Ethical statement
Trial registration
Not applicable.
