Abstract
Basiliximab is an important treatment for steroid-refractory acute graft-versus-host disease (SR-aGVHD). We performed this retrospective study to evaluate the efficacy and safety of basiliximab treatment in SR-aGVHD patients following matched sibling donor hematopoietic stem cell transplantation (MSD-HSCT) (n = 63). Overall response rate (ORR) was 63.5% and 54% at any time and at day 28 after basiliximab treatment. Grade III–IV aGVHD before basiliximab treatment predicted a poor ORR after basiliximab treatment. The rates of virus, bacteria, and fungi infections were 54%, 23.8%, and 3.1%, respectively. With a median follow-up of 730 (range, 67–3,042) days, the 1-year probability of overall survival and disease-free survival after basiliximab treatment were 58.6% (95% confidence interval [CI] = 47.6%–72.2%) and 55.4% (95% CI = 44.3%–69.2%), respectively. The 3-year cumulative incidence of relapse and non-relapse mortality after basiliximab treatment were 18.9% (95% CI = 8.3%–29.5%) and 33.8% (95% CI = 21.8%–45.7%), respectively. Comorbidities burden before allo-HSCT, severity of aGVHD and liver aGVHD before basiliximab treatment showed negative influences on survival. Thus, basiliximab was safe and effective treatment for SR-aGVHD following MSD-HSCT.
Keywords
Introduction
Acute graft-versus-host disease (aGVHD) remains an important complication for allogeneic hematopoietic stem cell transplantation (allo-HSCT) recipients1–5. Even in matched sibling donor (MSD) HSCT, the cumulative incidence of total and grade III–IV aGVHD could be as high as 25%–27% and 10%–14%, respectively6–8, and aGVHD accounts for approximately 15% of non-relapse mortality (NRM) after allo-HSCT 9 . Although corticosteroid is the first-line treatment of aGVHD10,11, nearly half of aGVHD patients could not show response to steroid treatment (ie, steroid-refractory aGVHD) 12 and clinical outcomes of these patients were poor13–15.
Interleukin-2 (IL-2) is critical in the development of aGVHD through activating cytotoxic donor T-lymphocytes 16 , and IL-2 receptor antagonist (IL-2RA) is an important method for SR-aGVHD treatment 17 . Several studies have reported the clinical outcomes of basiliximab treatment in SR-aGVHD patients18–22, but most of them did not provide the subgroup analysis for MSD-HSCT recipients. Only two studies reported the overall response rate (ORR) at day 28 after basiliximab treatment in MSD-HSCT recipients18,19; however, other clinical outcomes after basiliximab treatment were still unknown. For example, whether basiliximab treatment would increase the relapse risk after MSD-HSCT was unclear because some studies reported that graft-versus-leukemia (GVL) effect of MSD-HSCT was not as intensive as haploidentical-related donor (HID)-HSCT 23 . In addition, whether basiliximab treatment could decrease the incidence of chronic GVHD (cGVHD) in MSD-HSCT patients was still a problem. Although MSD-HSCT recipients have a lower incidence of aGVHD, the incidence of cGVHD of them could be as high as 53.8% 24 . Thus far, no studies focused on basiliximab treatment in a disease-specific cohort of MSD-HSCT recipients.
Thus, in this retrospective study, we aimed to investigate the efficacy and safety of basiliximab treatment in MSD-HSCT recipients with SR-aGVHD, and we also aimed to identify the prognostic factor and long-term clinical outcomes of these patients.
Methods
Patients
Data on patients undergoing MSD-HSCT from January 2015 to December 2021 were obtained from the Peking University Institute of Hematology (PUIH). Patients who had SR-aGVHD and received basiliximab treatment after allo-HSCT were included. The last follow-ups were conducted on June 30, 2023. Thirty-eight patients had been reported in BRIGHT study 18 , and all of them were further followed-up in the present study. The study design was approved by the ethics committee of Peking University People’s Hospital. Informed consent was obtained in accordance with the Declaration of Helsinki
Treatment of aGVHD
Methylprednisolone (MP, 1 mg/kg/day) was administrated as the first-line treatment once aGVHD was diagnosed. If aGVHD progressed within 3 days, or did not respond by 5 days after the initiation of MP (steroid-resistant) 25 , or flared during the slow MP taper (steroid-dependent), SR-aGVHD was defined and basiliximab treatment was initiated. Basiliximab (Simulect; Novartis Pharma AG, Basel, Switzerland) was administered intravenously at a dose of 20 mg (for adults or children weighing over 35 kg) or 10 mg (for children weighing less than 35 kg) on days 1, 3, and 8, and then repeated weekly.
End Points and Definitions
The primary end point of this study was ORR, which consists of complete response (CR) and partial response (PR) (defined in supplementary methods) 19 . The secondary end points included cGVHD, overall survival (OS), disease-free survival (DFS), relapse, and NRM. cGVHD was diagnosed according to National Institutes of Health Consensus Criteria 26 . OS was defined as the survival period from basiliximab treatment to death from any cause while DFS was defined as the survival period with continuous complete remission. Relapse was defined as morphologic evidence of underlying malignancy in peripheral blood, bone marrow, or extramedullary sites, or by the recurrence and sustained presence of pre-transplantation chromosomal abnormalities. NRM was defined as all deaths after basiliximab treatment except for recurrence of underlying disease.
Statistical Analysis
Grouped variables were compared using the χ2 and Fisher’s exact tests, and continuous variables were compared using the Mann–Whitney U test. OS and DFS were calculated using the Kaplan-Meier method. The cumulative incidence of ORR, cGVHD, relapse and NRM were estimated in the competing risks framework. Death and relapse were treated as the competing events for ORR and cGVHD, whereas relapse and NRM were treated as events competing with each other. The multivariable Cox proportional hazards regression with a backward stepwise model was used to estimate hazard ratios (HRs) for clinical outcomes. The considered variables included: patient age (< 18 years vs ≥ 18 years), gender, underlying disease (hematologic malignancies vs non-hematologic malignancies), donor–recipients gender matching (female to male vs others), hematopoietic cell transplantation-comorbidity index (HCT-CI) score 27 before allo-HSCT (high risk and intermediate risk vs low risk), graft type (blood marrow and peripheral blood harvests vs peripheral blood), nuclear cell counts and CD34+ cell counts (using the median value as the cutoff point), refined Minnesota Acute GVHD Risk Score 28 at diagnosis and at the beginning of basiliximab treatment (high risk vs standard risk), the stage (= 0 vs ≥ 1) of involved organs (skin, gut, and liver) and numbers of organs (1 vs 2–3) at the beginning of basiliximab treatment, and severity of aGVHD at diagnosis and at the beginning of basiliximab treatment (≥ grade III vs < grade III). The P values were two-sided. Independent variables with P < 0.1 were included for sequential evaluation, and P < 0.05 was considered to be significant. The statistics analysis was performed using SPSS 24 (SPSS Inc./IBM, Armonk, NY, USA) and the R software (version 3.6.2; http://www.r-project.org).
Results
Patient Characteristics
Sixty-three patients received MSD-HSCT between January 2015 and December 2021 in PUIH and developed SR-aGVHD were enrolled (Table 1). The diagnosis of aGVHD was confirmed by biopsies in 15 patients, or by classical clinical features in 48 patients. The median follow-up was 730 (range, 67–3042) days. The median time from transplantation to aGVHD was 40 (range, 11–98) days. The characteristics of aGVHD were summarized in Table S1, and 52 (82.5%) and 11 (17.5%) patients were considered as steroid-refractory and steroid-dependent, respectively. The first dose of basiliximab was administered 6 (range, 2–21) days after diagnosis of aGVHD. The median number of doses of basiliximab was 4 (range, 2–12) doses.
Patients Characteristics.
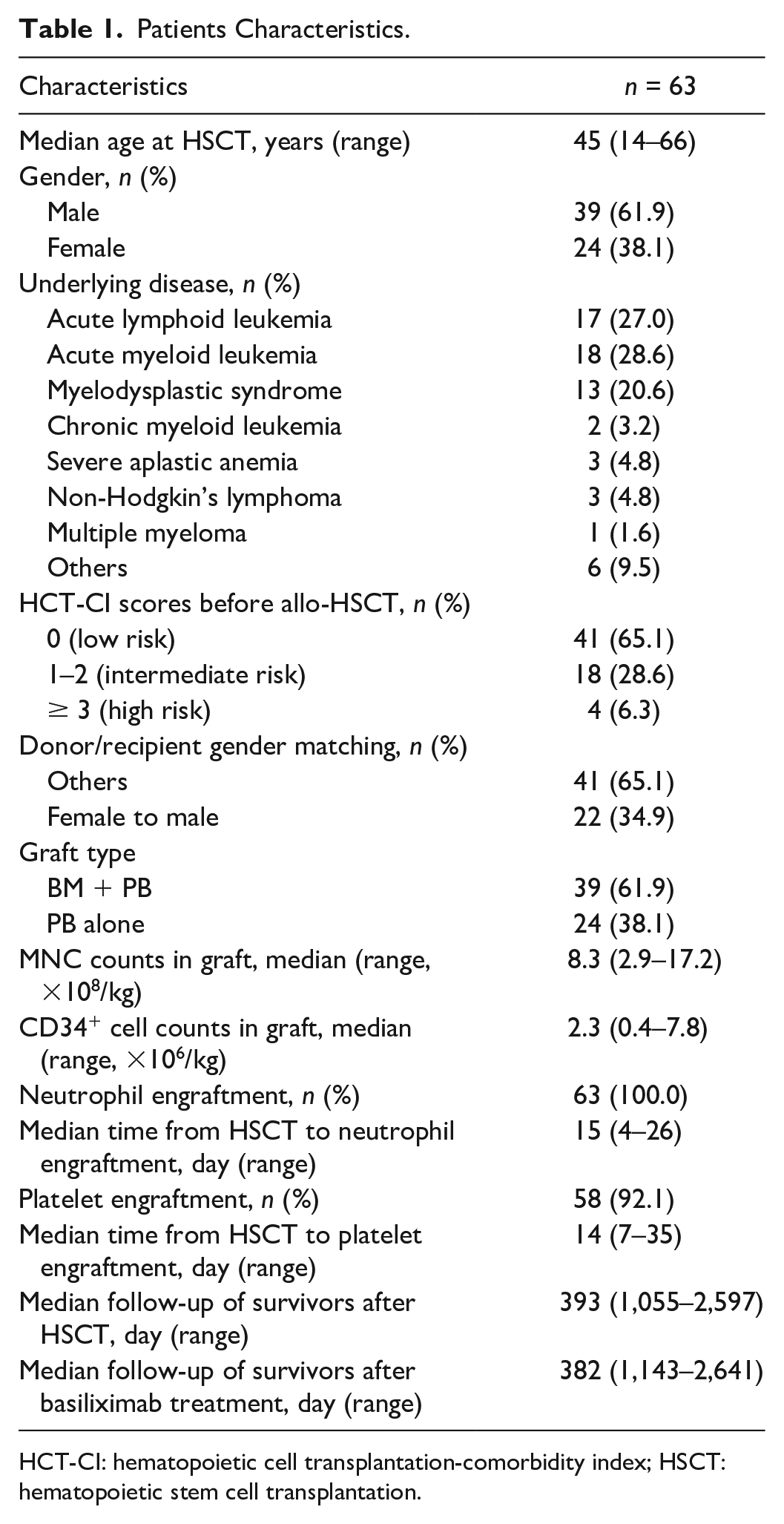
HCT-CI: hematopoietic cell transplantation-comorbidity index; HSCT: hematopoietic stem cell transplantation.
Response
ORR at any time was observed in 63.5% (40/63) of patients, with 54% (34/63) of responders achieved CR and 9.5% (6/63) of responders achieved PR. The median duration from the beginning of basiliximab treatment to ORR was 11 (range, 2–56) days. The cumulative incidence of ORR at day 28 after basiliximab treatment was 54.0% (95% confidence interval [CI] = 41.5%–66.4%), with 61.3% (95% CI = 43.7%–78.9%) in myelodysplastic syndrome/acute myeloid leukemia (MDS/AML) group and 47.1% (95% CI = 22.4%–71.8%) in acute lymphoid leukemia (ALL) group (P = 0.22).
Detailed information about response to basiliximab treatment is shown in Table S2. Some common factors, such as, graft type, donor/recipient gender matched, and nuclear cell count, were not associated with the ORR (Fig. 1). Patients with high-risk refined Minnesota aGVHD risk score or grade III–IV aGVHD at diagnosis showed a lower ORR. Meanwhile, high-risk refined Minnesota aGVHD risk score and grade III–IV aGVHD at the beginning of basiliximab treatment were also associated with a lower ORR (Fig. 1). In multivariate analysis, only grade III–IV aGVHD before basiliximab treatment was associated with a lower ORR (HR = 0.19, 95% CI = 0.09–0.38, P < 0.001).
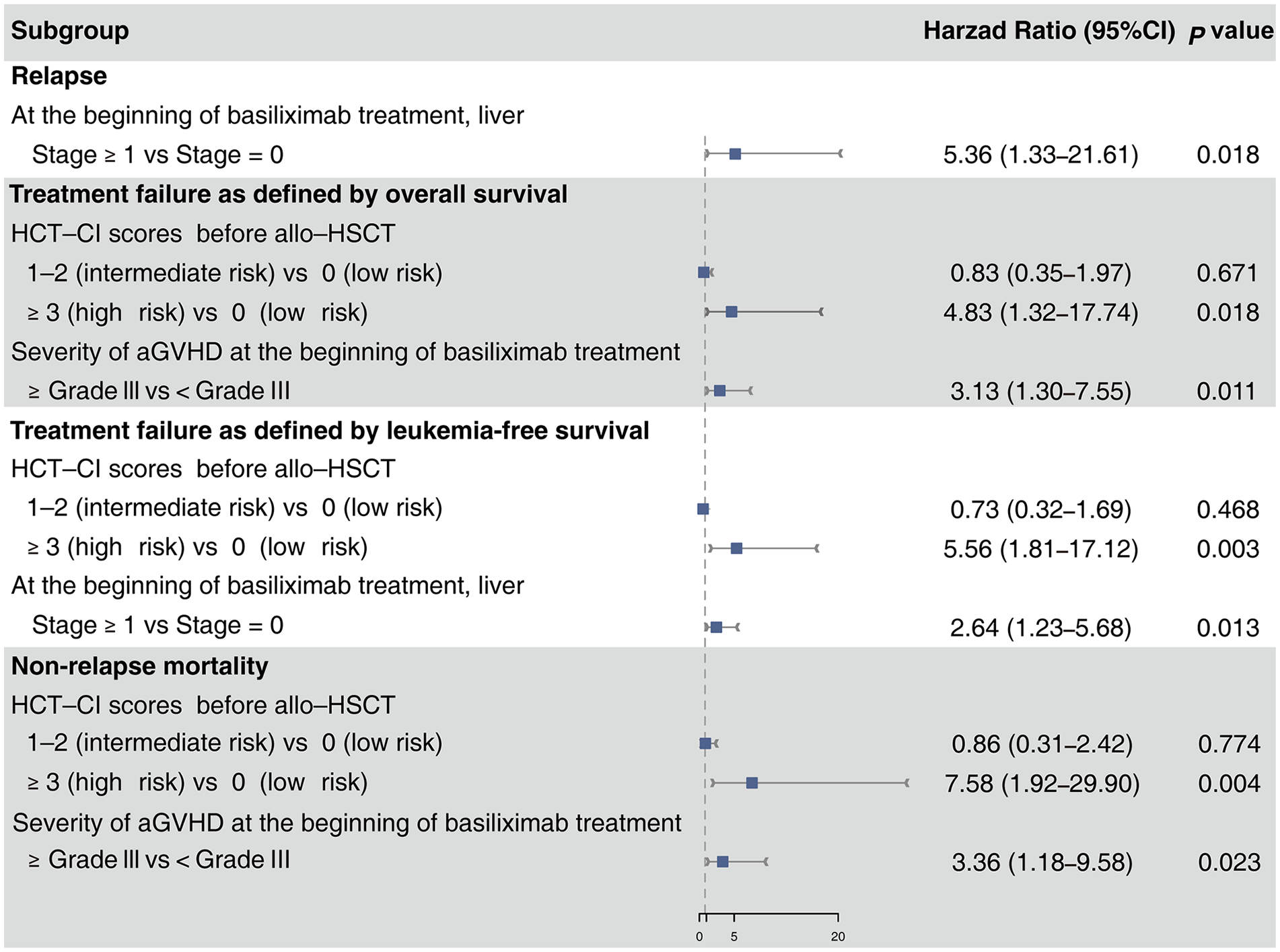
Univariable analysis of overall response at any time after basiliximab treatment.
Toxicities and Infections
No infusion-related toxicity or allergic reaction was observed after basiliximab treatment. Infection occurred in 41 (65.1%) patients (Table S3). The frequency of viral, bacterial, and fungal infection was 54.0%, 23.8%, and 3.1%, respectively. The median durations from basiliximab treatment to the occurrence of viral, bacterial, and fungal infections were 20 days (range, 4–89), 44 days (range, 25–1086), and 59 days, respectively. No hematologic toxicity was observed.
Long-Term Clinical Outcomes
Chronic GVHD
cGVHD occurred in 27 (42.9%) patients after basiliximab treatment, and their characteristics were summarized in Table S4. The cumulative incidence of total and severe cGVHD at 3 years after basiliximab treatment were 44.4% (95% CI = 31.1%–57.8%) and 14.5% (95% CI = 5.6%–23.4%), respectively.
Relapse and NRM
Eleven patients experienced relapse, and the median time from basiliximab treatment to relapse was 109 days (range, 3–987 days). The 3-year cumulative incidence of relapse after basiliximab treatment was 18.9% (95% CI = 8.3%–29.5%), with 16.1% (95% CI = 2.9%–29.3%) in MDS/AML group and 17.6 % (95% CI = 1.3%–36.5%) in ALL group (P = 0.88). Multivariable analysis showed that liver aGVHD at the beginning of basiliximab treatment was the only risk factor for relapse (Table 2).
Multivariable Analysis of Clinical Outcomes After Basiliximab Treatment.

aGVHD: acute graft-versus-host disease; CI: confidence interval; HCT-CI: Hematopoietic cell transplantation-comorbidity index; HSCT: hematopoietic stem cell transplantation.
Twenty-one patients died of NRM, and the median time from basiliximab treatment to NRM was 124 days (range, 34–658 days). The most common cause of NRM was GVHD (47.6%), followed by infection (28.6%) and intracerebral hemorrhage (14.3%, Table S5). The cumulative incidence of NRM at 3 years after basiliximab treatment was 33.8% (95% CI = 21.8%–45.7%). In multivariable analysis, high-risk HCT-CI score before allo-HSCT and severe aGVHD at the beginning of basiliximab treatment were associated with a higher risk of NRM (Table 2).
Overall Survival
The probability of OS at 1 year was 58.6% (95% CI = 47.6%–72.2%) (Fig. 2A). Median survival was 1,317, 425, and 155 days for CR, PR, and NR (no response) groups, respectively. Meanwhile, OS at 1 year was 64.5% (95% CI = 49.7%–83.8%) in MDS/AML group and 34.3% (95% CI = 17.5%–67.2%) in ALL group, but there was no statistical significance (P = 0.14, Fig. S1). In multivariable analysis, high-risk HCT-CI score before allo-HSCT and grade III–IV aGVHD before basiliximab treatment were associated with a worse OS (Table 2).
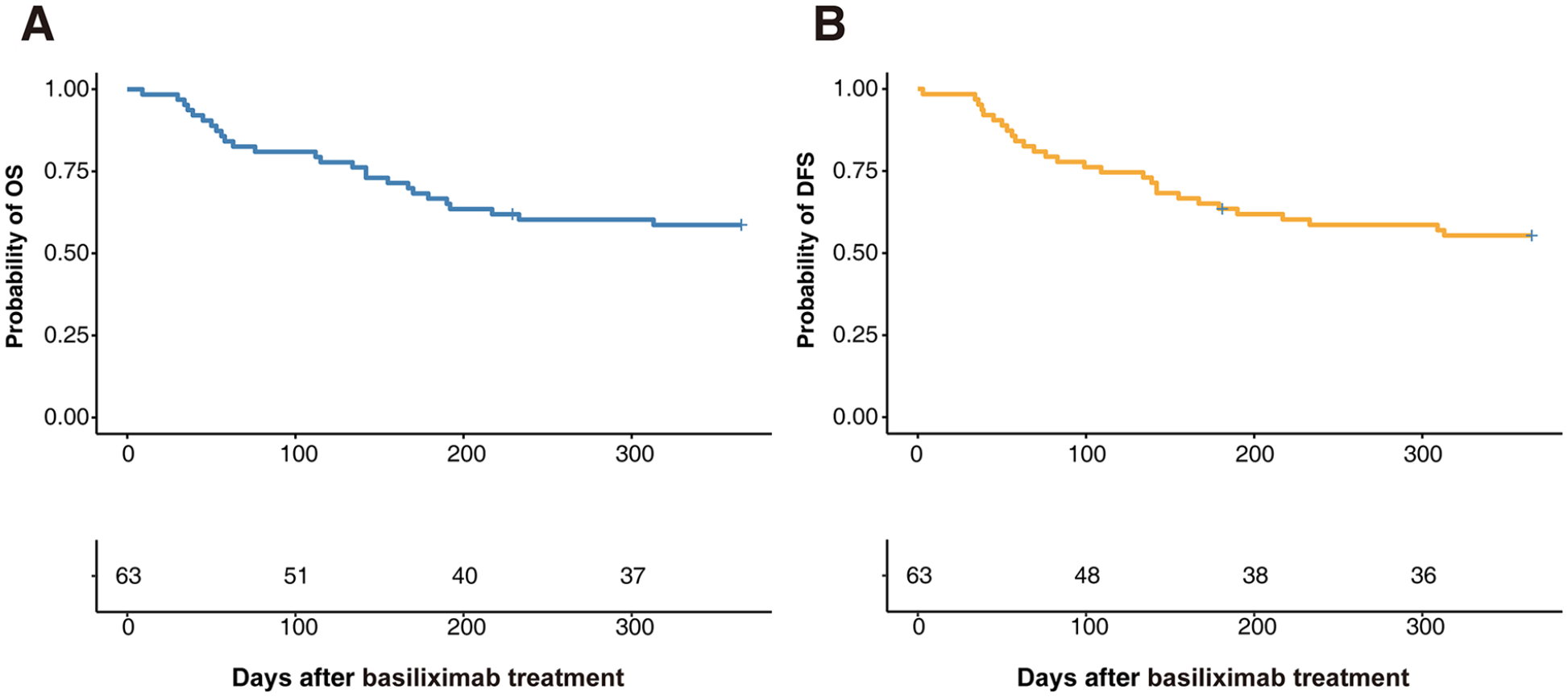
Overall survival (OS) and disease-free survival (DFS) after basiliximab treatment. (A) OS. (B) DFS.
Disease-free Survival
The probability of DFS at 1 year after basiliximab treatment was 55.4% (95% CI = 44.3% P 69.2%, Fig. 2B). In multivariable analysis, high-risk HCT-CI before allo-HSCT and liver aGVHD at the beginning of basiliximab treatment were associated with a poorer DFS (Table 2).
Discussion
To date, several studies have evaluated the efficacy of novel agents to manage SR-aGVHD, whereas few of them focused on MSD-HSCT recipients. We observed that the ORR was 63.5%, and the 1-year probabilities of OS and NRM were 58.6% and 33.3%, respectively, and there was no statistical difference between myeloid and lymphoid malignancies. This was the first study to investigate the clinical outcomes of MSD-HSCT recipients who had SR-aGVHD and received IL-2RA.
Infection was the most common complication after basiliximab treatment. The infection rate was 65.1% in this cohort, which was comparable with the results in previous studies using basiliximab treatment (59.6%–78.8%18,19,21,22,29,30). Our results were also similar with the studies using other IL-2RA blockades, such as daclizumab (38%–95%31,32) and inolimomab (62%–92.5%33–35). On the other hand, infections were also the most common complication for other treatments for SR-aGVHD, including ruxolitinib (61%–82.6%36–39), mycophenolate mofetil (MMF, 23%–66.7%40,41), anti-thymocyte globulin (ATG, 67%–92%42,43), tumor necrosis factor (TNF) blockades (72%–87%44–46), and mesenchymal stromal cells (MSCs, 45%–88%47,48). Therefore, basiliximab treatment did not lead to excessive risk of infections compared with other second-line treatments.
The rate of NRM was 33.8% in the present study, which was similar to the other studies using IL-2RA for SR-aGVHD (10.2%–44.8%18,21,30,49 for basiliximab, 25.9%–91.8%31,50,51 for daclizumab, and 24.2%–76.2%33,52,53 for inolimomab). Considering the other second-line treatment, the NRM rate after ruxolitinib, MMF, ATG, TNF blockades, and MSCs was 16.1%–52.9%37,38,54,55, 14.3%–48%41,56,57, 60.8%–83.3%56,58,59, 22%–72.7%53,56,60,61, 29.3%–77.6%30,48,62, respectively. This suggested that the safety of basiliximab was acceptable in MSD-HSCT recipients.
In this study, the cumulative incidence of total cGVHD at 3 years after basiliximab treatment was 44.4%, which was also similar to the other IL-2RA (25%–93.3%)31,32,35,51 and other second-line treatments, such as MMF (72.9%) 57 and TNF blockades (42%–90%)44,60, suggesting that basiliximab could also help to control the occurrence of cGVHD.
Some authors suggested that IL-2RA might diminish the GVL effect and increase the risk of relapse 63 . We observed that the relapse rate was 18.9% in 3 years, which was comparable with previous studies using basiliximab treatment for SR-aGVHD (9.4%–26%)18,21,29,30,49. In addition, it was also similar to the other IL-2RA (7%–23%)32,51 and the other second-line treatments, such as ruxolitinib (9.3%–49.3%)37,38,64, MMF (14.3%) 65 , and TNF blockades (12%–19.2%)44,60. These results indicated that basiliximab did not significantly diminish the GVL effect and increase the risk of relapse.
We observed the ORR at any time and at day 28 was 63.5% and 54%, respectively, in the present study. These findings are similar with previous studies (ie, ORR at any time: 65%–70% ORR at day 28: 54%–59%21,54,66). In addition, we found ORR at any time was ranging from 16.7% to 94%50,52 for other IL-2RA, 28%–81.5%35,36,64,67 for ruxolitinib, 30.8%–79%40,65,68 for MMF, 59% 59 for ATG, 28%–67%44,68,69 for TNF blockades, and 46.6%–58.7%62,70,71 for MSCs. Hence, data from this analysis support that this approach has a beneficial effect to SR-aGVHD patients after MSD-HSCT.
Nevertheless, the ORR seemed to be slightly lower than that in some studies using basiliximab treatment for SR-aGVHD, with an ORR ranging from 71% to 92%19,20,22,29,72–74 at any time. One of the common characteristics of these studies is that most of the patients receiving HID-HSCT. Actually, in our previous studies, the ORR for MSD-HSCT subgroup was 76.5% at any time 19 and 61% at 28 days 18 , which is also similar to the results in this study. This suggested that the ORR for HID HSCT recipients with SR-aGVHD might be better than MSD-HSCT. One exploitation may be because the conditioning regimen for HID-HSCT including ATG, which could effectively prevent aGVHD42,75. In addition, Perales et al. 32 also observed that the ORR of daclizumab for SR-aGVHD was 45% for MSD-HSCT recipients, which was lower than that of HID-HSCT recipients (79%). The impact of donor type on IL-2RA treatment should be further identified in SR-aGVHD patients.
Liver aGVHD before basiliximab treatment was a predictor of relapse and worse DFS. A series of studies reported that among other target organs, SR-aGVHD patients with liver involvement showed the lowest response20,74. Similar results were also been revealed in other IL-2RA blockades32,43, ruxolitinib36,38 and ATG 75 , indicating liver might be relatively insensitive to therapy. Pathologically, biliary ducts damage and loss may become irreversible during GVHD, and this might be one of the explanations for poor response to immunosuppressants in liver aGVHD 76 .
There were still some limitations. First, only 15 patients were diagnosed as aGVHD by biopsy. Second, the sample size, especially for patients with liver SR-aGVHD, was relatively small. The latter might be explained by the relatively low incidence of SR-aGVHD in MSD-HSCT recipients77,78. Actually, the sample of MSD-HSCT recipients was larger than the studies using basiliximab 54 or other second-line treatment for SR-aGVHD43,79,80. Thus, our study could still provide valuable information for basiliximab treatment in MSD-HSCT recipients.
In conclusion, our results suggested that basiliximab treatment was safe and effective for MSD-HSCT recipients with SR-aGVHD, which should be confirmed by randomized controlled trail in future.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241257568 – Supplemental material for Basiliximab Treatment for Patients With Steroid-Refractory Acute Graft-Versus-Host Disease Following Matched Sibling Donor Hematopoietic Stem Cell Transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897241257568 for Basiliximab Treatment for Patients With Steroid-Refractory Acute Graft-Versus-Host Disease Following Matched Sibling Donor Hematopoietic Stem Cell Transplantation by Xin-Ya Jiang, Xiao-Hui Zhang, Lan-Ping Xu, Yu Wang, Chen-Hua Yan, Huan Chen, Yu-Hong Chen, Wei Han, Feng-Rong Wang, Jing-Zhi Wang, Yu-Qian Sun, Xiao-Dong Mo and Xiao-Jun Huang in Cell Transplantation
Footnotes
Author Contributions
X-JH and X-DM were responsible for the study design. X-YJ and X-DM analyzed the data and wrote the manuscript. X-JH and X-DM interpreted the results and provided feedback. X-YJ, X-HZ, L-PX, YW, C-HY, HC, Y-HC, WH, F-RW, J-ZW and Y-QS collected the clinical data. All authors read and approved the final manuscript.
Data Availability
Data used to support the results of the study are available from the corresponding authors upon request.
Ethical Approval
This study was approved by the ethics committee of Peking University People’s Hospital.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (nos. 2022YFA1103300, 2022YFC2502606), Major Program of the National Natural Science Foundation of China (no. 82293630), Peking University Medicine Fund for world’s leading discipline or discipline cluster development (no. 71003Y3035), and the Tongzhou District Distinguished Young Scholars (JCQN2023009).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
