Abstract
Pressure injuries, or pressure ulcers, are a common problem that may lead to infections and major complications, besides being a social and economic burden due to the costs of treatment and hospitalization. While surgery is sometimes necessary, this also has complications such as recurrence or wound dehiscence. Among the newer methods of pressure injury treatment, advanced therapies are an interesting option. This study examines the healing properties of bone marrow mononuclear cells (BM-MNCs) embedded in a plasma-based scaffold in a mouse model. Pressure ulcers were created on the backs of mice (2 per mouse) using magnets and assigned to a group of ulcers that were left untreated (Control, n = 15), treated with plasma scaffold (Plasma, n = 15), or treated with plasma scaffold containing BM-MNC (Plasma + BM-MNC, n = 15). Each group was examined at three time points (3, 7, and 14 days) after the onset of treatment. At each time point, animals were subjected to biometric assessment, bioluminescence imaging, and tomography. Once treatment had finished, skin biopsies were processed for histological and wound healing reverse transcription polymerase chain reaction (RT-PCR) array studies. While wound closure percentages were higher in the Plasma and Plasma + BM-MNC groups, differences were not significant, and thus descriptive data are provided. In all individuals, the presence of donor cells was revealed by immunohistochemistry on posttreatment onset Days 3, 7, and 14. In the Plasma + BM-MNC group, less inflammation was observed by positron emission tomography–computed tomography (PET/CT) imaging of the mice at 7 days, and a complete morphometabolic response was produced at 14 days, in accordance with histological results. A much more pronounced inflammatory process was observed in controls than in the other two groups, and this persisted until Day 14 after treatment onset. RT-PCR array gene expression patterns were also found to vary significantly, with the greatest difference noted between both treatments at 14 days when 11 genes were differentially expressed.
Introduction
Pressure ulcers, more recently referred to as pressure injuries, are defined by the National Pressure Ulcer Advisory Panel (NPUAP) as localized damage to skin and underlying soft tissue, usually over a bony prominence as a result of intense and/or prolonged pressure or pressure in combination with shear 1 . Pressure ulcers arise mainly in people who are immobilized, and they are most often seen in association with spinal cord injury, other neurological dysfunctions, or hospitalization 2 . The most common pressure ulcer locations in adults are the bony prominences of the sacral and hip regions 3 . As well as causing morbidity and mortality, epidemiologic data indicate that this type of wound represents a major social and economic burden worldwide 4 .
Sustained pressure over a bony prominence provokes ischemia in the skin and underlying tissue. Once pressure is eliminated and this ischemia period ceases, the return of blood supply produces reperfusion. Studies have shown that several cycles of ischemia-reperfusion contribute significantly to the pathogenesis of pressure ulcers 5 . The injury induced causes the formation of reactive oxygen species leading to an inflammatory response and endothelial cell dysfunction 6 . This, in turn, disrupts cytokine synthesis and delays healing, causing massive tissue necrosis and ulcer development 7 .
For more complex ulcers, surgery is the treatment mainstay. However, this form of treatment gives rise to complications such as hematoma, infection, flap necrosis or dehiscence, and ulcer recurrence 8 . This determines a need for new treatments to resolve pressure ulcers and avoid or reduce their recurrence, the economic costs of treatment and long hospital stays. To further our understanding of the etiology of pressure ulcer formation, animal models are needed, and these models are also useful to monitor the clinical response to experimental treatments. Animal models of pressure ulcers are based on the use of different devices to apply localized pressure on surfaces overlying bone 9 . These models are therefore useful to investigate the effects of new treatments and facilitate the development of new and effective therapeutic modalities.
Among the most promising new treatment options for pressure ulcers is tissue engineering. This tool involves the use of cells seeded on scaffolds of natural or synthetic origin 10 . In general, there are three essential elements to consider in skin bioengineering: a source of cells, their regenerative capacity and adequate scaffolds 11 . Among suitable options for scaffolds, human body products including amniotic membrane or blood can be used 12 . The beneficial properties of these constructs include their biocompatibility, biodegradation, cell adhesion, low toxicity, and the induction of a discrete chronic inflammatory response 13 . In 2004, Llames et al. published a pioneering paper describing the successful treatment of major burns using “artificial skin.” This skin was developed using plasma combined with autologous fibroblasts and keratinocytes 14 . In effect, owing to its structure and properties such as biocompatibility, biodegradability, low cost and easiness to obtain, an autologous plasma gel could be an ideal way to provide structural and mechanical support for cells used to treat pressure ulcers. This pharmaceutical device avoids the risk of the cell loss that occurs when applying a cell suspension and is therefore of special interest for the treatment of large irregular-shaped wounds.
Bone marrow mononuclear cells (BM-MNC) are a heterogeneous cell population composed of lymphocytes (T, B, and NK cells), monocytes, dendritic cells, and progenitor and stem cells. These cells are critical components of the immune system and play a crucial role in humoral and cell-mediated immune responses. As a BM-MNC population, mesenchymal stem cells (MSCs), in particular, are considered clinically promising because of their differentiation capacity and pro-regenerative features 15 . BM-MNC have been used for years in regenerative medicine with satisfactory results in the treatment of several conditions such as asthma 16 , acute kidney injury preventing renal mitochondrial lesions 17 or pressure ulcers 18 , among others.
Considering the properties of plasma and MNCs, we speculated that the use of both combined would be of interest to treat pressure ulcers. Accordingly, the effectiveness of a plasma-based scaffold seeded with BM-MNC was tested in a mouse model to assess its possible translation to clinical practice. This treatment could help restore dermal layer structure achieving wound closure and improving the quality of life of affected persons.
Materials and Methods
Pressure Ulcer Model
The animal pressure ulcer model used was that described by Stadler et al. 19 Pressure was applied to the skin on the backs of anesthetized (with isoflurane) 3-month-old male Swiss nu/nu mice (Charles River, USA). To provoke ischemia/reperfusion cycles, the skin was gently pulled up and placed between two circular 12 × 3 mm (diameter × thickness) neodymium magnets (Supermagnete, Germany) for 8 hours a day over three consecutive days (Fig. 1A). The pressure created between the two plates was 100 mm Hg. Two tissue defects were created in each animal to minimize the number of animals needed in line with the three Rs principle for animal experimentation. To test the characteristics of the ulcers, some animals were sacrificed for use as histological and hematoxylin-eosin staining controls (Merck, Germany).
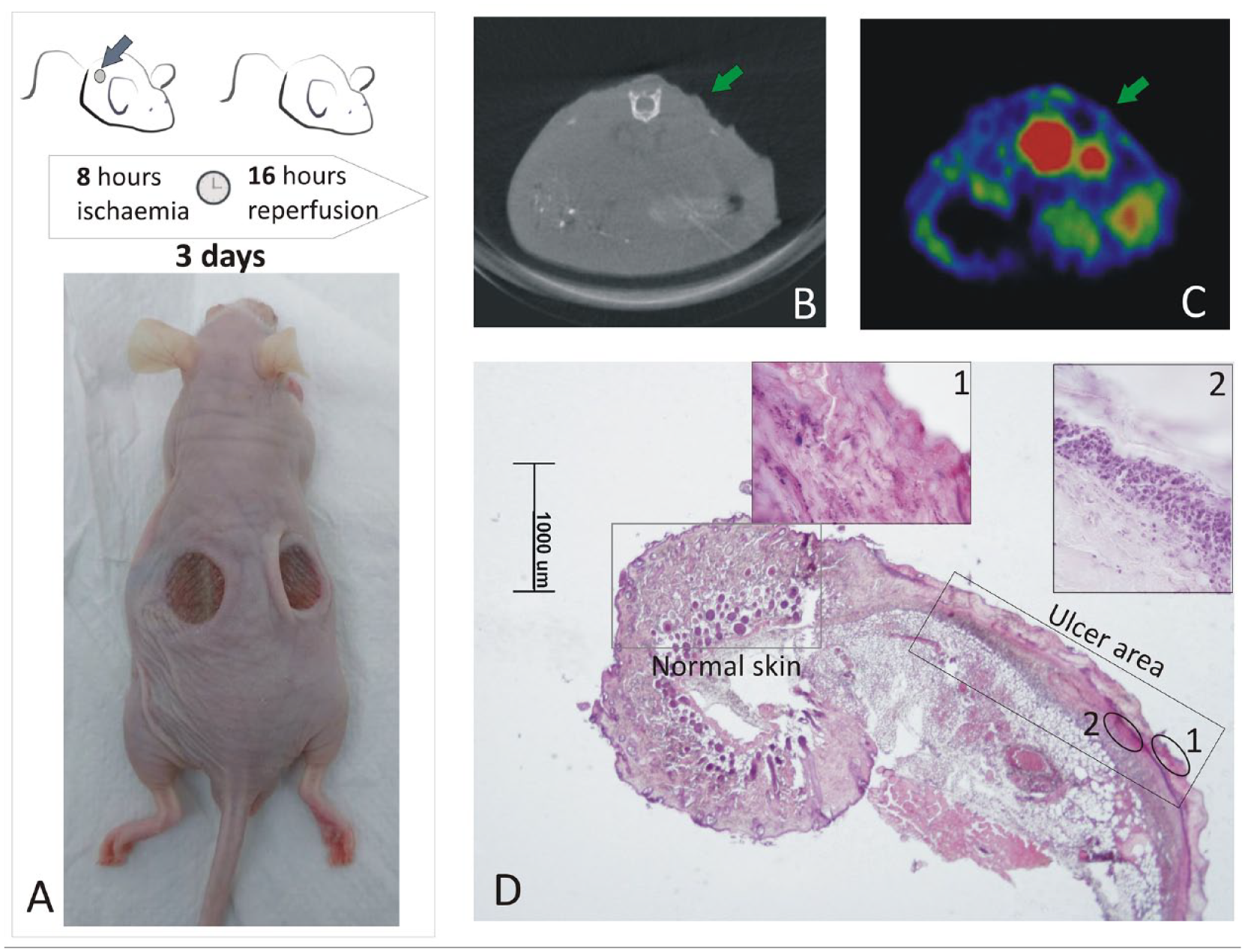
Ulcer model showing a much thinned dermis and an acute inflammatory process. Image A shows how the pressure ulcers are created in the mouse. The appearance of one of the ulcers at the end of the three cycles of ischemia/reperfusion is indicated with arrows in B (CT image) and C (PET image). Image D shows a hematoxylin and eosin–stained section at low magnification, in which both the ulcer area and the lesion can be observed. Inset 1 is a magnified image of an area of devitalized tissue. Inset 2 shows the lymphocyte infiltrate.
After this 3-day period, the ulcers (n = 46) performed in 23 animals were randomly assigned to one of three experimental groups: Control, nontreated ulcers (n = 15 ulcers); Plasma, treated with plasma scaffold (n = 15 ulcers); or Plasma + BM-MNC, treated with plasma scaffold containing BM-MNC (n = 15 ulcers). To monitor the course of the ulcers, each of these experimental groups was divided into 3 subgroups of 5 ulcers each subjected to follow-up exams after 3, 7, and 14 days of treatment. Ulcer diameters were measured using a caliper (Qfun, China) before treatment and after 3, 7, and 14 days of treatment.
Experiments were conducted in accordance with guidelines of the European Union (86/609/EU) and Spanish regulations (BOE 67/8509-12, 1988). Experimental protocols were approved by the Committee for Animal Care and Handling of the University of Oviedo (Permit number: 9-INV-2004). All surgeries were performed under isoflurane anesthesia and animals were sacrificed by CO2 narcosis. Every member of the project involved in the animal experiments had the pertinent official animal experimentation certificate (B and/or C). All efforts were made to comply with ARRIVE Guidelines 20 minimizing animal suffering.
Plasma Scaffold
Isolation from BM and flow cytometry characterization of MNCs
Five male mice (6 weeks old) of the commercial luminescent strain FVB/N-Tg (β-Actin-luc)-Xen mice (Caliper Life Sciences, USA) were used as a source of luminescence (Luc[+]) BM-MNC. All animals were sacrificed by CO2 narcosis. Immediately after slaughter, BM-MNC were isolated from luminescent mice by flushing femurs and tibias with sodium chloride 0.9% saline solution (Grifols, Spain). After performing BM-MNC counts, some samples were characterized by flow cytometry before being resuspended in human blood plasma (see section “Manufacture of plasma scaffolds with/without cells”). For cell characterization, an approximate 1 × 106 BM-MNC suspension was washed twice in phosphate-buffered saline (PBS, Biowest, France) supplemented with 3% fetal bovine serum (FBS, Gibco TermoFisher, USA). Next, cells were stained for 30 min at 4°C with APC-Cy7 anti CD105 (clone MJ7/18, Ref. 1399991182), PE-Cy7 anti CD45 (clone 30-F11, Ref. 1399991064), PE anti CD271 (clone ME20.4, Ref. 1399991063) (Immunostep, Spain) and Alexa Fluor-647 anti CD34 (clone SA376A4, Ref. 152205, Biolegend, USA) antibodies. PE isotype IgG1 (Ref ICIGG1 PE-50, Immunostep) was used as control. Once labeled, cells were centrifuged at 550G for 5 min and washed twice in PBS containing 3% FBS. They were then resuspended in 0.5 mL of PBS and conventional flow cytometry was performed in a FACSAria Flow IIu Cytometer (Becton Dickinson, United States). Data analysis was performed with FlowJo™ v10 Software.
Plasma
Human plasma was ceded by the blood bank of the Hospital Universitario Central de Asturias. These samples had been discarded for routine use as they were close to expiry and were conveniently anonymized.
Manufacture of plasma scaffolds with/without cells
BM cells were resuspended at a density of 1 × 106 cells/cm2 in 1 mL of plasma containing 2 mg of tranexamic acid (Amchafibrin; Fides-Ecofarm, Spain) and 100 µL of 0.913 mEq/mL of CaCl2 (B. Braun, USA) finally added. The mixture was placed at the bottom of a 12-well culture plate and allowed to solidify at 37°C in a 5% CO2 incubator for 30 min. Prior to its application, the plasma-based bioengineered scaffold was manually detached from the bottom of the culture dish and fixed to a gauze with an inorganic polymer glue (Histoacryl, B. Braun) to facilitate handling. Plasma scaffolds without cells were prepared following the same protocol.
In vitro bioluminescence imaging
The presence and viability of the BM-MNC embedded in the plasma scaffold were monitored by non-invasive bioluminescence imaging using an IVIS® Lumina system (Caliper Life Sciences, Germany) at different time points (after 0 h, 24 h, 48 h of incubation) with D-luciferin (Melford, UK) at 14.3 mg/mL dissolved in 0.9% sodium chloride (Grifols, Spain).
Scaffold application
Using tweezers, the scaffold was placed on the ulcer to be treated with the gauze facing upward and then covered with a thin, transparent adhesive film (Fixomull® Transparent, BSN Medical, Germany). To prevent scratching during the first 3 days, mice wore an Elizabethan collar and were housed independently.
Follow-Up
Wound closure
Ulcers were examined at the established time points. Measurements of wound length and width were made using a digital caliper positioned at the margins of each wound. Pictures of the ulcers were also taken. Wound size was determined based on calculating the area of an ellipse. The percentage of wound area reduction was calculated according to the formula: (Final area − Initial area)/Initial area*100.
In vivo bioluminescence imaging
The Lumina IVIS® system was used to assess cell viability in vivo at the plasma scaffold placed over the ulcers after an intraperitoneal injection of 150 mg/Kg D-luciferin diluted in saline solution. The bioluminescence signal was measured using Living Image® software (Perkin Elmer, USA) at the different time points (3, 7, and 14 days).
In vivo image study
For a detailed study of the ulcer resolution process, ulcers were subjected to positron emission tomography (PET) at the time point’s baseline and 7 and 14 days at the Preclinical Image Unit of the University of Oviedo’s animal house. Using this technique, we were able to compare the inner tissue regeneration process among the three groups. Briefly, all scans were performed with a dedicated small-animal PET/CT system (Argus-CT, Sedecal, Spain), which incorporates a dual-layer phoswich array of LYSO-GSO PET detectors and a flat-panel CT with CMOS technology. PET scanning was started 45 min after the injection of 18.5 MBq of FDG-18. The mice were anesthetized using 3%–4% isoflurane in oxygen at 1 L/min for induction, and 1.5%–2% isoflurane in oxygen at 3 L/min for maintenance. CT images were acquired at 150 µA and 45 kVp over 10 min. This was followed by the collection of PET data for 45 min with an X-ray beam intensity of 150 μA and a tube voltage of 40 kVp for attenuation correction.
Specimens
Skin biopsies were freshly harvested from nude mice at posttreatment onset Days 3, 7, and 14. Each specimen was divided into two: one portion was immediately embedded in OCT compound (Tissue Tek, Sakura Finetek, Spain) and then stored frozen at −80°C for molecular analysis, and the other portion was fixed in 4% paraformaldehyde (Sigma-Aldrich, USA) for histological analysis.
Histology
Skin biopsies were fixed in 4% paraformaldehyde in PBS for at least 48 h and then dehydrated in a graded series of ethanol dilutions (Panreac, Spain), cleared in xylene, and embedded in paraffin (VWR, USA). Sections 7- to 10-µm thick were obtained using a microtome H310 (Micron, Germany) and stained with hematoxylin and eosin and Masson’s trichrome. Other sections were reserved for immunohistochemistry. For all procedures, the paraffin sections were first deparaffinated in xylene and rehydrated in an ethanol series.
Hematoxylin and eosin staining
For histological examination of tissues, sections were stained with Harris hematoxylin (Merck, Germany) for 7 min and rinsed in tap water and then stained with 1% eosin (Merck) for 1 min. Finally, the slides were dehydrated in a graded series of ethanol dilutions starting at 96%, then cleared in xylene (VWR) and mounted using EntellanTM rapid mounting medium for microscopy (Merck).
Masson’s trichrome staining
Connective tissues were stained using the Masson Trichrome Stain Kit (Thermo Scientific, UK) according to the manufacturer’s instructions. Briefly, slides were placed in Bouin’s fluid at 56°C for 60 min. Slides were rinsed in tap water until sections were completely clear and then washed once in distilled water. The slides were stained with Weigerth’s iron hematoxylin for 7 min and rinsed in tap water. Next, they were stained with Biebrich Scarlet-Acid Fuchsin for 5 min and rinsed with distilled water followed by immersion in phosphomolybdic-phosphotungstic acid solution for 5 min. Without rinsing, the slides were treated with aniline blue solution for 5 to 10 min. The sections were then placed for 1 min in 1% acetic acid solution, to finalize staining, followed by washing in distilled water, dehydrating in a graded series of ethanol dilutions, clearing in xylene and mounting using Entellan™.
Immunohistochemistry
Briefly, paraffin-embedded sections were deparaffinated in xylene, rehydrated in an ethanol series, and placed in PBS. Endogenous peroxidase was blocked after 20 min with 1% hydrogen peroxide (Panreac, Spain). Possible background staining was blocked by incubating sections for 30 min in 5% bovine serum albumin (BSA, Merck) in PBS. Slides were then incubated with anti-luciferase primary antibody (clone LUC-1, Ref. L2164, Sigma-Aldrich) or collagen I primary antibody [EPR24331-53] (abcam Ref.270993) for 24 h in a humid chamber. The procedure used was the same for the two antibodies. Both antibodies were added at 1/1000 as the optimal dilution. Heat-induced epitope retrieval before immunohistochemical staining was performed using Envision Flex Target Retrieval Solution High pH (Dako, USA). The slides were rinsed, and the antigen localized using the REAL_ EnVision_ Detection system kit (Dako). This kit uses a DAB-like chromogen to stain luciferase-positive cells brown. All sections were counterstained with hematoxylin.
Molecular Study
RNA extraction and cDNA synthesis
Total RNA was isolated using TRI Reagent Solution (Invitrogen, Thermo Fisher) according to the manufacturer’s instructions. RNA quality was assessed through absorbance measurements made using a NanoDrop ND-1000 UV-Vis spectrophotometer (NanoDrop Technologies, USA). All the samples used in the PCR procedures showed a 260/280 nm absorbance ratio between 1.9 and 2.1. A ratio of approximately 2 is generally accepted as optimal for RNA.
Each RNA sample was reverse transcribed with the RT2 First Strand kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions and using the same amount of total RNA for each sample. The cDNA samples were then cooled at 4°C and stored at −20°C until use. To confirm the total absence of residual RNA, we performed a conventional PCR on β-actin (GenBank: E00829) (β-actin forward primer 5′-ACTACCTCATGAAGATCCTC-3′; β-actin reverse primer 5′-CGGATGTCCACGTCACACTTC-3′).
Quantification assay
Quantitative PCR was performed using the Mouse Wound Healing RT2 Profiler PCR Array PAMM-121ZA (Qiagen) containing 84 tested genes representative of seven different signaling pathways and five housekeeping genes, as well as controls for RT and PCR reactions. Whole volume synthesized cDNA was used in the qPCR reaction mixture. To each of 96 well-plates, 25 µl reaction mixture prepared using iQTM SYBR® Green Supermix (2X) (Bio-Rad, USA) were added. The thermal cycling conditions were as recommended by the array plate’s manufacturer for the CFX96 Real-Time PCR Detection System (initial denaturation at 95°C for 10 min followed by 40 cycles of 95°C for 15 s and 60°C for 60 s and a final step at 95°C for 15 s). Parameters testing array reproducibility, reverse transcription efficiency and genomic DNA contamination were adequate for all samples. Cycle threshold (Ct) values were used to calculate fold changes in terms of gene expression according to the 2−ΔΔCt method in all groups in relation to the control group.
Statistical Analysis
PCR array data were analyzed using GeneGlobe software (Qiagen) (https://www.qiagen.com/us/shop/genes-and-pathways/data-analysiscenter-overview-page/?akamai-feo=off) provided by Qiagen (Qiagen Sciences). In this analysis, we set the Ct cutoff at 35 cycles and the boundary to 2-fold. ΔCt was calculated as: Ct value of the gene of interest less the Ct value of the reference gene. The difference between ΔCt recorded for the treatment group and that for the control group is represented by ΔΔCT values, where the fold-change is calculated as 2(−ΔΔCT). Positive fold regulation values indicate upregulation of gene expression, while negative values (a negative inverse of the fold change) indicate downregulation. Values of 2−ΔΔCT for each gene in the treatment and control groups were compared using t-tests for independent samples.
Wound area measurements were compared among the groups using the Kruscal–Wallis test implemented in R Statistical Software RStudio (version 2023.06.2 + 561).
In all tests, significance was set at p < 0.05.
Results
Pressure Ulcer Model
The animal model established consisted of two similar-size pressure ulcers in each animal. Images of the ulcers created on the back of a mouse once the magnet was removed after 3 cycles of ischemia/reperfusion are provided in Fig. 1A. Histological examination of some sections revealed an ulcerated squamous epithelium settling on a thinned dermis with extensive necrosis. An intense inflammatory infiltrate can be observed showing the predominance of polymorphonuclear cells and remains of cell debris (Fig. 1D).
Flow Cytometry
Once cell debris (doublets and red blood cells) was discarded, CD271+ cells were selected as those showing low CD45 and negative CD34 expression. CD271 has been described as a versatile marker to selectively isolate and expand multipotent MSC 21 . Percentages of CD34+ are also shown. The gating strategy can be seen in Figure 2. This analysis was performed in five animals. Briefly, a mean value of 62.1% CD45+ cells was isolated from this population, 0.258% ± 0.15% corresponding to CD34+ cells and 0.01% ± 5.8 × 10−5% characterized as CD271+CD45lowCD34−. In addition, within the CD45− fraction, a mean of 0.93% ± 0.03% endothelial cells were selected as CD105+.
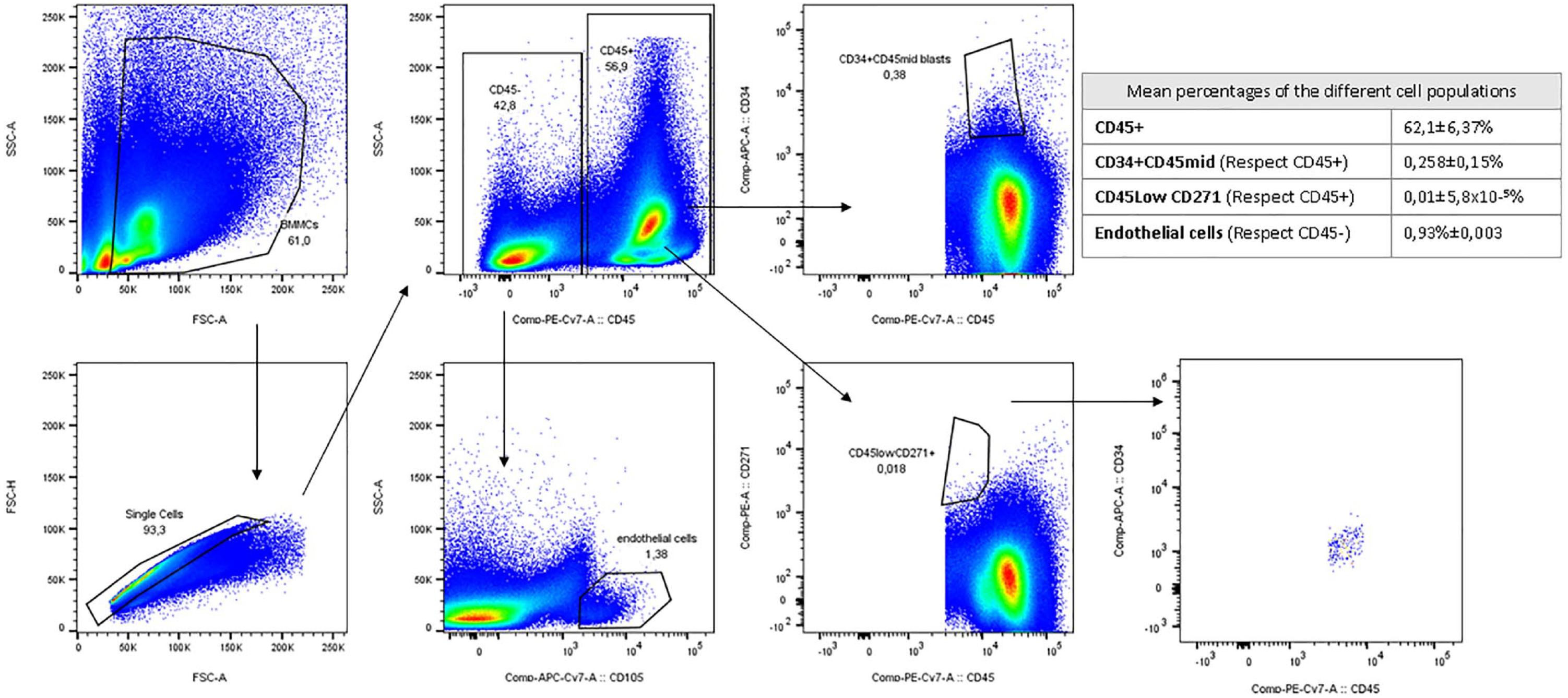
Gating strategy used to characterize the donor bone marrow mononuclear cell populations.
In vitro Bioluminescence Imaging
The cell activity of the plasma plus BM-MNC scaffold was confirmed after 24 h and 48 h of incubation with D-luciferin as a bioluminescence signal (Fig. 3B). Under standard culture conditions, 48 h after the scaffold was prepared BM-MNC were still present, although bioluminescence was reduced. The photon stream is proportional to the number of living cells expressing luciferase.
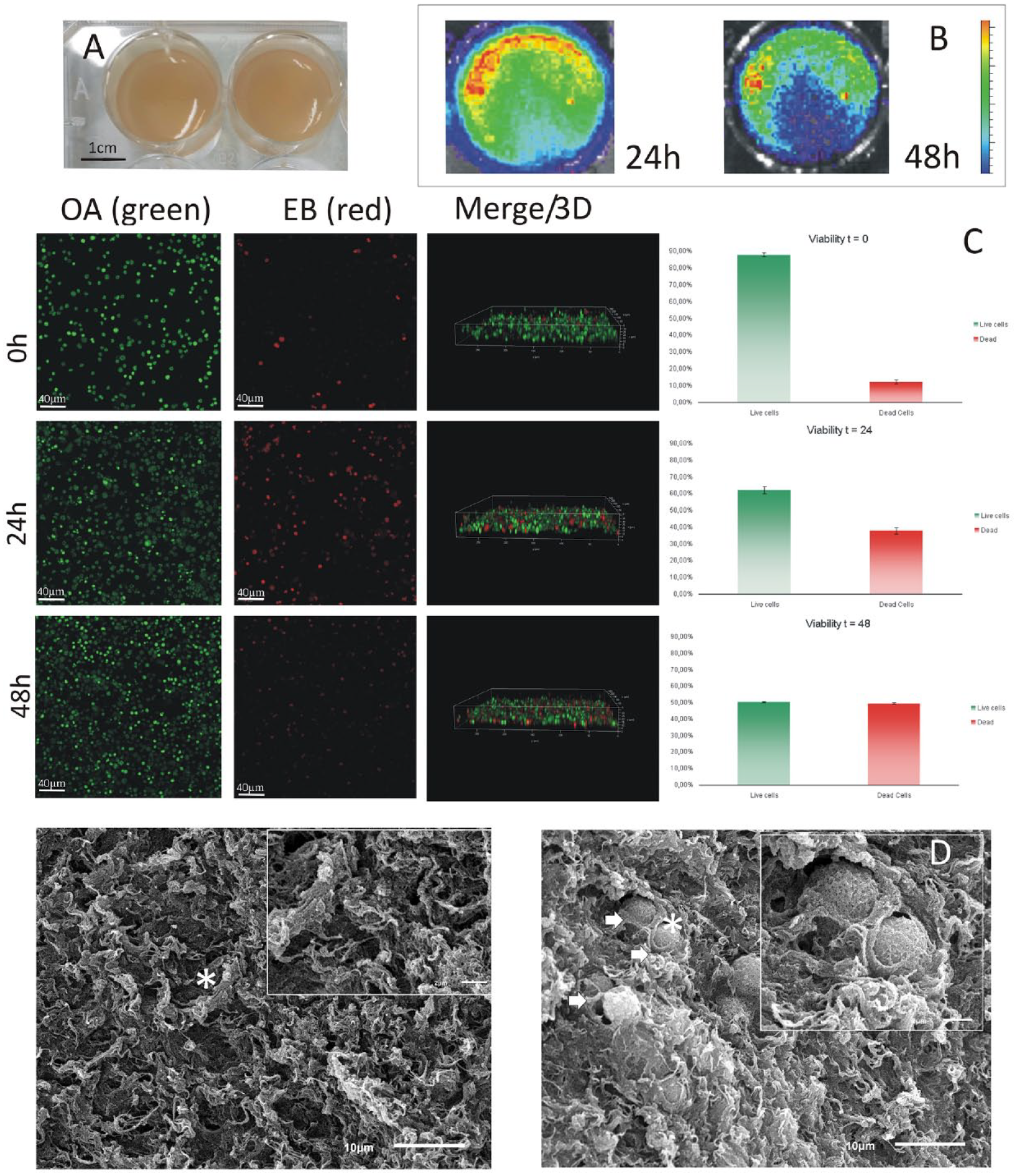
In vitro study of the scaffold. (A) Gel consistency of the plasma-based scaffold. (B) IVIS image of BM-MNC embedded in the plasma-based scaffold at 24 h and 48 h. (C) The images on the left show images acquired by confocal microscopy at different times, 0, 24, and 48 hours after scaffold fabrication. Live cells are shown in green and dead cells in red. The Merge/3D column shows a 3D reconstruction and shows how the cells are distributed. On the right, the livability is graphically represented in the three studied times. (D) On the left is an image of the scaffold without cells taken with the electron microscope, on the right is an image of the scaffold containing the cells. The arrows point to the cells and are seen at higher magnification in the right-hand box of the image.
Follow-Up
Ulcer closure
In each treatment group, wounds were measured with a caliper at baseline and on treatment days 3, 7, and 14. Percentage area reduction values for the three groups are provided in Fig. 4. After 3 days of treatment, these values were similar: 15.81 ± 2.79%, 18.28 ± 10.91%, and 18.24 ± 11.53% for the treatments Control, Plasma, and Plasma + BM-MNC, respectively. Corresponding values were 35.27 ± 10.98%, 43.32 ± 18.59%, and 51.40 ± 28.14%, respectively, at 7 days, and 64.23 ± 27.50%, 71.46 ± 27.98%, and 90.62 ± 8.68%, respectively, at 14 days. While these wound size reduction percentages were clearly higher in the groups treated with cells (mainly at 7 and 14 days), these differences were not significant according to the Kruskal−Wallis non-parametric test. When we subdivided the treatment groups according to the different time points, sample numbers were too small for statistical comparisons, so the data presented are merely descriptive.
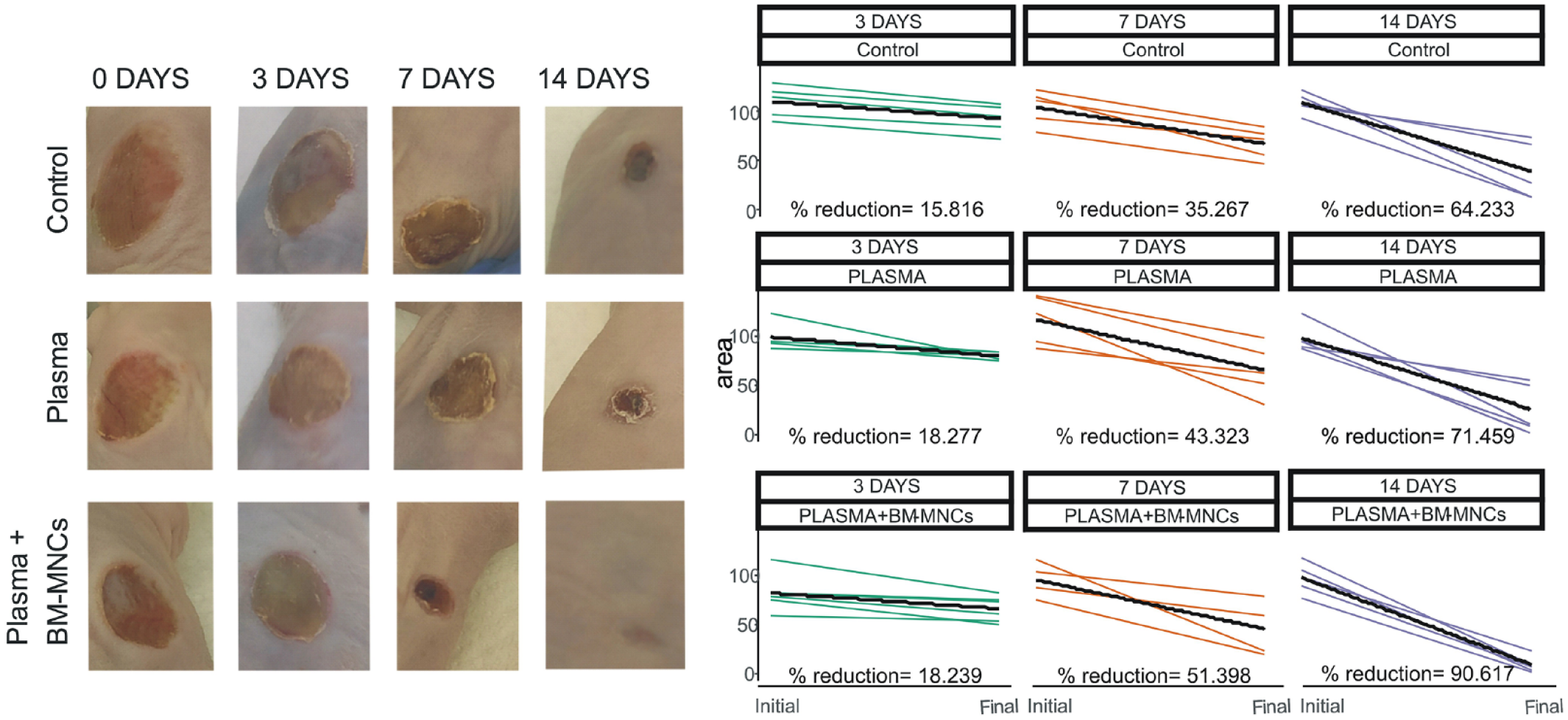
Visual appearance of the pressure ulcers showing clear improvement on Day 14 in the group treated with the plasma scaffold containing BM-MNC.
In vivo bioluminescence imaging
While several attempts were made to in vivo track the BM-MNC embedded in the plasma-based scaffold after treatment, no bioluminescence signal was detected.
In vivo imaging
Inflammation of the ulcer area after intravenous injection of FDG-18 tracer was assessed by PET/CT imaging. After 7 days of treatment, the extent of inflammation was considerably lower in the Plasma + BM-MNC group, while this variable was similar in the Plasma and Control groups. At 14 days, both the Control and Plasma + BM-MNC groups showed a complete morphometabolic response with no remaining residual inflammation, while there was persistent residual inflammation in wounds treated with the plasma-based scaffold alone (Fig. 5).
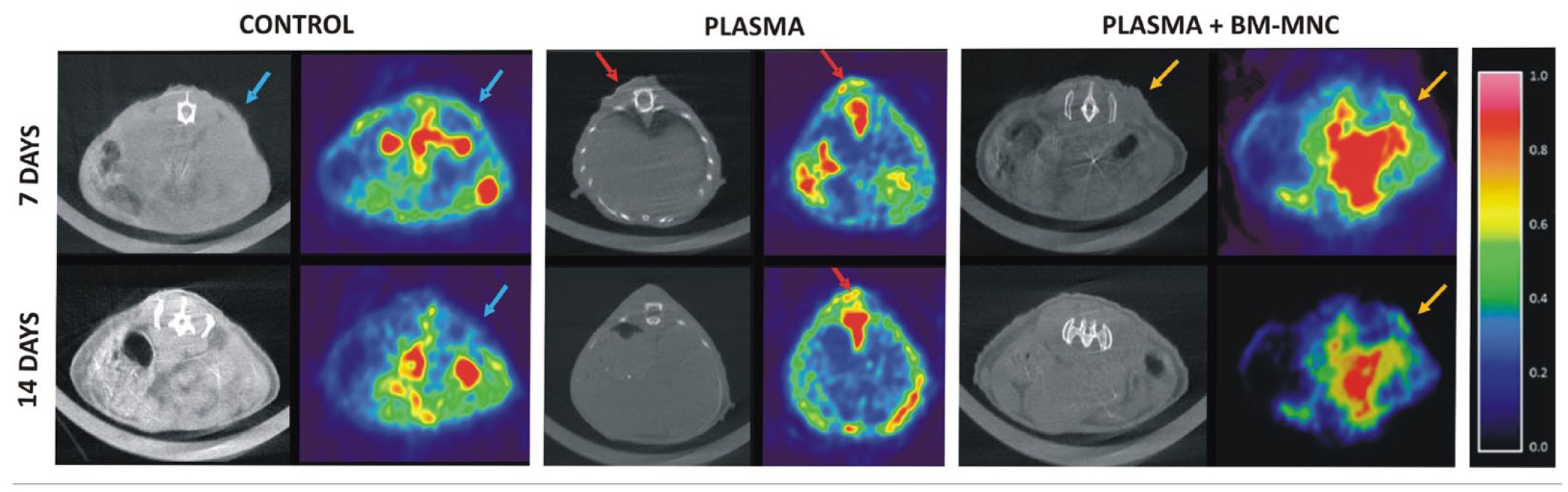
Imaging the level of inflammation of pressure ulcers.
Histologic Procedures
Histological analysis
Our histological findings indicated differences between the treatment groups at each of the established time points (Figs. 6 and 7). On Day 3, a very thin devitalized dermis was observed in the Control and Plasma groups along with intense lymphocyte infiltration and extensive necrosis. The Plasma + BM-MNC group also showed a thin devitalized dermis. However, despite the presence of devitalized tissue and necrotic debris in the plasma + BM-MNC group, this was not as evident as in the other two groups. Reactive changes were also more evident than in the control and plasma-based scaffold-treated animals. After 7 days of treatment, the beginnings of re-epithelialization of more peripheral areas could be observed in all three groups. However, in the Control group, epithelial thickness had not been re-established to the same extent as in the Plasma and Plasma + BM-MNC groups, in which the epithelium was less atrophic. At 14 days, despite complete re-epithelialization observed in each group, differences between groups were still considerable. In the Control group, the dermis appeared disorganized and more fragile, while in the two treatment groups, the thicknesses of the epidermis and dermis were practically normal and we detected loss of common adnexal skin structures. The main difference between these two groups was the presence of collagen fibers revealed by Masson’s trichrome staining and collagen I antibody immunohistochemistry, being denser in the Plasma + BM-MNC group.
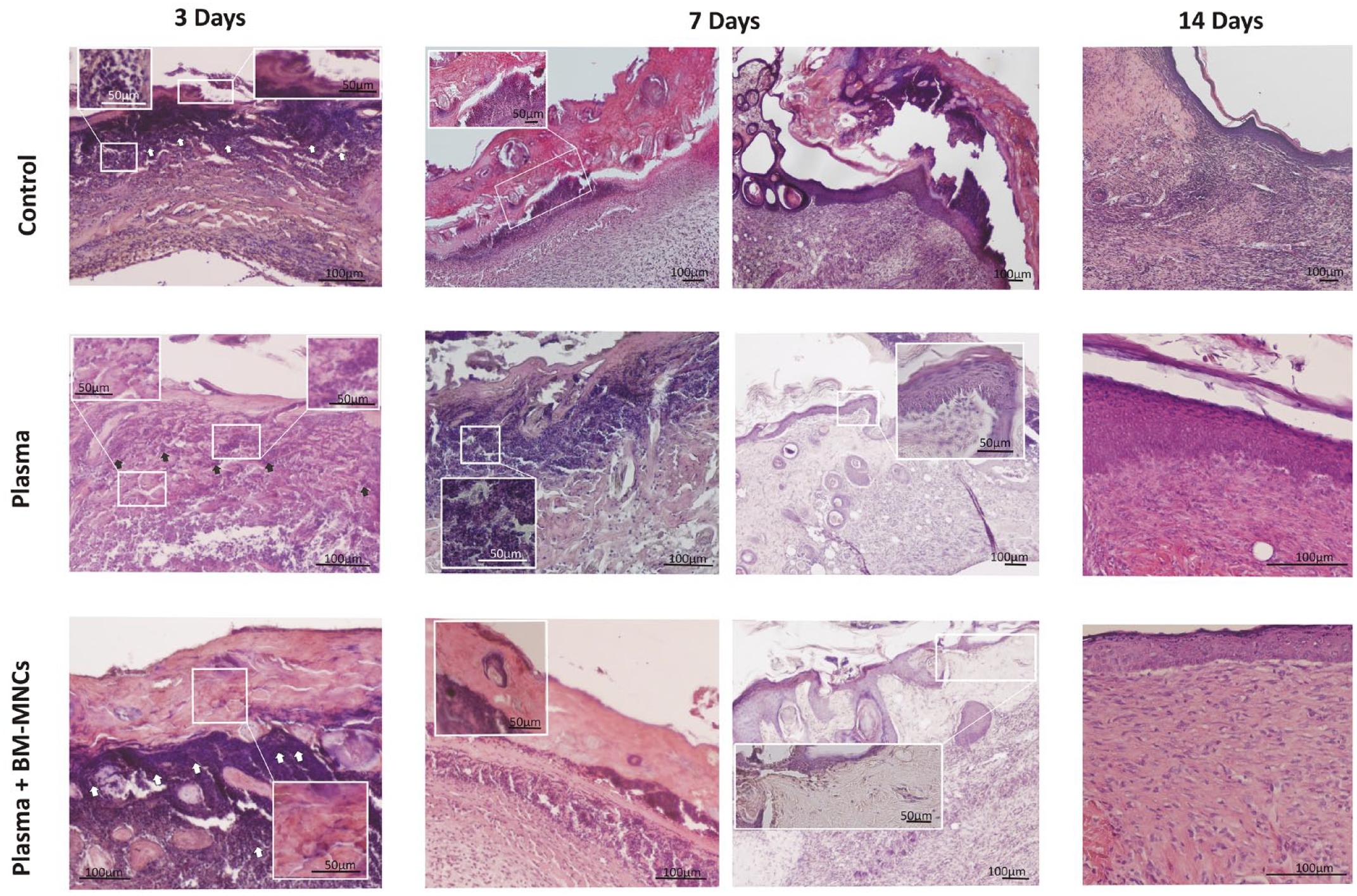
Micrographs of the pressure ulcers showing clear improvement on Day 14 in the Plasma + BM-MNC group. This figure shows the histological study performed by hematoxylin and eosin staining of sections obtained from the treatment groups at three time points (3, 7, and 14 days). Note that the ulcers treated for 14 days with the scaffold containing cells show a much more organized structure compared to the other groups. In the left column (3 days), the arrows show the lymphocyte infiltrate. The insets in the two columns on the left (day 3 and day 7) show magnified images of the devitalized tissue, as well as details of the lymphocyte infiltrate. The third column (day 7) shows the plasma and plasma + BM-MNC group displaying the start of re-epithelialization (insets provide magnified images) which was not observed in the control group.
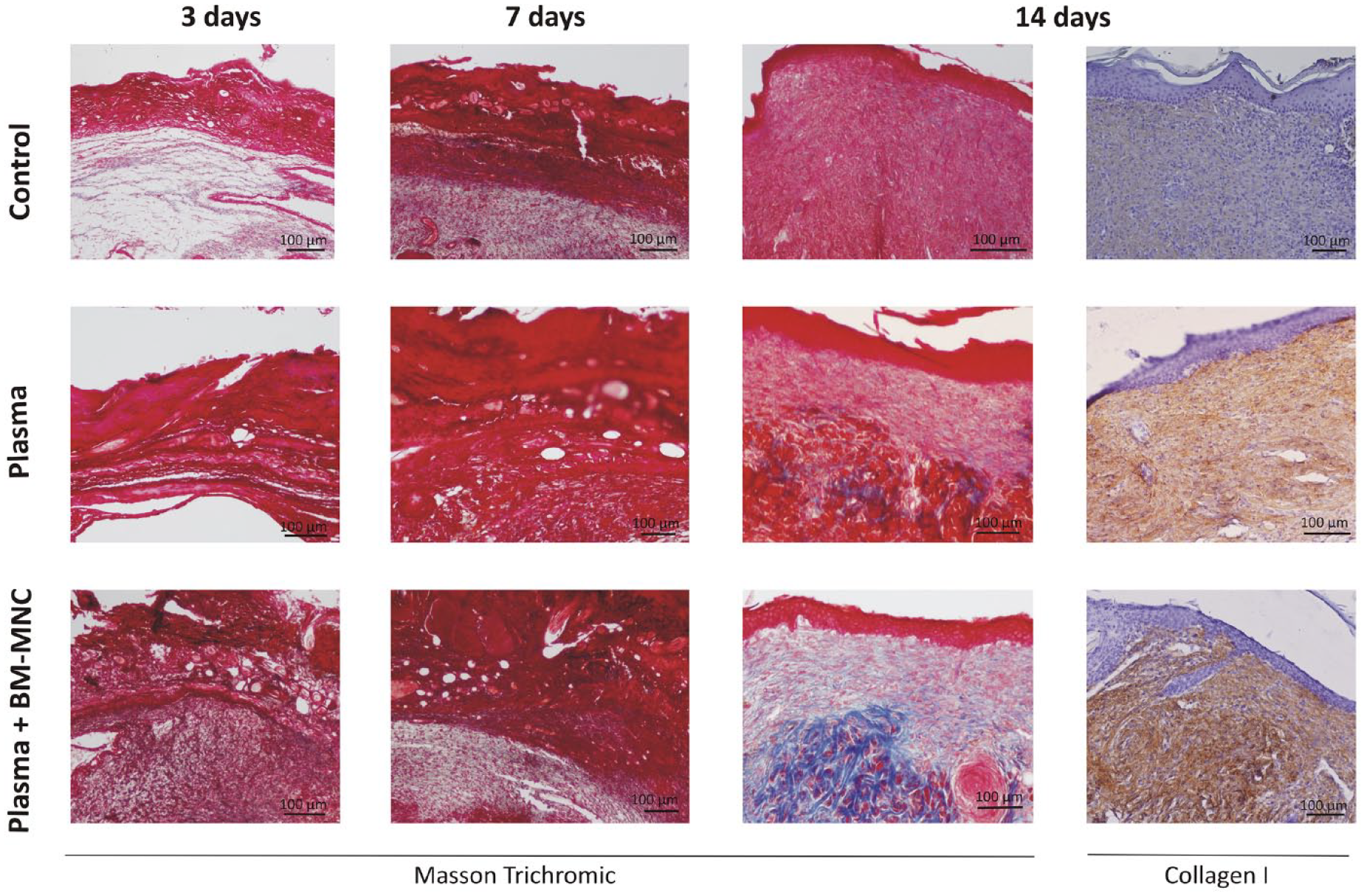
The presence of collagen fibers can be clearly seen on Day 14 in the group of ulcers treated with Plasma + BM-MNC. This figure shows the histological study performed by Masson’s trichrome staining of sections obtained from the treatment groups at three time points (3, 7, and 14 days). Note the blue staining of ulcers treated for 14 days with the plasma-based scaffold containing bone marrow mononuclear cells indicating the presence of collagen as it was confirmed at 14 days by immunohistochemistry.
Immunohistochemistry
An anti-luciferase antibody was used to test for the presence of donor cells. This marker was observed in all sections obtained from ulcers treated for 3, 7, and 14 days with the scaffold plus cells. On Days 3 and 7, labeled cells were detected across surface areas (Fig. 8), while on Day 14, these appeared also in deeper layers. It should be noted that while only low cell numbers were labeled, the marker was detected in every group.
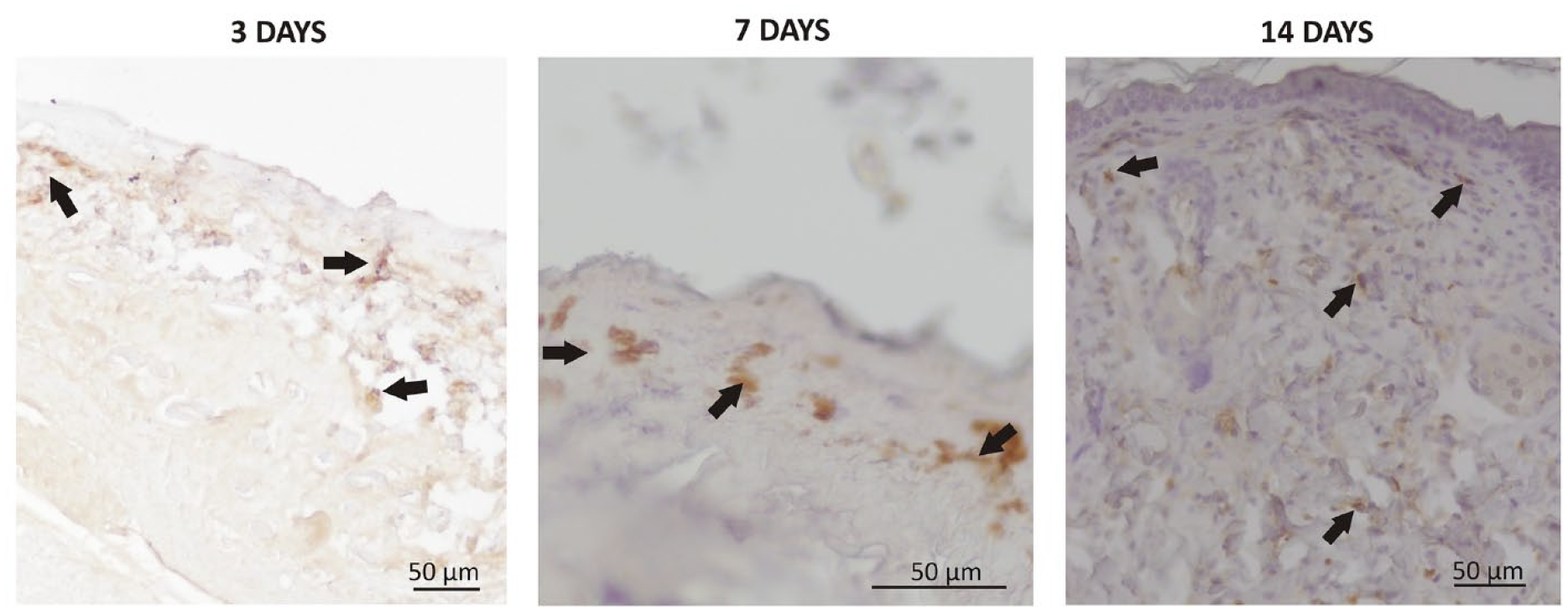
The presence of luciferase detected by immunohistochemistry by labeling with anti-luciferase antibody. Images are shown of pressure ulcers treated with the scaffold plus bone marrow mononuclear cells for 3, 7, and 14 days. Arrows indicate cells testing positive for the antibody.
Differential Expression of Wound Healing Genes
To examine factors that could play an important role in wound healing in our ulcer model, differential gene expression patterns were searched for using the PAMM-121ZA array. The genes considered were involved in different pathways such as those related to the extracellular matrix, remodeling enzymes, cell adhesion and cytoskeleton, inflammatory cytokines and chemokines, growth factors, and signal transduction. Ct values were obtained from the CFX96 PCR Real-Time detection System (Bio-Rad, California, USA), and data were then analyzed using the online analysis tool GeneGlobe software (QIAGEN, Hilden, Germany) provided by the array manufacturer. RNA profiles were compared between treatments at the different follow-up times.
First, we looked for significant (p < 0.05) 2-fold differences in gene expression in each treatment group relative to the control group 3, 7, and 14 days after scaffold placement. This analysis revealed that 16 of the 84 genes of the array were significantly differentially regulated in the three groups. The maximum difference between treatment groups was observed on Day 14, when 11 genes were differentially expressed (Col14α1, Igf1, Itgα1, IL10, Ptgs2, Actc1, Angpt1, Col1α1, Fgf10, Pdgfα, and Rac1), and this was followed by Day 3 when 3 genes were differentially expressed (Itgα4, transforming growth factor β1 [Tgfβ1], and tumor necrosis factor [Tnf]) and Day 7 when 2 genes were differentially expressed (Wisp1 and Tgfα) in the two treatment groups relative to the control group (Table 1).
List of significantly differentially expressed genes related to wound healing at the different time points examined. Positive fold values indicate up-regulation relative to the control group.
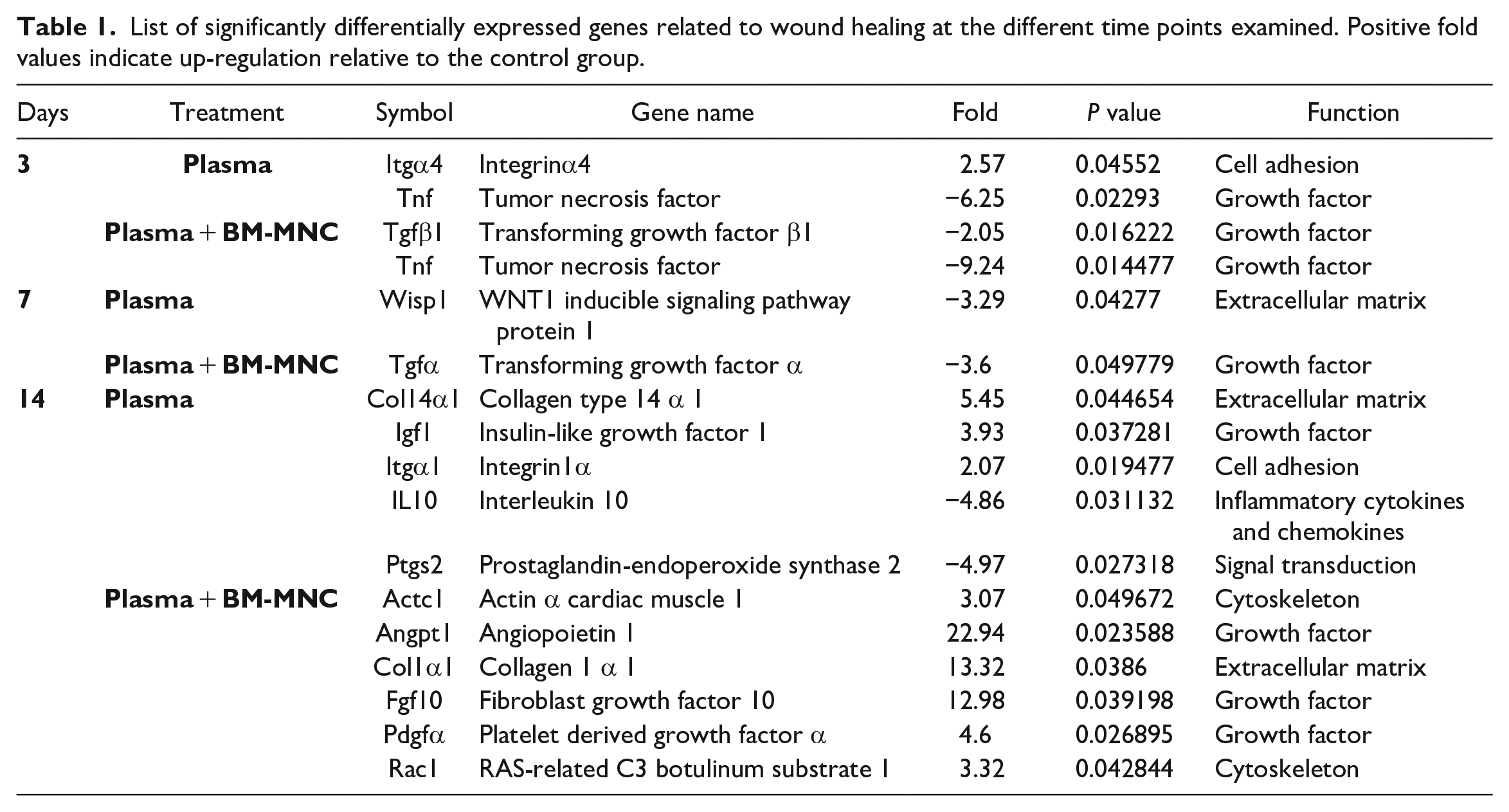
To complete our gene expression study, we also determined the numbers of genes that were over- or underexpressed relative to control. At 3 days, 2 genes were overexpressed and 24 were underexpressed in the Plasma group, and 4 genes were overexpressed and 22 underexpressed in the Plasma + BM-MNC group. At 7 days, these figures were 10 and 28 genes, respectively, in the Plasma group, and 23 and 9 genes respectively in Plasma + BM-MNC. Finally, at 14 days, corresponding numbers were 33 and 8, and 71 and 5 in the two groups, respectively (Fig. 9A).
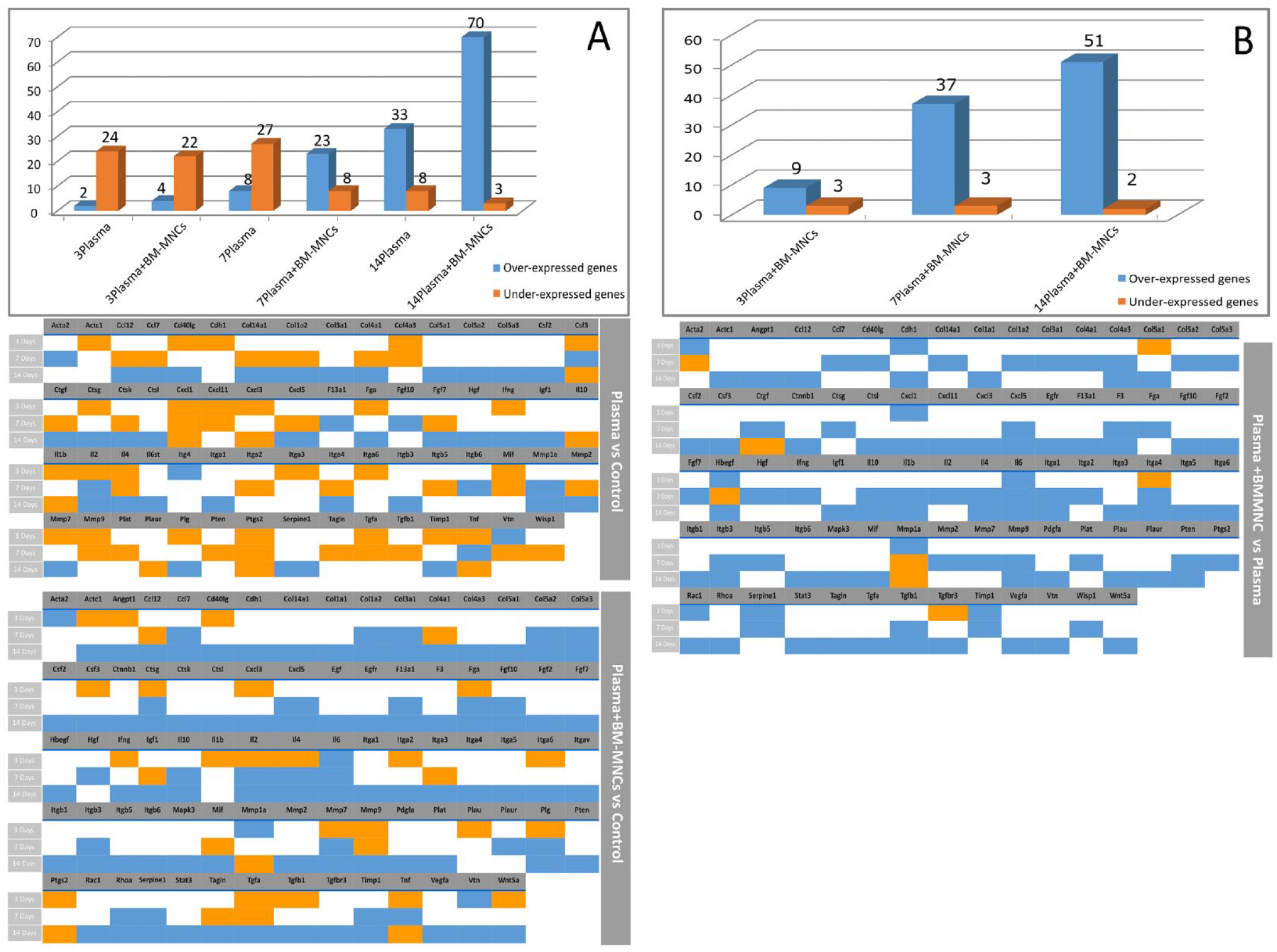
Genes showing differential regulation of mRNA expression. (A) Number of genes overexpressed and underexpressed relative to the control group in the two treatment groups (Plasma and Plasma + BM-MNC) at the different time points (3, 7, and 14 days). In the lower part of the image are represented the over- and underexpressed genes in each experimental group. (B) Number of overexpressed and underexpressed genes in Plasma + BM-MNC group versus Plasma group at different time points (3, 7, and 14 days). In the lower part of the image are represented the over- and underexpressed genes in the Plasma + BM-MNC group versus Plasma group in each time point.
To determine the role of the donor cells in ulcer resolution, we compared the results obtained in Plasma and Plasma + BM-MNC at the three time points (Fig. 9B). After 3 days of treatment, no genes were differentially expressed in the two groups, while at 7 and 14 days, 2 and 5 genes were significantly differentially expressed, respectively, the difference in angiopoietin 1 expression being the most remarkable (Table 2).
List of significantly differentially expressed genes related to wound healing at the different time points examined. Fold values indicate up-regulation in the Plasma+BM-MNC group relative to the Plasma group

Discussion
Wound healing is a complex highly organized process that takes place in stages: hemostasis/inflammation, cell proliferation, and remodeling 22 . These stages involve different mechanisms such as angiogenesis, stem cell recruitment and activation, and extracellular matrix remodeling. Any disruption in these processes impedes adequate wound repair, and this could lead to the development of a chronic lesion entailing keratinocyte proliferation and fibrosis as well as persistent inflammation 23 . In this context, chronic non-healing wounds are those in which the repair process is impaired and that need longer, about 3 months, for their resolution 24 . Chronic wounds include diabetic foot ulcers, venous leg ulcers, and pressure ulcers. Specifically, pressure ulcers are a kind of chronic wound that usually affects individuals with little mobility such as paraplegic or bed-ridden persons or those with underlying conditions like vascular disease or obesity. In addition, due to their high prevalence and costs of care, efficient treatments are urgently needed as they are currently still lacking 25 . In view of the complex nature of wound healing involving a cascade of events and various types of cells, researchers have centered their studies on somatic cell therapy to treat different skin conditions26,27. The use of cells to treat pressure ulcers seems interesting. However, sometimes because of ulcer size and/or location, it is not possible to administer a cell suspension. In this setting, tissue engineering, using scaffolds alone or combined with cells, offer some therapeutic advantages. This was the premise for the design of this study. A mouse pressure ulcer model was used to test a plasma-based scaffold combined or not with BM-MNC. After treatment (3, 7, and 14 days) ulcer progression was determined. This preclinical approach was used to test the safety and efficacy of this tissue engineered treatment, with the ultimate aim of assessing its potential as candidate treatment for a human clinical trial.
Since the start of this century, a similar kind of plasma-based scaffold has provided good outcomes when used to treat major burns 14 and rare skin diseases such as epidermolysis bullosa 11 . This bioengineered skin consists of a dermis composed of human fibroblasts embedded in a three-dimensional plasma support over which keratinocytes are seeded to create an epidermis. In the pre-clinical study by Llames, the viability of cells in the support and their tissue incorporation post-transplant were confirmed. For this study, as donor cells, we used BM-MNC from LUC mice, whose genome includes luciferase, as one of the reporter proteins more commonly used in research. Before application of the scaffold and with the purpose of confirming the viability of donor cells, the presence of luminescence was tested in vitro. Although the scaffold was applied to a pressure ulcer right after its preparation, cell viability was observed until 48 hours post-production. While more complete studies are needed, this finding could be of interest for translating the use of this plasma scaffold harboring BM-MNC to clinical practice. Once the scaffold model was appropriately set up and the previously described donor viability of its cells confirmed, the transplants were performed and we started to monitor the ulcer healing process. Wound repair was examined in vivo by measuring wound size over time and checking for wound reduction. Image-based techniques were also employed. On visual inspection, it was noted that in animals treated with the plasma scaffold plus BM-MNC ulcers had practically resolved after 14 days, with closure observed in more than 90%. This percentage was lower for ulcers treated only with the plasma scaffold and even lower in the control group. However, our sample size was too small for sufficient statistical power which is a main limitation of this study. The image results obtained by PET also revealed improved wound resolution in the mice treated with BM-MNC in terms of observed inflammation. However, variability in the size and depth of the ulcers treated could be confounding factors. Furthermore, the different grades of subclinical infection associated with the wound could also have an impact when trying to determine wound status. Finally, it must be taken into account that the reduced skin thickness of these mice could be decisive when interpreting images, as this could be within the equipment’s resolution limits.
Our histological results show how the wounds resolved structurally and served to confirm our macroscopic findings. In general terms, it was observed here that treatment with Plasma + BM-MNC accelerated wound healing. This result is in line with the findings of several research groups using a scaffold alone or in combination with cells 10 or even exosomes, such as recently reported by Wang et al. 28 . These authors used a collagen/platelet-rich plasma scaffold containing adipocyte-derived exosomes and detected faster wound healing than without treatment. One benefit of the scaffold tested here is that it could be completely autologous and applied the same day the cells were harvested without the need for culture. This avoids a potential risk of contamination. By analyzing in detail the results obtained at the different time points established, we can see how this acceleration of healing occurs.
At 3 days, the homeostasis and inflammation stage of wound repair is active in the mouse 29 . On Day 3, the percentage of wound reduction was macroscopically practically the same in all the treatment groups, only slightly higher for the ulcers treated with BM-MNC. Likewise, our histological study revealed the similar appearance of the ulcers in all three study groups, although the cell-treated ulcers showed a reduced level of tissue devitalization. Gene expression differences between treatment groups were compared using an RTqPCR wound healing array. At this time point, these significant gene expression differences were found: the integrin α4 (Itgα4) gene was overexpressed in the Plasma versus Control group, while Tnf and Tgfβ1 were under expressed in both Plasma and Plasma + BM-MNC versus Control, and this underexpression was more marked in Plasma + BM-MNC for Tnf. Briefly, Itgα4 plays a role in the onset and resolution of an inflammatory process 30 . Hence, its overexpression in the Plasma group could be related to a healing effect of the plasma scaffold. The genes Tnf and Tgfβ1 are related to the inflammatory response and scar tissue formation, meaning that their reduced expression could give rise to less intense inflammatory responses 31 and scar formation 32 in these treatment groups. This is a beneficial outcome as prolonged inflammation can lead to tissue damage if maintained over a long period. At this time point, no significant differences were observed between the Plasma and Plasma + BM-MNC group using the plasma group as control.
At 7 days, the proliferation stage is almost complete in the mouse 29 . When pressure ulcer specimens were analyzed on Day 7, the effects of treatment became more evident, with macroscopic differences observed between the three study groups. Clear differences emerged between controls and ulcers treated with BM-MNC, with wound area reductions of 35.267% and 51.398% recorded in the Control and Plasma + BM-MNC groups, respectively. Notwithstanding, sample sizes were too small for statistical treatment of these data so results are descriptive. At this time point, two genes were significantly underexpressed relative to controls in both treatment groups (Plasma and Plasma + BM-MNC). Tgfα was found underexpressed in the Plasma + BM-MNC group while Wisp1 was underexpressed in the Plasma group. The gene Tgfα is related to inflammation resolution 31 and also plays an important role in the early stages of re-epithelization 33 . Besides, Wisp1 is a gene whose expression is closely associated with healing and remodeling processes 34 . In effect, several studies have shown close interaction between Wisp1, Tgfβ, and extracellular matrix (ECM) proteins in regulating tissue remodeling35,36. As observed at 3 days, the underexpression of these genes could point to an effect of treatment on the wound repair response, determining the reduced expression of these gene products compared to the control group as their functions could be partly adopted by the implanted scaffold. Moreover, to establish the real contribution of the donor cells in the wound-healing process, differential gene expression patterns were examined in the Plasma and Plasma + BM-MNC groups using the Plasma group as control. At this time point of 7 days, the group treated with BM-MNC cells showed the significant overexpression of the genes Tgfβ1 and Wisp1 meaning that cell effects could be related to these two genes. Overall, at 7 days more evident macroscopic differences between treatments were observed very likely attributable to the synergistic effects of the cytokines in the plasma scaffold and paracrine factors released from the mononuclear/mesenchymal cells which enhance wound healing as mediators inducing angiogenesis and reducing inflammation 37 .
Finally, at 14 days, the last stage of tissue repair involving wound contraction and remodeling is completed in the mouse 29 . On day 14, ulcers in this Plasma + BM-MNC group had practically resolved (90.617%), a rate that was higher than that observed in the remaining groups. When ulcer samples were stained with hematoxylin and eosin or Masson’s trichrome this improvement was highlighted. The dermis formed in the control group was thinned and showed a scarce amount of collagen. Also, while an epidermis did form, it was atrophic. Those lesions treated with the plasma scaffold and plasma scaffold plus BM-MNC showed a practically normal epidermis and dermis, but the cell-treated group had denser collagen tracts than the scaffold-alone treatment group. This could translate to a more resilient dermis such that cell treatment both accelerates and improves healing. In both experimental groups, most differentially expressed genes were overexpressed (11 genes) in relation to controls. However, the highest level of upregulation was observed in the Plasma + BM-MNC group for three genes: Angpt1, Col1α1, and Fgf10. These observations are consistent with the other results for these ulcers such that mice treated with plasma and cells showed improved wound healing over that detected in the other groups, as expected considering their synergistic effects. Remodeling is the last stage of wound healing and requires a controlled balance between the synthesis and lysis of collagen, involving upregulation of type I collagen expression 38 as seen here as the predominant expression of this type of collagen. In addition, the expression of Fgf10 was also upregulated, in line with the notion that protein levels of this gene are robustly increased after tissue injury 39 . At this final time point, we compared gene expression patterns in the Plasma group as control and Plasma + BM-MNC group revealing five overexpressed genes in the cell treatment group, namely Angpt1, Col1α1, Fgf10, Pdgfα, and Rac1. The former three genes also showed higher levels of expression in Plasma + BM-MNC in relation to controls so these differences may be explained specifically by a cell effect. However, expression levels of the other two genes only varied significantly between Plasma and Plasma + BM-MNC, so we can hypothesize that the higher expression observed for both these genes could be attributed to the composition of the plasma scaffold.
As discussed above, our findings indicate the beneficial effect of combining cell therapy with the plasma-based support likely mostly a consequence of the antiinflammatory properties of these cells 40 . Notwithstanding, the non-hematopoietic components of BM, that is, MSCs, are capable of differentiating into different cell types 41 . With this in mind, CD271+/CD45low/CD34− populations were determined in BM-MNC from some donors. These markers have been used to establish fresh BM-MSC populations by several researchers including our group 21 . The number of cells with this phenotype is very small 42 so the effect observed here cannot be really attributed to this population alone. However, MSC have been reported to exert a paracrine effect on angiogenesis, increasing levels of factors such as angiopoietin1 37 , enhancing neovascularization of wounds, and improving wound closure and skin regeneration. This could be why we found that this was the gene that was most upregulated in the Plasma+BM-MNC group. Nevertheless, once the scaffold was topically placed on the mouse skin, cells were not visible with IVIS technology although several attempts were made to track them. A possible explanation for this could be that the number of donor cells was low. In addition, the constructs with embedded cells were placed directly on the backs of the animals. Once the cells are incorporated in the host tissue they spread out reducing their density and this could perhaps explain the absence of a bioluminescent signal. Unfortunately, this can be considered a limitation of our study. In contrast, positive immunolabeling for luciferase was detected in pressure ulcers in the Plasma+BM-MNC group (3, 7 and 14 days). Few cells were labeled and these were also localized in very superficial areas. This sparse and localized distribution of luciferase-positive cells in surface layers reinforces the hypothesis of the lack of luminescence, as luciferin injected intraperitoneally could have difficulty in reaching the target. Nevertheless, considering the better outcomes obtained in the group treated with cells, it seems that cell factors and cytokines carried out their functions suggesting the beneficial effects on wound healing of combined treatment with plasma and BM-MNC.
Conclusion
In a murine model of pressure ulcers, a plasma-based scaffold was found to improve ulcer healing and these therapeutic effects were ameliorated by the incorporation of BM-MNC in the scaffold. These beneficial effects were here associated with the over/under expression of genes related to healing and inflammation, highlighting the more than proven regenerative role of these cells. The clinical implications of our findings are that this new tissue engineering tool could be a therapeutic option for the treatment of pressure injuries and other chronic wounds.
Footnotes
Authors’ Contributions
SPL and A-VM conceived the project, performed all the experiments and actively participated in manuscript writing, LR-R assisted with murine surgical procedures, MP-B assisted with the experiments and edited the manuscript, JMG-G reviewed and edited the manuscript, PH-R assisted with the experiments, JM-G assisted in PET experiment including data analysis, R-VM and V-AB assisted with histological experiments and analyzed the associated data, AF-R and EM-R reviewed and edited the manuscript. All the authors have read and agreed to the published version of the manuscript.
Availability of Data and Material
The data that support the findings of this study are available on request from the corresponding author.
Ethical Approval
Experimental protocols were approved by the Committee for Animal Care and Handling of the University of Oviedo (Permit number: 9-INV-2004).
Statement of Human and Animal Rights
Animal experiments were conducted in accordance with guidelines of the European Union (86/609/EU) and Spanish regulations (BOE 67/8509-12, 1988).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been funded by Instituto de Salud Carlos III (ISCIII) through the project “PI19/01192” and co-funded by the European Union.
