Abstract
Systemic lupus erythematosus (SLE) is a multi-organ and systemic autoimmune disease characterized by an imbalance of humoral and cellular immunity. The efficacy and side effects of traditional glucocorticoid and immunosuppressant therapy remain controversial. Recent studies have revealed abnormalities in mesenchymal stem cells (MSCs) in SLE, leading to the application of bone marrow-derived MSCs (BM-MSCs) transplantation technique for SLE treatment. However, autologous transplantation using BM-MSCs from SLE patients has shown suboptimal efficacy due to their dysfunction, while allogeneic mesenchymal stem cell transplantation (MSCT) still faces challenges, such as donor degeneration, genetic instability, and immune rejection. Therefore, exploring new sources of stem cells is crucial for overcoming these limitations in clinical applications. Human amniotic epithelial stem cells (hAESCs), derived from the eighth-day blastocyst, possess strong characteristics including good differentiation potential, immune tolerance with low antigen-presenting ability, and unique immune properties. Hence, hAESCs hold great promise for the treatment of not only SLE but also other autoimmune diseases.
Introduction
The pathogenesis of systemic lupus erythematosus (SLE), an autoimmune disease affecting multiple organs and systems, is closely associated with defects in the bone marrow hematopoietic microenvironment. Studies have confirmed that the primary mechanism involves an imbalance between humoral immunity and cellular immunity, wherein hyperactive B lymphocytes generate a plethora of auto-antibodies, immune complexes, and cytokines. Consequently, functional regulatory T lymphocytes fail to sustain self-tolerance, leading to immune-mediated inflammation and tissue damage.
The conventional clinical management of SLE involves the administration of glucocorticoids and immunosuppressants. Despite achieving control over the condition and improving prognosis for most patients, there remains ongoing debate regarding their efficacy and safety profile. Inevitably, adverse reactions, such as infection, osteoporosis, tumor development, and ovarian failure cannot be entirely avoided. Moreover, some patients experience drug intolerance leading to a significant decline in treatment effectiveness, thereby impacting both quality of life and overall survival.
Recent studies have shown that there are unusual characteristics of mesenchymal stem cells (MSCs) in people with SLE1–5. MSCs that are derived from the bone marrow of SLE patients display impaired growth, are more vulnerable to cell death and aging, and secrete fewer cytokines. They also suppress the proliferation and differentiation of T and B cells. It has been suggested that the malfunctioning of MSCs may be responsible for SLE 6 . Animal models have confirmed this theory, as lupus mice treated with MSCs have shown a dose-dependent improvement in clinical symptoms and survival rates, especially when the treatment is administered in the early stages of the disease.
Advances in Stem Cell Therapy
Stem cells possess self-renewal ability, multi-directional differentiation potential, and multiple immune regulatory functions. They exhibit strong repair and regeneration capabilities, enabling partial or complete modification, and reconstruction of the immune system during their differentiation into immune cells. The pathogenesis of SLE is associated with defects in the bone marrow microenvironment primarily involving hematopoietic stem cells (HSCs) and MSCs. Through extensive research, stem cell-based therapies have demonstrated successful applications in treating SLE.
Hematopoietic Stem Cell Transplantation
Hematopoietic stem cell transplantation (HSCT) has been widely utilized in the treatment of SLE since Marmont et al. first reported successful autologous bone marrow stem cell transplantation (auto-BM-SCT) for severe SLE in 1997, demonstrating favorable therapeutic outcomes. HSCT commonly employs stem cells derived from bone marrow, peripheral blood, or umbilical cord blood and can be categorized as either autologous or allogeneic based on donor type. The primary mechanism underlying HSCs treatment involves immune reconstitution following pretreatment chemotherapy and HSCT. By eliminating abnormal immunity, HSCs facilitate the regeneration of normal cells and establish a new immune tolerance mechanism through thymus modification.
However, studies have identified several limitations associated with both auto-HSCT and allo-HSCT for SLE treatment. Auto-HSCT offers ease of sourcing, high safety profile, and effectiveness; however, due to its inability to alter the genetic progression of the disease, it is associated with a high recurrence rate. Furthermore, research has indicated that HSCT therapy struggles to reverse kidney damage caused by lupus nephritis—particularly structural pathological changes. On the other hand, allo-HSCT presents challenges, such as limited availability of suitable donors, increased risk of graft-versus-host disease (GVHD), transplant-related mortality (TRM), elevated costs, comparable recurrence rates to auto-HSCT treatments, and potential development of secondary autoimmune diseases—contrary to therapeutic objectives—which limits its acceptance among most patients and restricts its clinical applications.
Mesenchymal Stem Cell Transplantation
In recent years, MSCs have been tried out for the treatment of refractory and severe SLE, mainly from bone marrow, umbilical cord, fat, gingiva, and so on. Bone marrow-derived MSCs (BM-MSCs) transplantation is the most common. The main mechanism by which MSCs play a therapeutic role is that MSC can play an immune regulatory role through intercellular contact or secretion of cytokines, and can return to the kidney, lung, and other tissues to play a role, inhibit the proliferation of T cells, including Th17 and Tfh cells, participate in the regulation of Th1/Th2 balance, and promote the generation of Treg cells. It can also inhibit the function of B cells and macrophages, thus playing a role in immune regulation and tissue repair.
Subsequently, numerous studies7,8 have reported that allotransplantation of human BM-MSCs could effectively manage the disease and enhance the prognosis of refractory SLE patients. However, as research progresses, some scholars have discovered that although BM-MSCs are widely present in the hematopoietic microenvironment and play a crucial role in hemocytogenesis and immune regulation 9 , they exhibit dysfunction in SLE patients with reduced cytokine secretion and immune regulatory abilities, decreased proliferation and differentiation capacities, abnormal cell structure, and significantly increased intracellular oxidative stress 10 . Animal experimental studies 11 also revealed that autologous BM-MSCs from SLE mice failed to migrate to target organs after transplantation in vivo, while MSCs transplanted between allogeneic groups still exhibited donor degeneration, genetic instability, and immune rejection. Therefore, exploring novel sources of stem cells is imperative for overcoming clinical application limitations.
Human Amniotic Epithelial Stem Cell Transplantation
The amnion also referred to as the fetal membrane, is situated in the innermost layer of the placenta and constitutes the inner wall of the embryonic amniotic fluid cavity. It is a translucent and resilient membrane devoid of nerves, blood vessels, or lymphatic vessels. The amnion provides an optimal environment for embryo growth and development. With a thickness ranging from 0.02 to 0.5 mm, it can be anatomically divided into five distinct layers: epithelium, basal layer, dense layer, fibroblast layer, and sponge layer. The epithelial layer consists of a single sheet of amniotic epithelial cells (AECs), characterized by highly wrinkled surfaces on AECs. In total, there are approximately 200 million AECs that collectively cover an expansive surface area of up to 2 m2.
As early as 1981, Akle et al. 12 reported that human amniotic epithelial stem cells (hAESCs) did not express the A, B, C, DR antigens, and β2-microglobulin of human white blood cells. Therefore, the immunogenicity of the amniotic membrane is remarkably low with minimal risk of immune rejection. This exceptional immunological compatibility, combined with its abundant availability (obtained during childbirth), positions the amniotic membrane as a readily accessible natural polymer biomaterial. Since the beginning of the last century, it has been recognized that the amniotic membrane serves as an excellent surgical aid in treating skin injuries, such as burns, ulcers, scars, and surgical wound healing13–16.
Before the eighth day after fertilization, the epithelial stem cells in development share many characteristics with embryonic stem cells. According to a study by Miki et al. 17 . In 2005, amniotic epithelial stem cells have similar molecular markers to embryonic stem cells, except for telomerase expression. This suggests that they have the potential to differentiate into the three germ layers unique to embryonic stem cells. Further research has confirmed their ability to differentiate into nerve cells, cardiomyocytes, and hepatocytes, and to successfully treat animal models of diseases, such as Parkinson’s disease, myocardial infarction, and liver injury through transplantation18–20.
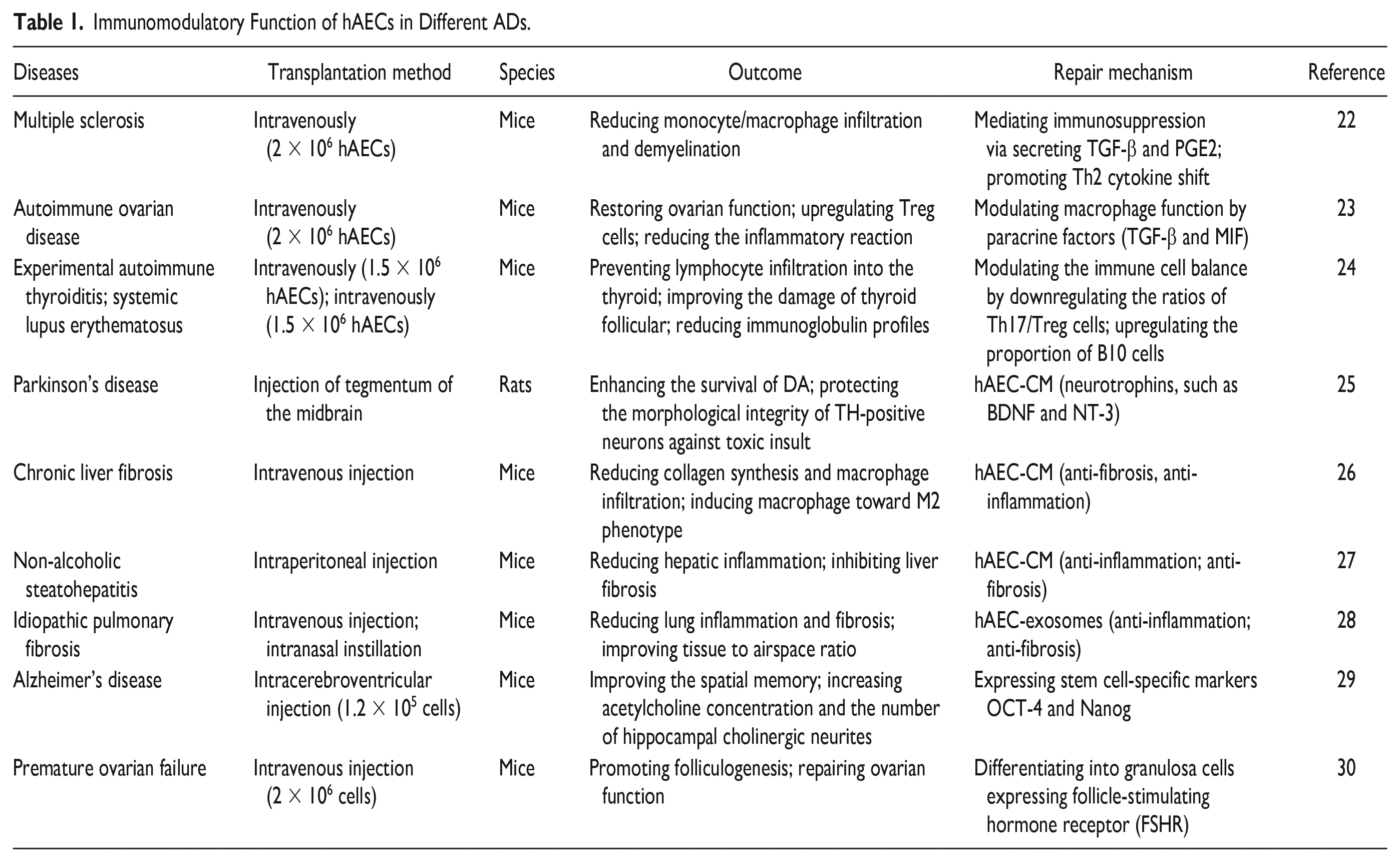
The findings suggest that hAESCs possess embryonic stem cell-like properties and exhibit robust differentiation potential into three dermal tissues. They demonstrate excellent immune tolerance without the need for human leukocyte antigen (HLA) matching or genotype compatibility, while also secreting a diverse range of anti-inflammatory factors with potent anti-inflammatory capabilities. Furthermore, due to their lack of telomerase activity, hAESCs do not undergo uncontrolled proliferation or pose a risk of carcinogenesis, ensuring the safety of transplantation. Clinical investigations conducted in the United States have demonstrated minimal instances of immune rejection following local or intravenous administration of hAESC in both healthy volunteers and patients. These results highlight the limited antigen-presenting ability and distinctive immunological characteristics exhibited by hAESCs. Notably, pioneering research conducted at the University of Texas Southwestern Medical Center in 2006 showcased the therapeutic potential of hAESCs in corneal injury treatment, revealing their capacity to repair damaged corneas and facilitate vision recovery 21 . Subsequently, researchers have successfully applied hAESC transplantation techniques to various autoimmune diseases, including multiple sclerosis, autoimmune ovarian disease, experimental autoimmune thyroiditis, SLE, Parkinson’s disease, chronic liver fibrosis, non-alcoholic steatohepatitis, idiopathic pulmonary fibrosis, Alzheimer’s disease, and premature ovarian failure22–30. And the immunomodulatory function of human amniotic epithelial cells (hAECs) in different Autoimmune diseases (ADs) is shown in Table 1.
Immunomodulatory Function of hAECs in Different ADs.
Comparison Between HSCT, MSCT, and hAESCT
The experience of patients with SLE treated by HSCs transplantation is reviewed and summarized: the curative effect is positive, but the incidence of adverse reactions is high, as well as the expense and the recurrence rate. Therefore, its clinical application is greatly limited. However, since 2007, Sun’s team for the first time successfully carried out allogeneic bone marrow-derived MSC transplantation (BM-MSCT) in the treatment of refractory SLE 31 . In the clinical study of patients with SLE, the long-term follow-up effective rate is 60%, and there is no obvious adverse reaction. There have been no clinical studies and only a few trials in SLE mice. These results demonstrated the immunoregulatory effect of hAECs for inflammation inhibition and injury recovery in SLE murine models 24 . The comparison between HSCT, mesenchymal stem cell transplantation (MSCT), and hAESCT is shown in Table 2.
Comparison Between HSCT, MSCT, and hAESCT for SLE.
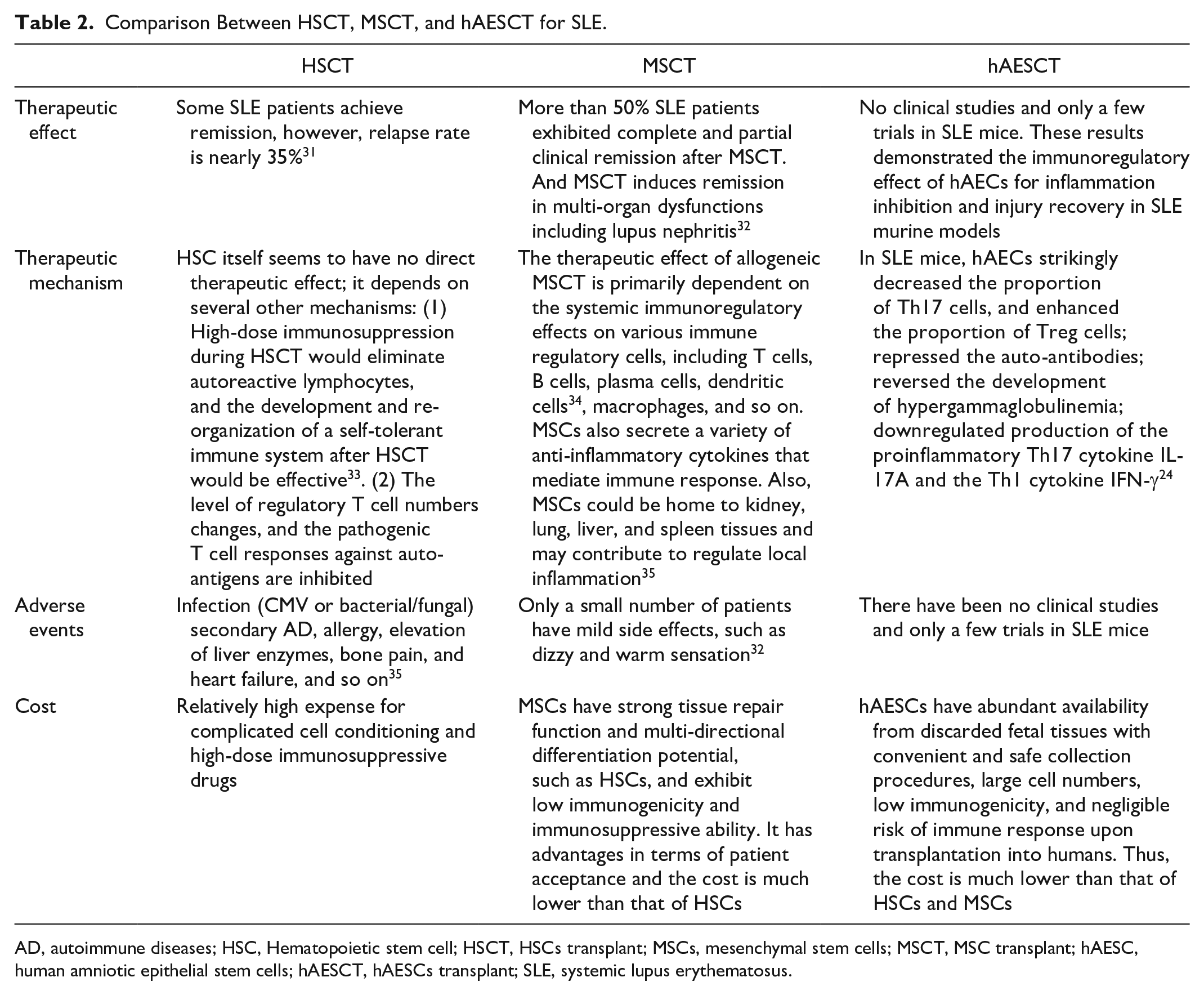
AD, autoimmune diseases; HSC, Hematopoietic stem cell; HSCT, HSCs transplant; MSCs, mesenchymal stem cells; MSCT, MSC transplant; hAESC, human amniotic epithelial stem cells; hAESCT, hAESCs transplant; SLE, systemic lupus erythematosus.
Conclusion
Based on the aforementioned research background, the author proposes that transplantation of hAESCs is a feasible and promising approach for treating SLE. To support this claim, the author refers to previous experimental studies conducted by other researchers using stem cell transplantation in SLE model mice 36 . To validate these findings, the author designs essential confirmatory experiments as follows:
The MRL/LPR mouse37–47, which is widely recognized as an animal model for studying SLE due to its consistent pathological changes with kidney pathology observed in human SLE patients (such as the presence of immune complexes in the mesangial region of glomerulus), was selected. This model has been extensively utilized in various fundamental experimental investigations related to SLE.
Green fluorescent protein (GFP)-labeled hAESCs were intravenously injected into the tail vein of MRL/LPR mice to investigate the reparative function of these cells on the kidneys. Initially, both control and experimental groups of MRL/LPR mice exhibited immune complexes in the mesangial region. It was anticipated that in the control group, tissue immune complexes would be distributed lamellarly and concentrated, whereas in the experimental group transplanted with human AECs, immune complexes would be distributed punctately, significantly lower than those in the control group treated with normal saline48–50. Furthermore, it was hypothesized by the author that within 10 weeks post-transplantation of hAESCs, there would be statistically significant differences observed in urinary protein levels between the two groups of MRL/LPR mice. Theoretically, the rate of increase in urinary protein concentration would be significantly higher in the control group compared with that in the experimental group. In addition, it could be inferred that serum ds-DNA antibody concentration an indicator for lupus nephritis activity would be notably lower in the experimental group as compared with controls.
In addition, it would be valuable to investigate the proportion of helper T cell subsets in the spleen of mice, specifically focusing on the Th1/Th2 ratio. Dendritic cells typically exert an immunomodulatory role by promoting Th1 cell proliferation and maintaining Th1/Th2 balance. Previous research has demonstrated a decrease in the proportion of Th1 cells and an increase in Th2 cells, resulting in a decreased Th1/Th2 ratio within the spleen of MRL/lpr mice 51 . Theoretically, treatment with hAESCs could lead to an increased proportion of both Th1 and Th2 cells, particularly enhancing the abundance of Th1 cells. Consequently, this may contribute to an elevated Th1/Th2 value, indicating that hAESCs possess the potential for promoting the proliferation of Th1 cells and facilitating transformation from Th2 to Th1 cells as a means to restore immune homeostasis.
By evaluating these core indicators through statistical analysis, it can be theoretically concluded that hAESCs have inhibitory effects on pathological injury associated with SLE mice while regulating the lymphocyte ratio between Th1 and TH2 subsets. This multifaceted approach suggests that hAESC therapy holds promise for treating SLE (Fig. 1).
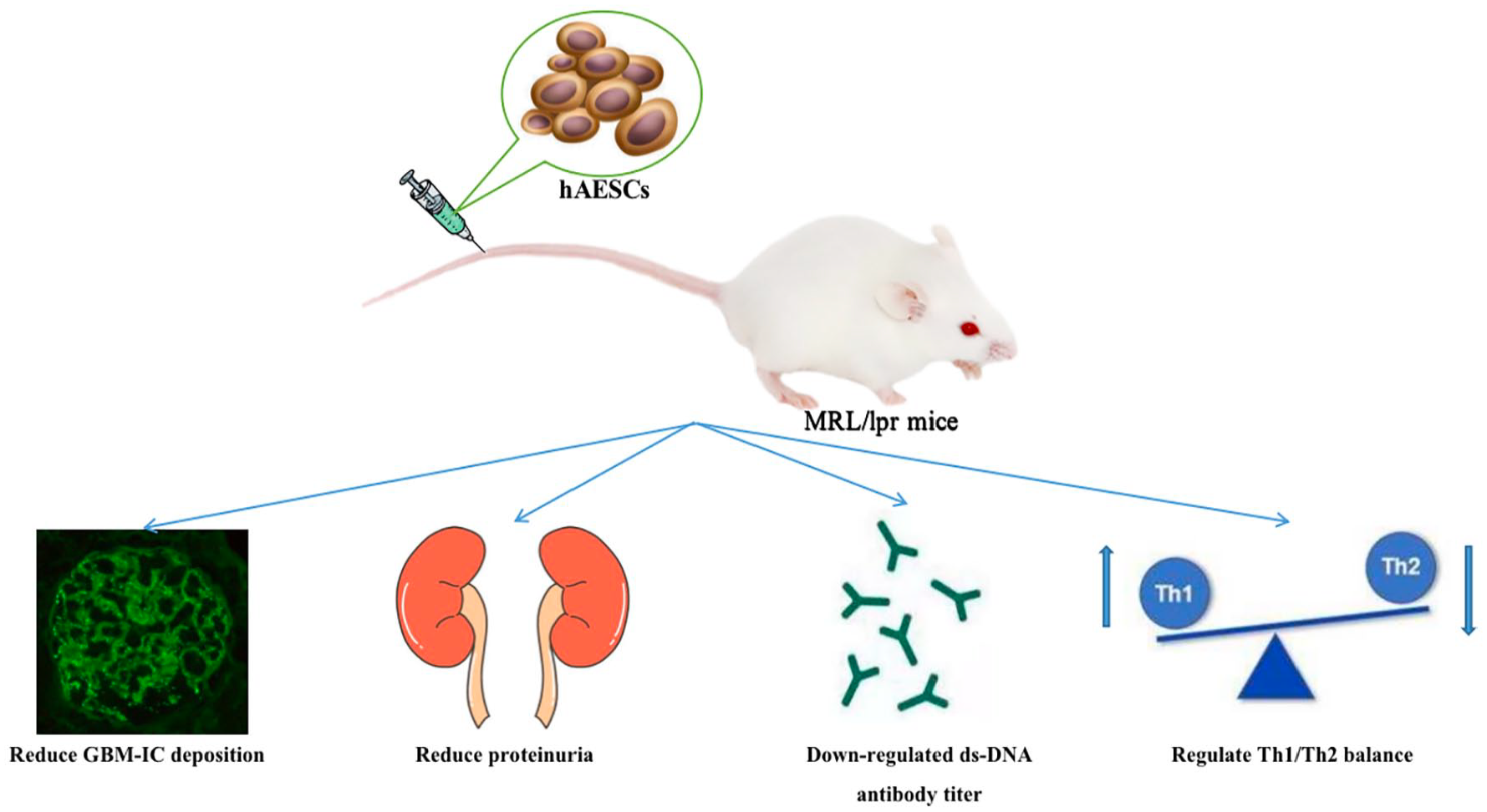
The hypothesis that transplantation of hAESCs exerts a therapeutic effect in MRL/LPR mice.
A lot of progress has been made in researching and developing hAESCs52–54, and Shanghai Siao Biotechnology Co., Ltd has been dedicated to this field for a long time. They focus on creating stem cell products that meet high clinical-grade standards, which is important for clinical investigations of hAESCs. The company has also formed strong collaborations with hospitals to conduct research on stem cell technology for treating related diseases, as well as studies on clinical applications and establishing an amniotic stem cell bank. Together with their partners, they have published research reports and obtained patent authorizations in China and the United States.
In conclusion, the author posits that prioritizing improvements in preliminary experimental studies is crucial. If the results confirm the immunomodulatory effects of hAESCs on immune complex deposition in the mesangial region of MRL/LPR mice, reduction in proteinuria, decrease in serum ds-DNA antibody titer and erythrocyte sedimentation rate (ESR) level, as well as regulation of Th1/Th2 balance can be achieved. Consequently, due to their abundant availability from discarded fetal tissues with convenient and safe collection procedures, large cell numbers, low immunogenicity, and negligible risk of immune response upon transplantation into humans; hAESCs emerge as a promising and superior source for stem cell transplantation therapy to treat SLE.
Footnotes
Authors’ Contributions
The literature review was conducted by XLP, while DQD conceived and planned the research. Article collection was performed by XLP, whereas data collection and analysis were carried out by ZY and LN. The initial manuscript was written collaboratively by XLP, JLN, and SXW with assistance from others. Finally, all authors read, corrected, and approved the final version.
Data Availability Statement
The data are available upon request from the corresponding author.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the seventh installment of the National Traditional Chinese Medicine Experts Academic Experience Inheritance Project (National Traditional Chinese Medicine Teaching Letter [2021] No. 272) and the Traditional Chinese Medical Science and Technology Project of Zhejiang Province (Grant No. 2022ZB130), and the Health Science and Technology Project of Zhejiang Province (Grant No. 2023KY870).
