Abstract
Aging, space flight, and prolonged bed rest have all been linked to bone loss, and no effective treatments are clinically available at present. Here, with the rodent hindlimb unloading (HU) model, we report that the bone marrow (BM) microenvironment was significantly altered, with an increased number of myeloid cells and elevated inflammatory cytokines. In such inflammatory BM, the osteoclast-mediated bone resorption was greatly enhanced, leading to a shifted bone remodeling balance that ultimately ends up with disuse-induced osteoporosis. Using Piezo1 conditional knockout (KO) mice (Piezo1fl/fl;LepRCre), we proved that lack of mechanical stimuli on LepR+ mesenchymal stem cells (MSCs) is the main reason for the pathological BM inflammation. Mechanically, the secretome of MSCs was regulated by mechanical stimuli. Inadequate mechanical load leads to increased production of inflammatory cytokines, such as interleukin (IL)-1α, IL-6, macrophage colony-stimulating factor 1 (M-CSF-1), and so on, which promotes monocyte proliferation and osteoclastic differentiation. Interestingly, transplantation of 10% cyclic mechanical stretch (CMS)-treated MSCs into HU animals significantly alleviated the BM microenvironment and rebalanced bone remodeling. In summary, our research revealed a new mechanism underlying mechanical unloading-induced bone loss and suggested a novel stem cell-based therapy to potentially prevent disuse-induced osteoporosis.
Introduction
A dynamic balance of bone remodeling exists in adult individuals, which means bone resorbed by osteoclasts is replenished by osteoblast-mediated new bone formation to ensure bone mass and mechanical strength1,2. This balance is maintained by precise coupling mechanisms, such as transforming growth factor (TGF)-β1-guided migration of bone marrow (BM) mesenchymal stem cells (MSCs) toward the resorbed bone surface3,4. Many external factors, such as hormone levels 5 , neural signals 6 , and mechanical signals7–10 regulate the coupling process. It has long been recognized that bone tissue can reconstruct morphological structure and strength according to mechanical stress to adapt to mechanical load. Excessive mechanical stimulation promotes osteoblastic differentiation and bone formation. However, decreased mechanical load under pathological conditions, such as long-term bedridden and aging leads to dysregulating of the coupling process, which enhances bone resorption and simultaneously depresses bone formation7–10. The mechanism by which bone formation is uncoupled from resorption under these conditions is vital to clinical prevention and treatment of disuse-induced bone loss.
To solve the above problems, it is necessary to consider how bone tissue senses mechanical stimuli and converts them into biochemical signals. Osteocytes embedded in the bone matrix are believed to be the primary mechanosensor, which regulates bone remodeling activities through secreting cytokines, such as RANKL (receptor activator of nuclear factor kappa B ligand), sclerostin, and Wnts11–17. However, increasing evidence shows that almost all osteo-lineage cells, including MSCs, progenitors, pre-osteoblasts, and mature osteoblasts, are mechanosensitive, indicating that these cells might also play parts in mechanical force-regulated bone remodeling. In recent years, it has been found that mechanical signal-regulated osteogenic differentiation of MSCs and their precursors is mediated by the mechanosensitive ion channel Piezo118–20. Meanwhile, the Piezo1 signal also inhibits osteoclast-mediated bone resorption by regulating the extracellular matrix in osteoblasts 18 . However, the Piezo1-transmitted mechanical signals and their downstream effects in the above cell types need further investigation.
Materials and Methods
Animals and Hindlimb Unloading Model
Three-month-old male Sprague–Dawley (SD) rats were purchased from Shanghai SLAC Laboratory Animal Co. Ltd (Shanghai, China) and were caged under standard conditions. Piezo1fl/fl mice were generated by Cyagen Corporation, China. LepRCre mice were maintained in the lab. Piezo1 KO animals were genotyped by polymerase chain reaction (PCR) with the following primer sequence: forward, 5ʹ-GCCTAGATTCACCTGGCTTC-3ʹ; reverse, 5ʹ -GCTCTTAACCATTGAGCCATCT-3ʹ.
For hindlimb unloading (HU), a strip of medical adhesion tape was applied along the proximal one third of the tail and attached to a metal bar on the tip of the cage. The suspension height was adjusted to prevent the hindlimbs from touching any supporting surface while the forelimbs could contact the grid floor for unrestricted movement. Weight-bearing (WB) rats were used as the control group. The medical tape was also used the same as the HU group, except that the hindlimbs were able to bear the body weight (Fig. 1A). All the experimental procedures were approved by the Committee of Animal Ethics and Experimental Safety of Zunyi Medical University.
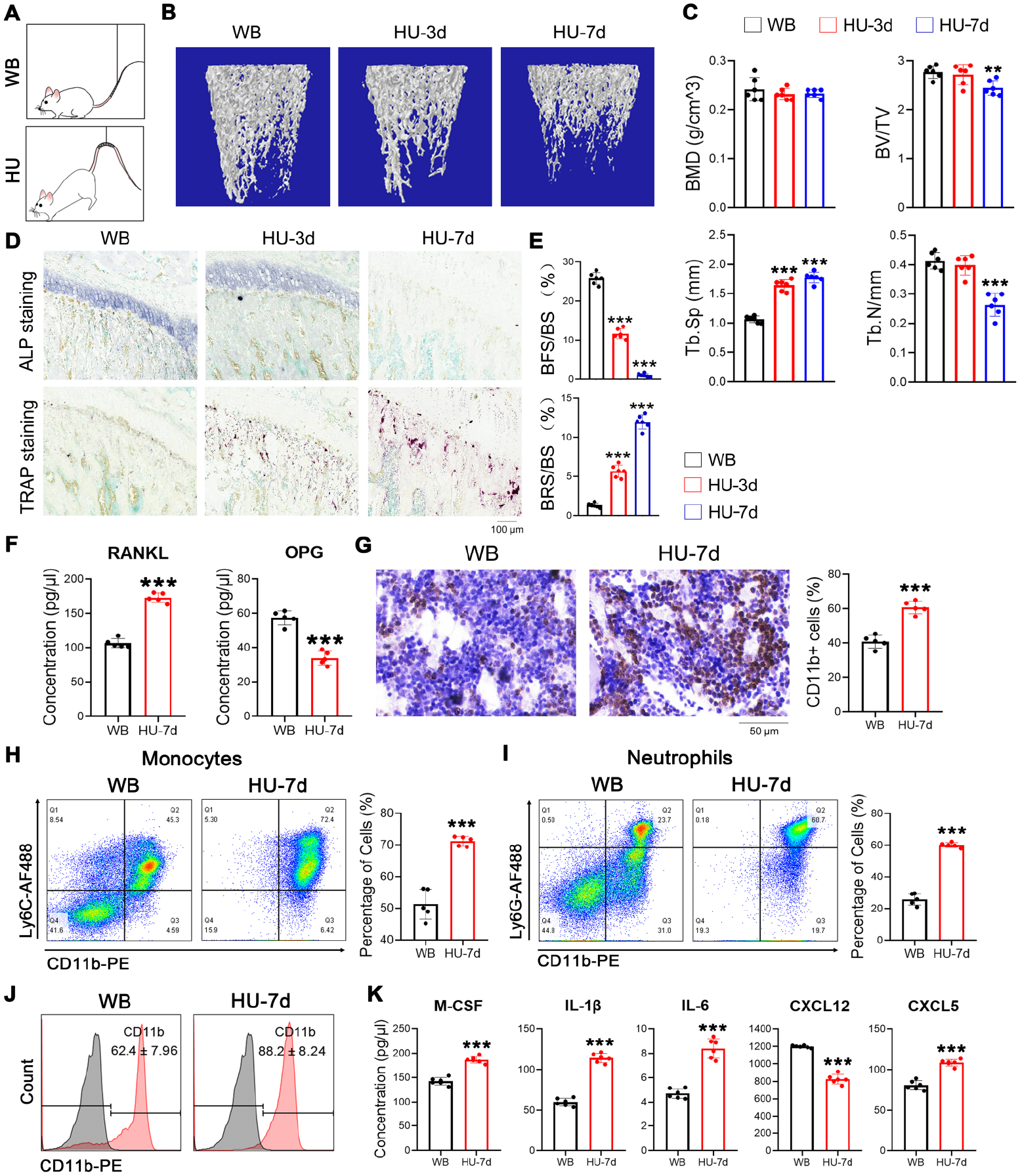
Mechanical unloading leads to BM inflammation. (A) Schematic of HU models and WB controls. (B) Representative images of micro-CT. Femur of rats unloaded for indicated time points were used for micro-CT detection. (C) Quantification of three-dimensional microstructural parameters from micro-CT was shown as BMD, BV/TV, Tb.Sp, and Tb.N. (D) Representative images of ALP/TRAP staining of tibia sections of HU rats as well as WB controls. (E) Quantification of ALP/TRAP staining was performed by measuring the positive areas in tibia sections, and data were presented as a percentage of ALP-positive surface (BFS) or TRAP-positive surface (BRS) to the total areas (BS). Data are means ± SD, n = 6 per group.
Bone Histomorphology
The femurs and tibiae were collected, and the attached soft tissue was removed before being fixed in 4% paraformaldehyde. The fixed femurs were scanned on SkyScan1076 (Bruker micro-CT, Kontich, Belgium). For each rat sample, 301 layers covering 5.478 mm were evaluated; for mouse sample, 201 layers covering 1.826 mm were evaluated. Three-dimensional models of the trabecular bone were reconstructed using SkyScan CT Analyzer version 1.17 to evaluate the alteration of bone and the structural parameters.
For bone remodeling analysis, tibia sections were subjected to tartrate-resistant acid phosphatase (TRAP) staining and alkaline phosphatase (ALP) staining with commercial kits (Beyotime Biotechnology, Shanghai, China) according to the manufacturer’s instructions.
Immunohistochemistry
Tibia sections were used for immunohistochemistry (IHC) analysis according to the standard method. Briefly, tibia sections were deparaffinized, and enzyme-induced epitope retrieval was performed for 30 min at 37°C in pepsin solution after blocking endogenous peroxidase in tissues with 0.3% H2O2/methanol for 10 min. Sections were washed with phosphate-buffered solution (PBS) three times and blocked with blocking buffer for 10 min before incubation with anti-CD11b antibody (Abcam, ab133357) for 1 h. Next, the tissue sections were washed twice with Tris-buffered saline-0.05% Tween20 (TBST) and incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit secondary antibody for 10 min. After washing, positive staining was shown with commercial IHC kits.
Flow Cytometry
For flow cytometric analysis, BM cells were flushed out from tibiae and femurs with PBS/2% fetal bovine serum (FBS) and collected by centrifuge at 300 × g for 10 min. After lysis of red blood cells with ammonium–chloride–potassium (ACK) buffer, an equal number of BM cells were subjected to immunostaining with the primary antibodies for 30 min and then washed with fluorescence-activated cell sorting (FACS) buffer. For rat monocyte/neutrophil staining, BM cells were incubated with AF488-anti-rabbit secondary antibody for another 10 min before washing with FACS buffer. After resuspending in 100 μl FACS buffer, the cells were subjected to FACS analysis with Guaca easyCyte 12 Base System (Millipore). Data were analyzed with FlowJo V10 software. BM monocytes were gated by CD11b-PE+/Ly6C-AF480+, while neutrophils were gated by CD11b-PE+/Ly6G-AF480+ according to published reports 21 .
For the peripheral blood analysis, blood was collected from rats via retro-orbital sinus, and red blood cells were lysed before the FACS staining procedure. Data were captured using a Guaca easyCyte 12 Base System (Millipore) and analyzed using FlowJo V10.
The antibodies used were listed as follows: anti-mouse CD11b-PE (Biolegend 101207), anti-mouse Ly6C-AF647 (Biolegend 128008), anti-mouse Ly6G-AF488 (Biolegend 127625), anti-rat CD11b-PE (Invitrogen 12-0110-82), anti-rat Ly6C (Biorbyt Orb654438), anti-rat Ly6G (GeneTex GTX40912), AF488-anti-rabbit antibody (Biolegend 406416).
BM-Fluid Preparation and Cytokine Detection
BM was flushed out from the tibiae and femurs with 0.5 ml PBS and centrifuged for 10 min at 1000 × g to remove the BM cells. After that, another centrifuge for 10 min at 12,000 × g was performed, and the supernatant was collected for cytokine detection. BM inflammatory cytokines were evaluated using Luminex-based multiplex assays (LX-MultiDTM-23 and LX-MultiDTR-23 were used for the detection of mouse and rat BM-fluid, respectively. Univ-Bio. Co, China).
Isolation of MSCs and Cyclic Mechanical Stretch Application
MSC isolation and in vitro culture were performed as previously reported 4 . Briefly, the BM cells were flushed out from the femurs and tibiae with growth medium (GM, α-minimal essential medium [MEM] supplemented with 10% FBS) into 10 cm dishes. Cells were allowed to adhere and grow for 3 days before the GM was replaced and continued to grow until confluence. Cells were not used beyond passage 5. For CMS application, MSCs were seeded on six-well BioFlex culture plates coated with collagen type Ⅰ (Flexcell International Corporation, Hillsborough, NC, USA). CMS at 0.5 Hz sinusoidal curve at indicated elongation was applied (80,000 με, Sin, 0.5 Hz, CMS) using an FX-5000T Flexercell Tension Plus unit (Flexcell International Corporation). The cells were incubated in a humidified atmosphere with 5% CO2 at 37°C while stretching. Cells were harvested immediately when the CMS application finished.
EdU Incorporation
For in vitro proliferating assay, a commercial kit (RiboBio) was used according to the manufacturer’s protocols. Briefly, EdU solution was diluted 1:1000 with culture medium and incubated with MSCs for 2 h. Blank controls were treated with dimethyl sulfoxide (DMSO). After incubation, cells were fixed with 4% polyformaldehyde. After washing with PBS, cells were permeabilized with 0.3% Triton X-100 for 10 min. The cells were washed twice with 3% bovine serum albumin (BSA) in PBS and then incubated with a freshly prepared Click-iT cocktail (Click-iT reaction buffer, CuSO4, Alexa Fluor 488, and reaction buffer additive) for 30 min. After washing, EdU+ staining was visualized under a fluorescent microscope.
Quantitative Real-Time PCR
Cells were collected and resuspended in TRIzol reagent, followed by RNA extraction. First-strand cDNA was synthesized from 1 μg of total RNA by incubating for 1 h at 42°C with Superscript III reverse transcriptase (Invitrogen, Mulgrave, Australia). Quantitative real-time PCR (qRT-PCR) was performed by LightCycler480 system (Roche, Mannheim, Germany) using SYBR Premix Ex TaqTM (Takara, Dalian, China) according to the manufacturer`s instructions. All amplifications were normalized by glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Data were analyzed using the comparison Ct (2–ΔΔCt) method and expressed as fold change compared with the respective control. Each sample was analyzed in triplicate. The primer sequences used in this study are listed in Table 1.
The Primer Sequences for Reverse Transcription Polymerase Chain Reaction in Rats.

ALP: alkaline phosphatase; Osx: Osterix; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Annexin V Assay
For cell apoptosis detection, an Annexin V kit was used according to the manufacturer’s instructions (Elabscience, Houston, TX, USA). Briefly, cells were harvested after treatment and washed twice with PBS, followed by staining with propidium iodide (PI) and annexin V–fluorescein isothiocyanate (FITC) dyes at room temperature for 5 min. After being washed, cells were resuspended in PBS, and a total of 10,000 cells were analyzed with Guaca easyCyte 12 Base System (Millipore).
Senescence-Associated Beta-Galactosidase Staining
Senescence-associated beta-galactosidase (SA-β-gal) staining was performed using a commercial kit (Beyotime Biotechnology, Shanghai, China) according to the manufacturer’s instructions. Briefly, cells were washed once with PBS before fixation at room temperature for 15 min. After being washed with PBS three times, cells were incubated overnight in the staining buffer at 37°C. β-gal-positive cells were visualized under a microscope.
RNA Sequencing
MSCs were seeded on six-well BioFlex culture plates and cultured for 12 h to adhere to the plates. After that, cells were subjected to 10% CMS treatment for another 24 h. Total RNA was collected by the TRIzol extraction method, and sample quality was assessed using the Nanodrop 2000 spectrophotometer (Thermo Scientific). LC-Bio Technology Co., Ltd., Hangzhou, China, performed RNA sequencing.
Osteoclastic Differentiation
BM cells were obtained and cultured in the presence of M-CSF (30 ng/ml; PeproTech) for three days to generate the BM-derived monocytes. To stimulate osteoclast formation, cells were treated with M-CSF and RANKL (75 ng/ml; PeproTech), and cultured for 6 days. TRAP staining was performed using a commercial kit (Solarbio, China) according to the manufacturer’s instructions.
In Vivo Transplantation
MSCs were isolated from 2-month-old male rats, and in vitro cultured to P3 before CMS application. After 24 h of CMS treatment, cells were immediately collected by trypsin digestion and resuspended in PBS at a density of 106 cells per ml; 105 cells in 100 μl PBS were transplanted into 2-month-old male wild-type rats intravenously by tail-vein injection once every 3 days. Control rats were injected with the same volume of PBS. Recipient rats were subjected to HU immediately after the first injection. BM fluids were collected on day 7 for the detection of inflammatory cytokines. Micro-CT analysis was performed on day 21 to assess bone morphological changes.
Statistical Analysis
Presentation and statistical analysis were performed in GraphPad Prism 8. Results are presented as individual animals and/or replicates with mean ± SD. When comparing more than two groups, analysis of variance (ANOVA) was performed with multiple comparisons, reported in the figure legend. When only two groups were compared, an unpaired Student’s t test was used unless otherwise stated. Statistical significance is reported as P value in the figure legend.
Results
Mechanical Unloading Increased Myeloid Cell Number, Inflammatory Cytokines in the BM, and Osteoclast-Mediated Bone Resorption
To analyze the cause of the unbalanced bone remodeling under microgravity, we applied the HU rat model (Fig. 1A). Seven days after unloading, no significant changes in bone mineral density (BMD) and bone volume (BV) were noticed in HU rats compared with WB controls; however, the trabecular number (Tb.N) was obviously decreased, and the trabecular space (Tb.Sp) was accordingly increased (Fig. 1B, C). Bone remodeling balance was changed after 3 days of unloading, as indicated by the staining of both alkaline phosphatase (ALP) and TRAP in tibia sections from unloaded rats (Fig. 1D, 1E). Consistently, RANKL concentration in the BM fluid significantly increased. In contrast, osteoprotegerin (OPG) concentration decreased (Fig. 1F). To clarify the reason for the rapid enhancement of bone resorption, we first detected the number of BM monocytes, the osteoclast precursors. IHC showed that CD11b+ cells increased dramatically in the BM of rats suspended for 7 days (Fig. 1G). Flow cytometry analysis showed that the number of both monocytes (CD11b+Ly6C+) and neutrophils (CD11b+Ly6G+) was markedly increased (Fig. 1H, I). In addition, we unexpectedly found a similar increase in CD11b+ cells in the peripheral blood (Fig. 1J), indicating that mechanical unloading-induced BM and systemic inflammation. This inflammatory state was further verified by elevated levels of inflammatory cytokines in the BM fluid, such as interleukin (IL)-1β, IL-6, CXCL5, and macrophage colony-stimulating factor (M-CSF; Fig. 1K). CXCL12, which is associated with the activities of hematopoietic stem cells (HSCs)22,23, was markedly decreased (Fig. 1K). These results suggested that the BM microenvironment was significantly altered and exhibited a low-level inflammation at the early stage of mechanical unloading. Under such conditions, it is possible that the proliferation and osteoclastic differentiation of monocytes were extensively promoted, resulting in enhanced bone resorption and final bone loss.
MSC-Specific Piezo1-Deficient Mice Exhibited Similar BM Inflammation Coupled With Enhanced Bone Resorption
We next investigated the reason for mechanical unloading-induced BM inflammation. As we noticed that CXCL12, the most critical maintaining factor of hematopoiesis derived from MSCs22,23, was dramatically decreased, we hypothesized that mechanical unloading indirectly affects the hematopoietic system by modulating the function of MSCs. To prove this, we established a Piezo1 conditional KO mouse model by crossing Piezo1fl/fl mice with LepRCre mice. Compared with littermate controls (Piezo1fl/fl), Piezo1 KO mice exhibited a bone loss phenotype similar to that of the HU model, as evidenced by a decrease in bone mass (Fig. 2A, B) and imbalanced bone remodeling (Fig. 2C). Compared with tail-suspended models, the enzyme-linked immunosorbent assay (ELISA) microarray revealed a more significant up-regulation of inflammatory cytokines in the BM fluid of the Piezo1-deficient mice under the WB conditions (Fig. 2D, Supplementary Table S1), with similar increases in monocyte and neutrophil counts (Fig. 2E). These results confirmed that the loss of mechanical stimuli in MSCs leads to BM inflammation identical to that in HU model, suggesting that mechanically stimulated MSCs play an essential regulatory role in maintaining the BM microenvironment.
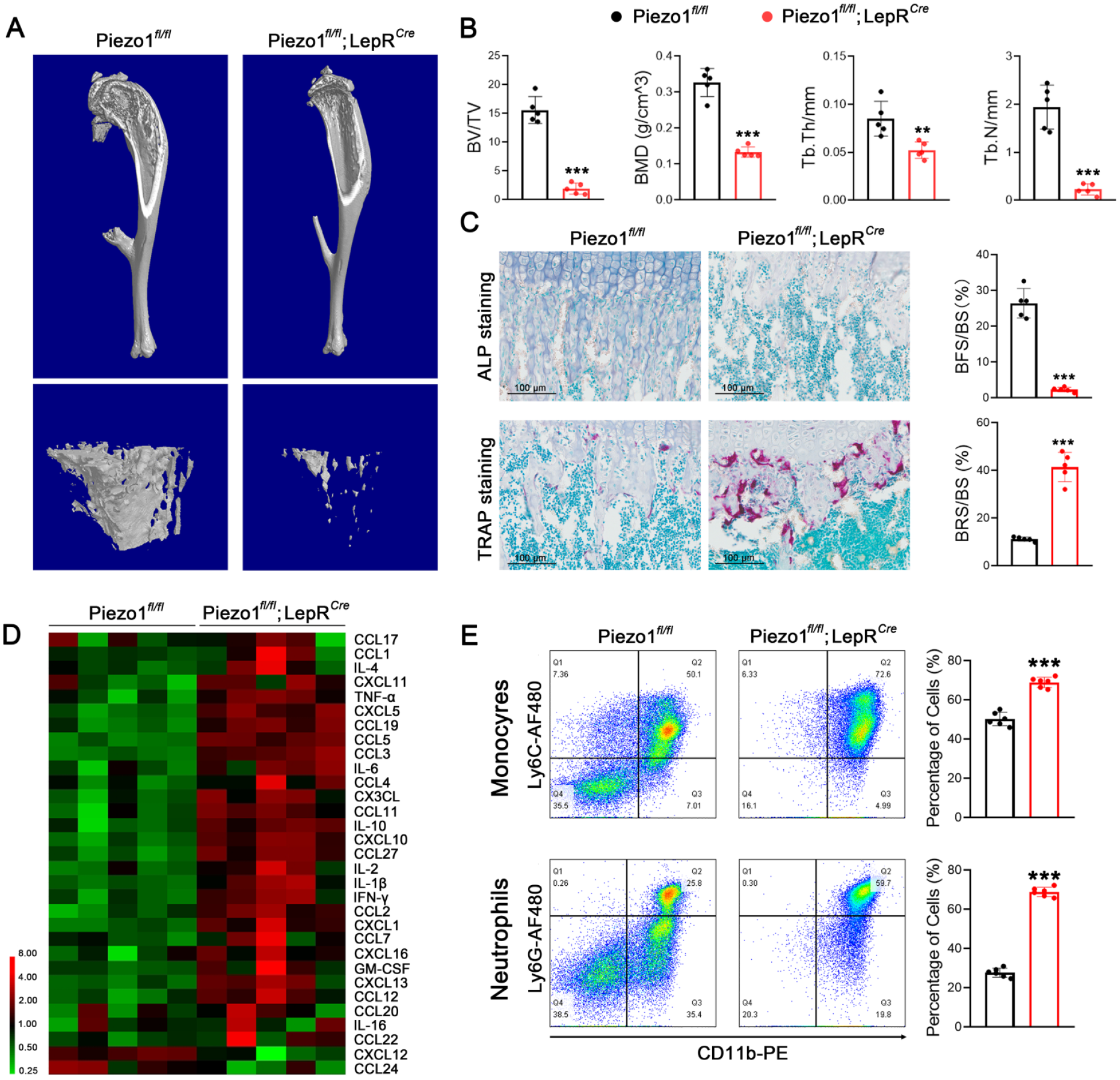
Conditional knockout of Piezo1 in MSCs leads to BM inflammation under the WB conditions. (A) Representative images of micro-CT of Piezo1 conditional KO mice (Piezo1fl/fl; LepRCre) and their littermate controls (Piezo1fl/fl). n = 5 per group. (B) Quantification of three-dimensional microstructural parameters from micro-CT scanned tibia. (C) Representative images of ALP and TRAP staining of tibia sections. Quantification data were presented as a percentage of ALP-positive area (BFS) or TRAP-positive area (BRS) to total bone surface (BS). n =5 per group. (D) Heatmap showed the protein levels of inflammatory cytokines in the BM fluid of Piezo1fl/fl; LepRCre mice compared with Piezo1fl/fl littermates. n = 5 per group. (E) Flow cytometry analysis of the percentage of monocytes and neutrophils. The statistical significance was assessed by a Student’s t test of three to six independent experiments. All data represented are means ± SD. *P < 0.05, **P < 0.01, ***P < 0.001. MSC: mesenchymal stem cell; BM: bone marrow; WB: weight-bearing; CT: computed tomography; KO: knockout; ALP: alkaline phosphatase; TRAP: tartrate-resistant acid phosphatase; BFS: bone formation surface; BRS: bone resorption surface; BS: bone surface.
10% CMS Stimulation Alters the Synthesis and Secretion of Inflammatory Cytokines in MSCs
To explore the molecular mechanism by which MSCs modulate the BM microenvironment under mechanical stimuli, we established a cyclic mechanical stretch (CMS) model using an FX-5000T Flexcell Tension Plus unit on in vitro cultured MSCs. We first noticed that 10% CMS promoted cell proliferation and the expression of osteoblastic genes Runx2, ALP, and Osterix (Osx) (Fig. 3A). To map the specific changes in gene expression, we performed high-throughput sequencing analysis immediately after 24 h of 10% CMS treatment. KEGG (Kyoto Encyclopedia of Genes and Genomes) analysis revealed that differentially expressed genes (DEGs) were mainly enriched in cytokine and receptor-related pathways, inflammation-related signaling pathways, such as rheumatoid arthritis, IL-17 signaling pathway, tumor necrosis factor (TNF) signaling pathway, and osteoclast differentiation pathway (Fig. 3B). Also, Volcano plots indicated that the top 20 DEGs were mainly cytokines (Fig. 3C), of which, inflammatory cytokines, such as IL-1α, CXCL2, and M-CSF were markedly down-regulated. Heatmap of DEGs enriched in cytokine-cytokine receptor interaction pathway showed significantly decreased expression of inflammatory cytokines (Fig. 3D, Supplementary Table S2) in 10% CMS-treated MSCs.
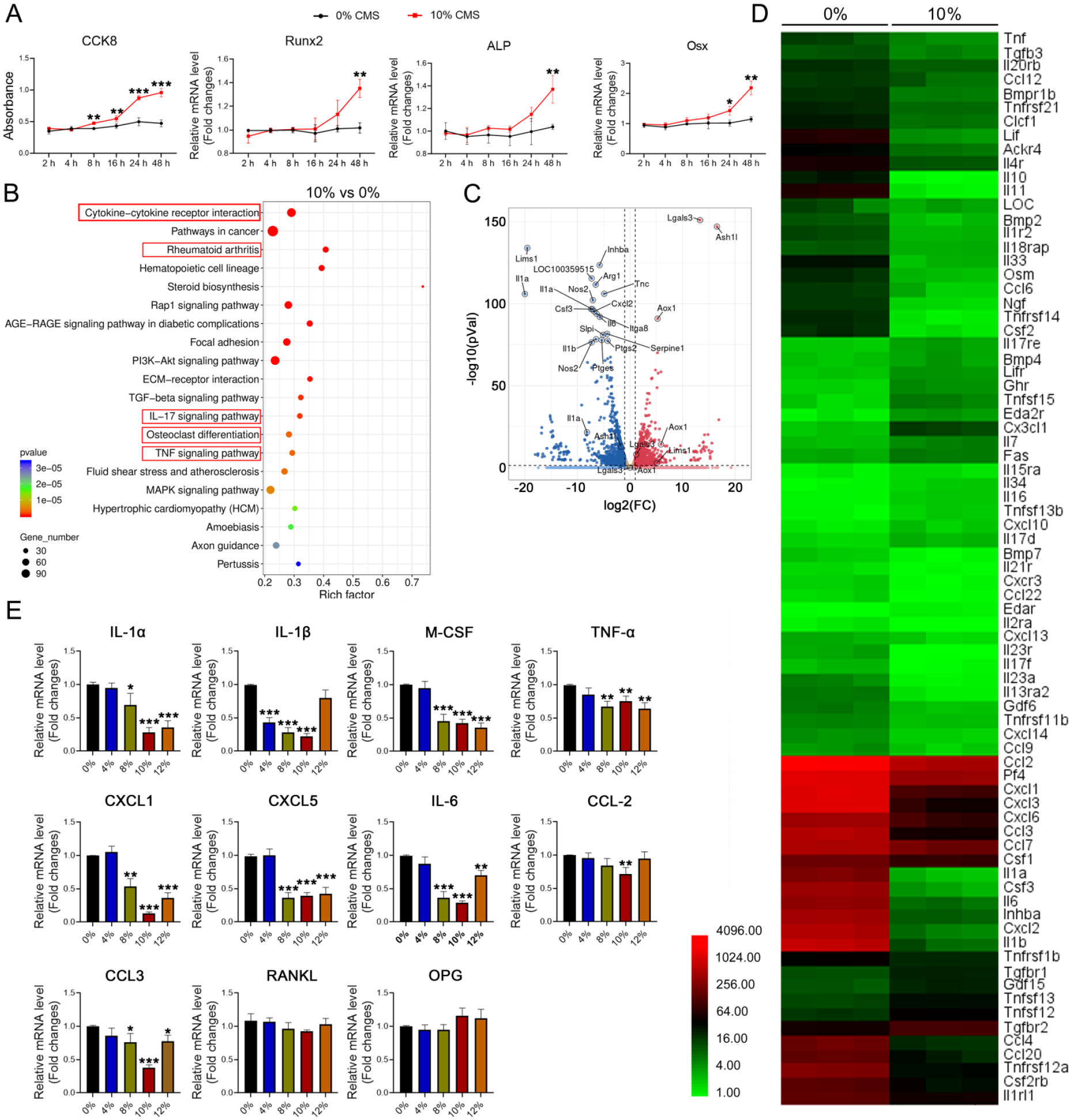
Ten percent CMS altered MSC secretome. (A) The time-dependent effect of 10% CMS on MSC metabolism and osteoblastic differentiation as indicated by CCK8 assay and the expression levels of osteoblastic genes Runx2, ALP, and Osx. n = 3. (B) Top 20 pathways enriched in cells treated by 10% CMS. (C) Volcano plot of DEGs identified by RNA-seq. The top 20 DEGs were indicated. (D) Heatmap of genes enriched in cytokines–cytokine receptor interaction pathways. (E) Quantitative RT-PCR showed the dose-dependent effect of CMS on the expression of selected cytokines. The statistical significance of experiments was assessed by a two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001 versus the respective nc group. Data are means ± SD of three independent experiments. Gene expression levels were normalized to GAPDH. CMS: cyclic mechanical stretch; MSC: mesenchymal stem cell; ALP: alkaline phosphatase; DEG: differentially expressed gene; RT-PCR: real-time polymerase chain reaction; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
To test whether the expressional changes of inflammatory cytokines exhibit a dose-dependent effect, different strengths of CMS were applied. We noticed significant decreases in the expression level of tested cytokines were manifested at 8% CMS, which reached their lowest point at 10% CMS (Fig. 3E). Notably, the expression levels of RANKL and OPG were not significantly altered (Fig. 3E), suggesting that the changes of these two factors in the BM after HU did not originate from MSCs 24 . In summary, these results indicated that adequate mechanical load on MSCs plays an essential role in regulating cytokine production.
CMS-Treated MSCs Inhibit Monocyte Proliferation and Osteoclastic Differentiation
As we noticed that DEGs after CMS stimulation were also enriched in the osteoclast differentiation pathway (Fig. 3B), indicating a correlation between MSC secretome and osteoclastic differentiation. As the expression levels of RANKL and OPG were not significantly changed in MSCs (Fig. 3E), we hypothesized that MSC secretome might affect the biological activities of monocytes. To prove this, the conditioned medium (CM) from MSCs after 24 h of 10% CMS application (CM-10%) was collected to treat RAW264.7 cells. Compared with the CM of MSCs in a static state (CM-0%), the CM-10% significantly inhibited the proliferation of RAW264.7 cells (Fig. 4A-C), but had no effect on cell apoptosis (Fig. 4D). Notably, CM-10% almost blocked osteoclast differentiation (Fig. 4E). It is reported that senescent cell releases large amounts of inflammatory cytokines known as senescence-associated secretory phenotype (SASP) 25 . To rule out the possibility that the secretome of MSCs was altered by CMS-induced cellular senescence, we performed SA-β-gal staining, and no significant differences were noticed after 24 h of CMS application on MSCs (Fig. 4F). Together, these results demonstrated that CMS-modified MSC secretome markedly inhibits the proliferation and osteoclastic differentiation of monocytes.
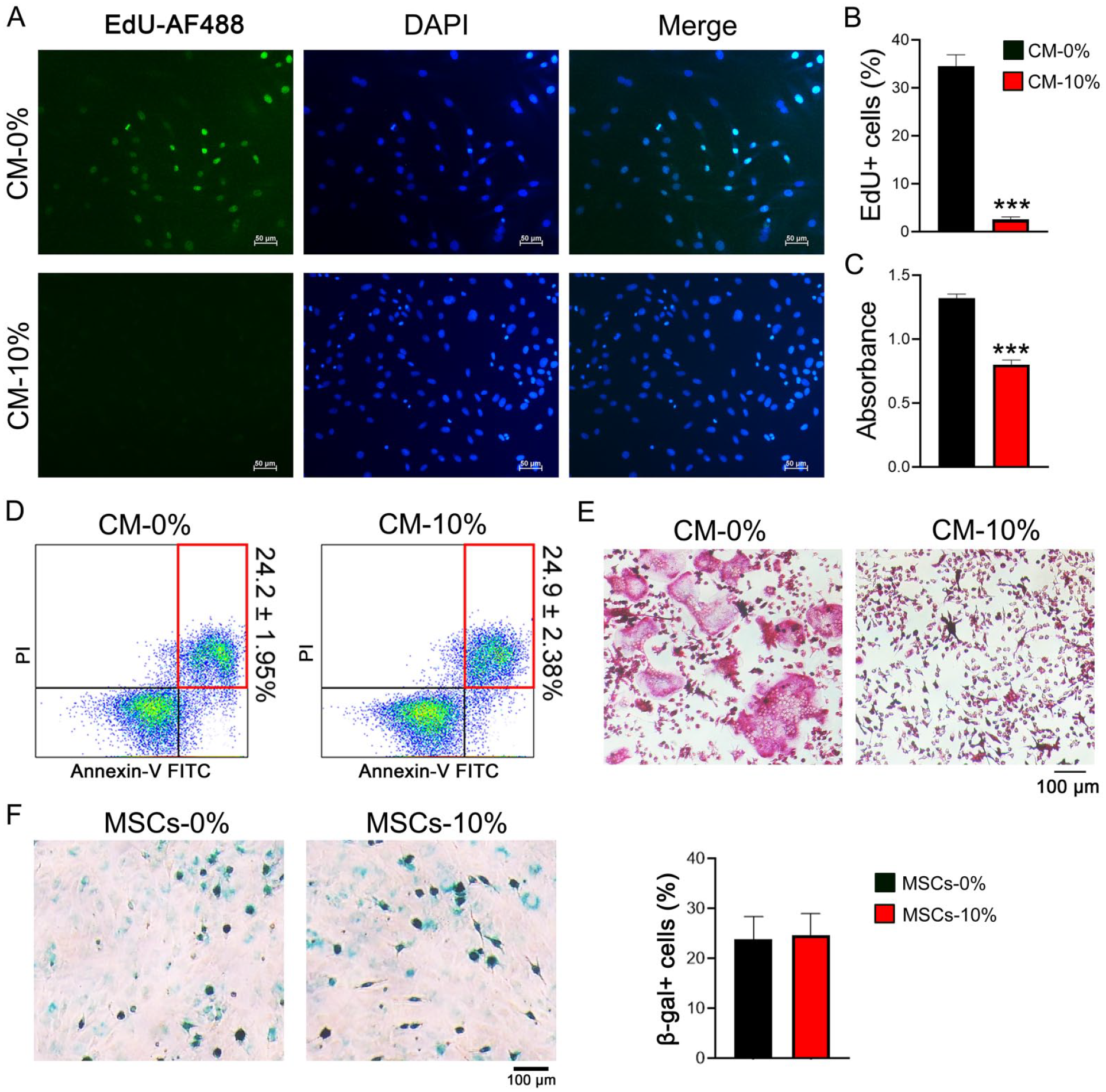
CM of 10% CMS-treated MSCs inhibited monocyte proliferation and osteoclastic differentiation. (A) Representative images of the EdU cooperation assay showed the proliferation rate of BM monocytes under the treatment of MSC-CM. (B) Quantification of EdU-positive cells presented as a percentage of total cells per field of view. (C) CCK8 assay showed the metabolic activity of monocytes under the treatment of MSC-CM. (D) Flow cytometry analysis of apoptotic cells treated by MSC-CM. (E) Representative images of TRAP staining of osteoclasts after 5 days of differentiation under the treatment of MSC-CM. (F) Representative images of SA-β-gal staining of MSCs 24 h after CMS application. Quantification data were presented as a percentage of β-gal-positive cells to total cells per field of view. The statistical significance was assessed by two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001 versus the respective nc group. Data are means ± SD of three independent experiments. CM: conditioned medium; CMS: cyclic mechanical stretch; MSC: mesenchymal stem cell; BM: bone marrow; TRAP: tartrate-resistant acid phosphatase; SA-β-gal: senescence-associated beta-galactosidase.
Transplantation of 10% CMS-Primed MSCs Ameliorated HU-Induced Bone Loss
As the inflammatory BM microenvironment and the enhanced bone resorption are partly due to functional changes of MSCs under mechanical-unloading conditions, we next tested the possibility of transplantation of CMS-primed MSCs as a therapeutic strategy for disuse-induced osteoporosis. We first detected how long CMS-induced phenotypic changes can last. To do this, MSCs were cultured in a static state after 24 h of 10% CMS application before the expression levels of osteogenic genes and cytokines were detected at different time points. No significant changes were noticed within 3 days after 10% CMS treatment (Fig. 5A). Similarly, CM collected within the first 3 days still significantly inhibited osteoclastic differentiation (Fig. 5B), suggesting that the effect of CMS on MSCs can last at least for 3 days.
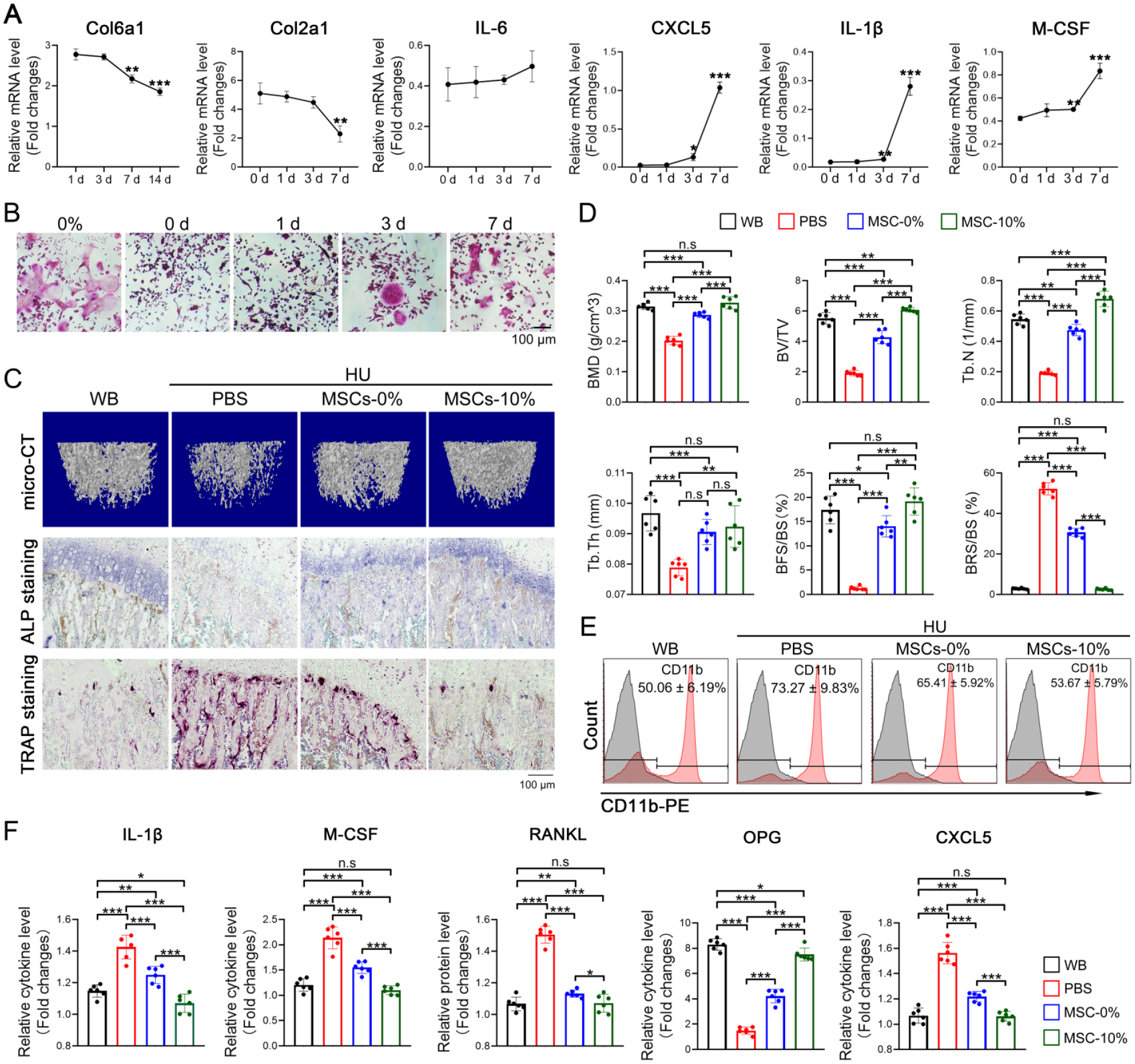
Transplantation of CMS-treated MSCs effectively rescued bone resorption and BM inflammation under mechanical-unloading conditions. (A) The expression of genes was tested by qRT-PCR at the indicated time points after CMS application for 24 h. (B) Representative images of TRAP staining of osteoclasts. The culture medium was changed after 24 h of CMS application, and MSCs were cultured in a static state for the indicated time point before the conditioned medium was collected for the treatment of osteoclast differentiation. (C) Representative images of bone histomorphology analysis of HU rats as well as WB controls. micro-CT was performed on femurs 21 days after suspension, and ALP and TRAP staining was performed on tibia sections 7 days after suspension. (D) Quantification (BMD, BV/TV, Tb.N, and Tb.Th) of three-dimensional microstructural parameters from micro-CT scanned femur. Quantification data of ALP and TRAP staining were presented as a percentage of BFS and BRS, respectively, to total bone areas (BS). n = 6 per group. (E) Flow cytometry analysis of the percentage of CD11b+ cells in the BM of rats treated as indicated. n = 6 per group. (F) ELISA showed the protein levels of cytokines in the BM fluid of rats treated as indicated. n = 6 per group. The statistical significance was assessed by two-tailed Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001. Data are means ± SD. Gene expression levels were normalized to GAPDH. CMS: cyclic mechanical stretch; MSC: mesenchymal stem cell; BM: bone marrow; qRT-PCR: quantitative real-time polymerase chain reaction; TRAP: tartrate-resistant acid phosphatase; HU: hindlimb unloading; ALP: alkaline phosphatase; BMD: bone mineral density; BV/TV: bone volume versus total volume; Tb.N: trabecular number; BFS: bone formation surface; BRS: bone resorption surface; BS: bone surface; ELISA: enzyme-linked immunosorbent assay; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Next, MSCs either treated with 10% CMS for 24 h (MSCs-10%) or cultured in a static state (MSCs-0%) were transplanted into rats by tail vein injection and started HU immediately. Both MSCs-10% and MSCs-0% were transplanted every 3 days. Twenty-one days after tail suspension, micro-computed tomography (CT) analysis revealed that both MSCs-10% and MSCs-0% transplantation effectively alleviated bone loss, while MSCs-10% almost totally reversed the bone loss phenotype (Fig. 5C, D). Likewise, bone remodeling was rebalanced, as indicated by enhanced bone formation, and simultaneously inhibited bone resorption (Fig. 5C, D). Flow cytometry analysis revealed that CD11b+ cell number (Fig. 5E) and inflammatory cytokine levels deceased after MSCs-0% transplantation (Fig. 5F), which were reversed to an extent almost equivalent to WB controls by MSCs-10% (Fig. 5E, F). Surprisingly, the levels of RANKL and OPG in the BM fluid were also restored (Fig. 5E), even though CMS did not significantly change their expressions in MSCs. These results confirmed that CMS-primed MSCs were more effective in improving the BM microenvironment and rebalancing bone remodeling under mechanical unloading-conditions, which suggested a promising stem cell-based therapy for disuse-induced osteoporosis.
Discussion
Bone constantly undergoes modeling and remodeling by responding to mechanical stimuli for the adaptation to mechanical loading7–10. However, this mechanically sensitive character leads to the loss of bone mass and decreased BMD under pathological conditions linked to mechanical unloading, such as long-term bedridden and aging7–10. Osteocytes in the canaliculi within the bone matrix are considered to be the main mechanosensory cells in bone11,12, which respond to the fluid flow shear stress in the lacuna-canalicular generated by mechanical load with increased secretion of bone anabolic factors, such as Wnts, OPG, TGF-β1, and so on13–15. Inadequate mechanical load leads to a decrease of these factors with the increased synthesis and secretion of RANKL and sclerostin16,17. Connexin 4326,27, cytoskeleton 28 , extracellular matrix 29 , and mechanical sensitive signals FAK and ErK30,31 were reported as the primary mechanisms for the cellular response to mechanical stimuli. Recently, the mechano-gated ion channel Piezo1 has been found to play a critical role in mediating mechanical signals on bone remodeling 18 . Lack of Piezo1 in MSCs, osteocytes, and osteoblasts leads to similar pathological phenotypes of mechanical unloading18–20, demonstrating that osteo-lineage cells, not just osteocytes, are mechanosensitive and contribute to mechanical-mediated bone remodeling.
MSCs have been paid great attention in the study of bone physiology. Like many types of stem cells, biological functions, such as proliferation and osteogenic differentiation are regulated by their niche, which is composed of niche cells, extracellular matrix, and soluble niche factors32,33. As mechanical strength also plays a critical role in regulating osteogenic efficacy, the mechanical microenvironment of MSCs cannot be neglected. However, the spatial location of MSCs in the BM has not yet been revealed; what kind of mechanical force regulates in vivo MSC activity and how MSCs sense mechanical signals need further investigation. It is reported that a specific MSC subset located at the mechanosensitive peri-arteriolar niche is responsible for bone formation 21 , which undergoes osteogenic differentiation by sensing the mechanical stretch transmitted from the arteriolar wall. This study suggests that the BM mechanical microenvironment generated by gravity load and exercise comprises multiple mechanical forces. However, there is currently no feasible model to calculate the strength of each mechanical force and to assess their combined effect on bone remodeling 34 .
In this research, we found that the remodeling balance is shifted to favor bone resorption at the early stage of mechanical unloading before bone loss phenotypes are manifested. Compared with the decrease in bone formation, the studies on bone resorption under such conditions were relatively few. Till now, the enhanced bone resorption under microgravity is attributed to regulatory factors (RANKL, OPG, sclerostin, etc) in the BM16–18. Here, we found that mechanical unloading significantly changed the BM microenvironment with increased myeloid cells and inflammatory cytokines. The inflammatory BM is advantageous in myeloid cell proliferation, BM homing, osteoclastic differentiation, and bone resorption35,36. Meanwhile, CD11b+ cells were also elevated in peripheral blood, indicating systemic inflammation, possibly leading to pathological changes in other tissues or organs. Nonetheless, this study demonstrated that mechanical unloading generates inflammation, which may render the BM microenvironment favorable for bone resorption instead of bone formation.
What needs to be addressed is the reason that causes BM inflammation. The biased differentiation of HSCs and the promoted proliferation of myeloid lineage are the possible underlying mechanisms 37 . Whether the hematopoietic system is mechanosensitive and is guided to favor myeloid differentiation under microgravity needs to be illustrated. However, as the primary niche cell of HSCs, mechanosensitive MSCs may be responsible for the expansion of inflammatory cells38,39. We noticed that the concentration of niche factor CXCL1222,23 secreted by MSCs is decreased, which, together with other reports 34 , indicates that mechanical unloading indirectly affects the hematopoietic system by deteriorating the regulatory functions of MSCs. The phenotype of Piezo1 conditional KO mice further proved this hypothesis. Consistent with other reports, Piezo1-KO mice exhibited bone loss and low bone density, with increased bone resorption. As expected, the BM microenvironment of KO mice is also characterized by increased myeloid cells and greatly elevated inflammatory cytokines.
MSCs exert essential regulatory effects on the local microenvironment by secreting cytokines and extracellular vesicles40,41. The extent to which MSC paracrine activity regulates the BM microenvironment has not been illustrated. It is reported that the secretome of MSCs is altered by mechanical stimuli, such as substrate stiffness42,43. We also noticed that the most DEGs in 10% CMS-treated MSCs were inflammatory cytokines, indicating that the expansion of myeloid cells was due to, at least in part, the altered MSC secretomes under mechanical-unloading conditions. The CM of CMS-treated MSCs significantly inhibited monocyte proliferation and osteoclastic differentiation, further proving the MSC secretome’s essential role in maintaining bone remodeling balance. The functional changes of MSCs may trigger a series of chain reactions that finally lead to BM inflammation and homeostasis deterioration. Our result is consistent with the previous finding 44 , which reported that MSCs inhibited osteoclastic differentiation by expressing non-coding RNAs transported by exosomes to monocytes after mechanical stimuli.
In summary, mechanical stimuli are essential for MSC proliferation, osteogenic differentiation, and the secretion of regulatory cytokines and extracellular vesicles that are indispensable for maintaining the phycological BM microenvironment. Mechanical unloading shifts the MSC secretome to favor inflammation, leading to myeloid proliferation, migration, and osteoclastic differentiation. As a result, bone formation is inhibited, while bone resorption is extensively promoted.
This study indicated that MSCs also work as a coupling regulator for maintaining bone remodeling balance. As this capability is hampered under mechanical-unloading conditions, we hypothesized that transplanting CMS-primed MSCs might be a promising therapeutic strategy. As expected, CMS-treated MSCs are more effective in restoring the BM microenvironment, decreasing the myeloid cells and inflammatory cytokines, and rebalancing bone remodeling in unloaded rats. Interestingly, MSCs with or without CMS treatment significantly promoted bone formation, indicating that MSC possesses the capability of treatment, which was enhanced by mechanical stimulation.
However, there are some limitations of this study. First, as we noticed that the immune microenvironment of the HU model was disrupted, we failed to investigate the possible changes in intrinsic HSCs. It is reported that lymphopoiesis and bacterial clearance were severely affected in the HU model 21 , which further proved that the hematopoietic system is also sensitive directly or indirectly to mechanical load. The underlying mechanisms and the possible role of MSCs in regulating HSCs under such circumstances need a lot more investigation. Second, we found that MSC transplantation was an effective way to prevent disuse-induced bone loss. However, the biological activities of transplanted MSCs were not investigated. Whether the improved bone formation was due to the direct contribution of osteoblastic differentiation of transplanted MSCs or indirectly through regulating BM microenvironment cannot be confirmed in this study. These concerns need to be addressed before the clinical usage of MSCs for disuse-induced bone loss.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241236584 – Supplemental material for Mechanical Unloading Promotes Osteoclastic Differentiation and Bone Resorption by Modulating the MSC Secretome to Favor Inflammation
Supplemental material, sj-docx-1-cll-10.1177_09636897241236584 for Mechanical Unloading Promotes Osteoclastic Differentiation and Bone Resorption by Modulating the MSC Secretome to Favor Inflammation by Wanyuji Wang, Xueling Zheng, Hehe Wang, Bin Zuo, Sisi Chen and Jiao Li in Cell Transplantation
Supplemental Material
sj-docx-2-cll-10.1177_09636897241236584 – Supplemental material for Mechanical Unloading Promotes Osteoclastic Differentiation and Bone Resorption by Modulating the MSC Secretome to Favor Inflammation
Supplemental material, sj-docx-2-cll-10.1177_09636897241236584 for Mechanical Unloading Promotes Osteoclastic Differentiation and Bone Resorption by Modulating the MSC Secretome to Favor Inflammation by Wanyuji Wang, Xueling Zheng, Hehe Wang, Bin Zuo, Sisi Chen and Jiao Li in Cell Transplantation
Footnotes
Author Contributions
Conceptualization, J.L.; methodology, J.L. and W.W.; validation, J.L. and B.Z.; formal analysis, H.W. and X.Z.; data curation, W.W., H.W., and X.Z.; writing–review and editing, J.L. and B.Z. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
All data included in this study are available upon request by contacting the corresponding author.
Ethical Approval
This study was approved by our institutional review board (approval no. ZMU21-2303-048).
Statement of Human and Animal Rights
All of the experimental procedures used in this study were conducted in accordance with the Experimental Animal Ethics Committee of Zunyi Medical University, Guizhou Province, China.
Statement of Informed Consent
This article did not involve human subject research and informed consent was not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by grants from the National Natural Science Foundation of China (grant no. 82060266).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
