Abstract
To overcome the difficulty of vascular regeneration in exposed tendon wounds, we combined human placenta-derived mesenchymal stem cells (hPMSCs) with an artificial dermal scaffold and assessed their role in promoting vascular regeneration and wound healing in vivo. hPMSCs were isolated from the human placenta and characterized based on their morphology, phenotypic profiles, and pluripotency. New Zealand rabbits were used to establish an exposed tendon wound model, and hPMSCs and artificial dermal scaffolds were transplanted into the wounds. The results of gross wound observations and pathological sections showed that hPMSCs combined with artificial dermal scaffold transplantation increased the vascularization area of the wound, promoted wound healing, and increased the survival rate of autologous skin transplantation. Following artificial dermal scaffold transplantation, hPMSCs accelerated the vascularization of the dermal scaffold, and the number of fibroblasts, collagen fibers, and neovascularization in the dermal scaffold after 1 week were much higher than those in the control group. Immunohistochemical staining further confirmed that the expression of the vascular endothelial cell marker, CD31, was significantly higher in the combined transplantation group than in the dermal scaffold transplantation group. Our findings demonstrated that hPMSCs seeded onto artificial dermal scaffold could facilitate vascularization of the dermal scaffold and improve tendon-exposed wound healing.
Keywords
Introduction
Exposed tendon wounds frequently occur following extensive surgery, deep burns, trauma, or chronic ulcers. Tendon-exposed wounds are common at joint connections and full-thickness skin defects1,2. Traditional treatments involve skin grafting and flap surgery to close wound surfaces. However, these treatments have several limitations, such as donor skin shortage, tendon adhesion, donor site morbidities and patient discomfort3–5. The repair of exposed tendon wounds and their function remains a crucial subject in the surgical field.
Biosynthetic skin substitutes have been an important treatment option for restoring skin function by reducing scar hyperplasia, inhibiting wound contraction, and reconstructing the skin structure 6 . The Lando® dermal scaffold is a double-layered artificial dermis skin structure. The upper layer is a semi-transparent medical silicone rubber layer made of polydimethylsiloxane. The lower layer is a crosslinked wet collagen layer composed of type I collagen, chondroitin sulfate, and phosphate buffer 7 . This product has been widely used in China to repair deep burns, traumatic full-thickness skin defects, and noninfectious wounds. Owing to the difficulty of scaffold fixation, long vascularization time, and high rate of local infection in tendon wounds, the survival rate of artificial dermal scaffolds remains low. Mesenchymal stem cell (MSC)-based therapies have been widely used recently in tissue engineering and regenerative medicine8–10. MSCs accelerate wound healing by inhibiting inflammation, facilitating angiogenesis, and promoting epithelialization11,12.
MSCs are multipotent, self-renewing stem cells isolated from various sources. The placenta, as an enriched vascular tissue, has attracted increasing attention as a source of MSCs for therapeutic angiogenesis 13 . Human placenta-derived mesenchymal stem cells (hPMSCs) secrete higher levels of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) and possess a higher endothelial differentiation ability than that of other MSC sources. The beneficial effects of hPMSCs on tendon-exposed wounds may be mediated by promoting granulation tissue and accelerating neovascularization 14 . We hypothesized that the combined application of hPMSCs and artificial dermal scaffolds would promote the survival rate of the artificial dermal scaffolds and accelerate the healing of tendon-exposed wounds.
In this study, we isolated hPMSCs from the human placenta and characterized their morphology, phenotypic profile, and pluripotency. We then explored the growth characteristics of hPMSCs on artificial dermal scaffolds. To test this hypothesis, we used an established model of exposed tendon wounds in New Zealand rabbits to evaluate the role of hPMSCs combined with an artificial dermal scaffold in promoting vascular regeneration and wound healing in vivo.
Materials and Methods
Culture and Identification of hPMSCs
The hPMSCs were sponsored by the Beijing Health & Biotech Corporation. hPMSCs were cultured in MSC basal medium containing MSC additives at 37°C and 5% CO2. hPMSCs were identified according to the standard criteria 15 and used on passages 3–5 for experiments.
Passage 3–5 hPMSCs were seeded at a density of 1 × 105 cells/well in a six-well plate. When the cells reached 100% confluence, hPMSCs were differentiated into osteogenic and adipogenic lineages using a differentiation medium kit (Cyagen, Guangzhou, China), according to the manufacturer’s instructions. Osteoblasts and adipocytes were identified after 3 weeks using alizarin red S, von Kossa, and oil red O staining, as previously described16,17.
Characterization experiments were performed by incubating hPMSCs with monoclonal phycoerythrin (PE)-conjugated antibodies against HLA-DR, CD13, CD34, CD45, CD90, CD105 (Biolegend, San Diego, CA, USA), CD44, and CD73 (BD Pharmingen, San Diego, CA, USA) for 20 min at 22–25°C. After washing twice, the cells were resuspended in 350 µl of phosphate-buffered saline (PBS). Cell fluorescence was evaluated using a BD LSRFortessa flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) and data were analyzed using FlowJo software.
Assessing the Characteristics of hPMSCs on Artificial Dermal Scaffold
To visualize the growth and distribution of hPMSCs on the artificial dermal scaffolds, lentiviruses expressing GFP (H1-MCS-CMV-EGFP, Shanghai Jikai Jiyin Medical Technology Co., Ltd., China) were transfected into hPMSCs. After 3 days of hPMSCs culture on the artificial dermal scaffold, growth characteristics were observed by fluorescence microscopy and scanning electron microscopy (SEM). The fluorescence of the hPMSCs on the artificial dermal scaffold was measured using an inverted fluorescent microscope (ZEISS Axio Vert.A1, Germany) with 488 nm lasers for excitation. Their ultrastructure was examined by scanning electron microscopy (SEM). The samples for SEM examination (HITACHI SU8100, Japan) were fixed with 2.5% glutaraldehyde solution and sputter-coated with gold. To assess the cell proliferation of hPMSCs on artificial dermal scaffold, the Cell Counting Kit-8 reagent was used. The artificial dermal scaffold was cut using a 6 mm diameter puncher and placed inside a 96-well plate. The hPMSCs (1 × 106 cells/cm2) were seeded into each well. After incubation, 10 µl of Cell Counting Kit-8 reagent (CCK-8, GLPBIO, Montclair, CA, USA) was added to each well, and the plates were cultured for 2 h. Cell viability was determined by measuring the absorbance at a wavelength of 450 nm. The experiment was repeated three times for seven consecutive days.
Animals
All experimental procedures were approved by the Animal Ethics and Welfare Committee of Jinan Central Hospital (approval no. JNCH2022-001). Eighteen skeletally mature New Zealand white rabbits of either sex (16 weeks, 2.2–2.5 kg) were used for this study. The animals were maintained under controlled conditions: temperature 24 ± 1°C, 45% relative humidity, air changes per hour, and a light/dark rhythm of 12 h 1 . The animals were single-housed with ad libitum access to food, water, environmental and food enrichment, and visual access to other animals 18 . Prior to surgery, all animals were acclimatized to their environment for 1 week.
Surgical Procedures
New Zealand white rabbits were deprived of water and food for 8 h before surgery, and their weights were recorded. For all surgical procedures, rabbits were anesthetized with a marginal ear vein injection of 3% pentobarbital sodium (3 ml/kg body mass). All intervention groups underwent tenotomy. The Achilles tendon area above the ankle joints of both hind limbs was selected as the surgical area, and the left and right legs were shaved and sterilized. Tendon-exposed wounds were then sliced 3.0 cm above the calcaneus by removing the cutis, subcutis, and fascia. The area of each tendon-exposed wound was 2.0 cm × 2.0 cm (Fig. 1A). After hemostasis, each wound was washed thoroughly with sterile saline solution, and the fascia and interrupted skin were sutured using a 4-0 braided silk suture1,18.
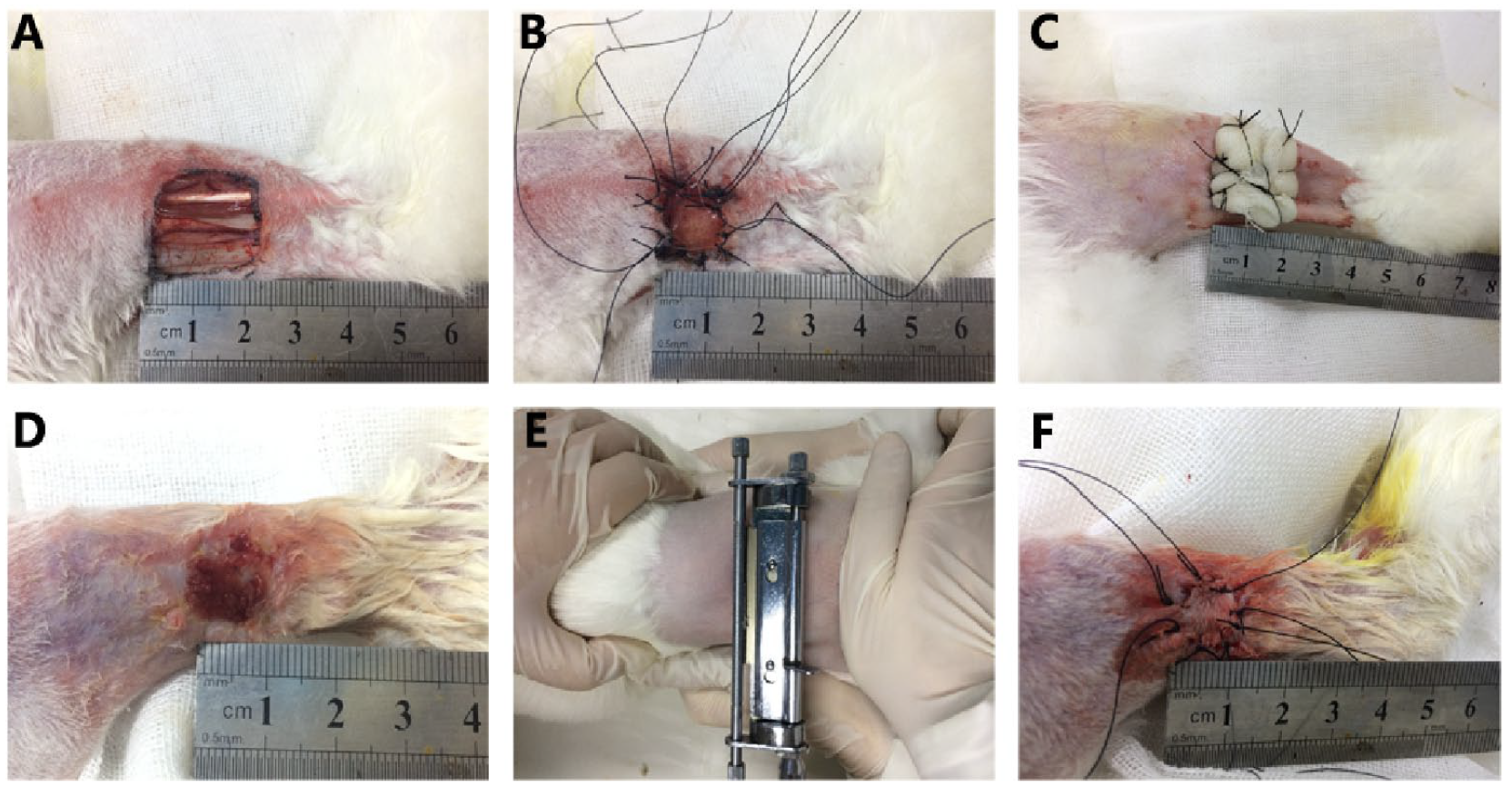
Model preparation and treatment process. (A) Tendon-exposed wound preparation. (B) Artificial dermal scaffold or dermal scaffold with human placenta-derived mesenchymal stem cells placed to cover the wounds. (C) Surface of the dermal scaffolds covered with Vaseline gauze. (D) Gross examination. (E) Autograft harvested from the rabbits’ dorsum as split-thickness skin grafts with a roller knife. (F) Autologous skin transplantation.
Treatment Groups
Each rabbit was assigned an ID number, and randomized into two study groups. Although the researchers were aware of the allocation during the surgeries and tissue harvest, the specimens were identified only by their ID number. Eighteen rabbits were randomly divided into two groups. The control group rabbits received artificial dermal scaffold (Lando®, Shenzhen Qikang Medical Equipment Co., Shenzhen, China) without hPMSCs, whereas the experimental group rabbits received a combined application of hPMSCs and artificial dermal scaffold.
In the experimental group, hPMSCs (1 × 107 cells/ml) were uniformly injected into the artificial dermal scaffold at a density of 1 × 106 cells/cm2 and left to stand for 5 min to allow full absorption. The dermal scaffold with cells was placed upward to cover the wounds with a silicone membrane by interrupted suturing (Fig. 1B). The surfaces of the dermal scaffolds were then covered with Vaseline gauze (Fig. 1C). A tie-over dressing with sterile gauze was used in the experimental group. Finally, the tendon-exposed wounds were covered with sterile gauze and wrapped with bandages for compression 7 . After 2 weeks, the wounds were grafted with 2 cm × 2 cm of autogenous split-thickness skin (Fig. 1D–F). The autograft was harvested from the dorsum of the rabbits using a roller knife 19 . The tendon-exposed wounds in the control group were treated with an artificial dermal scaffold without cells. All the other steps were similar to those used in the experimental group. The rabbits were then returned to their cages for routine care.
Wound Closure Measurements
Digital photographs of the wounds were taken at 7, 14, and 21 days after treatment. Wound closure, survival of the dermal scaffold, degree of vascularization, and autologous skin growth were observed and analyzed.
Histological Analysis
On days 7 and 21 following treatment, the rabbits were euthanized under deep anesthesia with sufficient pentobarbital. New granulation and skin tissue from the wounds were collected and fixed in 4% paraformaldehyde at 4°C for 48 h. After dehydration and embedding, tissue sections were cut into 3-μm transverse sections, and processed for hematoxylin and eosin (H&E) and Masson staining. Images were captured using an automatic digital slide scanner (Pannoramic MIDI, 3DHISTECH, Hungary).
Immunohistochemistry
After de-paraffinizing with xylene and rehydrating the sections (descending gradient of ethanol), primary antibodies against CD31 (1:1,000, OriGene, USA) or human nuclear antibody (1:500, Millipore, USA) were added at 4°C overnight, followed by incubation with horseradish peroxidase-conjugated affinipure goat anti-mouse immunoglobulin G (IgG) for 1 h at room temperature, followed by colorimetric detection using DAB (3,3′-diaminobenzidine) substrate, according to the manufacturer’s protocol 20 . CD31-positive expression was used to determine the microvessel count. The slices were observed under a high-magnification optical microscope (10 visual fields/slice, ×100) for the presence of vascular structures. The ImageJ software was used to perform the measurements.
Statistical Analysis
SPSS 22.0 software (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. The data were checked for normality using the Kolmogorov–Smirnov and Shapiro–Wilk tests. Variance homogeneity was checked using Levene’s test. Because the data were normally distributed and had no significant difference in variances, a parametric one-way analysis of variance (ANOVA) was performed for differences between multiple groups, independent-sample t test was used for comparisons of two groups. Statistical significance was set at P < 0.05. Values are expressed as means ± standard deviation.
Results
Characterization of hPMSCs
In vitro, hPMSCs exhibited a fibroblast-like morphology (Fig. 2A) and differentiated into adipocytes and osteocytes, after 3 weeks of adipogenic and osteogenic induction (Fig. 2B–D). Flow cytometry showed that undifferentiated hPMSCs expressed high levels of CD13, CD44, CD73, CD90, and CD105 and were negative for CD34, CD45, and HLA-DR (Fig. 2E).
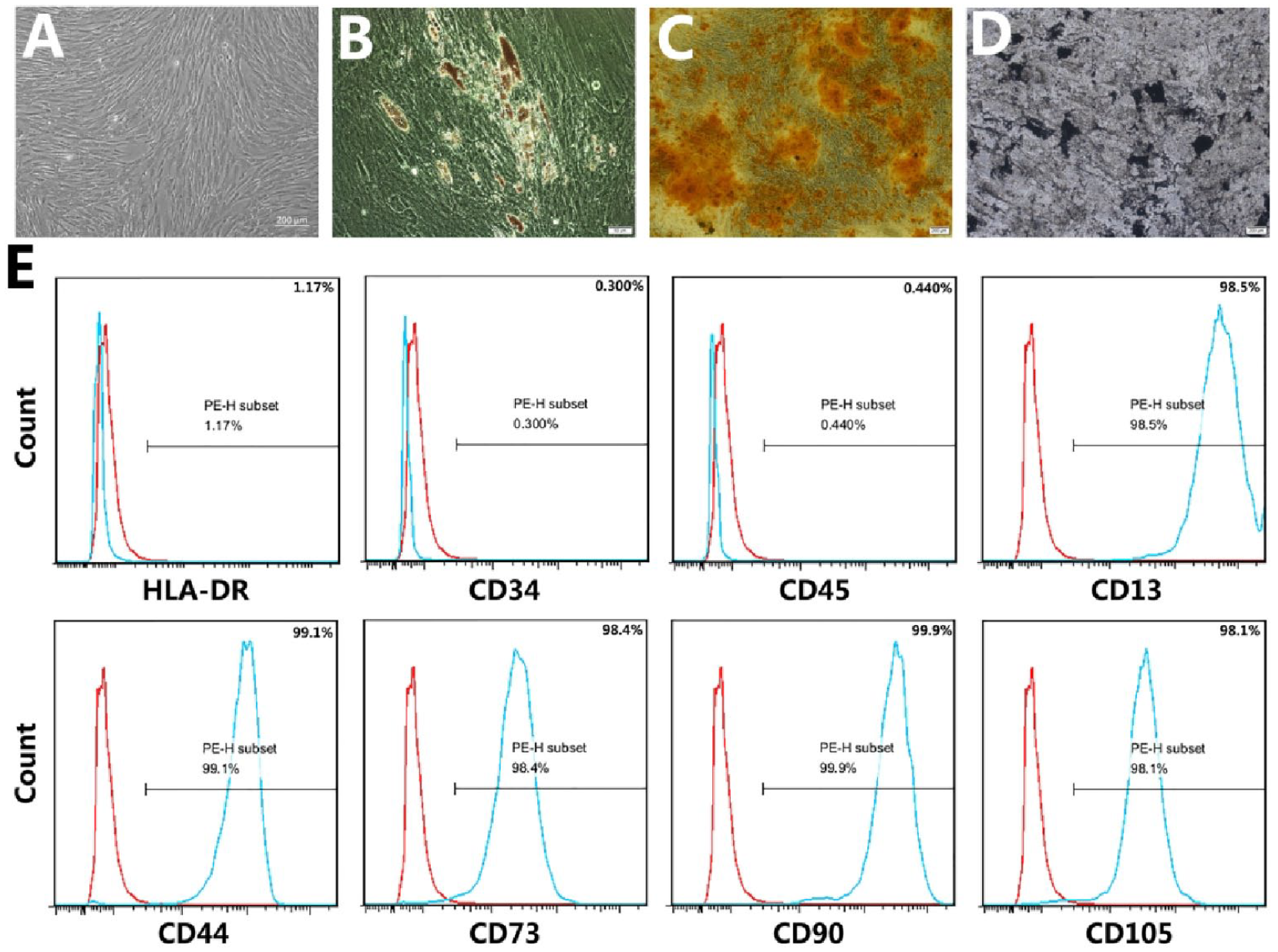
Characterization of hPMSCs. (A) Microscopic image showing spindle-shaped, third-passage hPMSCs. (B) Oil red O staining for adipocytes. (C) Alizarin red S staining for osteocytes. (D) Von Kossa staining for osteocytes. (E) hPMSCs characterization using flow cytometry showed high expression of CD13, CD44, CD73, CD90, and CD105 (positive) and very low expression of HLA-DR, CD34, and CD45 (negative) in the isolated hPMSCs. hPMSCs: human placenta-derived mesenchymal stem cells; PE-H: Phycoerythrin-Height.
Characteristics of hPMSCs on Artificial Dermal Scaffold
Fluorescence microscopy revealed that a large number of hPMSCs were attached to the artificial dermal scaffold as fibroblast-like cells (Fig. 3D), indicating that the scaffold has the potential to deliver a considerable number of hPMSCs. SEM imaging showed that the hPMSCs were consistently attached to the dermal scaffold (Fig. 3F). Analysis of hPMSCs proliferation on the artificial dermal scaffold revealed that the cells gradually expanded for 5 days and no growth was observed on the sixth and seventh days (Fig. 3G). These results indicate that the artificial dermal scaffold is suitable for hPMSCs proliferation.

Characterization of hPMSCs on artificial dermal scaffold. (A) Autofluorescence of artificial dermal scaffold. (B) Light microscopy of artificial dermal scaffold. (C) Ultrastructures of artificial dermal scaffolds were examined under scanning electron microscopy. (D) Fluorescence microscopy showing large numbers of hPMSCs attached to the artificial dermal scaffold. (E) Light microscopy of hPMSCs on artificial dermal scaffold. (F) Scanning electron microscopy imaging showing hPMSCs consistently attached to the dermal scaffold. (G) CCK-8 assays were performed to evaluate the cellular growth curves. *P < 0.05, day 1 vs day 2; #P < 0.05, day 2 vs day 3; &P < 0.05, day 3 vs day 4. Scale bar: 200 μm (light microscopy and fluorescence microscopy). Scale bar: 20 μm (scanning electron microscopy). hPMSCs: human placenta-derived mesenchymal stem cells; CCK-8: Cell Counting Kit-8.
Gross Examination
The double-layer structure of the artificial dermis in the control group was not tightly attached to the exposed area of the tendon at 7 days after surgery, and the dermal scaffold in some areas was yellowish white and intact. On the 14th day, the double-layer structure of the artificial skin was partially separated, the dermal scaffold was yellowish-red, the scaffold in the exposed area of the tendon was depressed, and the granulation tissue was shallow (Fig. 4A). The wound was drier, and granulation tissue grew better and smoother, with no obvious swelling or redness of the skin around the wound in the experimental group. The granulation tissue increased gradually in the experimental group, the double-layer structure of the artificial skin remained close together, and the dermal scaffold was light red with a good structure but did not closely adhere to the wound bed tissue on day 7 after treatment (Fig. 4B). The double-layer structure of the artificial skin was completely separated, the dermal scaffold adhered closely to the wound bed tissue, and the fresh red granulation tissue almost filled the entire scaffold on day 14 after treatment 7 . Seven days after autologous skin transplantation, all autologous skin grafts in the experimental group survived, whereas the autologous skin grafts in the control group partially survived (Fig. 4).
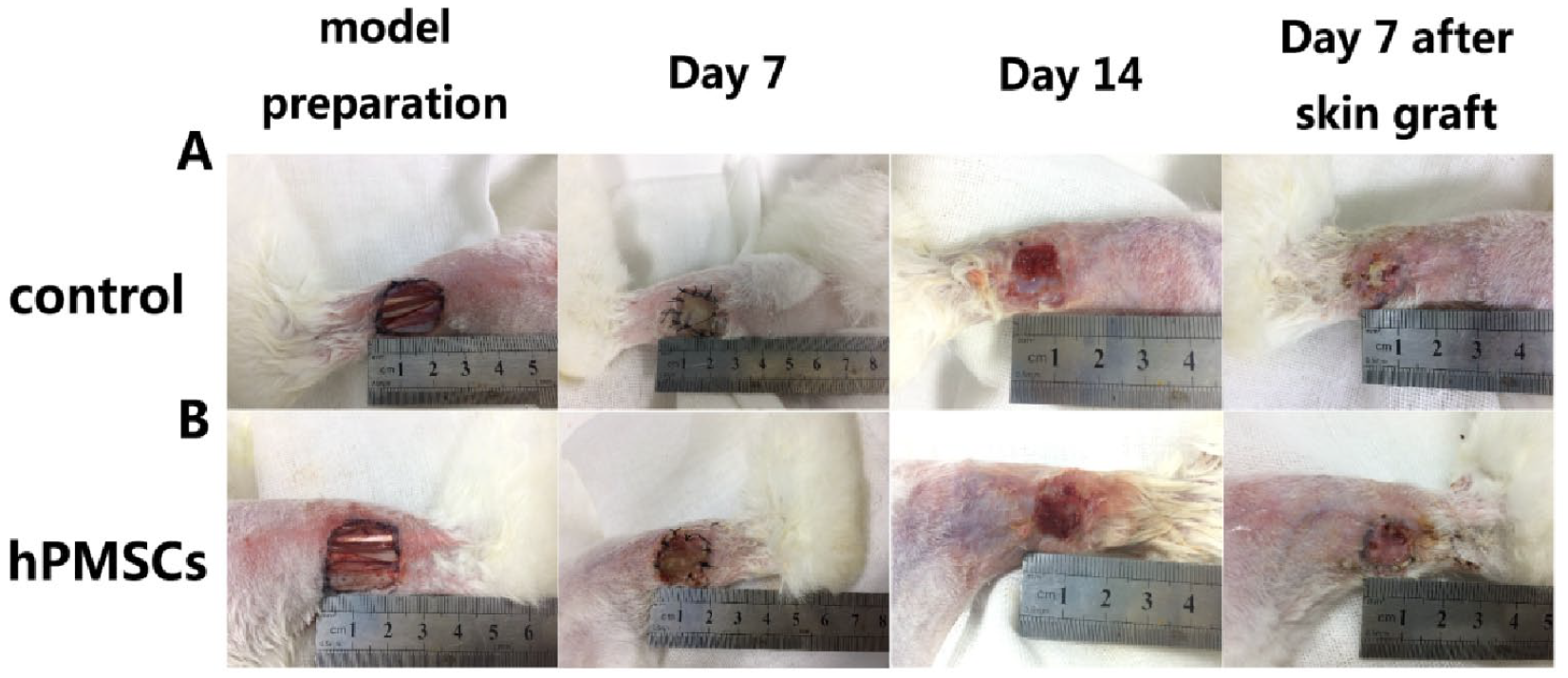
Rabbits were observed grossly at days 7 and 14 after injury, and day 7 after autologous skin transplantation. (A) Representative images of the gross examination in control group. (B) Combined application of hPMSCs and artificial dermal scaffold group. hPMSCs: human placenta-derived mesenchymal stem cells.
The vascularization area of the dermal scaffold was calculated 14 days after treatment. The results showed that the vascularization area of the experimental group was superior to that of the control group (70.27 ± 9.26% vs 41.81 ± 10.79%, P < 0.01) (Fig. 5A). After 3 weeks, the non-healing area of the experimental group was significantly smaller than that of the control group (0.087 ± 0.09 cm2 vs 0.30 ± 0.28 cm2, P = 0.027) (Fig. 5B). After autologous skin transplantation, the survival rates of autologous skin transplantation in the control and experimental groups were 27.8% and 61.1%, respectively (P = 0.04; Table 1).
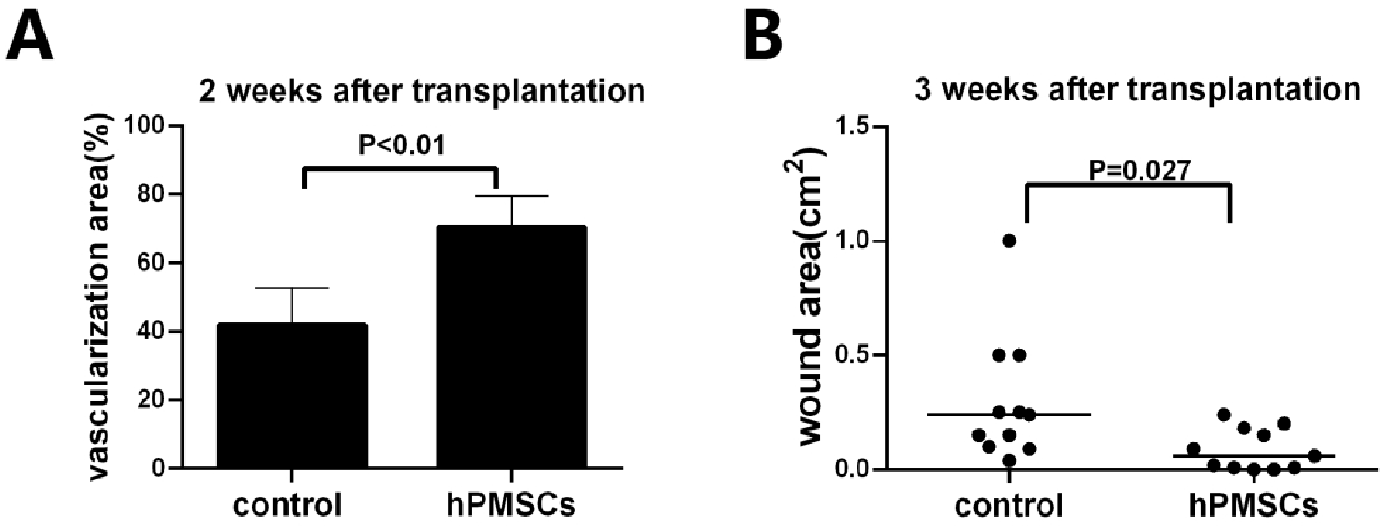
Combination of hPMSCs with artificial dermal scaffold facilitates vascularization degree of dermal scaffold and improves tendon-exposed wound healing. (A) The vascularization area of the wound was calculated 2 weeks after scaffold and cell transplantation. (B) hPMSCs improve tendon-exposed wound healing. Data are analyzed using independent-sample t test. hPMSCs: human placenta-derived mesenchymal stem cells.
Survival Rate of Autologous Skin Transplantation.

hPMSCs: human placenta-derived mesenchymal stem cells.
P = 0.04.
Histological Analysis
Histological analysis of wound tissues with H&E and Masson staining showed that the combined transplantation accelerated wound healing. The regenerated tissue in the experimental group was thicker than that in the control group on day 7 after treatment. Collagen fibers, fibroblasts, and neovascularization were observed inside the dermal scaffold pores (Fig. 6).
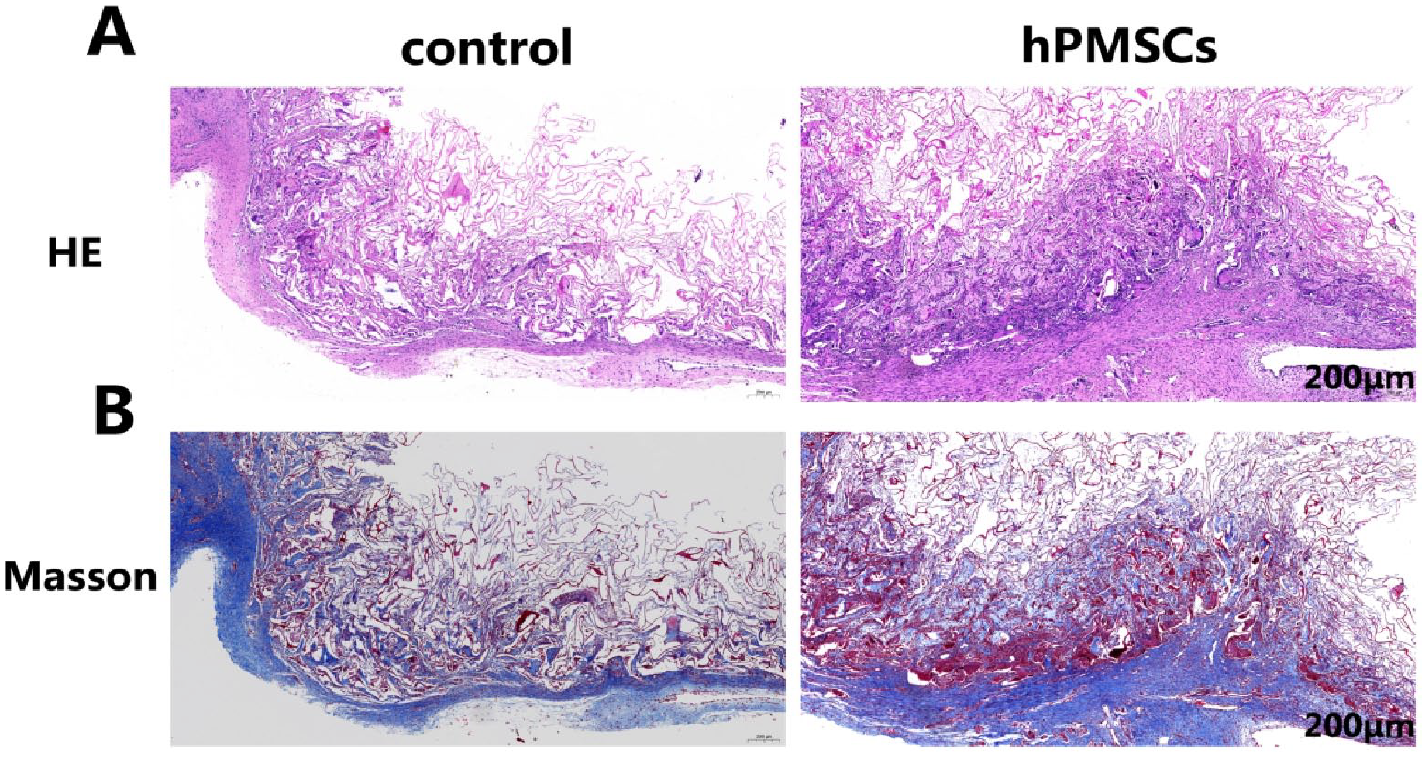
Histological analysis of wound tissues with H&E staining and Masson staining reveals the beneficial effect of hPMSCs on wound healing. (A) Representative images of wound sections with H&E staining on day 7 after dermal scaffold transplantation. (B) Representative images of wound sections with Masson staining (scale bar: 200 μm). H&E: hematoxylin and eosin; hPMSCs: human placenta-derived mesenchymal stem cells.
One week after autologous skin transplantation, H&E and Masson staining showed that parts of the dermal scaffolds in the control group were not covered by autologous skin, and the autologous skin grafts survived partially, whereas the autologous skin in the hPMSC-treated group almost survived on the scaffold (Fig. 7). In the experimental group, more nail protrusions, capillaries, and collagen fiber depositions were observed. The arrangement of fibroblasts and collagen was more regular (Fig. 7B) than in the control group.
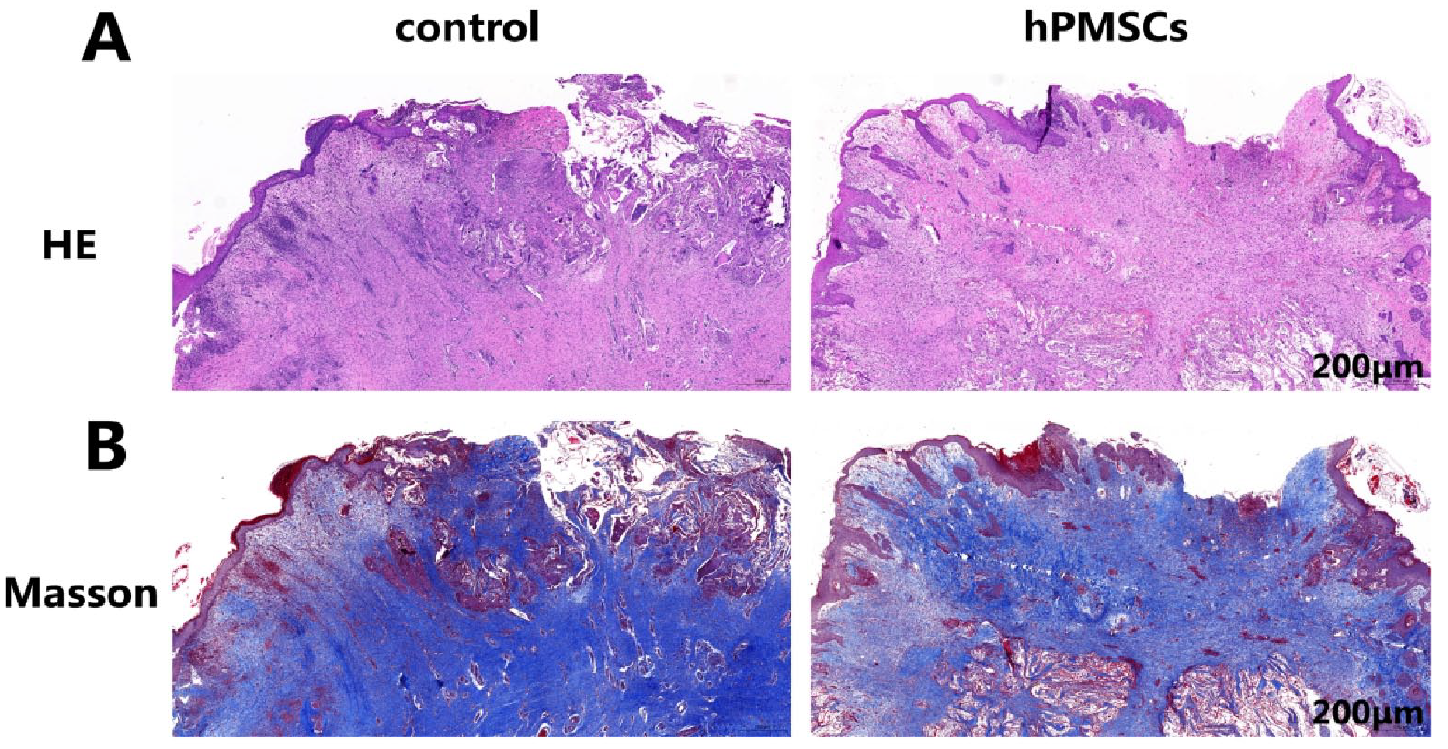
Histological analysis of wound tissues showing the beneficial effect of hPMSCs on autologous skin survival. (A) Representative images of wound sections with H&E staining on day 7 after autologous skin transplantation. (B) Representative images of wound sections with Masson staining (scale bar: 200 μm). hPMSCs: human placenta-derived mesenchymal stem cells; H&E: hematoxylin and eosin.
Expression of CD31 and Human Nuclei
Paraffin sections of skin lesions from autologous skin transplantation for 1 week were obtained, and endothelial cell CD31 and human nuclei were stained using immunohistochemistry. Vascular endothelial cells were marked by CD31 to confirm the degree of tissue neovascularization 21 . The results showed that CD31 was mainly expressed in the cytoplasm of microvascular endothelial cells, which were stained with brownish-yellow or brown particles (Fig. 8A). The degree of neovascularization with positive CD31 expression 1 week after autologous skin transplantation was higher in the experimental group than in the control group (P < 0.05) (Fig. 8B). Simultaneously, immunohistochemical staining of human nuclear antigens occasionally showed brown-yellow nuclei in the experimental group but not in the control group (Fig. 8A), indicating that hPMSCs can survive in the lesions.
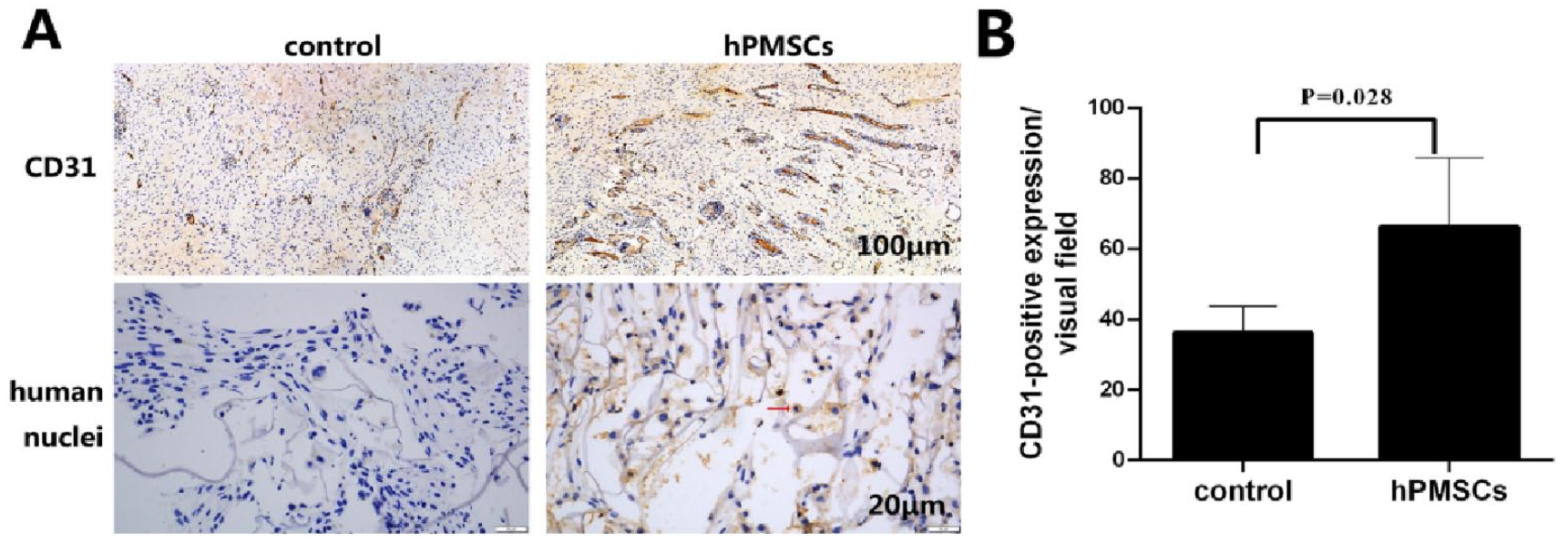
Expression of CD31 and human nuclei. (A) Paraffin sections of skin lesions from autologous skin transplantation for 1 week were obtained, and the endothelial cells CD31 and human nuclei were stained by immunohistochemistry. Scale bars: 100 μm (upper row), 20 μm (lower row). (B) Quantification of CD31-positive expression. hPMSCs: human placenta-derived mesenchymal stem cells.
Discussion
Prolonged vascularization, infections, and scar formation are clinical issues associated with exposed tendon wounds that negatively impact therapeutic efforts22–24. The clinical application of dermal substitutes was satisfactory, and composite transplantation with autologous split-thickness skin grafts can overcome defects in skin contracture and tendon adhesion25–28. The artificial dermis is a tool for soft tissue reconstruction developed by Yannas and Burke in the late 1980s29,30. This bilayer matrix is a composite graft with an inner porous layer made of cross-linked bovine tendon type I collagen and chondroitin-6-sulfate glycosaminoglycan and an outer layer made of a thin, non-resorbable, semi-permeable polysiloxane (silicone sheet). The biodegradable, porous bovine collagen layer serves as a scaffold for cellular invasion and capillary growth and is usually replaced within 3 weeks in dermal wounds. The silicone layer plays a role similar to that of the epidermis; it provides mechanical support, increases the tear strength of the matrix, and prevents excessive loss of moisture and bacterial invasion during the early stages of wound healing 5 .
Classically, a double layer of artificial dermis is transplanted onto the wound site for 2–3 weeks until adequate vascularization occurs, followed by a skin graft for eventual wound healing3,31. The degree of vascularization of the artificial dermis is a determining factor for the success of transplantation. However, tendon-exposed wounds lack subcutaneous fat, muscle tissue, and vascular tissue 32 , which results in a poor vascularization rate and a long interval between the first implantation of the artificial dermis and autogenous skin grafting. Therefore, to overcome the difficulty of vascular regeneration in the process of wound repair using an artificial dermal scaffold, we combined MSCs with an artificial dermal scaffold to assess their role in promoting vascular regeneration and wound healing in vivo.
In this study, placenta-derived MSCs were isolated and identified based on their phenotype and differentiation ability. Flow cytometry results showed that PMSCs strongly expressed CD13, CD44, CD73, CD90, and CD105, but not CD34, CD45, or HLA-DR. After in vitro induction, PMSCs differentiated into adipocytes and osteocytes. The hPMSCs offer an encouraging option for the reconstruction of full-thickness skin wounds owing to their active proliferative potential, low immunogenic profile, anti-inflammatory function, tissue repair, and angiogenic ability14,33–35. Kong et al. 36 reported that the injection of hPMSCs enhanced microvessel formation in the skin wounds of diabetic rats, and these cells secreted proangiogenic molecules, including VEGF, hepatocyte growth factor (HGF), bFGF, transforming growth factor beta (TGF-β), and insulin-like growth factor-1 (IGF-1). König et al. 37 reported that the paracrine effects of conditioned medium from hPMSCs enhanced endothelial cell viability, migration, and tube formation and elevated the secretion of proangiogenic proteins, such as angiogenin, angiopoietin-1, angiopoietin-2, and VEGF. This study aimed to ascertain whether hPMSCs improved the degree of vascularization of the dermal scaffold and promoted wound healing.
The incorporation of MSCs into artificial dermal scaffolds provides a suitable niche for transplanted MSCs to enhance their survival and therapeutic effects. To date, two methods are available for the combined transplantation of stem cells and scaffolds. The first method involves inoculating stem cells onto scaffolds, culture them in vitro for a certain period, form a cell-scaffold complex, and then transplant them to the wound surface38,39. The other method involves the local combination of stem cells and scaffolds on the wound without in vitro culture2,40. Stem cells were injected or sprayed into the wound bed. The second transplantation method was employed in our study. Our results showed that the artificial dermal scaffold has the potential to deliver a considerable number of hPMSCs and sustain their proliferation. hPMSCs were dripped onto an artificial dermal scaffold, allowed to stand for 5 min, and then the cell scaffold was transplanted into the tendon-exposed wound. Three weeks after transplantation, that is, 1 week after autologous skin transplantation, immunohistochemical staining results showed that human nuclear antigen could be detected in the wound lesions, indicating that hPMSCs could grow and survive in the transplantation area. Our transplantation method can reduce the time and difficulty of stem cell culture in the laboratory and provides a feasible scheme for patients with exposed tendons to obtain the best surgical time.
To further investigate the role of hPMSCs in the degree of vascularization of artificial dermal scaffolds, we constructed a model of a tendon-exposed wound in New Zealand white rabbits and transplanted hPMSCs and artificial dermal scaffolds into the wounds. The results of gross wound observations and pathological sections showed that hPMSCs combined with artificial dermal scaffold transplantation increased the vascularization area of the wound, promoted wound healing, and increased the survival rate of autologous skin transplantation. One week after artificial dermal scaffold transplantation, hPMSCs accelerated the vascularization of the dermal scaffold, and the number of fibroblasts, collagen fibers, and neovascularization in the dermal scaffold were much higher than those in the control group. Immunohistochemical staining further confirmed that the expression of the vascular endothelial cell marker, CD31, was significantly higher in the combined transplantation group than in the dermal scaffold transplantation group.
In this study, we present a novel approach for the reconstruction of tendon-exposed wounds using a combination of hPMSCs and an artificial dermal scaffold, and assess their role in promoting vascular regeneration and wound healing in vivo. Our experimental treatment demonstrated that hPMSCs seeded onto artificial dermal scaffolds could facilitate the vascularization degree of the dermal scaffold and improve tendon-exposed wound healing. This study demonstrates a feasible treatment option for tendon-exposed wounds. However, the survival time of hPMSCs in dermal scaffolds and optimization of the combination of hPMSCs with artificial dermal scaffolds need further validation.
Footnotes
Acknowledgements
Author Contributions
FL performed the experiments, analyzed the data, and drafted the manuscript. CG, KZ, and GBH contributed to the animal model and obtained specimens. GDS devised the protocol and conducted the study. KZ cultured the cells and performed in vitro experiments. HL performed the histological examination of the animal tissues, participated in the statistical analysis, and reviewed the manuscript. All the authors have read and approved the final version of this manuscript.
Data Availability Statement
All the data are presented in the main manuscript. Data and materials related to this study are available from the corresponding author upon reasonable request.
Ethical Approval
All experimental procedures were approved by the Animal Ethics and Welfare Committee of Jinan Central Hospital (approval no. JNCH2022-001).
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the animal experimental guidelines issued by the Animal Ethics and Welfare Committee of Jinan Central Hospital.
Statement of Informed Consent
Human placental tissues were obtained from healthy volunteers who provided informed consent, and hPMSCs were provided by the Beijing Health & Biotech Corporation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Plan of the Jinan Municipal Health Commission (2023-1-11), the Natural Science Foundation of Shandong Province (grant no: ZR2023MH235), the Youth Training Program for High-level Projects of Jinan Central Hospital (202105004), and the Youth Science Fund and Training Program of Shandong First Medical University (202201-130).
