Abstract
Hypoxia is a crucial factor with type diversity that plays an important role in stem cell transplantation. However, the effects of hypoxia on adipose-derived stem cells (ADSCs) are largely unclear in the autologous fat transplantation (AFT) model, which shows a special type of “acute-progressively resolving hypoxia.” Here, an AFT model in nude mice and a hypoxic culture model for ADSCs were combined to explore the link between hypoxia-inducible factor-1 α subunit (HIF-1α) and mitophagy under hypoxic conditions. The results showed that the activity of ADSCs in the first 7 days after grafting was the key stage for volume retention, and the expression of HIF-1α, light chain 3 beta (LC3B), and Beclin1 in ADSCs increased during this period. We also found that hypoxia for longer than 48 h damaged the differentiation and mitochondrial respiration of ADSCs in vitro, but hypoxia signals also activate HIF-1α to initiate mitophagy and maintain the activities of ADSCs. Pre-enhancing mitophagy by rapamycin effectively improves mitochondrial respiration in ADSCs after grafting and ultimately improves AFT outcomes.
Keywords
Introduction
Autologous fat transplantation (AFT) is an emerging clinical treatment to reconstruct soft tissue contours with grafted adipose masses that are enriched in mature adipocytes and adipose-derived stem cells (ADSCs) 1 . Although several practical details have been improved, the primary barrier to broad clinical application is still the low final retention rate after grafting2,3. Studies have shown that in the early postgrafting phase, ischemia within the recipient area causes an acute hypoxic and nutrient-deprived microenvironment around the postgrafting adipose mass (PGAM) 4 . In this hypoxic environment, the majority of mature adipocytes die quickly, whereas ADSCs are more likely to survive and progressively contribute to subsequent tissue remodeling and vascularization 5 . Thus, enhancing the viability and survival of ADSCs in this acute, progressively resolving hypoxic microenvironment is a primary concern in AFT.
It has been hypothesized that the viability of ADSCs in hypoxic conditions is highly related to mitochondrial functional homeostasis6,7. Mitochondria, the organelles responsible for energy metabolism in mammalian cells, are extremely susceptible to and highly reliant on oxygen concentration to conduct physiological functions8,9. Studies have shown that mitochondria undergo different functional adaptations depending on the type of hypoxia10,11. On the one hand, the dramatic decrease in intracellular oxygen concentration inhibits oxidative phosphorylation (OXPHOS) in the mitochondria, resulting in a decrease in ATP production and, more importantly, high production of reactive oxygen species (ROS) in the cell, which can damage many organelles or macromolecules and ultimately threaten cell survival 12 . On the other hand, mitochondria can be degraded by mitophagy (a special process of autophagy that selectively targets mitochondria as substrates) under the persistent stress of hypoxia and nutrient deprivation to preserve mitochondrial functional homeostasis and prevent apoptosis13,14. Thus, it is necessary to establish multiple types of hypoxia models to simulate and observe the changes in mitochondria under different conditions.
Recent studies have shown that these adaptive changes in the mitochondrial response to oxygen concentration are dependent on the function of hypoxia-inducible factor-1 (HIF-1) 15 . HIF-1 is a heterodimeric transcription factor that consists of an oxygen-sensitive α subunit (HIF-1α) and a stably expressed β subunit (HIF-1β). In normoxia, HIF-1α is hydrolyzed by the ubiquitin‒proteasome system, whereas in a hypoxic environment, the activity of the ubiquitin‒proteasome is inhibited. Under hypoxia, HIF-1α can bind with HIF-1β to form functional HIF-1, which is transported into the nucleus and reactively binds to hypoxia-responsive elements to regulate a series of physiologic and pathophysiologic processes, such as the tricarboxylic acid cycle, angiogenesis, and mitophagy16,17. Although the mechanism of mitophagy and HIF-1α under hypoxic conditions has been validated in several disease models, the majority of these studies have focused on the change in mature cells under acute-transient ischemia‒reperfusion injury (represented by myocardial infarction models) or chronic progressively aggravating hypoxia (represented by solid tumor models)18–23. In contrast, few studies have focused on the changes in stem cell–derived mitochondria under acute, progressively resolving hypoxia, which can be observed well with the AFT model.
Based on the aforementioned context, we designed this experiment to explore the mechanism of mitophagy and HIF-1α under hypoxic conditions after fat transplantation and their impact on fat transplantation outcomes. In this study, an AFT model was established in nude mice to imitate the clinical pathogenic process of acute progressive resolution of hypoxia. The interactions between mitochondria and hypoxia-related molecules in ADSCs were then investigated. In addition, we enhanced the volume retention rate of PGAM by regulating the mitophagy of ADSCs under hypoxic conditions. Our findings provide insights to improve the clinical outcomes of AFT.
Materials and Methods
Ethical Approval and Consent to Participate
Data from patients who were hospitalized in the Department of Oral and Maxillofacial Surgery, School of Stomatology, Fourth Military Medical University, from January 1, 2020, to December 31, 2021, were collected for analysis. The study followed the principles of the Declaration of Helsinki. All patients signed a written informed consent for the operation, agreeing that personal treatment information could be used for scientific research and article publication. The inclusion criteria were as follows: patients with soft tissue deficiency and AFT; for detailed conditions, see Supplemental Table 1. The animals and experimental protocols used in this study were obtained from the Laboratory Animal Care and Welfare Committee of the Fourth Military Medical University for approval (project name: “The hypoxic microenvironment affects the outcomes of AFT by regulating the mitophagy of ADSCs”; approval number: 2019(028); date of approval: February 28, 2019). The subjects were all adult patients with fat grafting needs who voluntarily participated in this study under fully informed conditions, and the procedure was approved by the School of Stomatology, Fourth Military Medical University Ethics Committee (project name: “Autologous fat particles with Stromal vascular fraction (SVF)/Platelet-rich fibrin (PRF) for soft tissue deformities in maxillofacial area”; approval number: IRB-REV-2018036; date of approval: April 17, 2018).
Inclusion and Exclusion Criteria for Experiments
A total of 26 subjects were screened prior to inclusion from January 1, 2020, to December 31, 2021, and 21 cases were ultimately included in the trial (see Supplemental Table 1 for screening criteria). There were 8 male and 13 female cases, aged between 18 and 40 years.
Harvest and Pretreatment of Adipose Tissue
Each patient was subjected to general anesthesia, and the surgical area was prepared according to conventional surgical standards. A short incision of approximately 0.5 cm in length was made in both lower limbs from the patient’s medial side, and approximately 200~300 ml of swelling fluid, 500 ml saline + 10 ml lidocaine (10 ml:0.1 g) + 0.5 ml epinephrine (1 ml:1 mg) + 5 ml sodium bicarbonate (250 ml:12.5 g), was slowly injected into the subcutaneous fat layer. After waiting for 5 min, a liposuction needle (opening aperture 0.5 cm) was used to probe the subcutaneous fat layer. Reciprocal pushing and pulling movements were made in a fan-shaped area with suction to obtain approximately 10~15 ml of subcutaneous white fat. The fascia, oils, and other impurities were carefully removed. After standing in the centrifuge tube for 10 min, the fat was centrifuged at 800 rpm for 5 min, excess blood and swelling fluid were discarded, and the upper layer of suspended adipose tissue was collected. The isolated fat was transferred to the laboratory in a 37°C incubator for pretreatment and subsequent processing according to experimental needs.
Establishment and Sacrifice of the AFT Model
Adipose tissue harvested from each subject was used in six nude mice to establish the AFT model. Eight-week-old nude mice were selected and anesthetized with inhaled isoflurane. After the anesthesia took effect, the dorsal region was disinfected with iodophor. The prepared adipose mass was loaded into a 1-ml syringe, and the needle was inserted from the lateral abdomen and slowly pushed into the subcutaneous layer of the back. This was performed bilaterally (0.5 ml on each side). In the last part of this study, rapamycin-treated PGAM cells were transplanted on the left side, while untreated PGAM cells were transplanted on the right side as a homologous control. After the mice awakened from anesthesia, they were kept under aseptic conditions. At each indicated time point (1 day, 2 days, 3 days, 7 days, 14 days, 21 days), five nude mice were anesthetized by inhalation, a longitudinal incision was made along the midline of the back, and additional transverse incisions were made at both the top and bottom of the longitudinal incision. The skin was turned over to expose the subcutaneous PGAM. After being photographed, the PGAM was peeled off completely, weighed, and its volume was measured using the immersion method.
Tissue Sectioning and Hematoxylin and Eosin Staining
The isolated PGAM was rapidly fixed in 4% paraformaldehyde (Beyotime, China, P0099) for 24 h and sequentially dehydrated, embedded, and sectioned at a thickness of 8 µm with a section embedding kit. The sections were subsequently processed according to the standard procedure for hematoxylin and eosin (HE) staining (LEAGENE, China, DH0006).
Immunohistochemical Staining
The hydrated sections were placed in a staining vat containing a sodium citrate antigen repair solution (ZSGB-Bio, China, pH 6.0) and heated to 98°C for 10 min to perform surface antigen repair. Then, antigen blocking (ZSGB-Bio, ZLI9056) and primary antibody staining (HIF-1α; Abcam, UK, ab51608, 1:100; light chain 3 beta [LC3B], CST, USA, 3868s, 1:200; Beclin-1, Abcam, ab210498, 1:100, cell adhesion molecule-1 [CD31], Novus Biologicals, USA, NB600-562, 1:25) were performed. After washing with a phosphate buffered saline containing 0.3% Tween-20 (PBST) buffer, the sections were reacted with enzyme-labeled secondary antibodies (ZSGB-Bio, SP-9000) for 15 min again and finally mounted. The results were observed with a microscope.
Establishment of Cell Culture Systems Under Normoxic and Hypoxic Conditions
The extracted adipose tissue was digested with 1% type I collagenase (Worthington, USA, 41J21478) at 37°C for 45 min. The digestion was terminated by adding an equal amount of alpha-minimum essential medium (α-MEM) (Gibco, USA, C12571500BT) containing 10% fetal bovine serum (Thermo, USA, 10091148) before centrifugation at 800 rpm for 5 min to obtain the precipitate at the bottom of the tube. The precipitate was aspirated well with 10 ml of α-MEM, and the suspension was inoculated into 100-cm2 cell culture dishes containing 10% fetal bovine serum. The cells were incubated at 37°C under 5% CO2 and 95% air (normoxia) or 5% CO2, 1.5% O2, and 93.5% N2 (hypoxia as control) according to the experimental grouping. Cells in logarithmic phase with a density of 80%~90% were harvested for subsequent experiments.
Flow Cytometry Assay
Cells were harvested with trypsin, collected into 1.5-ml Eppendorf tubes, and centrifuged at 350 × g for 5 min to harvest the precipitate. After resuspending the cells in phosphate buffered saline (PBS), the number of cells per sample was calibrated as 2 × 106. Antibodies were added at a concentration of 1:50, and the samples were incubated at 4°C for 30 min. CD29 (Biolegend, USA, RUO303003), CD105 (Biolegend, RUO323203), CD44 (Biolegend, USA, RUO397517), CD90 (Biolegend, RUO328107), CD34 (Biolegend, RUO34350325), and CD45 (Biolegend, ASR982316) were detected to analyze the markers of the sample cells. An unstained cell sample of each group was used as the blank control.
Cell Proliferation Assay
The ADSCs were incubated at 37°C under normoxia (control) or hypoxia for the specified period (depending on the experimental needs, the incubation period fluctuated from 12 h to 7 days). The ADSCs were then digested and transferred to 96-well plates at a density of 5 × 103/well in a normoxic environment and incubated for 1 day, 2 days, 3 days, 4 days, 5 days, and 7 days. At the selected time points, the medium was changed to a culture medium containing a 10% cell counting kit-8 (CCK-8) detection reagent (Yeasen, China, 40203ES80) and incubated for 40 min. The optical density (OD) value at 450 nm of each well was measured (BIO-TEK, USA, Epoch) to analyze the cell proliferation viability.
Cellular Lipogenic Differentiation Assay
Cells under normoxia (control) or hypoxia were digested and transferred to six-well plates at a density of 2 × 105/well and incubated under normoxia at 37°C. When the cell density reached 80%, the medium in the wells was replaced by human adipose mesenchymal stem cell lipogenic induction differentiation medium (Cyagen Biosciences, China, HUXMD-90031) for induction for 14 d, and the cell status was observed regularly. After sufficient lipid droplets could be seen under the microscope, the cells were treated with an Oil Red O Staining Kit (Cyagen Biosciences, HUXMD-90031) to analyze lipogenic differentiation viability.
Cellular Immunofluorescence Staining
After being treated, the cells on glass coverslips were treated in 4% paraformaldehyde for 30 min and then washed with PBST for 5 min three times. The cells were then treated with 0.5% Triton-X for 5 min and again washed with PBST for 5 min thrice. Then, BSA was used to perform antigen blocking for 1 h, after which the sections were incubated with primary antibodies (HIF-1α: Abcam, ab51608, 1:200; LC3B: CST, 3868s, 1:200; ICAM: Santa Cruz Biotechnology, USA, sc-8439, 1:500; Perilipin: CST, #9349, 1:200) at 4°C overnight. Excess primary antibody was washed away with PBST three times, and then the cells were incubated with secondary antibody for 1 h while protected from light (CST, mouse IgG594, 8890s, 1:1000; CST, rabbit IgG594, 8889s, 1:1000; CST, rabbit IgG488, 4412s, 1:1000). Immunofluorescence blockers containing 4’,6-diamidino-2-phenylindole (DAPI) were then added, and the samples were observed under a laser confocal microscope (Nikon, Japan, A1plus).
Regulation of Mitophagy With Rapamycin
For mitophagy-enhanced cell samples, rapamycin (MCE, China, HY-10219) was added at a concentration of 10 nM, 100 nM, or 1 μM. For mitophagy-enhanced adipose tissue samples, the adipose tissue mass was immersed in PBS containing 100-nM rapamycin and incubated at 37°C for 1 min before injection into the subcutaneous area.
Autophagy Analysis With LC3B-GFP
A Premo Autophagy Sensor LC3B-GFP Kit (Invitrogen, USA, P36235) was used to detect autophagy in ADSCs under hypoxic conditions. ADSCs were cultured in normoxia until they reached 70% density. The experimental group was transfected with LC3B-GFP under hypoxia, control group 1 was transfected with LC3B-GFP under normoxia, control group 2 was subjected to hypoxia, and control group 3 was subjected to chloroquine treatment to induce autophagy. Cells were seeded at a concentration of 5 × 105/well in 25-cm2 culture flasks and transfected when they reached 70% confluence. The LC3B-GFP (50 MOI) transfection volume was calculated using a formula and added to the complete medium. Chloroquine (30 mM) was also added to the medium. The cells were incubated under normoxia (5% CO2, 95% air) or hypoxia (5% CO2, 1.5% oxygen, 93.5% nitrogen) for 24 h and treated with 4% paraformaldehyde for 30 min. Images were collected under a fluorescence microscope (Nikon, A1plus).
Transfection of HIF-1α siRNA
ADSCs were incubated in six-well plates at a density of 20 × 104/well until they reached 70%–90% confluence, after which HIF-1α siRNA was transfected using a Lipofectamine 3000 Kit (Invitrogen, 2533476) at a final HIF-1α siRNA concentration of 50 nM. Cellular RNA was extracted at 12 h, 24 h, 48 h, and 72 h after transfection to analyze the transfection efficiency. The sequences were as follows: human-HIF-1α-siRNA-F: GGAAGAACUAUGAACAUAATT, human-HIF-1α-siRNA-R: UUAUGUUCAUAGUUCUUCCTT.
Seahorse Assay
Cells were incubated in 96-well plates at a density of 8 × 104 and pretreated according to the experimental groupings. A Seahorse assay was performed according to the Agilent Seahorse XFe24 (Agilent Technologies, Inc., USA) operating manual. The trifluoromethoxy carbonylcyanide phenylhydrazone (FCCP) concentration in this experiment was 2.75 μM. The concentration of oligomycin was 1 μM, and the concentration of rotenone/antimycin A was 2 μM.
Western Blot Analysis
Total protein was extracted from the pretreated cells. A bicinchoninic acid assay (BCA) protein quantification kit (Beyotime, P0010) was used to detect the protein concentration, and after adjusting the protein concentration, each sample was heated for denaturation. Then, the samples were electrophoresed, and the proteins were transferred to a membrane. The membrane was blocked and incubated with primary antibodies diluted with blocking solution (actin, Genetex, USA, GTX109639, 1:3000; HIF-1α, Abcam, ab51608, 1:1000; LC3B, CST, 3868s, 1:1000; Beclin-1, Abcam, ab210498, 1:1000) at 4°C overnight. The membrane was then incubated with secondary antibody at room temperature, and a luminescence solution was added to detect Beclin-1 and LC3B protein expression.
Statistical Analysis
GraphPad Prism (Prism 9.5.1 for Windows, GraphPad Software, La Jolla, CA, USA) software was used for statistical analyses. The data are expressed as the mean ± standard deviation (SD). For statistical analyses, we performed one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparisons test, and differences were considered significant at P < 0.05. All reported P values are two-sided. For statistical analysis in in vitro experiments, triplicate samples were used, whereas in in vivo experiments, five samples from each group were used. Photographs were analyzed with ImageJ (National Institutes of Health, USA, v1.53t).
Results
The First 7 Days After Transplantation Are Crucial for Volume and Mass Loss and Tissue Modification in PGAM
After the PGAM was transplanted into the recipient location, the volume and mass of the PGAM steadily decreased over time (Fig. 1A, B). The decreases were most obvious in the first 7 days and gradually became less pronounced after 14 days. HE staining of tissue sections revealed that the diameter of adipocytes in the PGAM was dramatically reduced during the first week after grafting and that this decrease was accompanied by the formation of inflammation-like structures (Fig. 1C, D). After 7 days, the morphology and diameter of adipocytes tended to stabilize. Consequently, this study focused on the first 7 days after grafting, when PGAM modifications occurred.
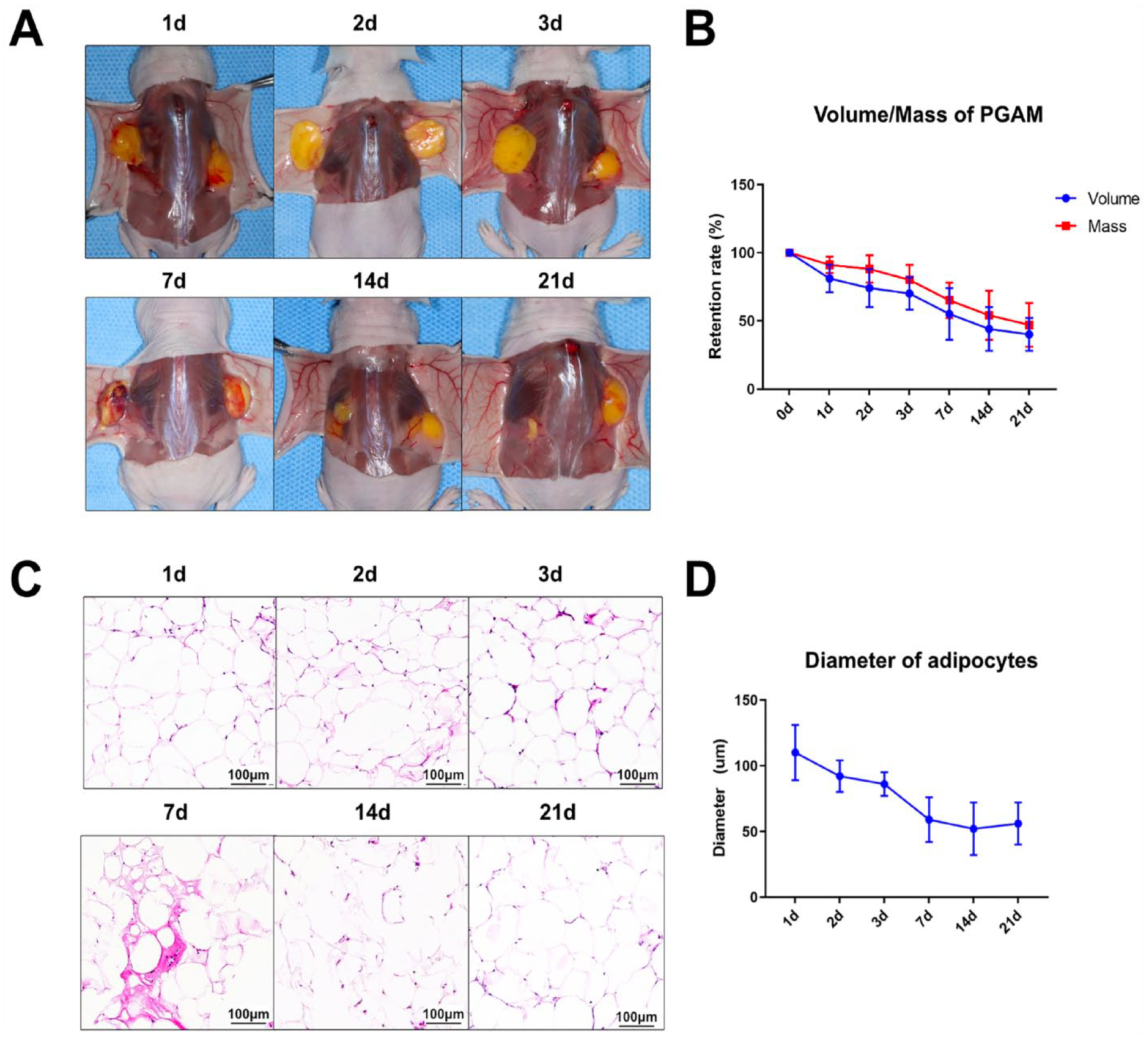
Establishment and observation of the AFT model with histological analysis at the 3-week follow-up. (A) Preprocessed human subcutaneous white adipose tissue was injected into the lower backs of nude mice (0.5 ml on each side, subcutaneous layer). During the 21 days of follow-up, general morphological and weight-volume changes were observed at each time point (1 day, 2 days, 3 days, 7 days, 14 days, and 21 days). (B) Volume and mass changes of PGAM during the 21 days. (C) Tissue sections with HE staining showing the histological features of PGAM at each time point. (D) Average diameters of adipocytes in PGAM sections based on HE staining to analyze the survival and regeneration conditions. AFT: autologous fat transplantation; PGAM: postgrafting adipose mass; HE: hematoxylin and eosin.
The Expression of Autophagy-Related Molecules (Beclin-1 and LC3B) and Hypoxia-Related Molecules (HIF-1α) Is Increased Within the PGAM During the Early Phase Postgrafting, and ADSCs Are the Main Cells Responding to Hypoxic Signaling
Immunohistochemical analysis revealed that the autophagy-related molecules Beclin-1 and LC3B, as well as the hypoxia-related molecule HIF-1α, were strongly expressed in PGAM and that this expression varied over time (Fig. 2A, B). Specifically, Beclin-1 expression was evident 1 day after grafting, while LC3B expression was detected at 3 days after grafting (which may have been related to the low LC3B expression at the tissue level). This finding indicates the initiation of autophagy in PGAM after grafting. Notably, the expression of HIF-1α was not only time-dependent but also region-dependent, with no detectable HIF-1α expression within 500 µm of the PGAM edge (Fig. 2C). These results demonstrate that HIF-1α is expressed mostly in the hypoxic microenvironment in the PGAM. According to immunofluorescence findings, ADSCs (marked by intercellular adhesion molecule, ICAM) and mature adipocytes (marked by perilipin) both expressed HIF-1α (Fig. 2D). Intriguingly, the expression of HIF-1α in ADSCs was most pronounced at 3 and 7 days. On the one hand, this indicates that HIF-1α is broadly expressed in the PGAM; on the other hand, it indicates that ADSCs are the active cells responding to HIF-1α signaling in the early stage after grafting. During the first week, the original mature adipocytes were unable to adjust to the hypoxic conditions and showed a progressive decline in proportion. Therefore, in this study, we used ADSCs in the early postimplantation period, which were more active in response to hypoxic signals than mature adipocytes, as the main targets.

Autophagy and hypoxia-related molecules in PGAM at the early stage after grafting (0, 1, 2, 3, and 7 days). (A) Immunohistochemistry showing the expression of Beclin-1 in the cytoplasm of PGAM (the arrow indicates Beclin-1). (B) Immunohistochemical staining showing the expression of LC3B in the cytoplasm of PGAM (the arrow indicates LC3B). (C) Immunohistochemistry and analysis showing the different expression levels of HI-1α in the center (top) and edge (bottom) areas of the PGAM (the arrow indicates HIF-1α). (D) Immunofluorescence staining to show the expression of HIF-1α in ADSCs (left, red for ICAM) and mature adipocytes (right, red for Perilipin). ADSC: adipose-derived stem cells; ICAM: intercellular adhesion molecule; PGAM: postgrafting adipose mass.
In Vitro, Hypoxia Induces the Expression of HIF-1α Signaling in ADSCs While Impairing Lipogenic Differentiation and Proliferation Capacity
By obtaining human-derived ADSC primary cells, we successfully established a hypoxic in vitro culture system and performed preliminary characterization (Fig. 3A). Flow cytometry showed that ADSCs had typical stem cell markers (CD34–, CD45–, CD29+, CD44+, CD90+, CD105+) (Fig. 3B). Immunofluorescence confirmed that the HIF-1α signal was detectable in ADSCs cultured for 12 h under hypoxic conditions, but the expression intensity of the signal did not increase further with time, which may have been related to a decrease in cell activity due to prolonged hypoxia (Fig. 3C). The western blot results further confirmed that HIF-1α was highly expressed in ADSCs after 12 and 24 h of hypoxic culture (Fig. 3D). Oil red O staining showed that hypoxic culture for 24 h or longer resulted in a significant decrease in the lipogenic differentiation of ADSCs (Fig. 3E, F). The CCK-8 results showed that more than 72 h of culture under hypoxic conditions significantly impaired the proliferative ability of ADSCs (Fig. 3G).
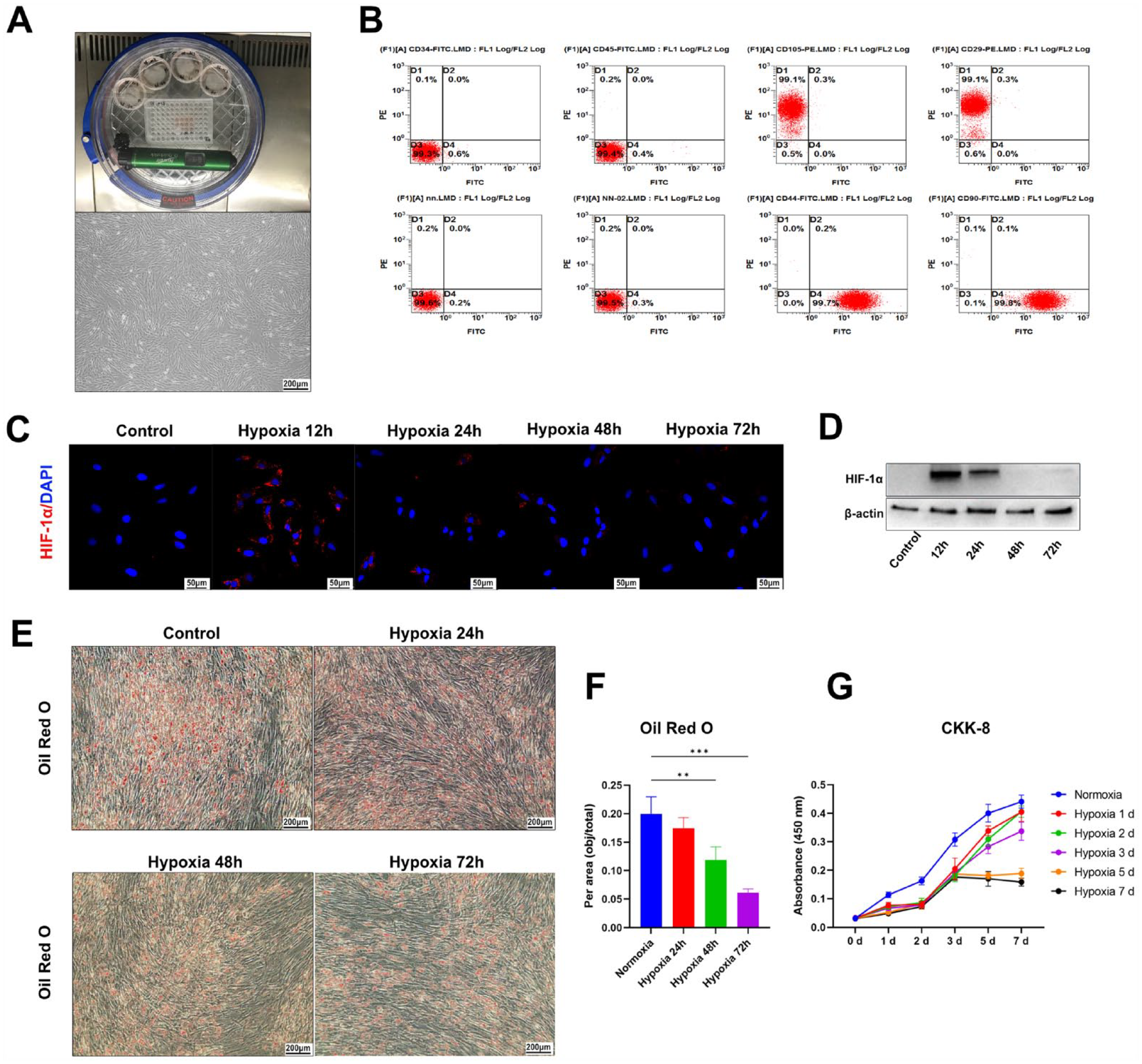
Effects of hypoxic conditions on the proliferation and adipogenic differentiation capacity of ADSCs in vitro. (A) Establishment of the cultivation system with an adjustable oxygen concentration (top) and the isolation and general characterization of ADSCs under an optical microscope (bottom). (B) Flow cytometric assay to confirm that ADSCs were CD34–, CD45–, CD29+, CD44+, CD90+, and CD105+. (C) Immunofluorescence staining to show the expression of HIF-1α in ADSCs cultured under hypoxia for 0, 12, 24, 48, and 72 h (blue indicates DAPI; red indicates HIF-1α). (D) Western blot to confirm HIF-1α expression in ADSCs cultured under hypoxic conditions. Full-length blots/gels are presented in Supplemental Figure 1. (E and F) Oil red O staining to show the adipogenic differentiation capacity of ADSCs cultured under hypoxic conditions. (G) CCK-8 (cell counting kit-8) experiment showing the proliferation capacity of ADSCs cultured under hypoxic conditions. ADSC: adipose-derived stem cells; HIF-1α: hypoxia-inducible factor-1 α subunit; DAPI: 4’,6-diamidino-2-phenylindole.
Mitochondrial Autophagy Within ADSCs Is Activated Under Hypoxic Conditions, While Mitochondrial Integrity and Respiratory Function Are Impaired
Immunofluorescence showed that LC3B was actively expressed within ADSCs cultured in hypoxic conditions for 24 h or longer (Fig. 4A). In adenovirus transfection ex-periments, chloroquine was used to block the binding of GFP-LC3B-marked autophagosomes with lysosomes, which resulted in the observation of many undegraded autophagosomes in ADSCs (Fig. 4B). This finding suggests that active autophagy does occur within ADSCs cultured under hypoxia for 24 h or longer. Further immunofluorescence results showed that GFP-LC3B appeared to co-localize with mitochondria (Fig. 4C). Western blotting also revealed that a large amount of cytochrome C released from mitochondria was detectable in the cytoplasm of ADSCs after 12 h of hypoxic culture, indicating that the integrity of mitochondria was damaged under hypoxic conditions (Fig. 4D). This suggests that mitophagy may be active in ADSCs cultured under hypoxic conditions. The Seahorse assay results also revealed a significant decrease in mitochondrial respiratory capacity after 12 h of hypoxia (Fig. 4E).

Effects of hypoxic conditions on autophagy and mitochondria in vitro. (A) Immunofluorescence staining to show the expression of LC3B in ADSCs under hypoxic cultivation for 0, 12, 24, 48, and 72 h (blue for DAPI, green for LC3B). (B) Immunofluorescence staining to assess autophagy in ADSCs cultured in hypoxic conditions for 24 h (blue indicates DAPI, and green indicates GFP-LC3B). (C) Immunofluorescence staining to show the subcellular location of LC3B and mitochondria (blue indicates DAPI; red indicates MitoTracker; green indicates GFP-LC3B). (D) Western blot to verify the activity of mitochondria of ADSCs cultured in hypoxic conditions for 0, 12, 24, 48, and 72 h by testing cytochrome C in the cytosol and mitochondria. Full-length blots/gels are presented in Supplemental Figure 2. (E) Seahorse analysis to test the effects of hypoxic conditions on the mitochondrial respiration activity of ADSCs. ADSC: adipose-derived stem cells; DAPI: 4’,6-diamidino-2-phenylindole.
Knockdown of HIF-1α Inhibits the Initiation of Mitophagy and Further Impairs the Lipogenic Differentiation Capacity and Mitochondrial Function of ADSCs Under Hypoxic Conditions
Reverse transcription (RT) PCR showed that siRNA constructs had high knockdown efficiency (Fig. 5A). The knockdown of HIF-1α significantly inhibited the lipogenic differentiation capacity of ADSCs under hypoxic conditions (Fig. 5B, C). Interestingly, the knockdown of HIF-1α showed a transient inhibitory effect on cell proliferation only at 2 days, while there were no significant differences between the groups at 5 days (Fig. 5D). Immunofluorescence showed that ADSCs with knockdown of HIF-1α did not exhibit active LC3B signals under hypoxic conditions for 24 or 48 h, suggesting that knockdown of HIF-1α inhibited the initiation of mitophagy in ADSCs under hypoxic conditions (Fig. 5E). Furthermore, the Seahorse assay showed that knockdown of HIF-1α further reduced the mitochondrial respiratory capacity of ADSCs under hypoxic conditions (Fig. 5F).
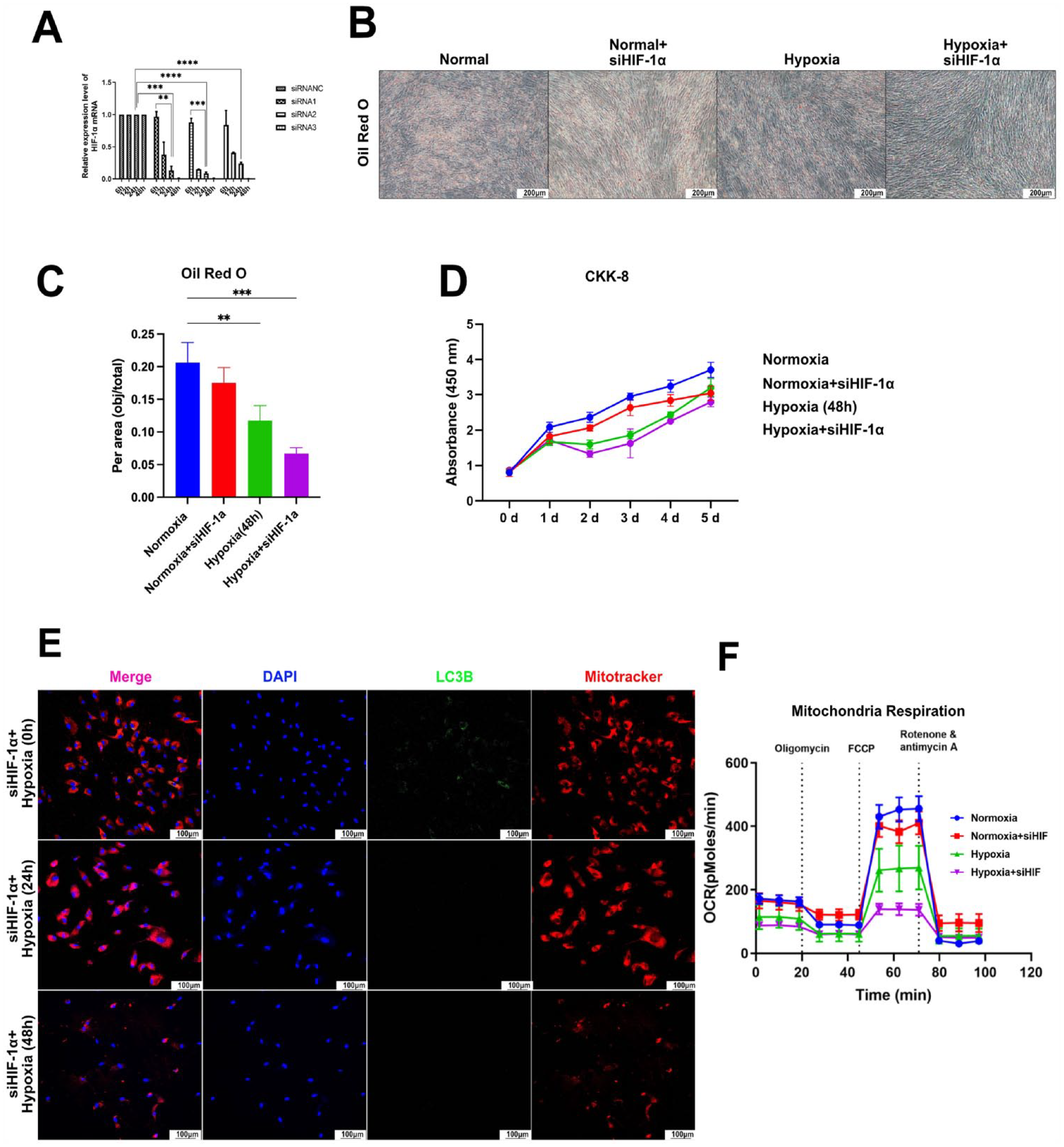
siRNA-mediated HIF-1α silencing to analyze the effects of HIF-1α on ADSCs and mitochondria. (A) Construction and efficiency validation of the siRNA. (B and C) Oil red O experiments to test the effect of HIF-1α on the differentiation capacity of ADSCs under hypoxic conditions. (D) CCK-8 experiments to analyze the effect of HIF-1α on the proliferation capacity of ADSCs under hypoxic conditions. (E) Immunofluorescence staining showing autophagy-related LC3B expression in ADSCs under hypoxic conditions. (F) Seahorse analysis to test the effects of HIF-1α on mitochondrial respiration in ADSCs under hypoxic conditions; CCK-8: cell counting kit-8.
Rapamycin Enhances Autophagy in ADSCs, Thereby Restoring the Lipogenic Differentiation Capacity and Mitochondrial Function of ADSCs Under Hypoxic Conditions
Immunofluorescence showed that the addition of rapamycin significantly increased autophagy in ADSCs under hypoxic conditions, with this enhancement peaking at 100 nM and decreasing again at 1 μM (Fig. 6A). To avoid the unknown effects of high doses of rapamycin, a 100-nM concentration of rapamycin was used as the standard concentration in the follow-up experiments of this study. Oil red O staining showed that ADSCs with 100-nM rapamycin in the medium maintained a high lipogenic differentiation capacity after hypoxic stimulation for 48 h (Fig. 6B). The CCK-8 assay showed that rapamycin did not significantly affect the proliferation activity of ADSCs cultured in hypoxia for 48 h (Fig. 6C). The Seahorse assay showed that the maximum mitochondrial respiration rate of ADSCs cultured in hypoxic conditions was significantly enhanced by the addition of rapamycin (Fig. 6D). This indicates that rapamycin can effectively reduce the adverse effects of hypoxia on the lipogenic differentiation ability of ADSCs by enhancing the autophagy of ADSCs under hypoxic conditions while partially restoring the respiratory capacity of mitochondria. However, under normoxia, rapamycin did not show significant effects on ADSCs or their mitochondrial activity.
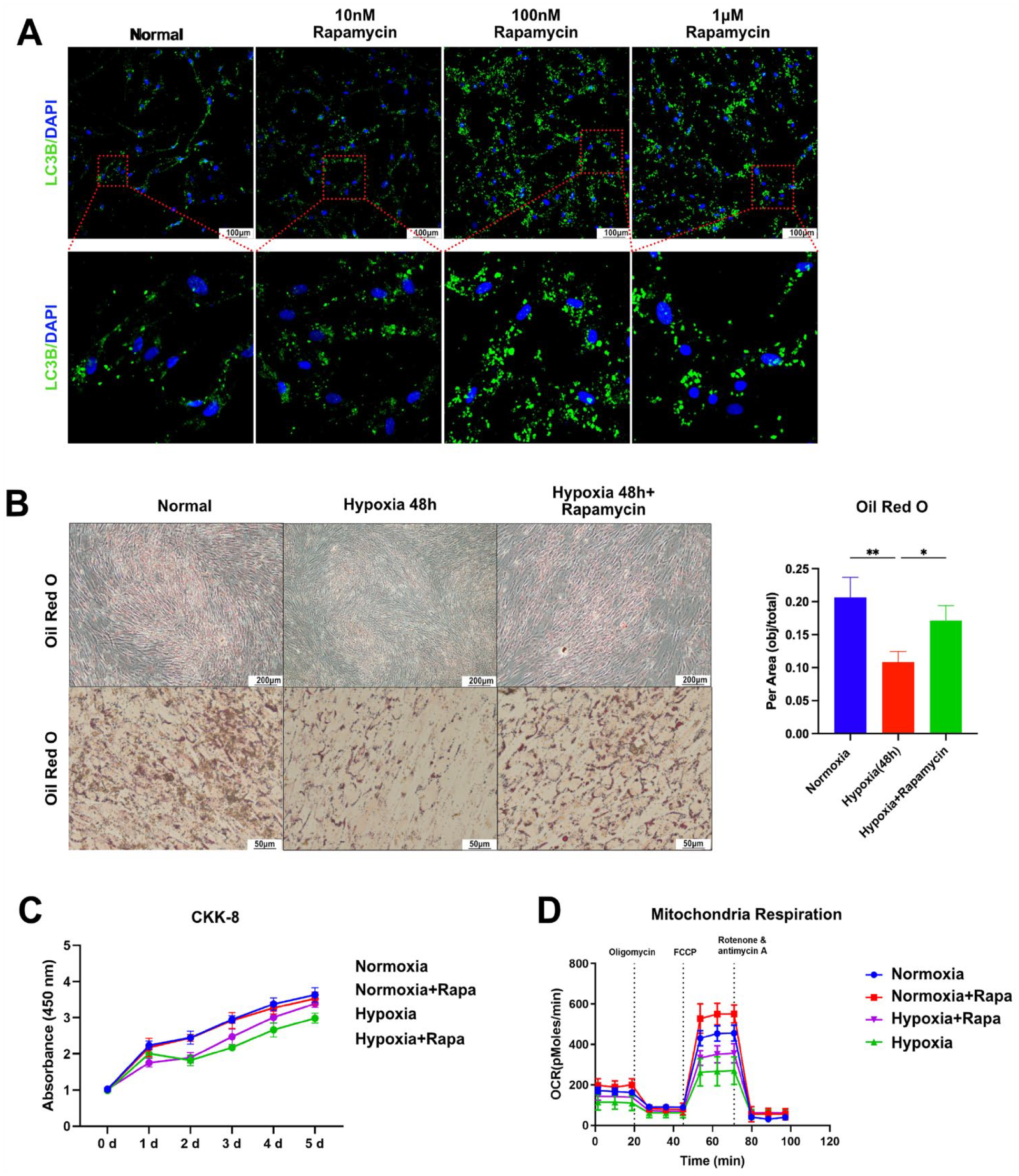
Effects of enhanced autophagy on ADSCs under hypoxic conditions. (A) Autophagy-related LC3B expression induced by different concentrations of rapamycin (0 nM, 10 nM, 100 nM, and 1 μM). (B) Oil red O experiments showing the differentiation capacity of autophagy-enhanced ADSCs under hypoxic conditions. (C) CCK-8 experiments to analyze the proliferation capacity of autophagy-enhanced ADSCs under hypoxic conditions. (D) Seahorse analysis to test the effects of enhanced autophagy on mitochondrial respiration in ADSCs under hypoxic conditions. ADSC: adipose-derived stem cells; CCK-8: cell counting kit-8.
Rapamycin Can Effectively Increase Autophagic Activity in the PGAM, Thus Alleviating the Volume Loss and Cell Morphological Changes After AFT
The results of AFT model experiments in nude mice showed that the mass and volume of rapamycin-treated PGAM were significantly higher than those of the control group at 21 d (Fig. 7A–C). This was largely attributable to suppression of the volume and mass loss during the first 7 d after grafting. HE staining revealed that rapamycin-treated PGAM showed a uniform and mature cellular morphology at 14 and 21 days, with fewer inflammatory-like structures than the control PGAM (Fig. 7D, E). This may have been because rapamycin enhanced the protective effects of autophagy, helping ADSCs resist the stress of hypoxic conditions. Immunohistochemistry showed that PGAM cells treated with rapamycin exhibited high expression of LC3B after 3 days, reflecting enhanced autophagic activity (Fig. 7F). Moreover, the expression of CD31 in rapamycin-treated PGAM was significantly higher than that in the control group, indicating a potential angiogenesis-related mechanism behind this protective effect (Fig. 7G).

Establishment and observation of the AFT model with autophagy-enhanced PGAM. (A) Preprocessed human subcutaneous white adipose tissue was injected into the lower backs of nude mice (0.5 ml on each side, subcutaneous layer; rapamycin-treated PGAM on the left side, PBS-treated PGAM on the right side). During the 21 days of follow-up, general morphological and weight-volume changes were observed at each time point (1 day, 2 days, 3 days, 7 days, 14 days, and 21 days). (B and C) Changes in the mass and volume of autophagy-enhanced PGAM over 21 days. (D) Tissue sections with HE staining showing the histological features of autophagy-enhanced PGAM at each time point. (E) Average diameters of adipocytes in autophagy-enhanced PGAM sections based on HE staining to analyze the survival and regeneration conditions. (F) Immunohistochemistry showing the LC3B expression level in the autophagy-enhanced PGAM at 3, 7, 14, and 21 days. (G) Immunohistochemistry showed a higher CD31 expression level in the autophagy-enhanced PGAM group than in the control group at 21 days. PGAM: postgrafting adipose mass; HE: hematoxylin and eosin; AFT: autologous fat transplantation.
Discussion
Autologous cell/stem cell transplantation is widely used in cell regeneration, tissue reconstruction, and functional modulation24–26. Numerous studies have demonstrated that transplanted cells/stem cells play important roles in multiple ways27,28. However, many studies have also identified multiple factors in the postgrafting microenvironment that can adversely affect transplanted stem cells during the therapeutic period, such as ischemia, hypoxia, inflammation, and growth factor deficiency 29 . Among the many factors, hypoxia is the primary problem that cells face in terms of survival in the early stage after grafting.
Numerous studies have demonstrated the beneficial impacts of ADSCs in fracture injuries and skin healing, with significant therapeutic implications30,31. However, due to the presence of other types of stem cells (e.g., bone marrow stem cells) around the recipient areas in disease models, the focus of these experiments has primarily been on the interaction and regression of ADSCs with the microenvironment after grafting, which partly hinders direct observation of the physiological patterns of ADSCs themselves 29 . In contrast, the AFT model has prominent advantages: Its recipient area is also dominated by adipose tissue, which supports a microenvironment similar to the donor location. Moreover, the PGAM remains in a state of ischemia and hypoxia for 7~10 days until vascularization is completed 32 , making AFT an ideal model for observing the functional changes in ADSCs under the pathological conditions of acute-progressively resolving hypoxia.
The three-layer survival theory of AFT was based on the nutrition condition of different layers and has declared a clear fate of ADSCs, which established the foundation of clinical treatment of AFT: protecting the outer and middle layers (survival and regeneration layers) while diminishing the inner layer (necrosis layer). Our results show that HIF-1α is mainly expressed (especially in the early stage after grafting) both in ADSCs and mature adipocytes in the center area of PGAM, which is well matched with the three-layer survival theory (Fig. 2C). Furthermore, the results of in vitro experiments show that hypoxic stimulation longer than 48 h can cause unrecoverable damage to the lipogenic differentiation and proliferation activity of ADSCs, which plays an important role in the outcomes of AFT (Fig. 3E–G). However, the boundary between the apoptotic and necrotic layers in vivo is dynamically changing, which makes it difficult to regulate the oxygen or nutrition conditions of a particular layer precisely and persistently.
The effects of the hypoxic microenvironment on mitochondria are dependent on the type of hypoxic environment10,11. In this experiment, we successfully simulated the acute, progressively resolving hypoxic environment by establishing an AFT model in nude mice and observed diminished mitochondrial respiratory function under hypoxic conditions. The function of mitochondria can be restored to some extent by enhancing mitophagy, suggesting that clearance of damaged mitochondria under this type of hypoxia is essential to maintain mitochondrial functional homeostasis (Fig. 6D). Notably, HIF-1α is not the only pathway mediating mitophagy. Studies have demonstrated that mitophagy can be categorized into ubiquitin-dependent and ubiquitin-independent types 33 . The former is mainly due to the aggregation and activation of phosphatase and tensin homolog deleted on chromosome ten (PTEN)-induced kinase 1 (PINK1) on the outer membrane of damaged mitochondria. Active PINK1 recruits the E3 ligase Parkin, which generates ubiquitin chains and further initiates autophagosome formation. Ubiquitin-independent mitophagy can be activated by numerous autophagy receptors located at the outer membrane of mitochondria, including BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3), BCL2/adenovirus E1B 19 kDa protein-interacting protein 3-like (BNIP3L, NIX), FK506-binding protein 8 (FKBP8), BCL2-like protein 13 (BCL2L13), and FUN14 domain-containing 1 (FUNDC1). Upon mitophagy induction, these receptors interact with LC3 and recruit mitochondria into preformed phagophores to perform degradation. In general, ubiquitin-dependent mitophagy is often viewed as a response to mitochondrial stress, and ubiquitin-independent mitophagy is thought to play important roles in mitochondrial quality control and metabolic function regulation, especially during cellular differentiation 34 . As one of the main regulators of mitophagy receptors, HIF-1α triggers an increase in BNIP3 and NIX, thus enhancing ubiquitin-independent mitophagy. Our observations show that the initiation of mitophagy under an acute-progressively resolving hypoxic environment is highly dependent on HIF-1α and that knockdown of HIF-1α results in an inability to initiate mitophagy. This finding suggests that HIF-1α is the primary initiator that regulates the level of mitophagy under acute, progressively resolving hypoxia conditions.
Rapamycin is a macrolide immunosuppressant widely used in organ transplantation and anticancer adjuvant therapy. This drug has been found to promote autophagy (including mitophagy) by inhibiting the activity of mammalian target of rapamycin (mTOR). However, the optimal dose and treatment schedules for rapamycin to exert its effects on different cell and tissue types are unclear; thus, there is a long way to go before rapamycin can be used to modulate cellular autophagy in vivo 35 . Our study reveals that a single but adequate pretreatment of the adipose mass with rapamycin before transplantation is effective in activating autophagy of ADSCs (Fig. 7F). The immunohistochemical staining showed a more significantly expressed CD31 that indicated active angiogenesis within the autophagy-enhanced PGAM and predicted the potential mechanisms by which rapamycin exerts its protective effects against hypoxia in AFT (Fig. 7G). It also suggests that the time before transplantation is an ideal time point for pharmacological intervention of the adipose mass in the acute, progressively resolving hypoxia model. Although hypoxic conditions last for several days, highly activated autophagy in ADSCs before transplantation is very important for resisting the adverse effects of impending hypoxic conditions. Although finding an optimal treatment schedule for rapamycin was not the focus of this study, we have shown that a single pretreatment with rapamycin is sufficient to influence the final fate of ADSCs in the AFT model. Furthermore, this one-time, low-dose topical administration circumvents the unknown effects of rapamycin on most other tissues and cell types. These findings provide theoretical references for the clinical application of rapamycin in stem cell transplantation (Fig. 8).

The influence of HIF-1α-induced mitophagy on the final outcomes of PGAM under hypoxic conditions. The ADSCs located in the middle layer of the PGAM are surrounded by a hypoxic microenvironment, which causes mitochondrial damage and results in massive ADSCs death, fibrogenesis, and oil cysts (outcome I). On the other hand, HIF-1α responds to hypoxic signaling and counteracts the damage of hypoxia by initiating mitophagy, which preserves mitochondrial function in ADSCs to a certain extent, thus allowing more ADSCs to survive and leading to PGAM with a poor retention rate (outcome II). In addition, pretreatment with rapamycin effectively activates mitophagy and protects mitochondrial function, thus greatly improving the survival of ADSCs and the retention rate of PGAM (outcome III). ADSC: adipose-derived stem cells; PGAM: postgrafting adipose mass.
We also found that the hypoxic signal started 2 days after hypoxia began and persisted until 7 days (Fig. 2C). During this process, the expression of the autophagy-related molecules Beclin-1 and LC3B was detected (Fig. 2A, B). Interestingly, the expression of the autophagy-associated molecule Beclin-1 in our study appeared to precede the expression of HIF-1α, which may have been related to the different expression levels of these two molecules (Fig. 2A, C). It can be speculated that low-level expression of HIF-1α causes a significant elevation in Beclin-1 expression through a cascade response or that other adverse factors (e.g., nutrient deprivation) that are concurrent with hypoxic conditions initiate mitophagy through an HIF-1α-independent pathway. Exploring how the effects of rapamycin on cells change over time and elucidating the possible schedules of multiple administrations will be part of our future research.
Conclusion
In this study, we constructed an AFT model in nude mice. We found that the activity of ADSCs after grafting was significantly decreased in the early stage of acute, progressively resolving hypoxia conditions, while the mitophagy process was significantly increased. In addition, we found that HIF-1α is the key molecule that initiates and maintains mitophagy in the early stage after grafting. Enhancing mitophagy with rapamycin before transplantation can improve the mitochondrial activity of ADSCs under hypoxic conditions to resist subsequent hypoxic conditions, thus increasing the activity and survival rate of ADSCs and ultimately improving the outcomes of AFT.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231210750 – Supplemental material for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897231210750 for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation by Kai Zhang, Dan Jin, Xin Zhao, Bin Lu, Weiwei Guo, Rui Ren, Simo Wu, Junrui Zhang and Yunpeng Li in Cell Transplantation
Research Data
sj-docx-2-cll-10.1177_09636897231210750 – Supplemental material for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation
Supplemental material, sj-docx-2-cll-10.1177_09636897231210750 for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation by Kai Zhang, Dan Jin, Xin Zhao, Bin Lu, Weiwei Guo, Rui Ren, Simo Wu, Junrui Zhang and Yunpeng Li in Cell Transplantation
Research Data
sj-docx-3-cll-10.1177_09636897231210750 – Supplemental material for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation
Supplemental material, sj-docx-3-cll-10.1177_09636897231210750 for HIF-1α-Induced Mitophagy Regulates the Regenerative Outcomes of Stem Cells in Fat Transplantation by Kai Zhang, Dan Jin, Xin Zhao, Bin Lu, Weiwei Guo, Rui Ren, Simo Wu, Junrui Zhang and Yunpeng Li in Cell Transplantation
Footnotes
Author Contributions
Y.L. and K.Z. conceived the project. Y.L. and K.Z. designed the experiments. Y.L. and K.Z. supervised the research. J.Z., B.L., K.Z., D.J., and R.R. provided key reagents, methods, and technologies. D.J., X.Z., W.G., S.W., J.Z., B.L., K.Z., and R.R. performed the experiments. D.J., X.Z., W.G., S.W., J.Z., B.L., K.Z., and R.R. analyzed the data. Y.L. and K.Z. interpreted the results. Y.L. and K.Z. wrote the paper; all the authors have read and approved the manuscript.
Availability of Data and Material
The data, results, and conclusions of this study are all original, authentic, and have not been published in any other academic journals or conferences.
Ethics Approval
This study was approved by the Laboratory Animal Care and Welfare Committee of the Fourth Military Medical University, Shaanxi Province, China.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Laboratory Animal Care and Welfare Committee of the Fourth Military Medical University, China, and School of Stomatology, Fourth Military Medical University Ethics Committee, China (approval number: 2019(028); date of approval: February 28, 2019). The subjects were all adult patients with fat grafting needs who voluntarily participated in this study under fully informed conditions, and the procedure was approved by the School of Stomatology, Fourth Military Medical University Ethics Committee. Project name: “Autologous fat particles with SVF/PRF for soft tissue deformities in maxillofacial area” (approval number: IRB-REV-2018036; date of approval: April 17, 2018).
Statement of Informed Consent
All patients in this study signed a written informed consent form for the operation, agreeing that personal treatment information could be used for scientific research and article publication.
Consent for Publication
All authors have read the final manuscript and agreed to publish this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81970988) and Natural Science Foundation of Shaanxi Province, China (2023-GHZD-20). All the funding bodies played no role in the design of the study and collection, analysis and interpretation of data, and in writing the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
