Abstract
Human umbilical cord mesenchymal stem cells (hUCMSC) have shown promising potential in ameliorating brain injury, but the mechanism is unclear. We explore the role of NogoA/NgR/Rho pathway in mediating hUCMSC to improve neurobehavioral status and alleviate brain injury in hypoxia/ischemia-induced CP (cerebral palsy) rat model in order to promote the clinical application of stem cell therapy in CP. The injury model of HT22 cells was established after 3 h hypoxia, and then co-cultured with hUCMSC. The rat model of CP was established by ligation of the left common carotid artery for 2.5 h. Subsequently, hUCMSC was administered via the tail vein once a week for a total of four times. The neurobehavioral status of CP rats was determined by behavioral experiment, and the pathological brain injury was determined by pathological staining method. The mRNA and protein expressions of NogoA, NgR, RhoA, Rac1, and CDC42 in brain tissues of rats in all groups and cell groups were detected by real-time quantitative polymerase chain reaction (RT-qPCR), Western blot, and immunofluorescence. The CP rats exhibited obvious motor function abnormalities and pathological damage. Compared with the control group, hUCMSC transplantation could significantly improve the neurobehavioral situation and attenuate brain pathological injury in CP rats. The relative expression of NogoA, NgR, RhoA mRNA, and protein in brain tissues of rats in the CP group was significantly higher than the rats in the sham and CP+hUCMSC group. The relative expression of Rac1, CDC42 mRNA, and protein in brain tissues of rats in the CP group was significantly lower than the rats in the sham and CP+hUCMSC group. The animal experiment results were consistent with the experimental trend of hypoxic injury of HT22 cells. This study confirmed that hUCMSC can efficiently improve neurobehavioral status and alleviate brain injury in hypoxia/ischemia-induced CP rat model and HT22 cell model through downregulating the NogoA/NgR/Rho pathway.
Introduction
Cerebral palsy (CP) describes a group of non-progressive functional disorders that occur in a developing fetus or infant, resulting in limited mobility and permanent developmental disorders of posture 1 . The global incidence of CP ranges from 1.5 to 4 per 1,0002–4. Although treatment strategies for CP are varied (including various surgeries on muscles, tendons, bone and nerves, medications, electrical stimulation, patterning, conductive education, and orthoses)5–7, all treatment strategies only provide symptom relief but do not repair damaged brain tissue 8 . Therefore, it is urgent to develop new therapeutic techniques and repair the damaged brain tissue, the pathogenic source of CP. At present, stem cell transplantation is considered a promising treatment; many clinical trials have reported the effectiveness and safety of different types of stem cells for CP treatment, including cord blood mononuclear cells 9 , neural stem cells 10 , bone marrow mesenchymal stem cells 11 , and umbilical cord mesenchymal stem cells 12 . However, the mechanism of action of stem cell therapy for CP remains unknown.
As we know, adolescent brains possess high plasticity, but adult brains develop multiple mechanisms to control plasticity 13 . Nogo (a.k.a. Reticulon-4, RNT4) is one of these key factors and belongs to the ubiquitous reticulon protein family14,15 with multiple functions, including the development and maintenance of neurons and oligodendrocytes, axon growth, myelination, synaptic plasticity, and apoptosis16,17. NogoA/RNT4A, one of the different splicing variants of Nogo/RNT4, is specifically expressed in the central nervous system (CNS) 18 . Binding of NogoA to specific receptor NgR can activate Rho/ROCK signaling pathway; induce cone collapse, axon growth collapse, and atrophy; and inhibit axon regeneration19–21. The key molecules of the Rho/ROCK signaling pathway include NogoA, NgR, RhoA, Rac1, and CDC42 22 . Rac1 GTPase is essential for axon formation during hippocampal neuron development and can stimulate axon growth expansion, axon guidance, dendrite growth, and neuronal polarity of nerve cells22,23. CDC42 can regulate microfilaments, mediate the formation of filamentous pseudopods, participate in vesicle transport, and regulate axon and dendrite formation and synaptic plasticity 23 . The expression patterns and levels of members of the NogoA pathway change with age, which is associated with cognitive resilience in older animals 24 . In vivo experiments showed that blocking NogoA expression may result in an increase in dendritic spine density21,25. Therefore, we speculate that NogoA, as a subtype of Nogo/RTN4 that is specifically distributed in CNS, plays an important role in CP development and stem cell intervention in CP through the NogoA/NgR/Rho pathway.
Materials and Methods
Cells and Animals
Human umbilical cord mesenchymal stem cells (hUCMSC) were obtained by umbilical cord tissue adherent culture. Prior to umbilical cord collection, all participants signed a written informed consent and were informed that umbilical cords were used only for hUCMSC culture and subsequent animal study. The cell line HT22, hippocampal neuronal cell line, was purchased from Procell Life Science & Technology Co., Ltd (Wuhan, China). The adult Sprague-Dawley (SD) rats were purchased from Hunan SJA Laboratory Animal Co., Ltd, China, for breeding rat pups.
Bioinformatics Analysis
Data set GSE23317 Global transcriptomic profiling of hypoxic presumption in an in vivo neonatal mouse model (cortex) was downloaded from the GEO database, GEO2R was used to screen differentially expressed genes, the screening criteria for P < 0.05 and |log FC| > 0.3. The volcano map was plotted in Hiplot (https://hiplot.com.cn/home/index.html) and Gene Ontology (GO) enrichment analysis was conducted for differential genes. The relative expression of RTN4 was extracted from the expression matrix of GSE23317 and analyzed statistically.
Primary Culture and Identification of hUCMSC
The hUCMSC used in this study were isolated from human umbilical cord tissues from voluntarily donated, which were obtained from rigorously screened healthy pregnant women at the First Affiliated Hospital of Gannan Medical University in Jiangxi Province, China. The umbilical cord was transferred to a biosafety cabinet, cut into sections, soaked in sterile phosphate-buffered saline (PBS) containing 1% penicillin and streptomycin, washed several times to remove the blood, and then the outer membrane and blood vessels were removed. The separated Wharton’s Jelly was cut in pieces and placed in a culture dish for 10 min, then slowly added into low-sugar medium Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (FBS) (Gibco, Auckland, New Zealand), and cultured in an incubator at 37°C and 5% CO2 for 14 days. When the adherent cells reached 80% to 90% integration, the culture medium was removed. The cells were digested and separated with 0.25% trypsin at 37°C and sub-cultured at a density of 5 × 103/cm2. The hUCMSC from passage 3 were characterized with flow cytometry (BD, Franklin Lakes, NJ, USA). The cells were added with rabbit polyclonal fluorescent antibodies conclude CD73, CD44, CD90, CD105, CD34, CD45, and HLA-DR (Invitrogen, Carlsbad, CA, USA) (1:100 dilution) to detect the surface antigens of hUCMSC8. The multi-potency of hUCMSC was tested by OriCell® Osteogenesis (Cyagen, Shanghai, China) and Adipogenesis (Cyagen, Shanghai, China) differentiation Kits (37°C, 5% CO2) according to the instructions.
Cell Model
The HT22 cells were divided into three groups: the cells of control group were cultured in DMEM containing 10% FBS (Gibco, Auckland, New Zealand) at 37°C with 5% CO2; the cells of hypoxia group were transferred into 1% oxygen concentration hypoxia incubator cultured for 3 h, and then cultured in conventional culture for 24 h; the cells of hypoxia+hUCMSC group were treated with hypoxia, and a co-culture chamber with 2 × 105 hUCMSC was added to the six-well plate for 24 h.
Immunofluorescence
The cells were fixed in 4% formaldehyde at room temperature for 15 min, then added with primary antibody solution (1:200; Proteintech, Wuhan, China) and incubated at 4°C overnight. After washing, the sample was covered with the second antibody working solution (anti-goat Alexa 488, anti-mouse Alexa594, 1:500; Proteintech, Wuhan, China) and incubated at room temperature for 2 h. Seal the sample with a sealant containing DAPI anti-fluorescence quencher. The images obtained are viewed under a fluorescence microscope (Zeiss, Oberkochen, Germany).
Animal Model
The rat pups were anesthetized with 1% pentobarbital sodium via intraperitoneal injection at 7 days age. The left carotid artery of rat pups was ligation, and bupivacaine was used for pain relief. The pups were placed with their mothers for an hour after the surgery. Then, the rat pups were immediately placed in an anoxic chamber at 37°C, and filled with a mixture of 8% oxygen and 92% nitrogen with a flow rate of 1 L/min for 2.5 h 26 . The control group rat pups only isolated the left carotid artery, but with neither ligation performed nor hypoxia treatment 27 .
Behavioral Testing
Behavioral testing was performed 3 weeks after surgery and 7 days after the last hUCMSC administration to validate the model or assess the effect of the intervention. The scoring rules were as follows: Suspension test 28 : the front paws of the rat pups were placed on a glass rod with a diameter of 0.5 cm and a distance of more than 1 m from the ground. The duration of hanging on the glass rod was recorded. The duration: < 10 s was one point, 10–30 s was two points, 31–119 s was three points, 2–5 min was four points, and >5 min was five points. Slope test 29 : the rat was placed on a plate tilted 45°, with its head facing the tilt direction, and the time it took to turn in the opposite direction was observed. Open field test 29 : a box with 36 cm length and width was divided into nine equal size lattices at the bottom of the box. When more than half of the body part of the rat entered, the adjacent grid was 1 point, and the hind limb of the rat stood was 1 point. The sum of these two parts was recorded as the total score.
Cell Transfusion
The successfully modeled rats were divided into three groups (CP, CP+PBS, CP+hUCMSC), 10 rats in each group. The CP group without other treatment. CP+hUCMSC group rats were injected with 1 × 106 hUCMSCs by tail vein at 7, 14, 21, and 28 days after surgery, respectively. The CP+PBS group and the control group only received the same volume of PBS through tail intravenous injection.
TTC, HE, and Nissl Staining
The rats were killed by neck breaking, the brain tissue was cut into five coronal sections with 2 mm thickness and immediately soak in 2% TTC (2,3,5-Triphenyl-2H-Tetrazolium Chloride) solution and incubate at 37°C for 30 min. Rats were intraperitoneally injected with 1% sodium pentobarbital and then injected with 4% paraformaldehyde through systemic circulation. Then, the brain tissue of rats was taken and paraffin embedded after dehydration. According to the requirements of HE (hematoxylin-eosin) and Nissl dyeing kit (Beyotime, Shanghai, China), the wax blocks were cut into 5 μm slices for subsequent dyeing.
Real-time Quantitative PCR
The total RNA of rat brain tissue and vitro cells were extracted using Trizol (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Reverse transcription (cDNA reverse transcription kit; TransGen, Beijing, China) was performed after nucleic acid quantification. Real-time quantitative polymerase chain reaction (PCR) analysis was performed using Bio-rad PCR Systems, according to the instructions of SYBR Green supermix (Bio-rad, Berkeley, CA, USA). The primers used in this study were synthesized and purchased from Sangon Biotech, Shanghai, China (Table 1).
Primer Sequences for Polymerase Chain Reaction.

Western Blot
The total protein of rat brain tissue and vitro cells were extracted by the RIPA (radioimmunoprecipitation assay) buffer (Beyotime, Shanghai, China) with 1% phenyl methyl sulfonyl fluoride (PMSF). The proteins were separated by 10% SDS-polyacrylamide gel electrophoresis and electrotransferred onto polyvinylidene fluoride (PVDF) membrane. The membrane was blocked by sealing fluid for 15 min, after incubation with the antibody (β-actin, TUBB3, NogoA, NgR, RhoA, Rac1, CDC42 antibody, dilution ratio of 1:1000; Proteintech, Wuhan, China) for 12 h at 4°C. Then, the membrane incubated with secondary antibodies (1:5000; Proteintech, Wuhan, China) for 1 h at room temperature. Finally, the membrane was incubated with the chemically luminescent solution for exposure.
Statistical Analysis
Statistical analysis of the data was conducted using GraphPad Prism 9. Data were presented in the form of mean ± SE (standard error). When the two groups were compared, the data conform to normal distribution and the variance was equal, and the Student’s test was used; otherwise, the Mann–Whitney test was used. Ordinary one-way analysis of variance (ANOVA) and Tukey’s multiple comparisons test were used for multiple analysis. P < 0.05 was considered to indicate a statistically significant difference.
Results
RTN4 Was Highly Expressed in the Cortex of Ischemic Hypoxic CP Rats
A total of 798 differential genes were screened after removal of repeated probes (Fig. 1A). GO enrichment analysis of these differential genes showed that they were closely related to the biological processes of neuronal death and apoptosis, and the regeneration of neurons and glial cells (Fig. 1B). Subsequently, we selected RTN4, which we were interested in, and observed its expression between the two groups. The results showed that the expression level of RTN4 in the HI+PC-24h group was significantly higher than that in the Sham-24h group, P < 0.05 (Fig. 1C).

Bioinformatics analysis of GSE23317: (A) Volcanic map; (B) GO enrichment map of differential genes; (C) relative expression of RTN4 in GSE23317, *P < 0.05.
Morphological Observation and Identification of Primary Cultured hUCMSC
After 7 days of umbilical cord tissue culture, primary hUCMSC began to crawl out. At this time, the number of cells was small and the morphology was diverse, as shown in Fig. 2A (P0 hUCMSC). When the degree of cell fusion reaches 90%, the cells were passaged. The morphology of the passage 1 cells is shown in Fig. 2A (P1 hUCMSC), the cell morphology gradually unified, and hUCMSC purity increased. When the cells are passaged into passage 2, the cells are in the same morphology; all are long fusiform and arranged in vortex shape, as shown in Fig. 2A (P2 hUCMSC). Specific surface markers of hUCMSC at passage 3 were identified by flow cytometry (Fig. 2B). The hUCMSC were positive for CD73 (97.2%), CD44 (98.2%), CD90 (97.9%), CD105 (98.8%), and were negative for CD34 (0.1%), CD45 (0.1%), and HLA-DR (0.1%). A large number of calcium nodules can be seen after osteogenic induction of hUCMSC (Fig. 2C), indicating that hUCMSC has good osteogenic differentiation ability. In addition, the large number of lipid droplets induced by adipogenic differentiation of hUCMSC indicated that hUCMSC also could differentiate into adipocytes well. These results showed that the quality of hUCMSC obtained in the experiment meets the requirements of subsequent experiments.

Identification of hUCMSC: (A) The morphological observation of hUCMSC in P0, P1, and P2; (B) the flow cytometry analysis results of hUCMSC-specific surface markers; (C) the osteogenic and adipogenic differentiation results of hUCMSC.
hUCMSC Can Improve Hypoxic Injury of HT22 Cells Via NogoA Signaling Pathway
Tubulin beta-III (TUBB3) is a neuron-specific marker that may be involved in axon guidance and maintenance 30 . Both immunofluorescence (IF) and western blotting (WB) results showed that compared with the control group, TUBB3 expression decreased (Figs. 3A, B, and 4G), NogoA and NgR expression increased (Figs. 3C, D, 4B, C) in the HT22 hypoxic treatment group. After adding the Transwell insert containing hUCMSC, compared with the hypoxia group, the expression of TUBB3 was increased (Fig. 3A, B), and the expression of NogoA and NgR was decreased (Figs. 3C, D, 4B, C). It has been reported that NogoA binding with NgR can activate RhoA/ROCK pathway 31 , which upregulates the expression of RhoA and decreases the expression levels of Rac1 and CDC42. In this study, WB results were consistent with those reported above. Compared with the control group, RhoA in the hypoxia treatment group was significantly increased (Fig. 4D), while Rac1 and CDC42 expression levels were significantly decreased (Fig. 4E, F). However, after hUCMSC addition, abnormally elevated RhoA was inhibited (Fig. 4D), and the corresponding Rac1 and CDC42 expression levels were significantly increased (Fig. 4E, F). These results indicate that hUCMSC intervention can improve the state of HT22 after hypoxia treatment, and NogoA and its downstream molecules play an important role in this process.
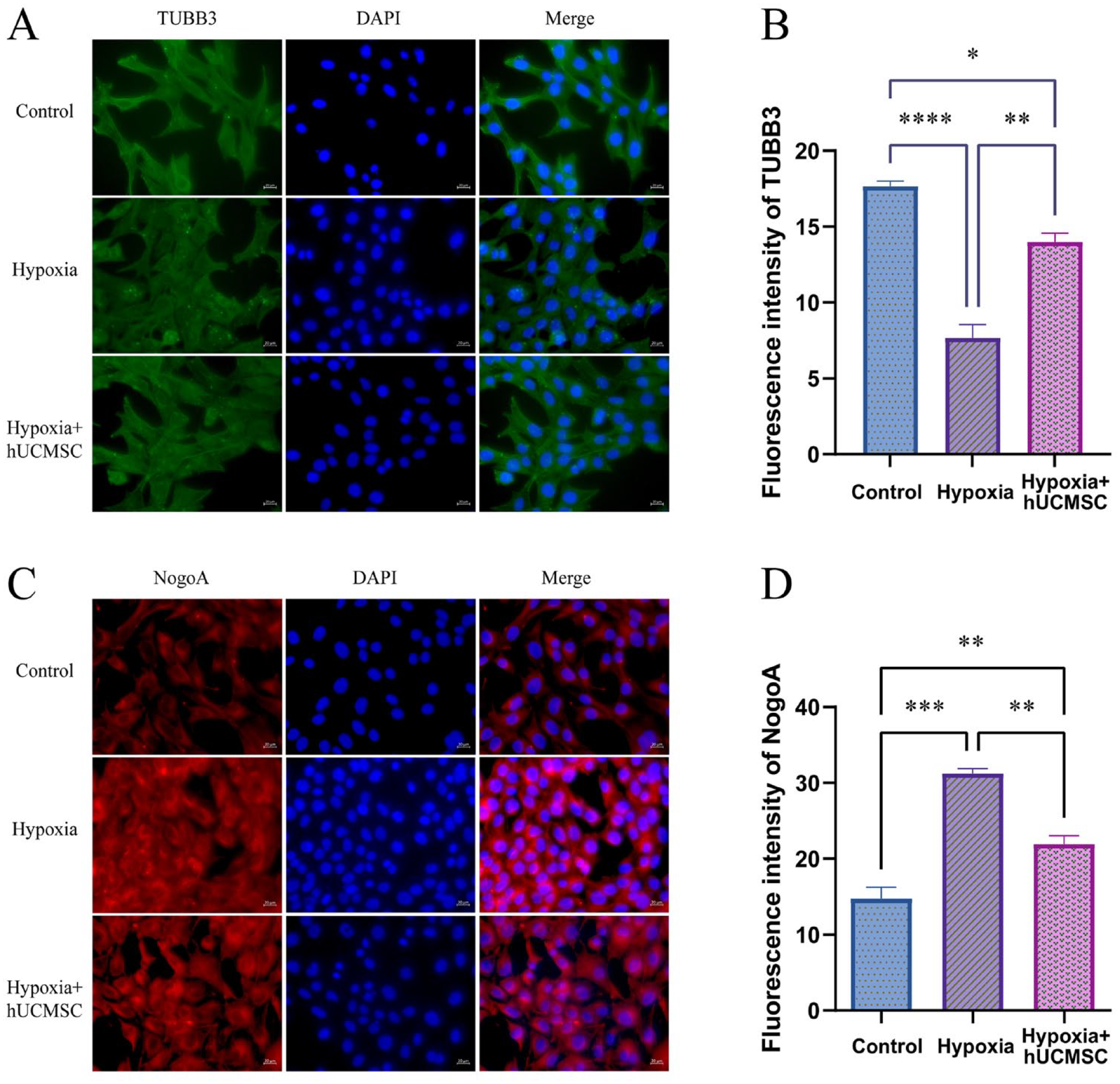
(A) Representative images of immunofluorescence staining for TUBB3; (B) fluorescence intensity quantitative analysis of TUBB3; (C) representative images of immunofluorescence staining for NogoA; (D) fluorescence intensity quantitative analysis of NogoA. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
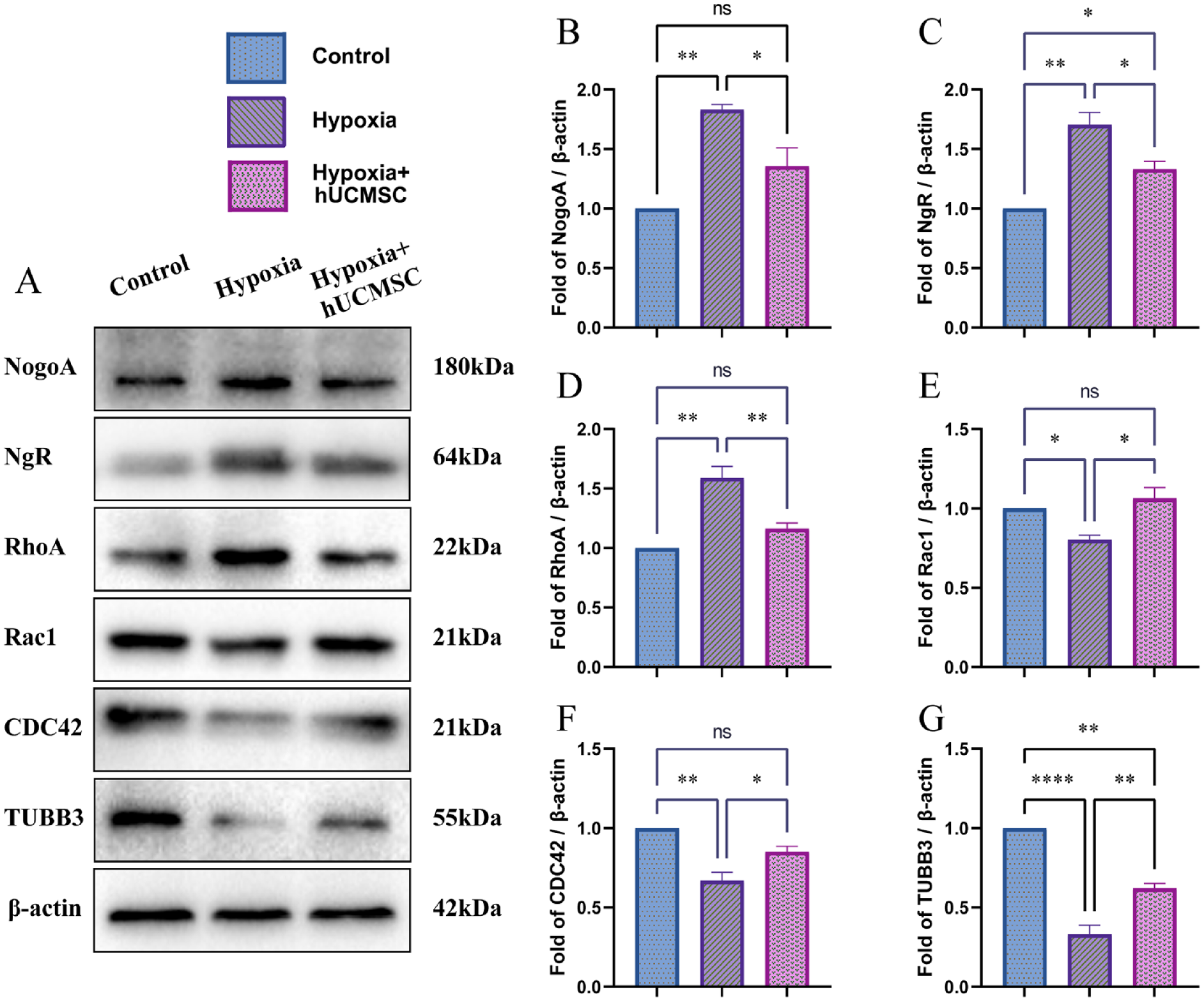
(A) The expression of NogoA, NgR, RhoA, Rac1, CDC42, and TUBB3 protein in HT22 by Western blot. The relative expression analysis by gray value of (B) NogoA. (C) NgR. (D) RhoA. (E) Rac1. (F) CDC42 and (G) TUBB3. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Characterization of CP Model
After the establishment of the CP model, TTC staining of ischemic and hypoxic rat brain sections showed obvious ischemic areas (Fig. 5A). HE staining (Fig. 5B) results suggested that the brain structure was damaged and the white matter was disordered in the ischemic hypoxia group. Compared with the control group, the nuclei were stained more deeply, the nuclear envelope was incomplete or absent, axonal dendrites were absent, and microglia infiltrated. The results of the suspension experiment showed that the CP group hung on the glass rod for a shorter time than the control group. The results of the open field test showed that the open field scores in the CP group were lower than those in the control group rats. The results of the oblique board experiment showed that the CP group spent longer time turning the head to the tail on the oblique board than the control group (Fig. 5C). To sum up, the results revealed that rat CP models were successfully established.

Assessment of CP rat. (A) Rat brain tissue stained with TTC staining. (B) HE staining of rat brain tissue. (C) Behavioral testing results of suspension test, slope test, open field test. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
HUCMSC Improved Cerebral Neurobehavioral Status and Pathological Injury in CP Rats
After transplantation of hUCMSC in CP rats, the results of the suspension test suggested that the time hung on the glass rod in the CP and CP+PBS group rats was lower than that in the CP+hUCMSC group (Fig. 6A). The results of an open field test showed that the score in the CP and CP+PBS groups was lower than that in the CP+hUCMSC group (Fig. 6B). The results of the oblique board test showed that the turnaround time of the CP group and CP+PBS group was significantly longer than that of the CP+hUCMSC group (Fig. 6C). The above results showed that the neurobehavioral state of CP rats was significantly improved after four hUCMSC transfusions, indicating that hUCMSC is effective in treating the CP model of ischemia and hypoxia.

(A) Behavioral testing results of suspension test; (B) open field test; (C) oblique board test. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
The results of HE staining are shown in Fig. 7A. In the control group, the neuronal cells of the brain were intact with regular arrangement. The CP and CP+PBS group rats’ brain tissue structure was destroyed, which was presented with multiple irregular cells’ swelling, degeneration, disordered arrangement, inflammatory cell infiltration, and partial round softening foci. After transplantation of hUCMSC in CP rats, the degree of cell swelling was reduced, inflammatory infiltrating cells were decreased, and cystic degeneration was decreased; the pathological degree of brain tissue was further alleviated and the cells were arranged neatly. The results of Nissl staining are shown in Fig. 7B. In the control group, the neuronal cells arranged closely and the cell structure was complete. In the CP and CP+PBS group, the neuronal cells were arranged disorderly and loosely, the nuclei were deeply stained and pyknotic, some cell membranes had disappeared, and the number of intact cells decreased significantly. Compared with the CP group, the neuronal cells in CP+hUCMSC were more orderly and the number of intact cells was higher.
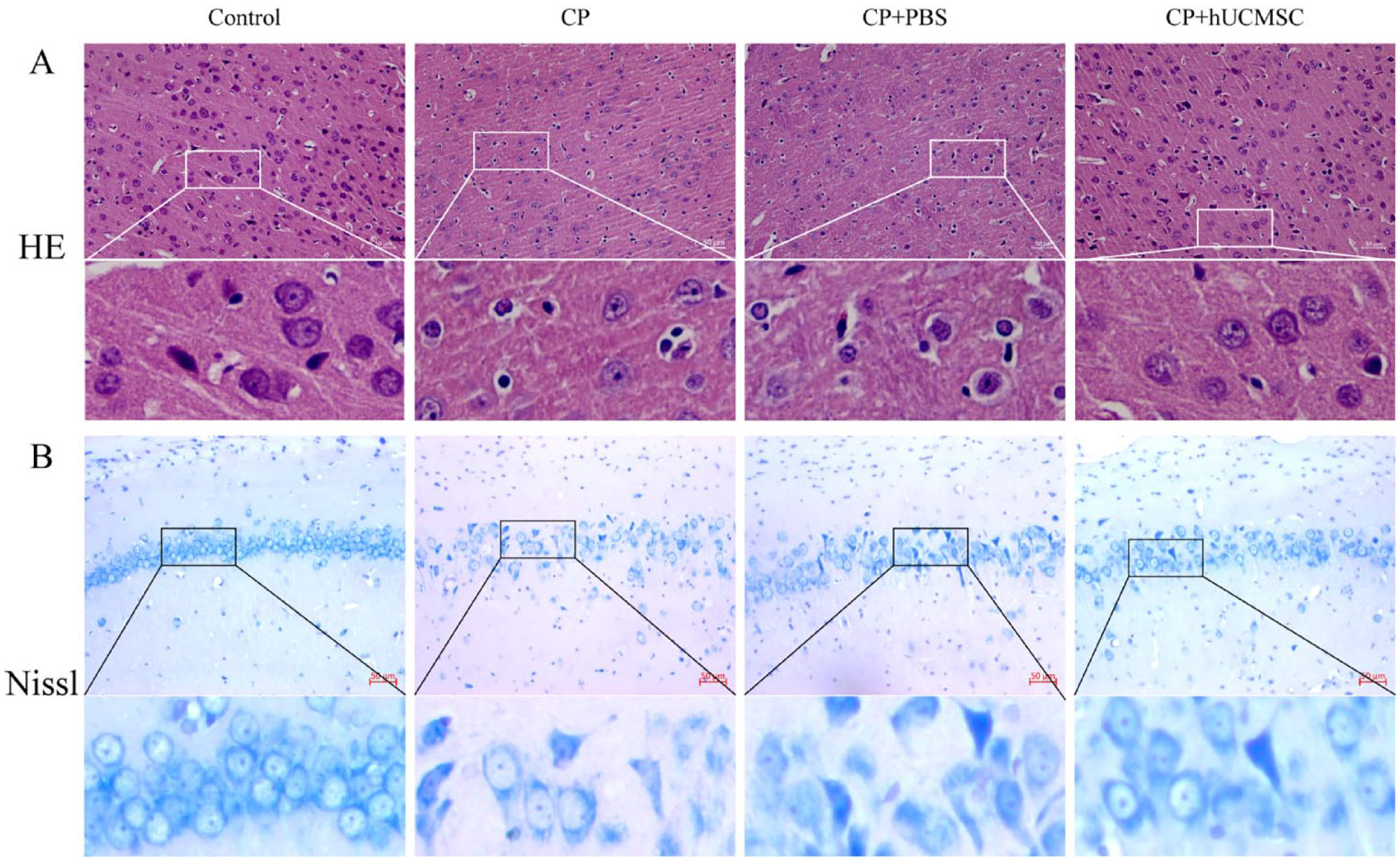
The rat brain tissues slice stained with HE staining (A) and Nissl staining (B).
HUCMSC Improves the Status of CP Rats Through the NogoA/NgR/Rho Pathway
Compared with control group and CP+hUCMSC groups, the expression of NogoA, NgR, RhoA relative mRNA and protein in the CP group and CP+PBS groups was significantly increased in the brain tissues (Figs. 8A–C and 9A–D). However, the expression of Rac1 and CDC42 relative mRNA and protein in the CP group and CP+PBS groups was significantly decreased (Figs. 8D, E and 9E, F). These results are consistent with the results of the above cell experiments, revealing that the NogoA/NgR/Rho pathway is involved in the improvement process of hUCMSC in CP rats.

The mRNA expression of (A) NogoA. (B) NgR. (C) RhoA. (D) Rac1, and (E) CDC42 in brain tissues of rats by RT-PCR. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
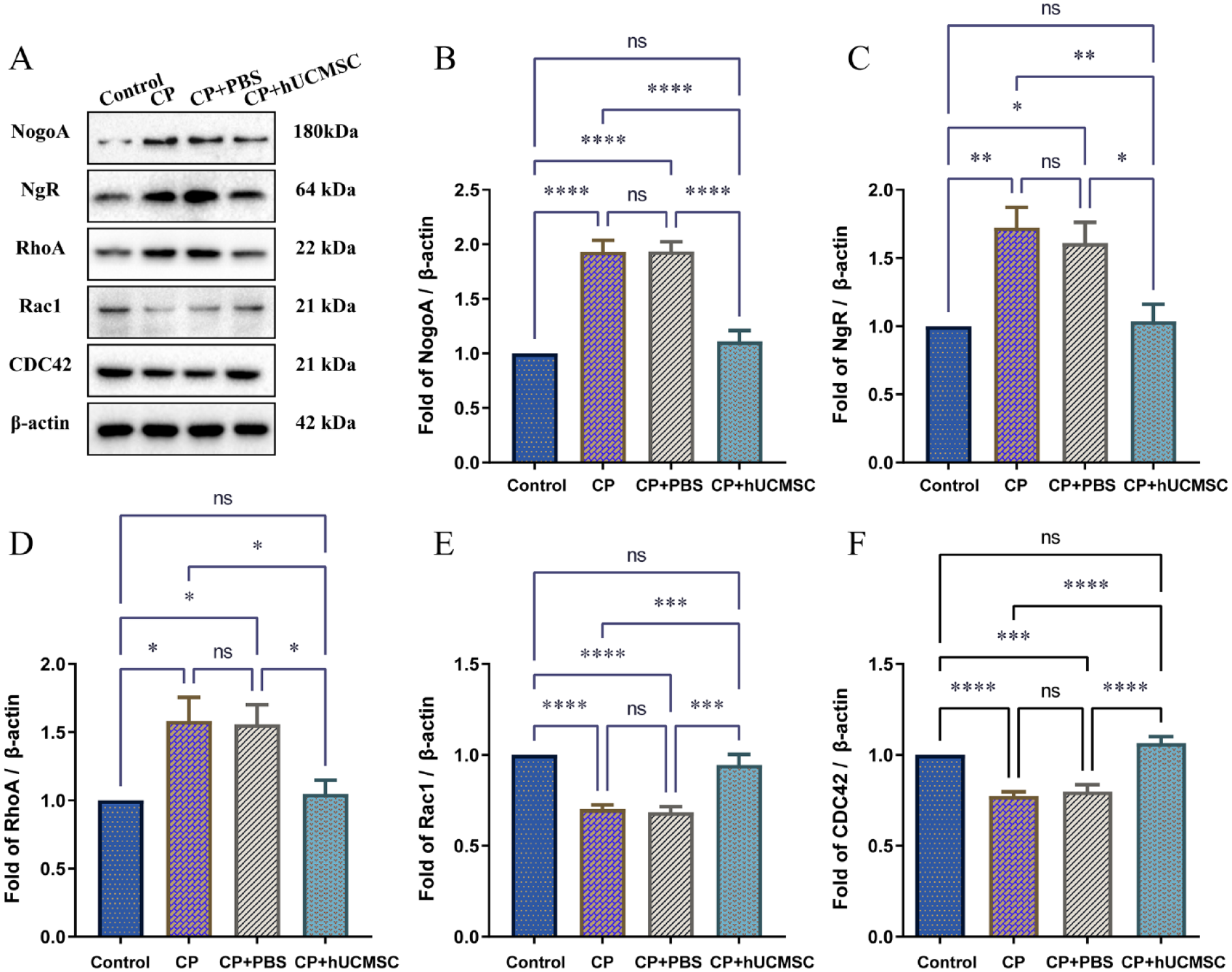
(A) The expression of NogoA, NgR, RhoA, Rac1, and CDC42 protein in brain tissues of rats by Western blot. The relative expression analysis by gray value of (B) NogoA. (C) NgR. (D) RhoA. (E) Rac1 and (F) CDC42. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Discussion
In this study, we used neonatal rats on the seventh day after birth to make models of hypoxia ischemia, which is the most commonly used method to mimic CP disease in humans32–34. CP rats are obviously sluggish and lack normative movement ability. After stem cell transplantation, the CP rats significantly improve the neurobehavioral situation, alleviate brain pathological damage, and promote nerve regeneration. Several studies32,33,35 have shown that stem cell transplantation is effective in animal models of early brain injury, which is consistent with our results.
NogoA is the most important axon growth inhibitory factor in the CNS, which can bind to specific receptor NgR to inhibit axon regeneration and play a negative regulatory role in central nervous injury28,36. The immunofluorescence staining of TUBB3 showed that the axonal dendrites of HT22 cells were greatly damaged after hypoxia. When co-cultured with hUCMSC (Figs. 3 and 4), some damaged or missing axons of nerve cells were repaired. In addition, after transplantation, CP rats showed significant improvement in neurobehavioral status, HE staining showed significant reduction of brain pathological injury, Nissl staining showed shorter and damaged axons, signs of repair. Zhang et al. 37 research showed that the highest expression levels of NogoA protein were observed in the spinal cord injury (SCI) model group, and NogoA levels were decreased after neural stem cells transplantation. Wang et al. 38 research shows that adipose-derived stem cell transplantation could promote the recovery of neurological functions, partly by inhibiting the expression of NogoA in the middle cerebral artery occlusion (MCAO) rat’s brain. Our results showed that hUCMSC significantly inhibited the expression level of NogoA, NgR, and RhoA, and increased the expression level of Rac1 and CDC42. Therefore, we believe that hUCMSC can improve the state of hypoxia/ischemia CP rats by downregulating the NogoA/NgR/Rho pathway, and its therapeutic effect may be related to axonal regeneration.
In this study, stem cells can inhibit the expression of nerve regeneration inhibitor NogoA and promote the regeneration of nerve cell axons. However, we believe that this inhibitory pathway is not single and may be the result of hUCMSC exerting various regulatory functions. Boghdadi et al. 39 demonstrated that NogoA expression is associated with astrocytes at the same time point after ischemic stroke in marmosets and humans. In addition, they found that NogoA antagonism may restrict the fence of infiltrating macrophages mediated by astrocytes, and NogoA blocking may have an indirect immunomodulatory effect at an early point in time 39 . Although immune infiltration is critical for removal of harmful cells and myelin debris 40 , inflammatory consequences may exacerbate secondary neuronal cell death. The immunomodulatory function of hUCMSC has been widely reported as its unique property. Xian et al. 41 showed, in pilocarpine induced epileptic mice, MSC-derived exosomes treatment could repair astrocyte changes induced by inflammatory reaction. One obstacle to endogenous repair after neonatal ischemia is the differentiation of neuroblast into astrocytes in the postnatal subventricular region 42 . Velthoven et al. 43 reported that MSC treatment after HI brain injury in newborns increased differentiation of neuroblast into neurons and oligodendrocytes, and reduced proliferating inflammatory cells. In conclusion, inflammation, the increase of reactive astrocytes, and the increase of NogoA expression may interact. Evidence suggests that mesenchymal stem cells–derived exosomes exhibit functions similar to MSCs with low immunogenicity and no tumorization44–46. We found that 24 h after injection of red fluorescently labeled stem cells through the caudal vein in CP rats, injected exogenous stem cells were observed in the cortex (Supplementary File 1), indicating that some cells migrated to the brain, but the number was very small, which also indicated that the therapeutic effect of exogenous mesenchymal stem cells may not depend on differentiation into damaged cell types. We speculate that hUCMSC play a therapeutic role in CP by exerting its immunomodulatory capacity on one hand and promoting endogenous neural stem cells to differentiate into neurons and oligodendrocytes on the other hand. These changes inhibit the expression of NogoA, further regulate inflammation, and affect the expression of the downstream Rho GTPase family, ultimately improving the defective axons.
In summary, we confirmed that the NogoA/NgR/Rho pathway is involved in hUCMSC treatment of CP rats, providing a new idea for clarifying the therapeutic mechanism of hUCMSC. But some limitations of the current study are worth discussing. The components and mechanisms of hUCMSC in vivo are complex. Although we have confirmed that the NogoA pathway is involved in the hUCMSC treatment of CP, whether there is a direct causal interaction between this pathway and the therapeutic effect of hUCMSC needs further proof, which is also the direction of our follow-up research. In addition, whether hUCMSC regulate the NogoA pathway by inhibiting overactivation of inflammation and altering astrocyte reactivity remains to be further explored.
Supplemental Material
sj-tif-1-cll-10.1177_09636897231210069 – Supplemental material for Human Umbilical Cord Mesenchymal Stem Cells Improve the Status of Hypoxic/Ischemic Cerebral Palsy Rats by Downregulating NogoA/NgR/Rho Pathway
Supplemental material, sj-tif-1-cll-10.1177_09636897231210069 for Human Umbilical Cord Mesenchymal Stem Cells Improve the Status of Hypoxic/Ischemic Cerebral Palsy Rats by Downregulating NogoA/NgR/Rho Pathway by Yaoling Luo, Jiayang Qu, Zhengyi He, Minhong Zhang, Zhengwei Zou, Lincai Li, Yuxian Zhang and Junsong Ye in Cell Transplantation
Footnotes
Acknowledgements
We would like to thank Qianqian Xu and Junming Chen for their help in animal experiments, as well as the Department of Obstetrics and Gynecology of the First Affiliated Hospital of Gannan Medical College and umbilical cord donors for their support.
Author Contributions
Conception and design of study: Y.L., J.Y.; Acquisition of data: Y.L, J.Q., Z.H., L.L., Y.Z.; Analysis and/or interpretation of data: Y.L., J.Q., M.Z., Z.Z.; Drafting of the manuscript: Y.L., J.Q., Z.H.; Revising the manuscript critically for important intellectual content: J.Y., Z.H.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research Ethics and Patient Consent
All experiments in this study were approved by the Ethics Committee of Gannan Medical University, Jiangxi Province, China (Approval Number: 2020057). Prior to umbilical cord collection, all participants signed a written informed consent and were informed that umbilical cords were used only for hUCMSC culture and subsequent animal study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the financial support received from The Foundation of Jiangxi Educational Committee (GJJ180791, GJJ201535), The Science and Technology Project of Jiangxi Provincial Health Commission (202130652, 202130627, 20191079), The Open Project of Key Laboratory of Prevention and treatment of cardiovascular and cerebrovascular diseases, Ministry of Education (XN201913), The Foundation of Technology Innovation Team of Gannan Medical University (TD201806), The Foundation of Technology Innovation Team of First Affiliated Hospital of Gannan Medical University (2021CXTD-08), Key Project Foundation of Gannan Medical University (ZD201831), First Affiliated Hospital of Gannan Medical University, Doctor Start-up Fund (QD066, QD076), The Science and Technology Project of Ganzhou (202101034530). The Science and Technology Project of First Affiliated Hospital of Gannan Medical University Fund (YJYB202005).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
