Abstract
The 2019 novel coronavirus disease (COVID-19) has lasted for over 3 years and has seriously affected the regular life trajectory of human beings. The severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) has caused non-negligible harm to people’s respiratory systems and multiple organs. Although the pathogenesis of COVID-19 has been fully elucidated, there is still a lack of effective and specific treatment for COVID-19. Mesenchymal stem cells (MSCs) or MSC-derived extracellular vesicles (MSC-EVs) by far have become the most promising candidates in preclinical studies and clinical trials, and MSC-related therapies have shown their potential to treat severe COVID-19. The multidirectional differentiation potential and immunomodulatory function of MSCs have allowed the cells to exert multiple cellular and molecular actions on different immune cells and organs. It is critical to understand the therapeutic roles of MSCs before entering the clinics for COVID-19 and other diseases. This review summarizes the recent progress of particular mechanisms underlying the immunomodulatory and tissue regenerative effects of MSCs toward COVID-19. We focused on discussing the functional roles of MSC-mediated effects on immune cell responses, cell survival, and organ regeneration. Besides, the novel discoveries and recent findings of MSC clinical application in patients with COVID-19 were highlighted. This will provide an overview of the current research on the rapid development of MSC-based therapies not only for COVID-19 but also for other immune-mediated/immune-dysregulating diseases.
Keywords
Introduction
The novel coronavirus disease 2019 (COVID-19) is an infectious disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) and is characterized by acute pneumonia and other symptoms of infection. According to World Health Organization, up until May 2023, COVID-19 has had a significant impact on global health and society, leading to the loss of 6.92 million lives, separation of families, and disruptions to daily life. However, it is important to note that the actual number of deaths caused by COVID-19 may be higher due to wide variations in the excess death estimates across the six World Health Organization regions 1 . The pandemic has highlighted the issue of extreme inequality worldwide, and the emergence of different virus variants has made the situation more challenging. Due to its ability to continually adapt to different hosts and produce mutations during replication, SARS-CoV-2 is expected to coexist with humans for the foreseeable future.
The SARS‑CoV‑2 is a β-coronavirus with a diameter of 60 to 140 nm. The particles are generally oval or spherical envelope viruses with a 30 kb positive single-stranded RNA genome, which causes the infection of the airway and alveolar epithelial cells and vascular endothelial cells2,3. There are four structural proteins: spike (S), envelope (E), membrane (M), and nucleocapsid (N) proteins in the virions. The E and M proteins form the viral envelope; the N protein binds to the virus’s RNA genome; and the S is responsible for receptor binding and virus–host membrane fusion via a specific receptor, the human angiotensin-converting enzyme 2 (ACE2) on the epithelial cells and endothelial cells 4 . SARS-CoV-2 infection commonly leads to COVID-19 pneumonia, which results in breathing difficulties due to fluid accumulation in the lungs. This is caused by exaggerated inflammatory hyper-responses and cytokine storms, which can lead to damage of the pulmonary tissue, impaired air exchange, and acute respiratory distress syndrome (ARDS). Studies have shown that cytokine storm is directly associated with an unfavorable prognosis in ARDS 5 . While COVID-19 primarily affects the lungs, it can also cause harm to other organs in the body such as the heart, brain, kidneys, intestine, liver, pharynx, and other tissues. Organ damage may result in long-term health problems post-COVID, including long-term breathing problems, heart complications, chronic kidney damage, stroke, and Guillain–Barre syndrome, which possibly affects people’s health in the very long term6–8. At least 65 million individuals worldwide are estimated to have long COVID, with cases increasing daily. However, current diagnostic and treatment options are insufficient8,9.
It is critical to find a safe and effective treatment for patients with severe COVID-19 10 . Although the pathogenesis of COVID-19 has been fully elucidated, there is still a lack of effective and specific treatment for COVID-19, and most treatments are limited to supportive care. The primary therapies to treat the disease are antiviral, anti-inflammatory and respiratory therapy 11 . Stem cells are endowed with the properties of self-renewal, multilineage differentiation potential and immunomodulation, making them an attractive form of clinical cell therapy. Stem cell therapy might be an active and essential part of SARS-CoV-2 infection treatment. Nowadays, among all stem cell therapies for COVID-19, mesenchymal stem cells (MSCs) or MSC-derived extracellular vesicles (MSC-EVs) by far have become the most promising candidates in preclinical studies and clinical trials12,13. MSCs have been found to possess regenerative properties, as well as immunomodulatory effects, which have been explored in various clinical trials aimed at treating conditions such as allergic inflammation, diabetes, rheumatoid arthritis, neurological disorders, and so on14–16. MSCs have been shown to have a potential therapeutic effect in reducing immune collateral damage by modulating the cytokine storm and immune overactivation17,18. And MSC implantation has been found to significantly reduce mortality in ARDS patients 19 . In addition, preclinical models and clinical trials have indicated the feasibility of MSC therapy for acute lung injury, suggesting its potential utilization in treating COVID-19. Therefore, MSC-related therapies have shown their potential to treat severe COVID-19 that cannot be treated.
As of May 2023, there were over 100 registered clinical trials in ClinicalTrials.gov investigating the potential of MSC and MSC-EV therapy for COVID-19. Despite the challenges posed by the disease, the promising therapeutic effects of MSCs and their established safety suggest that MSC therapy could be a viable option for improving outcomes in severe COVID-19 patients. However, the clinical application of MSC-based products still faces several challenges related to the heterogeneity of cell sources, varying passage numbers, optimal cell dosages, and frequency of MSC transplantation. These factors can significantly affect the efficacy of the treatment and need to be carefully considered and standardized to ensure consistent and reproducible results. This review summarizes the associated symptom and pathological responses in COVID-19, along with the recent progress of particular mechanisms underlying the immunomodulatory and tissue regenerative effects of MSCs toward COVID-19, highlighting the rationality of MSC utilization in the treatment of the disease.
COVID-19-Associated Syndrome, the Involved Pathology, and Immune Responses
COVID-19 Associated Syndrome
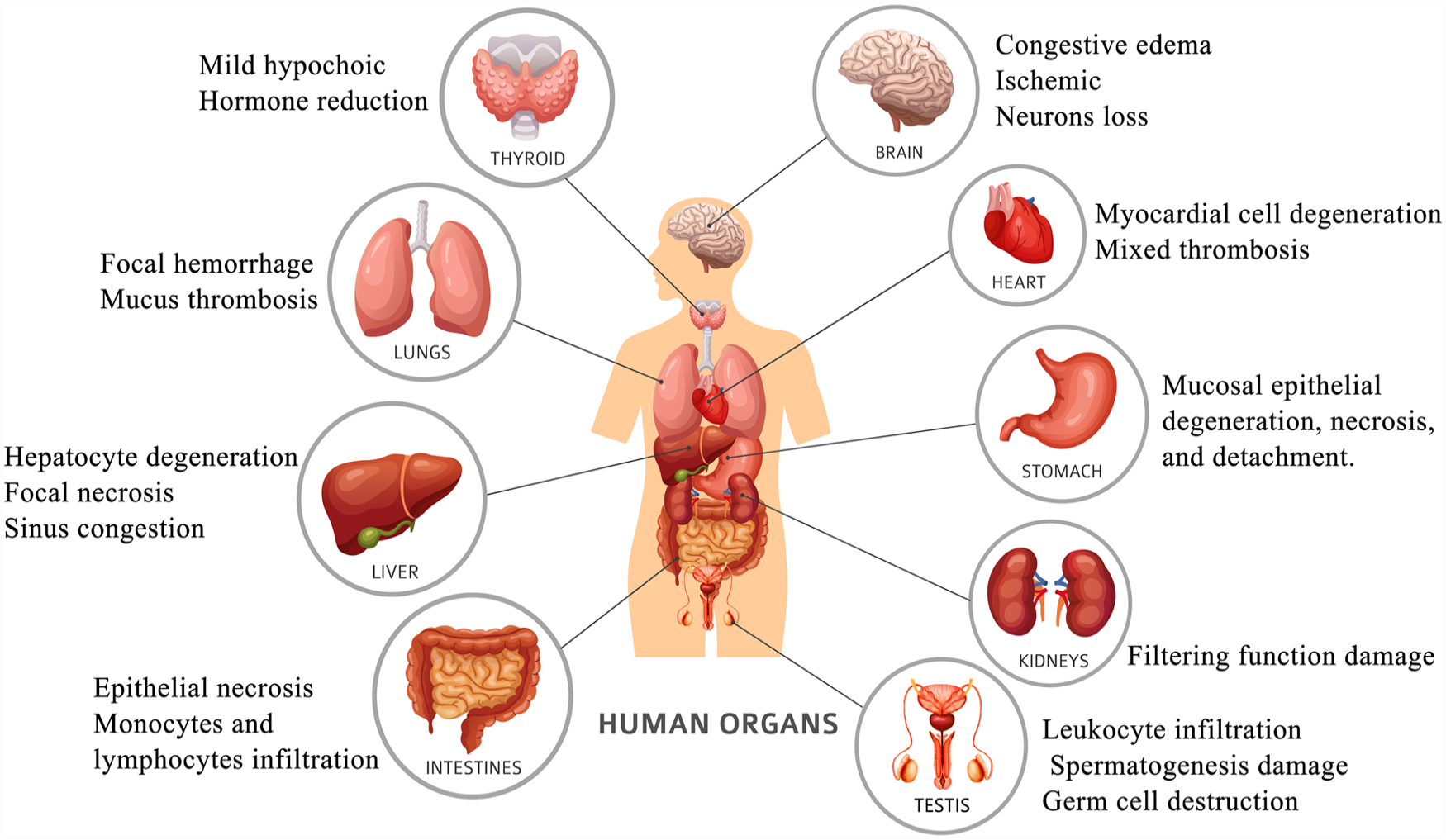
The main symptoms of COVID-19 were fever, dry cough, and fatigue after infection. The first symptoms in some patients can be also olfactory, sense of taste or smell loss. A few patients may also develop additional symptoms, such as nasal congestion, runny nose, sore throat, conjunctivitis, muscular soreness, diarrhea, and other symptoms. ARDS and multiple organ failure can quickly develop in extreme cases of individuals who develop dyspnea and/or hypoxemia within a week of the start of the condition (Fig. 1).
Pathological changes of organs caused by SARS-CoV-2. Following invasion through the ACE2 receptor, SARS-CoV-2 proliferates within cells, leading to the release of PAMPs and DAMPs and the secretion of excessive pro-inflammatory cytokines. This release is then detected by macrophages and dendritic cells in lung tissue, which triggers the production of type I IFN and other pro-inflammatory cytokines. This, in turn, leads to the production of cytokines and chemokines such as IL-6, IL-8, MCP-1, and IP-10, which promote local inflammation in the lung tissue and recruit neutrophils and monocytes from circulation to the infected area. After immunization, DCs presenting SARS-CoV-2 peptides activate Th1 cells, promoting the production of IFN-γ, TNF, and IL-2. These factors act on CD8+ T cells and B cells. Activated CD8+ T cells recognize and target cells presenting viral antigens through MHC class I molecules and perform cytotoxic killing. Activated B cells differentiate into plasma cells, which synthesize and secrete specific antibodies against the spike protein of SARS-CoV-2 to neutralize the virus. ACE2: angiotensin-converting enzyme 2; DAMP: damage-associated molecular pattern; DC: dendritic cell; IFN: interferon; IL: interleukin; IP-10: IFN-γ-induced protein-10; MCP-1: monocyte chemoattractant protein-1; MHC: major histocompatibility complex; PAMP: pathogen-associated molecular pattern; SARS‑CoV‑2: severe acute respiratory syndrome coronavirus 2; TNF: tumor necrosis factor.
The lungs of patients with COVID-19 had varied degrees of consolidation in their lungs, which predominantly manifested as exudative alveolitis and widespread alveolar damage 20 , alveolar loss of elasticity, and patchy peripheral hemorrhage in the lungs. Alveolar septum vessels exhibit edema, widening, hyperemia, and dilatation in segment 21 . In the alveolar cavity, serosity, fibrinous exudation, and hyaline membrane development were observed. At every level of the airway, some bronchial mucosa epithelium was observed peeled off. Mucus and exudate could be seen in the lumen. Mucus plugs are easy to form in small bronchus and bronchioles. It is possible to observe thrombosis (mixed thrombosis, translucent thrombosis), thromboembolism, and pulmonary vasculitis. In addition, hemorrhagic infarction, bacterial and/or fungal infections, and focal bleeding can be found in lung tissue8,9.
The ACE2 receptor, which allows the virus causes COVID-19 to infect cells, is more strongly expressed in the thyroid gland than in the lung22,23. A total of 75% of the COVID-19 patients showed diffuse mild hypoechogenicity on thyroid ultrasound examination 24 . The thyroid hormone in the patient declines at various rates depending on the various disease stages 23 . Specifically, the complications associated with COVID-19 are thyrotoxicosis (subacute thyroiditis, thyroxine thyrotoxicosis, Graves’ disease), hypothyroidism (central and primary hypothyroidism), and non-thyroidal illness.
Patients with COVID-19 had partial neuronal degeneration, edema, and congested brain tissue8,25; peripheral neuropathies, hemorrhagic and ischemic strokes, generalized central nervous system (CNS) disorders (seizures, meningitis, encephalitis), and other associated complications were observed in some patients 26 .
COVID-19-induced hypoxemia decreases the oxygen supply to cardiac tissue 27 . Degeneration, necrosis, interstitial hyperemia and edema of some myocardial cells, endothelial cell shedding, intimal or full-thickness inflammation, mixed thrombosis, thromboembolism, and infarction can all be found in the small blood arteries of the body’s major organs.
Alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) exhibited mild to moderate increases in the early phase of the disease in COVID-19 patients. Several patients’ blood albumin levels dropped while their serum bilirubin levels increased 28 . Hepatocyte degeneration, neutrophil-infiltrating localized necrosis, hepatic sinus congestion, lymphocyte and monocyte infiltration in the portal region, and microthrombosis were symptoms observed in some of the patients9,29.
Patients with COVID-19 had varying degrees of stomach mucosal epithelium degeneration, necrosis, shedding, lamina propria and submucosal monocytes, and lymphocyte infiltration. Some patients could also have acute pancreatitis, acute appendicitis, intestinal blockage, intestinal ischemia, bleeding peritoneum, or abdominal compartment syndrome9,30.
The tubular damage that COVID-19 causes to the kidneys is typically accompanied by observable anomalies in urine analysis. Moreover, impaired glomerular filtration function occurs, which typically manifests as apparent microthrombosis, high blood creatinine, raised urea nitrogen levels, and congested renal interstitial space 31 .
COVID-19 disrupts the blood–testis barrier through the induction of inflammatory cytokines and disruption of junctional proteins 32 . The patients had symptoms, including spermatogenesis damage, germ cell destruction, basement membrane thickening, hypogonadism, germ tube damage, interstitial cell reduction, and so on 33 .
Pathological and Immune Responses Involved in COVID-19
Patients with mild COVID-19 can successfully fend off viral invasion due to their quick immune responses. However, patients with COVID-19 often have abnormal hematological changes, including leukopenia, reduced blood plates, and significantly reduced lymphoid cells in peripheral blood, especially T lymphoid cells. CD4+, CD8+, T lymphocyte subtypes were significantly decreased34–36. In patients with severe cases, the immune response that is delayed in the early stages is unable to eliminate the virus, while an excessively late immune response can lead to organ damage and even death 37 . As shown in Fig. 2, the SARS-CoV-2 invades cells by recognizing and binding the specific receptor on the surface of target cells, ACE2, through spinous process protein 38 . The surface of lung cells, endothelial cells, alveolar macrophages, and dendritic cells (DCs) express ACE2, which makes these cells target cells for SARS-CoV-2 in the lung 39 . SARS-CoV-2 infiltrated host cells, where it started to reproduce and multiply continually, resulting in cell death after the invaded cells secreted excessive pro-inflammatory cytokines 40 . Cells damaged by pyroptosis release pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), which can be identified and recognized by macrophages and DCs resident in lung tissue 41 . Then, the macrophages and DCs are initiated to express Type Ⅰ interferon (IFN) together with other pro-inflammatory cytokines 42 . In the early stages of infection, type I IFN can effectively prevent the virus’s ability to replicate and spread through tissue cells 43 . The production of additional pro-inflammatory cytokines and chemokines, such as interleukin-6 (IL-6), IL-8, monocyte chemoattractant protein-1 (MCP-1), and IFN-γ-induced protein-10 (IP-10), is encouraged by the activation of such recognition events and will result in local inflammation in lung tissue 44 . This procedure will recruit neutrophils and monocytes from the circulation to the infected area 45 .

Process of SARS-CoV-2 invasion and human immune responses. In COVID-19 patients, focal hemorrhage is easy to be seen in lung tissue, and mucus thrombosis is easy to be seen in bronchioles. Thyroid ultrasound showed diffuse mild hypoechoic, and thyroid hormone decreased in different levels. The epithelium of intestinal mucosa degenerates, necroses, and falls off to varying degrees, and the lamina propria and submucosa monocytes and lymphocytes infiltrate. Cerebral congestion and edema, degeneration, ischemic changes and loss of some neurons, and occasional ganglia. Some myocardial cells degenerated and necrosed, and mixed thrombus was formed in blood vessels. Degeneration, necrosis, and abscission of gastric mucosa epithelium in different degrees. Renal tubules and glomerular filtration function are damaged. Leukocyte infiltration, spermatogenesis damage, and germ cell destruction in testis. ACE2: angiotensin-converting enzyme 2; DAMP: damage-associated molecular pattern; IFN: interferon; IFN-γ: interferon gamma; IL: interleukin; IP-10: IFN-γ-induced protein-10; MCP-1: monocyte chemoattractant protein-1; PAMP: pathogen-associated molecular pattern; SARS‑CoV‑2: severe acute respiratory syndrome coronavirus 2; TNF: tumor necrosis factor; WJ-MSCs: Wharton’s jelly–derived mesenchymal stem cell.
The CD8+ T cell and CD4+ type 1 T helper, or Th1 cell response are activated after immunization with DCs presenting SARS-CoV-2 peptides on their major histocompatibility complex (MHC) class II molecule on the cell membrane. One of the crucial events at the beginning of specific immunity is the activation of Th1 cells, which boosts the production of interferon-gamma (IFN-γ), tumor necrosis factor (TNF), and IL-2, which act on CD8+ T cells and B cells. Activated CD8+ T cells can recognize the target cells presenting viral antigens through MHC class I molecules and execute cytotoxic killing actions. Activated B cells differentiate into plasma cells that synthesize and secrete particular antibodies against the spike protein of SARS-CoV-2 to neutralize the virus 46 . The majority of patients with mild COVID-19 had their lungs cleaned of the virus by these immune cells, and both the body’s natural defenses and the adaptive immune response decreased 47 . However, in a subset of patients, evasion of innate immunity occurs and a later excessive/dysfunctional immune response is triggered 48 . SARS-CoV-2 infection may trigger dysregulated neutrophil cell death and neutrophil extracellular trap (NET) formation in both circulatory and infiltrating neutrophils, resulting in pulmonary injury, extensive inflammation, and the formation of a typical COVID-19 thrombus 49 . In addition, severe and critical cases of COVID-19 exhibit significantly lower levels of peripheral CD4+ and CD8+ T cells, particularly in cases with lymphocytopenia, impaired type I IFN production, and excessive activation of Th1 and inflammatory monocytes 16 . And regulatory T cells (Tregs) were found significantly reduced 50 . These decreases in immune cell numbers may be further exacerbated by a high viral load and immune imbalance, which can intensify the ongoing inflammatory response and elevate the risk of respiratory failure.
MSCs and Their Therapeutic Potential in COVID-19
MSCs are a heterogeneous subset of stromal cells that can be isolated from the fetal, adult, and other types of tissues, such as bone marrow, adipose tissue, viscera, amniotic fluid, amniotic membrane, umbilical cord, placenta, and so on51–54. The International Society for Cellular Therapy (ISCT) proposed a set of minimal criteria for the identification of MSCs in 2006. These criteria include (1) MSCs must adhere to plastic surfaces when in culture; (2) MSCs must express specific surface markers, including CD73, CD90, and CD105, and must lack the expression of CD34, CD45, CD11b, CD19 or CD79α, and HLA-DR; and (3) MSCs must be capable of differentiating into osteoblasts, adipocytes, and chondroblasts when cultured under specific conditions 55 . In addition, MSCs can differentiate into other embryonic lineage cells such as hepatocytes, insulin-releasing cells, and neuron-like cells14,51. It should be noted that MSCs are rather mesenchymal multipotent stromal cells than classical stem cells. The ISCT has recommended the use of the term mesenchymal stromal cell instead of MSC, as it more accurately reflects the multipotent nature of these cells and their ability to support tissue regeneration through mechanisms other than differentiation 56 . MSCs are presently considered to be viable candidates for cell therapy due to their high proliferation, multidirectional differentiation potential, and immunological modulatory functions 57 .
Function of MSCs
The effective therapeutic potential of MSCs is mainly based on two mechanisms. The first involves replacing damaged tissue with functioning tissue. MSCs injected into the body will naturally recognize the damage signal, migrate, and then differentiate into new cells at the damaged site 58 . However, the precise mechanisms behind this process are still not fully understood. There is a considerable amount of data indicating that transplanted MSCs are cleared rapidly in the recipient’s body59–61. The extent of in vivo MSC differentiation is still under investigation. The second mechanism through which MSCs promote the self-regeneration of damaged tissues is immunomodulation, which is believed to be their primary mechanism of action 62 . MSCs are able to actively interact with immune cells through paracrine activity and cell–cell contact. By paracrine activity, MSCs generate a significant quantity of soluble cytokines, chemokines, and growth factors that can aid in tissue healing 57 . Also, the capacity of MSCs to migrate and secrete is a major factor in how well they modulate the immune system 57 . MSCs interact with cells of the innate and acquired adaptive immune systems to control a number of effector activities in vivo. For instance, MSCs migrate to regions of wounded tissue and promote peripheral tolerance following in vivo delivery. By preventing the production of pro-inflammatory cytokines, tolerance bodies can aid in the survival and healing of injured tissue 51 . The immunomodulatory function of MSCs is unique and has been studied as a potential treatment for various immune diseases.
The Immunomodulation of MSC on the Immune Cells Which Is Involved in COVID-19
When infected, the count of inflammation markers and chemokines in COVID-19 patients significantly increase, such as C-reactive protein (CRP), lactic dehydrogenase, inflammatory cytokines, IL-1β, IL-6, IL-7, IL-2, TNF-α, granulocyte-colony stimulating factor (G-CSF), IP-10, and MCP-1 63 . These data show that the cytokine storm causes to increase in inflammatory factors which results in acute lung injury in patients. Both innate immunity and acquired immunity can be regulated by MSCs (Fig. 3). It is mainly involved in immune regulation through intercellular reactions with immune cells, including T cells, B cells, macrophages, monocytes, DCs, neutrophils, and so on. MSCs can also secrete chemokines, growth factors, cytokines, and EVs to exert their immunomodulatory properties 64 . These molecules are paracrine factors that are also found to be encapsulated in extracellular vesicles (EVs). MSC-EVs exhibit similar immunoregulatory functions to maternal MSCs65,66. Given the current extensive research and therapeutic potential of MSCs, it is recommended that MSCs may be functional in treating COVID-19 patients with compromised immune systems and damaged organs. Many preclinical and clinical data have proven the feasibility of MSCs.
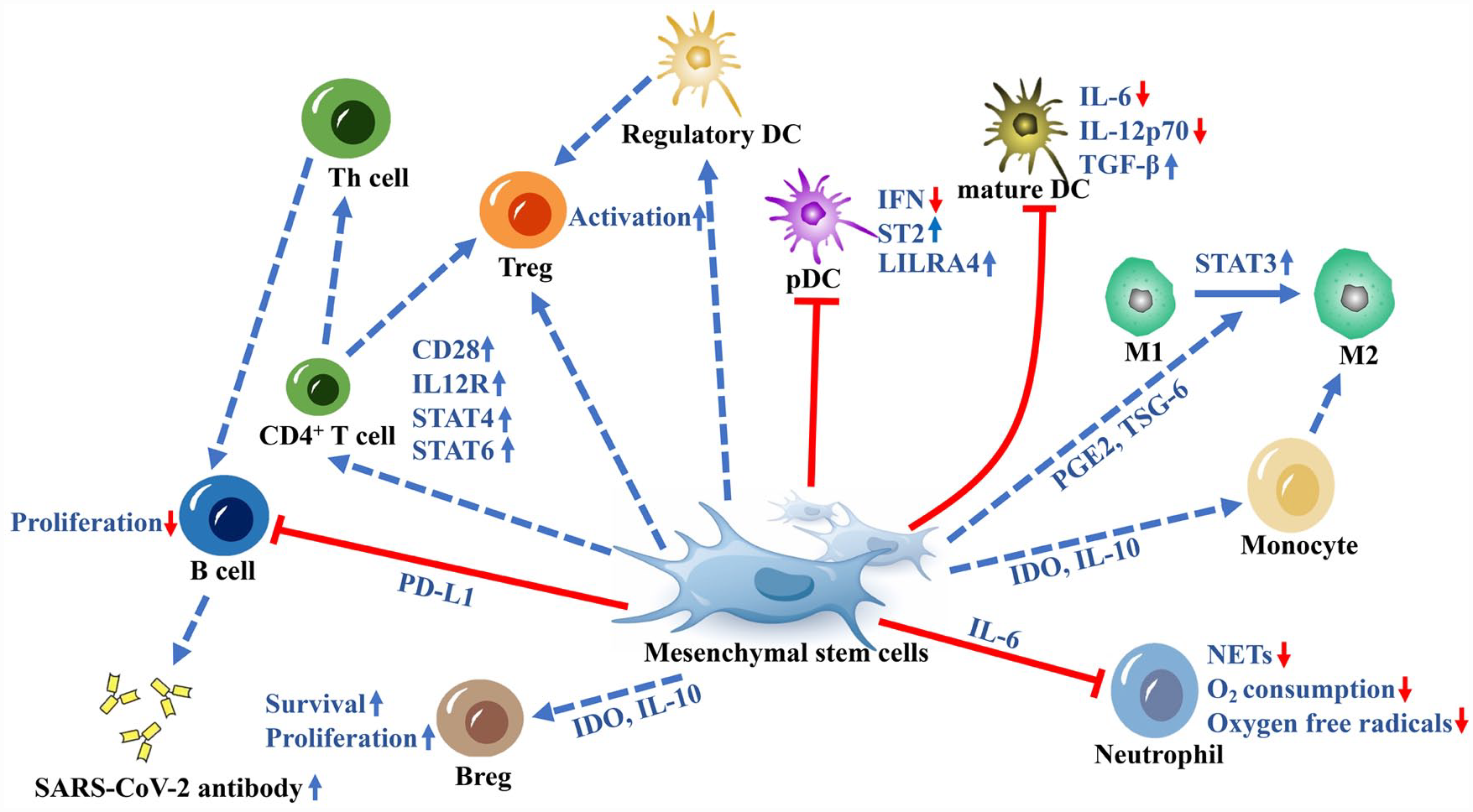
A schematic overview of the immunomodulation of MSC on the immune cells which are involved in COVID-19. MSC intervention hinders the maturation process of immature DCs, resulting in reduced secretion of IL-6 and IL-12p70, while promoting TGF-β production in DCs. By upregulating LILRA4 and ST2 expression on pDCs, MSCs decrease the presence of IFN-producing pDCs. Furthermore, MSCs induce the generation of regulatory DCs and facilitate the proliferation of Tregs. MSCs play a role in transforming M1-type macrophages into M2-type macrophages through the production of PGE2 and TSG-6. They also aid in the conversion of monocytes to M2 macrophages via IDO and IL-10. In terms of neutrophils, MSCs inhibit excessive oxygen consumption and oxygen radical production, thereby reducing the levels of NETs in patients. In addition, MSCs impede the proliferation of B cells through the action of PD-L1. They enhance the expression of CD28, IL12R, STAT4, and STAT6 in CD4+ T cells, promoting their differentiation into Th cells that can activate B cells for the production of SARS-CoV-2-specific antibodies. Moreover, MSCs directly enhance the activity of Tregs. By secreting IDO, which works in concert with IL-10, MSC can enhance the survival and proliferation of Bregs. Bregs: regulatory B cells; DC: dendritic cell; IDO: indoleamine 2,3-dioxygenase; IFN: interferon; IL: interleukin; MSC: mesenchymal stem cell; NET: neutrophil extracellular trap; pDC: plasmacytoid DC; PD-L1: programmed cell death ligand 1; PGE2: prostaglandin E2; STAT: signal transducer of transcription; TGF-β: transforming growth factor-β; Th cells: helper T cells; Tregs: regulatory T cells; TSG-6: tumor necrosis factor–stimulated gene 6 protein.
MSC effects on antigen-presenting cells
After infection with SARS-CoV-2, a high number of antigens contribute to a dramatic increase in DC levels inside the organism, which intensifies the cytokine storm and causes patients to experience respiratory failure, severe lung tissue destruction, and multiple organ failure, as well as new coronary pneumonia and eventually death. DC differentiation and maturation may be affected by MSCs. It was discovered that MSC interference prevented immature DCs from maturing into mature DCs, which affected the ability of DC endocytosis. At the same time, the secretion of IL-6 and IL-12p70 in DCs decreased, and the production of transforming growth factor-β (TGF-β) increased. Hence, it was unable for DCs to efficiently activate naive T cells or promote T lymphocyte proliferation 67 .
A particular cell type in the innate immune system is the plasmacytoid dendritic cell (pDC), often known as natural IFN-producing cells. During a viral contact, pDCs’ toll-like receptors 7 and 9 allow them to identify viral RNA and DNA and quickly produce huge amounts of type I IFNs. The production of type I IFNs by pDCs leads to the maturation of these cells into specialized antigen-presenting cells (APCs) that can stimulate T cells in the adaptive immune system 68 . By increasing LILRA4 (ILT7, a crucial inhibitory receptor of pDC), and ST2 (an activator of LILRA4) expression on pDCs, MSCs can reduce pDC production of IFN and pro-inflammatory cytokines 69 .
MSCs may induce a new Jagged-2-dependent population of regulatory DCs and promote their differentiation. MSCs can induce the production of regulatory DCs, Scal-1+ bone marrow–derived mesenchymal stem cell (BM-MSC)-driven DCs (sBM-DCs), from bone marrow–derived hematopoietic stem cells by suppressing IRF8 expression in an RBP-J-dependent way and by regulating histone H3 modifications. It is significant to note that sBM-DCs are a phenotypically distinct population of regulatory DCs that may inhibit IL-12 production in a way that is reliant on RBP-J by expressing less IRF8 and promoting the proliferation of CD4+CD25+ Tregs. In patients with COVID-19, the enhanced Tregs will lessen the severe inflammatory response by preventing CD4+ T cells from proliferating in vivo 70 .
MSC effects on macrophages
It was found that MSCs can not only recruit macrophages but also inhibit the pro-inflammatory activity of macrophages 71 . Song et al. 65 found that MSCs enter the body and briefly remain in the lungs before being quickly ingested by monocytes and migrating to other body areas. Monocyte phenotype and function will also be altered after phagocyting MSCs, indicating that MSC phagocytosis is able to regulate the acquired immune system cell 72 . Moreover, MSCs had the ability to create immunosuppressive components such as prostaglandin E2 (PGE2), TNF-stimulated gene 6 protein (TSG-6), lactate, kynurenic acid, and spermidine that help M1 macrophages switch from a pro-inflammatory to an anti-inflammatory phenotype 73 . For example, signal transducer of transcription-3 (STAT3) activation by PGE2 will cause a phenotypic transition in M2 anti-inflammatory macrophages, which can increase IL-10 production 74 . According to research by Aggarwal et al. 75 , PGE2 synthesis inhibitors greatly reduced the immunological effects of MSC by increasing the levels of TNF-α and IFN-γ in vivo. Moreover, MSC can inhibit macrophage entrance into inflammatory regions in a TSG-6-dependent way 76 . Indoleamine 2,3-dioxygenase (IDO) produced by MSC also regulates innate immune function by causing monocytes to develop into M2-like macrophages. These M2-like macrophages are associated with the regulation of T-cell proliferation in an IL-10-independent way, alleviating inflammation and promoting the immunomodulatory role of MSC 77 . Furthermore, apoptosis of adipose-derived MSCs (AD-MSCs) has also been demonstrated to change the phenotype of macrophages toward a regulatory and anti-inflammatory phenotype. Ghahremani Piraghaj et al. 78 found that apoptotic MSCs might enhance the expression level of IL-10 and decrease the production of TNF-α and nitric oxide (NO) in macrophages after being cultured with macrophages.
MSC effects on neutrophils
When the patient is infected with COVID-19, neutrophils are inappropriately attracted to the infection site under the stimulation of validated chemokines, which causes acute lung damage in target tissues 63 . According to clinical research, the number of neutrophils is correlated to the level of COVID-19 infection 79 . Due to their capacity for tissue repair and immunomodulation, MSCs can reduce excessive neutrophil recruitment and prevent neutrophil infiltration. MSCs have the ability to produce IL-6, which can stop neutrophils from consuming a significant amount of oxygen and generating oxygen-free radicals, which might cause the respiratory burst phenomena 80 . Furthermore, neutrophils can release NETs composed primarily of bactericidal granule proteins and chromatin fibers, which break down virulence components and eliminate bacteria. These inappropriately regulated NETs may propagate inflammation and microvascular thrombosis in COVID-19 patients81,82. Zhu et al. 69 discovered that MSCs were effective in promoting the production of SARS-CoV-2-specific antibodies in addition to lowering NET levels in the plasma of COVID-19 patients.
MSC effects on T lymphocytes
MSCs are well known to suppress effector T-cell activation and proliferation. At the same time, MSCs promote the development of Tregs by keeping macrophages, monocytes, and DCs in an immature or anti-inflammatory state 83 . MSC can directly influence T cells by secreting immunosuppressive molecules like hepatocyte growth factor (HGF), galectins, heme oxygenase 1 (HO1), IDO, leukemia inhibitory factor (LIF), NO, programmed cell death ligand 1 (PD-L1; also known as B7H1), PGE2, and TGF-β. It was shown that TGF-β secretion is necessary for both MSCs’ induction of Tregs and their regulation of T lymphocyte activation73,75,84. It was found that COVID-19 patients treated with MSCs had a significantly increased proportion and absolute number of CD4+ T cells and Tregs in their bodies 85 . MSCs can stimulate the maturation of T cells, as evidenced by the upregulation of early T-cell markers CD25 and CD69 under co-culture with MSCs 86 . In addition, RNAseq data from studies by Zhu et al. 69 and Bukreieva et al. 87 have demonstrated that MSCs upregulate the expression of genes involved in the inflammation response. These findings indicate that MSCs not only possess anti-inflammatory properties but also exert immune stimulatory effects on T lymphocytes. MSCs also have the ability potential to alter the different T-cell subsets, which could contribute to recovery from SARS-CoV-2 infection. MSC treatment increased the expression of IL12R, STAT4, and STAT6 in CD4+ T cells, and prevented T cells from secreting IFN-γ and TNF-α, which switched the cells from a pro-inflammatory state to an anti-inflammatory state13,88. Besides, MSCs can also suppress T-cell activation and proliferation, through direct cell-to-cell contact, which reduces the number of inflammatory factors. MSCs penetrate the aggravation site and become engaged with recovery through selectin and integrin. All things considered, by regulating the amount of T-cell-secreted inflammatory cytokines, MSCs can regulate inflammatory activity at the damaged site 89 .
The complex processes of T-cell development, activation, and differentiation include kinases from the mitogen-activated protein kinase (MAPK) family 90 . MSCs can regulate the MAPK-ERK/JNK signaling pathway, which in turn controls the percentage of CD28+ T cells in humans. MSCs can function as ERKi and JNKi to support T-cell function by inhibiting ERK and JNK phosphorylation 69 . By increasing the amount of intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1), MSCs have been demonstrated in vitro to reduce memory T cells’ responsiveness to APCs. This can effectively limit the overactivation of T cells 91 . Related studies have reported that ICAM-1 can increase the immunosuppressive ability of MSCs and contribute to the migration of MSCs in vitro. In addition, ICAM-1 may also affect the differentiation of MSCs by regulating the MAPK signaling pathway 92 .
MSC effects on B lymphocytes
Studies have shown that the severity of the disease is correlated with considerably high numbers of B cells in COVID-19 patients 93 . In contrast to COVID-19 patients who received a placebo, Zhu et al. 69 found that peripheral blood mononuclear cells (PBMCs) from COVID-19 patients treated with MSCs contained a lower proportion of B cell. By increasing both costimulatory and coinhibitory receptors in patients’ B cells, MSCs can control B-cell activation to levels adequate to block COVID-19 69 . MSCs exert immunomodulatory functions on B cells through both intercellular contact and paracrine effects 65 . First, MSCs can cause B-cell apoptosis by binding the Fas ligand (Fas-L) on MSCs to Fas receptors on B cells, which will cause downstream activation of Fas-associated domains and caspases. In addition, PD-L1 on MSCs will interact with the PD-1 receptor on B cells, preventing B-cell proliferation 94 . Through the intervention of T cells, MSCs can also prevent B-cell proliferation and antibody secretion while influencing the tendency of B cells. MSCs promote CD4+ T-cell differentiation into Th cells to promote B-cell activation, which leads to the production of antibodies specific to SARS-CoV-2 69 . Second, MSCs can regulate B cells by secreting soluble cytokines. For instance, MSCs can inhibit Caspase 3-mediated B-cell apoptosis by increasing the production of vascular endothelial growth factor (VEGF) in a cell contact-dependent manner 95 . One research, revealing that amelioration of refractory chronic GvHD by MSCs is mediated by an increase in CD5+ regulatory B cells, indicates that B-cell responses are likely to contribute to the immunomodulatory effects of MSCs. By secreting IDO, which works in concert with IL-10, MSCs can enhance the survival and proliferation of CD5+ regulatory B cells 96 .
MSC Effects on COVID-19-Associated Syndrome
Several studies have shown that SARS-CoV-2 infection would likely result in a catastrophic immunological response in humans due to the virus’s huge invasion of the body. This will cause the patient to produce a significant number of inflammatory factors, which will result in a cytokine storm97,98. The appearance of MSCs can reduce pro-inflammatory cytokines and increase anti-inflammatory cytokines 99 . Such approaches can accelerate the healing of the patient’s autoimmune system, control immunological response in a timely manner, and enhance cell activity, aiding in the regeneration of injured tissues97,100,101. After being activated, MSCs can proliferate and produce immune regulatory and anti-apoptotic signal factors to limit inflammation, so that the damaged site can be managed, and there is enough time to repair and rebuild, giving the right conditions for the recovery of the body (Fig. 4). Therefore, MSCs can have a favorable influence on the treatment of SARS-CoV-2 or other comparable viruses and speed up the recovery of the body 102 .

A schematic overview of the therapeutic effects of MSCs on the organs and tissues infected by SARS-CoV-2. In the lungs, they promote organ-like cell development and prevent pulmonary fibrosis. In the thyroid, MSCs reduce pro-inflammatory cytokine production and lower thyroid antibody levels. In the brain, MSCs help restore blood–brain barrier integrity and produce TGF-β1 to prevent neuronal death. MSC-EVs increase neurogenesis and angiogenesis. In the heart, MSCs improve heart function through TSG-6 secretion. MSCs can also reduce atherosclerosis and thrombosis in the cardiovascular system. In the liver, MSCs can differentiate into hepatocyte-like cells and inhibit B-cell activation, thereby reducing inflammatory factors. In the stomach, MSCs differentiate into gastric interstitial cells, secrete angiogenesis-promoting proteins, reduce inflammation, and enhance neovascularization. In the intestine, MSCs enhance epithelial cell proliferation and angiogenesis, reduce inflammation, oxidative stress, and intestinal cell apoptosis and pyroptosis. In the kidneys, MSCs regulate endothelial survival, and reduce inflammation, oxidative stress, and serum creatinine levels. In the testicles, MSCs boost testosterone levels, improve testicular size, and rejuvenate reproductive tissues through growth factor secretion. They also reduce acute oxidative damage, inflammation, and improve sperm concentration and motility. MSC: mesenchymal stem cell; MSC-EV: mesenchymal stem cell–derived extracellular vesicle; NRG1: neuregulin 1; SARS‑CoV‑2: severe acute respiratory syndrome coronavirus 2; TGF-β: transforming growth factor-β; TSG-6: tumor necrosis factor–stimulated gene 6 protein.
The Effects of MSCs on the Lungs Infected by SARS-CoV-2
Due to the connection between MSCs and inflammation, it can help COVID-19 patients recover from lung injury by partially inhibiting the inflammatory response brought on by virus invasion 21 . Damage to lung epithelial cells is the primary cause of the development of lung disorders. Therefore, the treatment of alveolar can be critical for the repair of lung injury and restoration of normal lung function. According to experimental research, introducing MSCs to a culture of lung progenitor cells can promote the development of an organ-like cell from distal lung epithelial cells (Epcam+Sca-1+). Epcam+Sca-1+ cells that have been stimulated by MSCs can develop lung cell-like organs that resemble lung cells, including bronchioles and alveolar cells 103 . MSCs can also secrete relevant signal factors to recruit lung progenitor cells, which can correctly repair lung injury. Relevant studies have demonstrated that the EVs secreted by MSC in an MSC-conditional medium (CM) are beneficial for lung repair103,104. At the same time, MSC-derived CM can activate pathways that help with lung healing, such as anti-inflammatory, survival-promoting, and anti-apoptotic pathways 105 .
MSC may also release a range of soluble media in inflamed lung tissue to lessen inflammation and safeguard alveolar epithelial cells 99 . To create a non-inflammatory microenvironment that is conducive to repairing lung epithelial cell damage and promoting alveolar fluid clearance, MSC’s immunomodulatory effect can decrease the production of pro-inflammatory cytokines and increase anti-inflammatory cytokines. This results in an increase in alveolar space volume and a decrease in alveolar thickening99,106. Studies have shown that 2 days following intravenous transplantation of MSCs, the clinical complaints of COVID-19 patients have greatly improved97,98,104,107. Intravenous injection of MSC can help the human body repair the lung microenvironment, protect alveolar epithelial cells, prevent pulmonary fibrosis, and treat pulmonary dysfunction and COVID-19 pneumonia. Such effects were achieved by the immune regulation function of MSCs together with a large number of cells in the lung101,104,108. In addition, a significant reduction in lung damage during a 1-year follow-up was observed in COVID-19 patients who received MSC administration, indicating MSC administration can exert positive effects on lung lesions and symptom relief with good tolerance over a long term87,109.
The Effects of MSCs on the Thyroid Gland Infected by SARS-CoV-2
Studies have shown that MSCs can perform a repair in animals with autoimmune thyroiditis, which is an organ-specific T-cell-mediated disease110–112. It was found that MSCs reduced pro-inflammatory cytokine production and regulated Th1/Th2 balance by reducing Th1 cytokine in autoimmune thyroiditis by studying the effects of MSCs and CTLA4Ig gene-transduced MSCs 113 . In addition, human umbilical cord mesenchymal stem cells (hUC-MSCs) were shown beneficial in rats with experimental autoimmune thyroiditis. hUC-MSCs significantly reduced serum thyroid antibody levels, ratios of IL-17α+/CD25+FOXP3+ cells, and serum IFN-γ/IL-4 in the experimental rats 112 . Treatment of hUC-MSCs can also elevate the expression of PTPN2 protein in autoimmune thyroiditis rat spleen lymphocytes, which will further weaken the phosphorylation of STAT3 in CD4+ T cells.
The Effects of MSCs on the Brain Infected by SARS-CoV-2
SARS-CoV-2 can cause persistent infection in the CNS in addition to brain damage 114 . Relevant data indicate that there are several cases with more severe neurological symptoms. Brain and spinal cord injuries, as well as other acute and chronic neurodegenerative illnesses, are thought to have their origins in CNS inflammation. Sun et al. 115 found that hUC-MSC-derived EVs can decrease IFN-γ, IL-6, TNF-α, and macrophage inflammatory protein (MIP)-1α levels in the CNS; delay CNS inflammation; and promote spinal cord injury recovery. Another study demonstrated that once the blood–brain barrier is compromised, intravenous MSC transplantation can help restore the blood–brain barrier’s integrity. Moreover, MSCs can block microglial activation, decrease the production of pro-inflammatory cytokines, and subsequently move into the substantia nigra pars compacta to produce TGF-β1, which can prevent neuronal death 116 . MSCs can thereby promote neuroprotective effects. Besides, MSCs and MSC-EVs produce neurotrophic factors that help the damaged spinal cord regain its neuromotor function 117 . Moreover, MSCs support neurovascular recovery and plasticity. EVs produced by MSCs can increase neurite remodeling, neurogenesis, and angiogenesis, as well as functional recovery and neurovascular function118,119.
The Effects of MSCs on the Heart and Blood Vessels Infected by SARS-CoV-2
The study discovered that on echocardiography, patients with cardiac injury caused by SARS-CoV-2 infection had an extremely low ejection fraction. When transplanted with MSCs, the patient’s heart function may return to normal 114 . This implies that MSCs may have a therapeutic effect on a heart affected by SARS-CoV-2. According to studies, BM-MSCs can greatly improve the effectiveness of myocardial production and heart function120,121. By direct injection, MSCs defined by CD34lowc-kit+CD140a+Sca-1high may differentiate into cardiomyocytes, endothelial cells, and pericytes or smooth muscle cells in humans 121 . Research indicates that intravenous injection of hMSCs can enhance heart function after myocardial infarction by reducing inflammation and infarct size through TSG-6 secretion 122 . According to recent research, endothelial progenitor cells may exchange mitochondria to regenerate and mend injured myocardium, and MSCs can make use of this ability to transfer mitochondria to assist in the therapy of heart disease or other organ problems123–126. Related research showed cardiomyocytes can be returned to a state similar to progenitor cells by fusing permanently or partially with stem cells and receiving functional mitochondria from MSCs 127 .
The SARS-CoV-2 can also lead to endothelial damage, vascular disease, associated vasculitis, and other conditions128–133. AD-MSCs can activate cell regeneration and heal through autocrine and paracrine pathways; they can quickly migrate into the injured site and differentiate into dermal fibroblasts, endothelial cells, and keratinocytes, which enhance the development of new blood vessels 134 . Human placental MSCs (hPMSCs) can reduce atherosclerosis and thrombosis by inhibiting agonist-induced platelet function 135 . MSC-derived EVs were also proven can prevent atherosclerosis by their contained miR-145 136 . In Kawasaki disease endothelial inflammation, hUC-MSCs can control inflammation and regulate the expression levels of CD54 and CD105 in vascular endothelial cells, which lessens inflammation in the blood vessels 137 .
The Effects of MSCs on the Liver Infected by SARS-CoV-2
There is a risk of liver damage from medicine used in the course of therapy for SARS-CoV-2, as well as immune-mediated liver damage or dysfunction brought on by the virus28,138–140. The study found that MSCs can migrate to the damaged liver and differentiate into hepatocyte-like cells101,141–143. MSCs have the ability to reduce liver inflammation by suppressing the production of inflammatory mediators, increasing the secretion of anti-inflammatory factors, and improving cell–cell contacts. In addition, they can facilitate liver regeneration and stimulate hepatocyte proliferation within the body142,143. MSCs can release soluble factors such as NO, PGE2, IDO, IL-6, IL-10, and human leukocyte antigen G to downregulate the infiltration of T cells, B cells, and monocytes in the liver141,144. These MSC-derived cytokines participate in the maintenance of interaction between immune cells and MSCs and the regulation of immune responses 141 . The MSCs can also inhibit B-cell activation, reduce immunoglobulin levels, polarize inflammatory macrophages toward surrogate macrophages, and release soluble factors such as IL-10 and IL-1Ra that help repair liver damage 141 . In addition to secreting a vast number of molecules into the extracellular space, such as soluble proteins, nucleic acids, lipids, and EVs, MSCs are capable of effectively repairing tissue damage and adjusting to fluctuations in physiological and pathological conditions. Paracrine action of their secretions regulates the liver immune microenvironment and promotes epithelial repair 143 . Under certain stimulation, hUC-MSC-derived EVs are able to highly express miR-455-3p, which inhibits macrophage infiltration, reduces inflammatory factors in the serum, and improves liver histology and systemic disease 145 .
The Effects of MSCs on the Stomach Infected by SARS-CoV-2
COVID-19 also affects the gastrointestinal tract, resulting in diarrhea, anorexia, nausea, vomiting, and abdominal pain146–148. MSCs have the functions of re-epithelialization, granulation tissue regeneration, TGF-β1 secretion, and inhibition of inflammation 149 . Studies have shown that intraperitoneal injection of BM-MSCs can accelerate the healing of gastric ulcers and repair mucous membranes in injured gastric mucosa150–152. It was shown that submucosal injection of AD-MSCs can accelerate the healing of stomach damage, MSCs differentiated into gastric interstitial cells while secreting angiogenesis-promoting proteins, by which to make the gastric glands fully repaired, reduce inflammatory infiltration, enhance neovascularization, and speed up damage repair149,153.
The Effects of MSCs on the Intestines Infected by SARS-CoV-2
The SARS-CoV-2 infection affects gut microbial homeostasis, reduces the richness and diversity of gut microbiota, causes immune dysregulation, and affects the lung–gut axis (a bidirectional interaction between the respiratory mucosa and the gut microbiota)154–156. Soontararak et al. found that MSC treatment significantly enhanced intestinal epithelial cell proliferation, the quantity of Lgr5+ intestinal stem cells, and intestinal angiogenesis in a mouse inflammatory bowel disease model. At the same time, MSC therapy partially improved intestinal repair and recovered the altered gut microbiota 157 . MSCs can help repair intestinal ischemia-reperfusion injury by reducing inflammation, relieving oxidative stress, and inhibiting intestinal epithelial cell apoptosis and pyroptosis158,159. It was found that MSC-derived neuregulin 1 (NRG1) can induce proliferative gene signatures, promote the formation of organoids from progenitor cells and improve gut healing after injury 160 . MSCs have the potential to provide niche factors that aid in maintaining the homeostasis of epithelial stem cells and bolstering intestinal defense mechanisms 161 . Research has shown that MSC EVs have the ability to influence the behavior of Th2 and Th17 cells in the mesenteric lymph nodes (MLNs). In addition, these EVs can help to restore the mucosal barrier and promote intestinal immune homeostasis through the action of TSG-6, a protein that supports the repair of damaged intestines. Overall, the use of MSC-EVs may hold promise as a therapeutic approach for conditions related to immune dysregulation in the gut162,163.
In the face of acute pancreatitis, MSCs can elevate Foxp3+ Tregs in lymph nodes and the pancreas while decreasing the expression of inflammatory markers such as TNF-a, IL-1β, and IL-6 in the pancreas164,165. It was found that MSCs can modulate acinar apoptosis through the chemokine receptor type 4 (CXCR4) axis. This interaction leads to the activation of both the death receptor protrusion and mitochondrial pathways, which ultimately promotes angiogenesis and tissue repair164,166. Placental chorionic plate–derived MSCs (CP-MSCs) have been found to have a remarkable ability to induce macrophage polarization from M1 to M2. This is achieved through the secretion of TSG-6, a protein that helps to relieve gland damage and systemic inflammation. It is particularly beneficial in chronic inflammation such as inflammatory bowel disease167,168.
The Effects of MSCs on the Kidney Infected by SARS-CoV-2
SARS-CoV-2 can cause significant damage to the kidneys, particularly through renal tubular damage. Patients with underlying kidney diseases are at increased risk of complications and mortality31,169. It was found that MSCs are able to regulate renal blood flow, capillary permeability, and endothelial survival, which will improve kidney repair170,171. By reducing the production of adverse factors and inhibiting excessive immune responses, MSCs have the potential to improve kidney function and promote tissue repair and regeneration in a range of kidney diseases172–175. Infusion of autologous BM-MSCs has been demonstrated to reduce serum creatinine levels in patients with polycystic kidney disease. This is because MSCs can inhibit the increase of blood urea nitrogen levels and relieve renal interstitial congestion, which could reduce inflammation and oxidative stress in the kidney 175 . Besides, TGF-β secreted by MSCs may play a critical role in fibrosis by promoting the epithelial-mesenchymal transition of tubular cells 176 . In addition to MSCs, MSC-EVs also have the ability to partially alter renal fibrosis and attenuate renal inflammation. Studies have demonstrated that the anti-inflammatory response, repair, and regeneration of renal tissue are all enhanced by the combination of EVs and soluble factors released by MSCs177,178.
The Effects of MSCs on the Testis Infected by SARS-CoV-2
The male reproductive tract and testis may experience histological or functional alterations as a result of the SARS-CoV-2 infection, which will have an adverse effect on male fertility33,179,180. Men who have SARS-CoV-2 may have sperm damage; however, it has been discovered that hPMSCs can mitigate this damage, encourage changes in sperm parameters, and boost testosterone levels, testicular size, and other factors. MSC therapy significantly increased the expression of proliferation genes (PCNA and KI67) and decreased the apoptotic genes (γ-H2AX, BRCA1, and PARP1) in the damaged testis 181 . Also, it was shown that BM-MSCs can improve sperm concentration and lower the possibility of defective spermatogenesis 182 . Men who are infected with SARS-CoV-2 may develop hypogonadism; however, MSCs can help cure the condition and restore normal gonadal function 183 . Bhartiya et al. 184 found that MSCs can rejuvenate reproductive tissues by secreting growth factors, which also promote the progenitor cells in reproductive tissues to differentiate into sperm. Research has demonstrated that MSCs may develop into progenitor cells and cells that resemble the male genitalia under specific conditions, aiding in the proper generation of sperm 185 . MSCs can develop into germ cells in the seminiferous tubules of the testis after transplantation. The structure of the seminiferous tubules, including spermatogonia, primary and secondary spermatocytes, spermatocytes, Sertoli cells, and interstitial cells can be improved by MSCs cultivated in a hypoxic environment186–188. Intratestis injection of BM-MSCs can effectively improve sperm count and motility 189 . By upregulating proliferating cell nuclear antigen and downregulating cyclin-dependent kinase inhibitor 2A (p16), MSCs can reduce aging and damage to reproductive organs 190 . MSCs can also boost levels of anti-inflammatory cytokines and prevent tissue damage. Studies have demonstrated that hUC-MSCs can preserve spermatogenic cells by reducing acute oxidative damage and inflammation191,192.
Therapeutic Potentials of MSC in COVID-19 Treatment
There are several treatments available for COVID-19; however, the effect of these treatments might not be effective and could be dangerous. So far, MSC therapy has been proven by multiple clinical trials to be very promising and effective to treat COVID-19. ARDS and cytokine release syndrome in COVID-19 are associated with high mortality 193 . The immunomodulatory and tissue repair capabilities of MSCs make them potential to treat COVID-19. Clinical studies have found that infused hUC-MSCs in subjects with COVID-19 and ARDS can significantly reduce inflammatory cytokines [granulocyte-macrophage colony-stimulating factor (GM-CSF), IFN-γ, IL-5, IL-6, IL-7, TNF, platelet-derived growth factor (PDGF), and RANTES (CCL5)]. Compared with controls which received two infusions of vehicle solution, researchers have not observed serious adverse events related to hUC-MSC infusions 194 . A phase I clinical trial conducted by Meng et al. 195 showed that UC-MSC is safe and effective in treating COVID-19, and no serious adverse events occurred. The patients who are infused with hUC-MSCs have their lymphocytes restored to ordinary levels in a short time 97 . Some researchers have found that in patients in the hUC-MSC treatment group, their clinical symptoms involving chest tightness, shortness of breath, and fatigue were significantly relieved in a shorter time than those in the control group. COVID-19 patients treated with hUC-MSCs have shown a significant reduction in signs of lung damage and shorter hospital stays 69 . Furthermore, studies have reported that patients with diabetes and COVID-19 complications who infused hUC-MSCs significantly reduced the exogenous insulin that they need 196 . These data suggest that infusing hUC-MSC treatment is safe and beneficial for COVID-19 patients.
In addition to hUC-MSCs, Leng et al. 98 demonstrated a significant improvement in clinical symptoms in patients with COVID-19 two days after transplantation of ACE2- MSCs. Following treatment, peripheral lymphocyte levels in patients were increased, the CRP decreased, and the CXCR3+CD4+ T cells, CXCR3+CD8+ T cells, and CXCR3+ NK cells, which immune cells were overactive and secreting cytokines, disappeared in 3 to 6 days. Two days after MSC transplantation, the levels of TNF-α in patients were significantly reduced in the experimental group compared with those of the control group 98 . In a phase I–II clinical trial using BM-MSCs for treating COVID-19, the treatment was found to be well tolerated and resulted in higher survival rates compared with the control group, with all patients surviving. While the CRP value of the two groups was similar, the D-dimer value was significantly lower in the MSC group 197 . Similarly, in another study involving the use of Wharton’s jelly–derived MSCs (WJ-MSCs) to treat severe COVID-19 patients, inflammation was reduced, and expressions of IL-10 and SDF-1 increased, while the expression of VEGF, TGF-β, IFN-γ, IL-6, and TNFα decreased. Antibodies to COVID-19 increased, chest imaging improved, and no serious complications were reported 198 . In a clinical trial conducted by Xu et al. 199 , where 26 COVID-19 patients were treated with human menstrual blood-derived MSCs, the mortality rate was lower in the MSC group compared with the control group, and improvements were observed in dyspnea and chest imaging results. Taken together, these prove that MSCs have beneficial therapeutic effects and therapeutic feasibility for patients with COVID-19 complications.
To date, there are 72 in progress or completed clinical trials using MSCs for the treatment of COVID-19 in ClinicalTrials.gov (Table 1). Among these clinical trials, 24 used hUC-MSCs, 17 used AD-MSCs, 7 used BM-MSCs, 4 used WJ-MSCs, 2 used dental pulp-derived MSCs, 1 used hPMSCs, and 17 used MSCs from other or unknown sources. In the vast majority of these clinical trials, MSCs were administered by the intravenous route with different dosages. Following the infusion of MSC, these cells can migrate to various tissues, with the majority initially accumulating in the lungs before a small proportion disseminates to other organs and tissues, which is advantageous for optimizing their role in the body’s immune response 200 . Although there are 21 completed clinical trials, most clinical trial outcomes remain unpublished. Due to the available published results, it is evident that MSC therapy has improved the survival rates in patients with severe/critical COVID-19, as well as those who have developed ARDS16,199,201. The timing of the MSC therapy is critical, for early initiation of MSC treatment may lead to a higher rate of extubation in severe COVID-19 patients202,203. However, there is still a lack of systematic comparison regarding treatment differences (i.e., cell source, administration route, dosage, etc.) with various MSCs.
MSC-Based Clinical Trials in Treating COVID-19 (Based on ClinicalTrials.gov).

AD-MSCs: adipose-derived mesenchymal stem cells; ARDS: acute respiratory distress syndrome; BM-MSCs: bone marrow mesenchymal stem cells; DW-MSCs: embryonic stem cell (SNUhES35)–derived mesenchymal stem cells; hPMSCs: human placental mesenchymal stem cells; hUC-MSCs: human umbilical cord mesenchymal stem cells; LMSCs: Longeveron mesenchymal stem cells; WJ-MSCs: Wharton’s jelly–derived mesenchymal stem cells.
Currently, the initial results of clinical trials mainly rely on improving lung function, serum indexes, hospital stay, and mortality. In COVID-19 patients, the expression of IP-10, MCP-1, MIP-1α, MIP-1β, PDGF, TNF-α, and VEGF were found to be severely out of the normal range 13 . But the evidence about the decrease in the concentration of pro-inflammatory cytokines was not general204,205. A meta-analysis done by Zhang et al. 206 showed that MSCs transplantation for COVID-19 has a remarkable effect on efficiency without altering blood levels of CRP, IL-2, and IL-6. Interestingly, transplantation of MSCs at phase 2 studies found no significant changes in IL-6, although the indicators of pneumonia on computed tomography imaging improved69,87,204,205. MSC transplantation did not affect IP-10 levels in critical COVID-19 patients204,207. Furthermore, it was shown that MSC transplantation in severe COVID-19 patients led to the elevation of plasma levels of pro-inflammatory cytokines such as IP-10, MIP-1α, G-CSF, and IL-10 without disease aggravation 87 . While IL-10 is generally associated with anti-inflammatory properties, it is worth mentioning that some studies have reported a pro-inflammatory effect of IL-10 in COVID-19 patients208–210. It has been shown that MSCs upregulate the expression of genes related to immune response development, specifically involving neutrophil activation, leukocyte migration, and phagocytosis. In addition, MSCs decrease the expression of genes associated with apoptosis in PBMCs obtained from COVID-19 patients87,211,212. There are still limited data on what MSCs play vital roles in the activation and maintenance of the immune response to SARS-CoV-2 infection either in vivo or in vitro. Therefore, more clinical data are necessary to elucidate the detailed mechanism and potential therapeutic targets of MSC clinical applications.
The Effects of MSC-EVs on COVID-19
When used as the kind of multifunctional stromal cells, MSCs are an essential choice for tissue repair, immunoregulation, and propagation due to their differentiation capability and paracrine properties 213 . The paracrine functions of MSCs are applied by secreting soluble cytokines and releasing EVs 214 . MSC-EVs are involved in immunological control, regeneration, and the transmission of miRNAs, mRNAs, and proteins from MSCs to the target cells. 213 . Because of their distinct characteristics, MSC-EVs can be used to treat respiratory diseases, especially COVID-19 and severe acute respiratory syndrome 215 . In the pathogenesis and epidemic mechanism of COVID-19, SARS-CoV-2 infected cells may be directed to the EV pathway, which packages virus components for secretion, further exacerbating the condition and its complications by facilitating viral spread and immune evasion216,217. In contrast, MSC-EVs can treat COVID-19-damaged endothelium and alveolar cells and reduce unproductive inflammation (cytokine storm) by providing anti-inflammatory chemicals 18 . It has been demonstrated that human MSC-EVs prevent DC maturation and prevent immature monocyte-derived DCs from uptaking antigens. MSC-EVs promoted the return of DCs to normal levels, thereby reducing the occurrence of inflammation in patients with COVID-19. These therapeutically beneficial EVs can be engineered into drug delivery platforms or vaccines, and can also be used to inhibit EV biogenesis and uptake to combat COVID-19 218 .
So far, there have been nine in progress or completed clinical trials using MSC-EVs for the treatment of COVID-19 (Table 2). Among these clinical trials, four were using BM-MSC-derived EVs, two were using hUC-MSC-derived EVs, two were using Zofin (EV fraction of human amniotic fluid), one was using AD-MSC-derived EVs, and another one that is not clear. EVs derived from allogeneic BM-MSCs (ExoFlo) have been proven the potential for the treatment of severe COVID-19 because of their safety, capacity to restore oxygenation, ability to downregulate cytokine storm, and capability to reconstitute immunity 219 . A single dose of ExoFlo has improved the patient’s clinical status and oxygenation, and their absolute neutrophil counts and T lymphocytes were significantly improved. Likewise, acute phase reactants, including CRP, ferritin, and D-dimer significantly declined 220 . Chu et al. 221 treated COVID-19 patients with EVs derived from hUC-MSCs, which resulted in improved absorption of lung lesions and reduced hospitalization time in mild patients, no allergic reaction or adverse event was observed. These results suggest that MSC-EVs may also be a promising approach for COVID-19 treatment.
Clinical Trials of MSC-EVs for the Treatment of COVID-19 (Based on ClinicalTrials.gov).

AD-MSC: adipose-derived mesenchymal stem cell; ARDS: acute respiratory distress syndrome; BM-MSC: bone marrow mesenchymal stem cell; EV: extracellular vesicle; hUC-MSC: human umbilical cord mesenchymal stem cell; MSC-EV: mesenchymal stem cell–derived extracellular vesicle.
Challenges and Perspectives
MSCs have been demonstrated to have great potential for the treatment of severe acute respiratory syndrome of SARS-CoV-2. Based on several early exploratory research and clinical applications, MSCs have been shown to be a potential innovative therapeutic protocol for the treatment of COVID-19, which has grown to be a severe threat to human health globally.
Many investigations and clinical trials have demonstrated that MSCs may be utilized to treat a wide range of common lung disorders, including idiopathic pulmonary fibrosis, acute lung damage, chronic obstructive pulmonary disease, asthma, and so on222–224. MSCs play a more active role in their clinical treatment, which is particularly evident in the treatment of diverse lung illnesses by boosting lung repair and controlling inflammation, despite the fact that the pathophysiology of the aforementioned diseases differs greatly from one another 19 . With the capacity for migration and homing, differentiation, immune regulation, anti-apoptotic characteristics, antiviral effect, EV secretion, and other mechanisms. It is possible to treat COVID-19 in a novel and efficient manner due to the different mechanisms of MSC therapy in a variety of lung diseases as well as other organ diseases.
The therapeutic effects of MSCs against COVID-19 are being intensively studied. And the utilized MSCs were isolated from different human tissues, including bone marrow, adipose, placenta, and so on. Most human tissues can be used for MSC isolation; however, it is still unclear which types of human tissues will yield the highest extraction efficiency, best results for tissue regeneration, or highest levels of safety. Preclinical experiments in acute lung injury models have shown that hUC-MSCs and BM-MSCs are more likely to be effective than AD-MSCs 225 . Further research and confirmation are still required, which will again challenge the medical application of MSCs.
MSCs localize to the injured site and can rapidly prevent additional tissue damage, effectively suppress the generation of pro-inflammatory mediators, and dramatically reduce collagen deposition in the extracellular matrix, which are some of the key strategies to successfully support tissue repair 226 . The anti-inflammatory, anti-apoptotic, and anti-oxidant properties of MSCs are thought to be strongly connected to their ability to support lung cell repair. This helps to maintain the integrity of the pulmonary endothelial-epithelial barrier 227 . It effectively aids in surfactant recovery and the decrease of atelectasis and alveolar edema. To further improve tissue healing and immunological control, MSCs can also release paracrine factors with anti-inflammation, anti-apoptosis, and anti-fibrosis properties. MSCs can actively interact with different immune cells to regulate innate and adaptive immunity. The immune regulation started by MSCs can quickly target different immune cells for adaptive immunity in response to the immune escape mechanism of SARS-CoV-2. Furthermore, MSCs prevent the invasion of the virus by paracrine function, the intervention and regulation of this immune response are enhanced by the secretion of soluble factors and EVs 13 . Taken together, by controlling the actions of immune cells taking part in pro-inflammation, MSCs promote the healing of tissue damage brought on by inflammation or other types of injury.
MSCs have emerged as a significant candidate and clinical standard for the treatment of COVID-19 due to the adaptive immune regulatory mechanism. It is important to keep in mind that the therapeutic use of MSCs to directly treat COVID-19 is still in the early stages of study although it has been 3 years since of the outbreak COVID-19. The clinical use of autologous MSCs is more suited for the safety and efficacy of this treatment because of the absence of long-term safety prediction and adequate data comparison. It will be extremely difficult to find the necessary number of MSCs in a timely manner without sacrificing quality. Treatment of severe COVID-19 patients with MSCs will encounter significant difficulties in the current situation.
Despite the fact that it is still challenging to determine the precise cause of this progressive and widespread lung parenchymal lesion, several studies have demonstrated that MSCs have great therapeutic potential and promising treatment options for idiopathic pulmonary fibrosis. In principle, hypoxic preconditioning of MSCs for transplantation might successfully increase the survival period of transplanted MSCs to boost the therapeutic efficacy of MSCs as MSCs can constantly release anti-inflammatory, anti-apoptotic, and anti-fibrotic substances under hypoxia 4 . Thus, it is suggested to use a hypoxic pre-condition to increase the implanted MSCs’ efficacy and extend their post-transplantation survival time. In addition, over-expression of anti-oxidant genes, anti-apoptotic genes, or growth factors in graft MSCs can significantly increase the survival rate of MSCs after transplantation in addition to hypoxic pre-treatment of the MSCs228,229. To facilitate the expression of cytoprotective genes by MSCs, it is helpful to have them undergo sublethal high stress in advance. Hypoxia, physical stimulation, small-molecule pharmaceutical preparations, heat shock, cytokines, and other treatments are examples of sublethal pre-treatments for MSCs in this situation 230 . Moreover, genetic modification can be used to increase the expression of anti-apoptotic proteins, chemokine receptors, and growth hormones, or to promote the transfection of survival genes to prepare MSCs for transplantation. These procedures, referred to as MSC pre-activation and microenvironment response, may be used to increase and/or modify the therapeutic potential of MSCs.
The therapy for COVID-19 is already widespread, and antiviral therapeutic approaches for this pneumonia are well-developed. However, severe viral pneumonia damage to lung tissue and other tissue is an irreversible process. Despite the fact that specific antiviral therapies are currently developing at a steady pace, clinical evidence has shown that to be sufficiently effective, these antiviral therapies must be used in the early stages of the pathological course, and they are of very little use in the late stages of infection231,232. This implies that patients with severe pneumonia may still only be treated using standard, all-purpose, and non-targeted methods, such as respiratory support and assistance, antipyretic medications, and analgesics, which may have little therapeutic outcomes and a challenging healing process. Following extensive research and analysis, MSCs have indeed been confirmed to be able to treat COVID-19 as a more direct and targeted treatment. MSC-based therapy has emerged as a significant new and ground-breaking approach to treating COVID-19: MSCs utilization may alleviate tissue damage caused by viral infection at the fundamental level. With the self-renewal, multilineage differentiation potential, and immunomodulatory properties, MSCs can directly reduce organ damage caused by SARA-CoV-2. MSCs have shown great prospects in curing severe viral pneumonia. Still, there are many gaps in the knowledge of MSCs for the cure of COVID-19. For instance, steroid therapy is currently the standard care item for COVID-19 patients; however, relevant data suggest that steroids may attenuate the beneficial effects of MSCs, which will negatively affect the therapeutic effect of MSCs233,234. Besides, the most suitable source type of MSCs, input dose, and the most suitable treating population are still under investigation.
Conclusion
At present, COVID-19 or other severe epidemic diseases will not be completely disappeared, and it is an urgent challenge for us to find a more innovative, effective, and secure treatment method for these diseases. MSCs have shown their therapeutic potential in COVID-19, which revealed an important breakthrough and effective treatment method for this pneumonia. However, more studies and investments are required to verify the concrete practice of MSCs in clinical application for not only COVID-19 but also other epidemic diseases. There are still many challenges to be overcome before MSCs can truly realize their value and prospect in the treatment of COVID-19 and its associated syndromes as well as other diseases.
Footnotes
Acknowledgements
Not applicable.
Author Contribution
Y.F., P.L., L.T., J.C., Y.C., L.S., and W.T. wrote the manuscript, X.F. designed and wrote the manuscript.
Availability of Data and Material
Not applicable.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research ethics and patient consent
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from NSFC (81900919), the Natural Science Foundation of Guangdong Province (2022A1515011817, 2017A030313105), the Special Fund for Science and Technology Innovation Strategy of Guangdong Province (pdjh2023b0388, pdjh2022b0381), the National College Student Innovation and Entrepreneurship Training Program (202314278012).
