Abstract
Epithelial regeneration is critical for barrier maintenance and organ function after intestinal radiation injury. Accumulating evidence indicates that the interleukin family members play critical roles in intestinal stem-cell-mediated epithelial regeneration. However, little is known about the relationship between interleukin 33 (IL-33)/ST2 axis and intestinal regeneration after radiation injury. We demonstrate here that IL-33 expression significantly increased after radiation treatment. Deficiency of IL-33/ST2 promotes intestinal epithelial regeneration, resulting in a reduction of mortality during radiation-induced intestine injury. Using ex vivo organoid cultures, we show that recombinant IL-33 promotes intestinal stem cell differentiation. Mechanistically, the effects of IL-33 were mediated by activation of transforming growth factor-β signaling. Our findings reveal a fundamental mechanism by which IL-33 is able to regulate the intestinal crypt regeneration after tissue damage.
Introduction
The gastrointestinal (GI) mucosa is a rapid turnover system, driven by mitotic activity of its stem cells characterized by expression of the Wnt target gene Lgr5 (leucine-rich-repeat-containing G-protein-coupled receptor 5, also known as Gpr49) 1 . Acute exposure to ionizing radiation causes lethal damage to the GI tract, a condition called the GI syndrome2,3, which has no Food and Drug Administration–approved effective treatment. Furthermore, the extreme sensitivity of GI cells to radiation toxicity largely determines the adverse side effects of anticancer radiation therapy and chemotherapy. Pathophysiology of GI tract damage involves depletion of the pool of stem cells residing at the bottom of the crypts of Lieberkühn, which impairs the regeneration of villi/crypt units and comprises the mucosal integrity and function4,5. The loss of epithelial integrity causes the malabsorption, fluid loss, electrolyte imbalances, and infection by resident enterobacterial flora, characterized clinically as anorexia, vomiting, diarrhea, dehydration, systemic infection, and in extreme cases, septic shock and death. Novel therapeutic principles are urgently needed to treat the radiation-induced intestinal injury. Recent published data have implicated that interleukin family members (IL-22, IL-11, and so on) are important in the maintenance of tissue homeostasis and play a vital role in tissue regeneration after injury6–9. However, the detailed functional role of the IL-33/ST2 axis in the intestinal regeneration after radiation injury is still ambiguous.
IL-33 is an IL-1 cytokine family member that was originally discovered in 2003 as a nuclear factor expressed by high endothelial venules 10 . IL-33 is widely expressed by different cell types, such as epithelial cells 11 , endothelial cells, fibroblasts 12 , and inflammatory cells, supporting a predominant role in the generation and development of inflammatory diseases. IL-33 has been described as an intracellular alarmin with broad roles in the pathogenesis of asthma 13 , rheumatoid arthritis 14 , cancer15,16, central nervous system disorders17,18, and inflammatory bowel diseases (IBDs) 19 . IL-33 is a bi-functional protein that functions as a traditional cytokine in the form of mature IL-33 or as a transcriptional regulator in the form of full-length IL-33. In fact, the mature form of IL-33 has significantly increased biological activity 20 . IL-33 acts as a nuclear factor in the normal state and binds to nucleosome proteins to exert transcriptional inhibition 10 . The IL-33 receptor ST2 (also known as IL1RL1), encoded by the IL1RL1 gene, comprises two primary isoforms: transmembrane ST2 isoform (ST2L) and soluble ST2 isoform. ST2 was considered as an “orphan” receptor 21 for many years, lacking a specific ligand, until Schmitz et al. 22 demonstrated that IL-33 is a ligand for ST2 in 2005. ST2 has structural features common to the IL-1 family of protein receptors, and extracellular is the three IgG-like domains used to recognize the receptor and the intracellular TIR domain. After it recognizes the ligand IL-33, it recruits the IL-1RAcP, a co-receptor of the IL-1 family protein, to generate downstream signals 23 . IL-33 activates downstream signaling molecules through the receptor ST2L acting on its target cell membrane, or directly enters the target nucleus as a binding factor to exert its biological effects. Accumulating evidence shows that IL-33 was involved in several intestinal diseases characterized by a Th2-type immune response, such as IBD 24 , ulcerative colitis 25 , trinitrobenzene sulfonic acid (TNBS)-induced enteritis 26 , and dextran sulfate sodium salt (DSS)-induced acute colitis 27 models. Although substantial progress has been made toward understanding the role of IL-33/ST2 axis in the intestinal diseases, there is little insight into the IL-33 regulating the intestinal regeneration after radiation damage.
Here, using IL-33/ST2 knockout (KO) mice and an intestinal organoid-based functional model, we found that the IL-33/ST2 axis represents a key regulator of intestinal regeneration after radiation injury. More importantly, IL-33 activates the transforming growth factor-β (TGF-β) signaling and subsequently promotes the intestinal stem cell (ISC) differentiation.
Materials and Methods
Mice
C57BL/6 and BALB/c mice were provided by the Shanghai Model Organisms Center, Inc. ST2-KO (ST2−/−) mice in the background of BALB/c were provided by Dr Andrew McKenzie at the MRC Laboratory of Molecular Biology, Cambridge, United Kingdom. IL-33-KO (IL-33−/−) mice in the background of C57BL/6 were provided by Dr Hiroshi Kiyonari at the Laboratory for Animal Resources and Genetic Engineering, Center for Developmental Biology, Institute of Physical and Chemical Research, Kobe, Japan. All mice used in this study were 8–10 weeks of age and of both sexes. Mice were maintained in a specific pathogen-free facility and exposed to a 12 h/12 h light/dark cycle. All animal protocols were approved by the institutional animal care and use committee of Fudan University.
Irradiation
Mice were irradiated with ionizing radiation using an X-RAD 320 Biological Irradiator (Precision X-ray, Inc, Madison, CT, USA) at a dose rate of 246 cGy/min (50 cm from the radiation source [source to skin distance, SSD] with 250-kVp X-rays, using 12 mA and a filter consisting of 2.5-mm Al and 0.1-mm Cu). Mice were irradiated with subtotal-body irradiation (SBI) as described previously 28 . Unanesthetized mice were held in jigs and placed under lead shielding to protect the head and front limbs.
Crypt Microcolony Assay
The microcolony assay was performed as described by Withers and Elkind 29 . Briefly, at 3.5 days after irradiation, small intestines were obtained and stained with hematoxylin and eosin as described previously. Surviving crypts were defined as containing 10 or more adjacent chromophilic non-Paneth cells, at least one Paneth cell, and a lumen. The circumference of a transverse cross-section of the intestines was used as a unit. Number of surviving crypts was counted per circumference.
Crypt Isolation and Organoid Culture
Small intestine tissue were collected and washed with cold phosphate buffer saline (PBS, PB2004Y; TBD, Tianjin, China). After digested by 10-mM ethylenediaminetetraacetic acid for 40 min, the collection was passed through a 100-μm-cell strainer (251300; SORFA, Zhejiang, China) to remove the villus. Crypts were washed and harvested by centrifuging at 4°C at 200 g. The collection of crypts was mixed with Matrigel (Corning, 356231; Glendale, AZ, USA) and cultured in standard organoid media containing Noggin (100 ng/mL, 50688-M02H; Sino Biological Inc., Beijing, China), R-spondin1 (500 ng/mL, 11083-HNAS; Sino Biological Inc., Beijing, China), and epidermal growth factor (EGF) (50 ng/mL, 50482-MNCH; Sino Biological Inc., Beijing, China). A different concentration of murine recombinant IL-33 (PeproTech, 210-33; Cranbury, NJ, USA) was added in supernatants, and the entire medium was changed every 3 days 30 .
Survival of Mice After Irradiation and Evaluation of Cause of Animal Death
Actuarial survival of animals was calculated by the product-limit Kaplan–Meier method. Cause of death was evaluated by autopsy, performed within 60 min of animal death or when terminally sick animals showing an agonal breathing pattern were euthanized by hypercapnia asphyxiation as per 28 . Tissue specimens were fixed in formaldehyde and stained with hematoxylin as above. GI damage was diagnosed as the cause of death when the small intestines showed denuded mucosa with nearly no villi or crypts apparent. Bone marrow damage was diagnosed as the cause of death when the marrow showed extensive matrix necrosis, widespread hemorrhage, and complete depletion of hematopoietic elements.
Histology and Immunofluorescence Staining
Intestinal tissue samples were fixed in 4% paraformaldehyde for 16–18 h and embedded in paraffin blocks. Tissue sections were subjected to H&E, immunohistochemistry (IHC), and immunofluorescence (IF) staining using standard procedures as described 31 . For IHC, the antibodies anti-Ki67 antibody (1:200, #12202; Cell Signaling Technology, Danvers, MA, USA) and anti-SOX9 antibody (1:100, A19710; Abclonal, Wuhan, China) were used, and the images were captured by using an Olympus DP73 microscope (Tokyo, Japan). For IF staining, goat polyclonal anti-IL-33 antibody (10 µg/mL, AF3626; R&D, Minneapolis, MN, USA) was used. All H&E and IF images were captured by using an upright widefield Zeiss Axio Imager 2 microscope.
Western Blot
Epithelial cells were isolated from small intestines of control and Irradiated mice. Total protein from epithelial cells was isolated using radioimmunoprecipitation assay (RIPA) buffer (PC 101; Epizyme, Shanghai, China). Lysates were centrifuged and separated on a gradient gel (8%–12%) via sodium dodecyl sulphate–polyacrylamide gel electrophoresis, blotted onto a polyvinylidene membrane, and analyzed using specific antibodies against murine IL-33 (0.5 µg/mL, AF3626; R&D, Minneapolis, MN, USA), ST2 (1 µg/mL, PA5-20077; Thermo Fisher Scientific, Carlsbad, CA, USA), and β-actin (1:10,000, HRP-60008, Proteintech, Wuhan, China). The detection was performed with a Millipore Immobilon Western Chemiluminescent HRP Substrate (WBKLS0500; ECL, Darmstadt, Germany) according to the manufacturer’s instructions.
RNA Scope
Intestinal tissue samples were fixed in 4% paraformaldehyde for 2 h dehydrated with 30% sucrose/PBS overnight and embedded in an optimal cutting temperature compound. RNAscope hybridization was performed with an RNAscope Multiplex Fluorescent Reagent Kit v2 (Advanced Cell Diagnostics, Newark, CA, USA) according to the manufacturer’s instructions. Fixed frozen samples were cut at 5 μm thickness. The tissue sections were treated with hydrogen peroxide for 10 min at room temperature, target retrieval reagent for 5 min at 100°C, and protease III digestion for 30 min at 40°C. The slices were incubated with a custom synthesized St2RNAscope probe (targeted 20 pairs of the St2 mRNA NM_001025602.3) for 2 h at 40°C, followed by amplifying hybridization processes and fluorophores staining steps.
RNA Sequencing
Small intestinal organoids were collected for RNA Seq after 6 days of in vitro culture with or without recombinant murine IL-33. Total RNA was extracted by using TRIzol (15596026; Invitrogen, Carlsbad, CA, USA). RNA-seq libraries were prepared using NEBNext UltraTM RNA Library Prep Kit for Illumina (New England Biolabs; Ipswich, MA, USA) following the manufacturer’s instructions, and index codes were added to attribute sequences to each sample. Briefly, ribosome-depleted RNA samples were fragmented and then used for first- and second-strand cDNA synthesis with random hexamer primers. The cDNA fragments were treated with a DNA End Repair Kit (New England Biolabs, Ipswich, MA, USA) to repair the ends, then modified with Klenow to add an A at the 3′ end of the DNA fragments, and NEBNext Adaptor (New England Biolabs, Ipswich, MA, USA) with a hairpin loop structure was ligated to prepare for hybridization. Purified double-stranded DNA was subjected to 12 cycles of PCR amplification, and the libraries were sequenced by an Illumina sequencing platform on a 150-bp paired-end run. Gene expression levels were calculated by the fragments per kilobase (fragments per kilobase of transcript per million mapped reads). The differentially expressed genes between each group were analyzed using the DESeq2 R package (1.16.1). The significant candidates were extracted with fold change (median) >2 or <0.5 and P value < 0.05. A heatmap analysis showed differential genes on the TGF-β pathway in control and treated groups. Gene set enrichment analysis (GSEA) was used to investigate the enrichment of TGF-β signal pathway.
Statistical Analysis
All data are given as mean ± standard deviation for various groups. Differences were analyzed by Student’s t test. Statistical significances were calculated and displayed compared with the control group. P ≤ 0.05 was considered significant.
Results
IL-33 Expression Is Increased After Radiation Treatment
Given that IL-33 appears to play a critical role in the intestinal regeneration after tissue damage, we sought to determine the expression pattern of IL-33 by staining intestinal tissues from untreated mice and mice treated with radiation. A histological analysis showed that an intense regeneration process is ongoing along the intestinal mucosa, as evidenced by the presence of regenerating crypts at 3.5, 5, and 10 days after 12-Gy radiation (Fig. 1A). IL-33 has been reported to be mainly expressed by pericryptal fibroblasts in the small intestine 32 . Consistent with this, we found that IL-33 expression was significantly elevated in the population of subepithelial cells neighboring the crypt base at day 5 after radiation (Fig. 1B, C). In contrast, the increase in ST2 expression was not observed after radiation (Fig. 1D, E). Finally, IL-33 upregulation in pericryptal fibroblasts was confirmed at the protein level using western blotting (Fig. 1F–H). Collectively, our data suggest that IL-33 can be induced and secreted by pericryptal fibroblasts in response to radiation-caused tissue damage in proximity to the ISC niche, thereby indicating an important role of IL-33 in regulating stem cell self-renewal and differentiation.
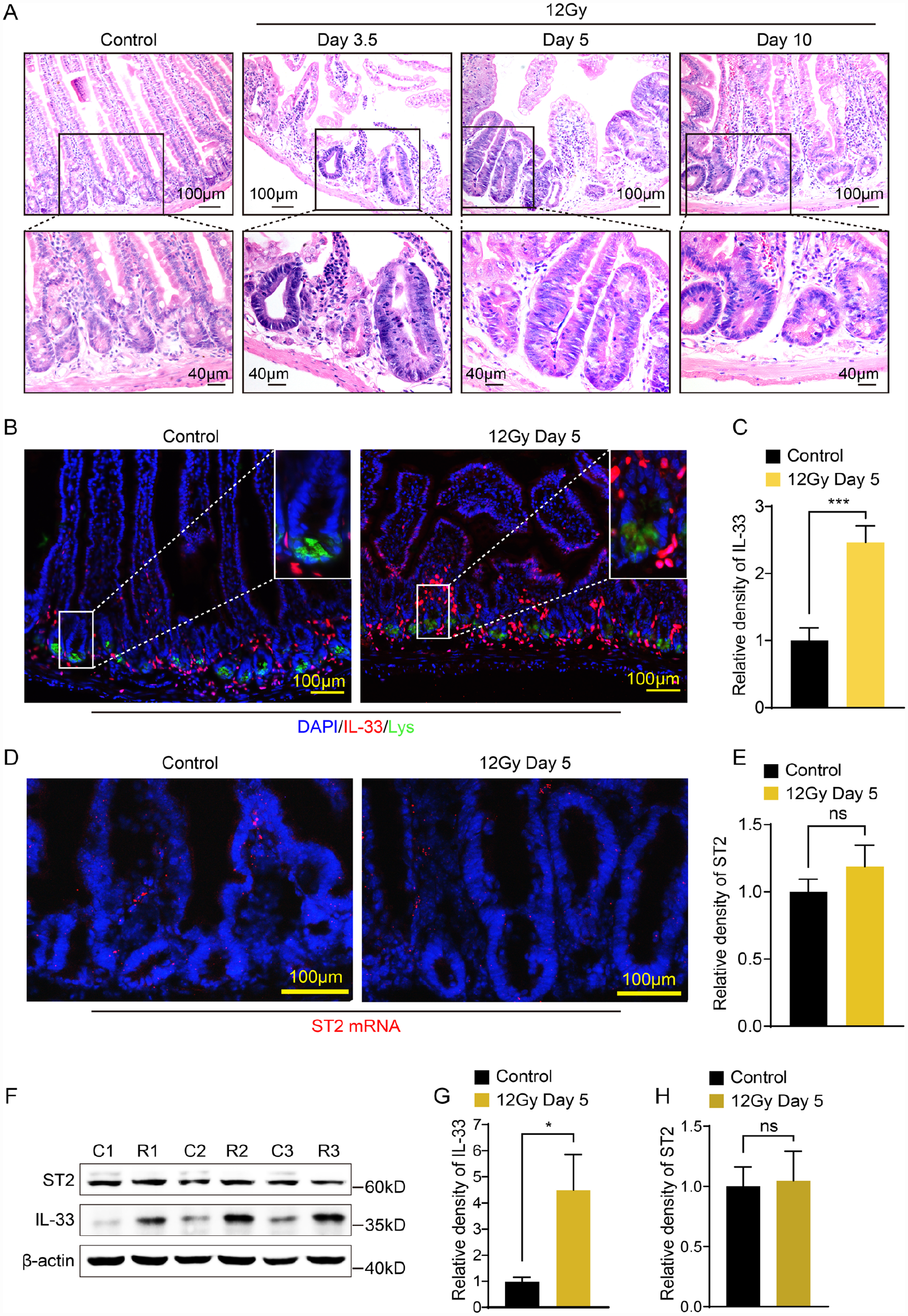
IL-33 expression increased after radiation treatment. (A) Regeneration process of intestinal crypt at different time points after 12-Gy irradiation. (B) IL-33 detection by immunofluorescence staining on small intestine cross-sections of wild-type and irradiated mice. (C) Quantification of IL-33 fluorescence intensity by Image J. ***P < 0.001 as calculated by unpaired t test. Error bars represent the mean ± standard deviation from three independent experiments. (D) ST2 mRNA detected by RNA Scope on small intestine cross-sections of wild-type and irradiated mice. (E) Quantification of ST2 mRNA fluorescence intensity by Image J. *P < 0.05 as calculated by unpaired t test. Error bars represent the mean ± standard deviation from two independent experiments. (F) Epithelial cells were isolated from small intestines for western blot. Blots are representative of three independent experiments. (G, H) Quantification of the gray values of the strips by image J. *P < 0.05 as calculated by unpaired t test. Error bars represent the mean ± standard deviation from three independent experiments.
IL-33 Deficiency Promotes Intestinal Epithelial Regeneration
In order to access the role of IL-33/ST2 axis in intestinal regeneration after damage, we used an established acute radiation-induced GI damage model in Il-33−/− mice in the C57BL/6 background and Il1rl1−/− mice in the BALB/c background. Given that radio-sensitivity varies in different inbred strains, in the initial experiments, we compared survival using WT mice on the C57BL/6 background and WT mice on the BALB/c background. Survival data showed that most BALB/c mice died from GI syndrome at 11 Gy, whereas C57BL/6 mice died at 14 Gy (Fig. S1). Therefore, 14-Gy and 11-Gy radiation were chosen for the following survival experiment in IL-33 KO mice and ST2 KO mice, respectively. We first demonstrated the absence of IL-33 had no effect on intestinal morphology and number of crypts under homeostasis (Fig. S2). Mice were exposed to SBI (13–14 Gy) after shielding the thorax, head, and neck, thus protecting the partial bone marrow. Hair graying induced by irradiation at 90 days was restricted to the exposed abdominal part, serving as a biological marker of the incident beam (Fig. 2A). Under this experimental condition, IL-33-deficient mice are better able to regenerate epithelial crypts after injury as assessed by the microcolony assay, developed by Withers and Elkind 29 , which measures the number of regenerating crypts per intestinal circumference at 3.5 days after radiation. The microcolony assay serves as a surrogate for ISC survival. Compared with their WT littermates, IL-33-deficient mice showed increased number of surviving crypts. Fig. 2B, C show that WT littermates contain 8.42 ± 0.82 regenerating crypts/circumference, and that increased to 15.06 ± 1.01 crypts after IL-33 knocking out. To test whether IL-33 deficiency promotes crypt regeneration through promoting proliferation, we stained the proliferation maker, which is also the marker of ISC self-renewal, Ki67, by IHC. The results showed that 3.5 days after 14-Gy radiation treatment, the number of Ki67 positive cells per crypt significantly increased in IL-33 KO mice compared with that in their WT littermates (Fig. 2D, E). This indicates that IL-33 deficiency promotes the intestinal regeneration after radiation damage through promoting proliferation.

IL-33 deficiency promotes intestinal epithelial regeneration. (A) Hair graying induced by irradiation was restricted to the exposed abdominal part. (B) Representative cross-sections of proximal jejunum from wild-type and IL-33 KO mice treated with 14-Gy SBI at 3.5 days. (C) Crypt microcolony assay in wild-type and IL-33 KO mice was analyzed at 3.5 days after 14-Gy radiation. Data (mean ± standard deviation) were quantified from 3 mice/group, with >40 circumferences/mouse. (D) Ki67 was stained in small intestines of C57 WT and IL-33 KO mice 3.5 days after 14-Gy radiation treatment by immunohistochemistry (IHC). (E) The numbers of Ki67 positive cells per crypt in (D) were counted and analyzed. (F) Actuarial survival of wild-type and IL-33 KO mice treated with 14-Gy SBI. Actuarial survival was calculated by the Kaplan–Meier Method. Number of animals/group is shown in parentheses. (G) C57BL/6 wild-type mice and IL-33 KO mice were treated with 13-Gy SBI, and body weight loss was recorded. The difference in body weight loss was calculated by the ratio of day 0 in each mouse. Error bars represent mean ± standard deviation. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 as calculated by unpaired t test. SBI: subtotal-body irradiation; KO: knockout.
To examine whether IL-33 deficiency also improves the animal survival after exposure to high-dose radiation, the effect of 14 Gy SBI on WT mice was first tested. A single-dose fraction of 14 Gy SBI induces GI syndrome and death of animal after exposure, verified by the autopsy results showing total or near total depletion of the crypt compartment and no extensive matrix necrosis in bone marrow (Fig. S3). Animal survival was evaluated by Kaplan–Meier curves. With a single dose of 14 Gy, 100% of IL-33 KO mice continued to survive beyond 90 days (Fig. 2F) compared with 50% of WT mice survival. To observe whether IL-33 KO affects the changes of body weight of mice after radiation, we reduced the radiation dose to 13 Gy to obtain more survived mice. Consistently, radiation-treated IL-33 KO mice had less weight loss over time than similarly radiation-treated WT mice (Fig. 2G). Thus, IL-33 deficiency promotes intestinal epithelial regeneration and animal survival after radiation damage.
ST2 KO Leads to Increased Intestinal Epithelial Regeneration
To further assess the role of IL-33/ST2 axis on the intestinal regeneration after tissue damage, we applied ST2 KO mice on the BALB/c background. ST2 deficiency does not affect intestinal homeostasis (Fig. S4). In consistence with IL-33 deficiency, ST2 KO mice showed more regenerating crypts than BALB/c wild-type mice at 3.5 days after 11-Gy X-ray abdominal radiation (Fig. 3A, B). Similar data were obtained in 10-Gy and 13-Gy experiments (Fig. 3B). With a single dose of 11 Gy, 83% of ST2 KO mice continued to survive beyond 90 days compared with 16% of WT mice survival (Fig. 3C). Furthermore, sublethal dose radiation-treated (10 Gy) ST2 KO mice had less weight loss over time compared with similarly radiation-treated WT mice (Fig. 3D).

ST2 deficiency leads to increased intestinal epithelial regeneration. (A) Representative cross-sections of proximal jejunum from wild-type and ST2 knockout mice treated with 11-Gy SBI at 3.5 days. (B) Quantitative comparison of regenerative crypts in small intestine of wild-type and ST2 knockout mice treated with 10, 11, and 13 Gy. Data (mean ± standard deviation) were quantified from two mice/group, with 40 circumferences/mouse. (C) Kaplan–Meier survival curves of wild-type and ST2 knockout mice treated with 11-Gy SBI. The number of animals per group is shown in parentheses. (D) BALB/c wild-type mice and ST2 knockout mice were treated with 10 Gy, and body weight loss were recorded. Error bars represent mean ± standard deviation. *P < 0.05; **P < 0.01; ***P < 0.001, as calculated by unpaired t test. SBI: subtotal-body irradiation.
IL-33 Promotes the ISC Differentiation by Activating TGF-β Signaling
Our previous studies found that IL-22 directly targeted ISCs, inducing STAT3 phosphorylation and promoting the ISC expansion 6 . Those studies lead us to hypothesize that IL-33 may also targets ISCs. Recent development of an ISC organoid culture system allows us to visualize these cells long term in vitro and define their functional and molecular phenotypes after IL-33 treatment. We isolated small intestinal crypts and cultured them under standard condition with or without IL-33. We examined whether IL-33 could regulate the ISC differentiation by using the organoid model. Interestingly, IL-33 significantly increased the budding of organoids after just 6 days (Fig. 4A, B). Furthermore, as with crypt-derived organoid, ST2 deletion abolishes the effect of IL-33 on crypt budding (Fig. 4C, D). The organoid model showed that IL-33 promotes the ISCs’ differentiation. To further reveal the role of IL-33 in the differentiation of ISCs from another perspective, we performed IHC staining Ki67 (for transient-amplifying cells) and SOX9 (for progenitor cells). After analyzing the number of Ki67- and SOX9-positive cells in crypts, we found that IL-33 deficiency significantly augmented the number of transient-amplifying cells and progenitor cells, indicating that IL-33 deficiency significantly promoted ISC self-renewal, and in other words, significantly diminished ISC differentiation (Fig. 4E–H). To uncover the underlying mechanism by which IL-33/ST2 axis regulates the differentiation of ISCs, we collected the organoids from the IL-33-treated group and control group after 6 days and applied them to RNAseq analysis. Comparative gene expression profiling by RNAseq of the 11 well-known TGF-β target genes showed an upregulation in IL-33-treated organoids (Fig. 4I). To evaluate whether TGF-β signaling was activated by IL-33, we performed a GESA, assessing the expression of TGF-β target genes. Expression of the TGF-β target gene signature was significantly enriched in IL-33-treated organoids (Fig. 4J). Thus, we found a promising clue for the mechanism that IL-33 promotes the ISC differentiation by activating TGF-β signaling.

IL-33 promotes the intestinal stem cell differentiation by activating TGF-β signaling. (A) Representative pictures of organoids cultured from crypts in C57BL/6 wild-type mice with/without IL-33 for 6 days. (B) The number of buds was calculated in organoids cultured with/without IL-33 (n = 200 organoids per group). (C) Representative pictures of organoids from BALB/c wild-type or ST2 KO mice cultured with/without IL-33(100 ng/ml) for 6 days. (D) Organoid buds from BALB/c wild-type and ST2 KO mice with or without IL-33 (WT: n = 152, WT + IL-33: n = 100, ST2 KO: n = 117, ST2 KO + IL-33: n = 78). (E, G) Representative images of IHC staining Ki67 and SOX9 in small intestines derived from C57 WT and IL-33 KO mice. (F, H) The numbers of Ki67 and SOX9 positive cells per crypt in (E, G) were counted and analyzed. (I) The heatmap of significantly upregulated gene in TGF-β signaling pathway analyzed by RNAseq of intestinal organoids cultured with or without IL-33 for 6 days. (J) GSEA of TGF-β targeted genes in organoids treated with IL-33. Error bars represent the mean ± standard deviation. ***P < 0.001; ****P < 0.0001 as calculated by unpaired t test. SBI: subtotal-body irradiation; KO: knockout; GSEA: gene set enrichment analysis; TGF-β: transforming growth factor-β.
Discussion
IL-33 is a newly discovered cytokine of the IL-1 family and plays an important role in the development and progression of IBD 33 . Most studies focus on the roles of IL-33 in regulating the intestinal immunity, as an alarmin 34 ; little is known whether IL-33 could directly target the ISC compartment. Sedhom et al. 35 reported that IL-33 impairs epithelial permeability independent of intestinal inflammation by negatively regulating wound healing time in the colon. Given IL-33 expression was significantly elevated in pericryptal fibroblasts neighboring the crypt base, this finding indicates the pericryptal fibroblasts might serve as mucosal sensors of radiation damage, translating danger signals to neighboring ISCs by secreting IL-33. Stem cell niches are critical for damage-induced intestinal regeneration 36 . Thus, in addition to the stromal and epithelial components of the ISC niche that are essential for normal ISC maintenance, IL-33 provides evidence for an immunological contribution to the ISC niche that is activated to regulate the intestinal regeneration after damage.
In this report, we found that IL-33 links immunity to epithelial regeneration by acting directly on ISC differentiation. Deficiency of IL-33/ST2 promotes intestinal epithelial regeneration, resulting in a reduction of mortality during radiation-induced intestine injury. However, our data do not rule out the possibility that, in vivo, the augmentation of epithelial regeneration by IL-33 deletion might be further supported by other IL-33 responding immune cells. Indeed, to answer this question, advanced mouse models with conditional deletion of ST2 (such as ST2-villin-cre mice 37 ) in intestinal epithelial cells are necessary. Recombinant IL-33 activates the TGF-β signaling, and promotes the ISC differentiation. By acting directly on ISC differentiation, the immune system is thus able to regulate intestinal regeneration. However, the potential mechanisms underlying how IL-33 activates TGF-β signaling remain unknown and will require further studies for elucidation.
In summary, our data show an unexpected role of IL-33 in the intestinal regeneration after radiation damage. These data identify the IL-33/ST2 axis as a potential therapeutic target in radiation-induced intestinal injury.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231177377 – Supplemental material for Interleukin-33 Potentiates TGF-β Signaling to Regulate Intestinal Stem Cell Regeneration After Radiation Injury
Supplemental material, sj-docx-1-cll-10.1177_09636897231177377 for Interleukin-33 Potentiates TGF-β Signaling to Regulate Intestinal Stem Cell Regeneration After Radiation Injury by Ruoyu Guan, Mengxue Pan, Xiaoya Xu, Lixia Du, Xinxin Rao, Guoxiang Fu, Tao Lv, Long Zhang, Yuanchuang Li, Peiyuan Tang, Yi Zhou, Yanqing Wang, Zhen Zhang, Jianjun Gao, Hong Zhou, Wenli Mi and Guoqiang Hua in Cell Transplantation
Supplemental Material
sj-docx-2-cll-10.1177_09636897231177377 – Supplemental material for Interleukin-33 Potentiates TGF-β Signaling to Regulate Intestinal Stem Cell Regeneration After Radiation Injury
Supplemental material, sj-docx-2-cll-10.1177_09636897231177377 for Interleukin-33 Potentiates TGF-β Signaling to Regulate Intestinal Stem Cell Regeneration After Radiation Injury by Ruoyu Guan, Mengxue Pan, Xiaoya Xu, Lixia Du, Xinxin Rao, Guoxiang Fu, Tao Lv, Long Zhang, Yuanchuang Li, Peiyuan Tang, Yi Zhou, Yanqing Wang, Zhen Zhang, Jianjun Gao, Hong Zhou, Wenli Mi and Guoqiang Hua in Cell Transplantation
Footnotes
Acknowledgements
We thank Dr Andrew McKenzie (the MRC Laboratory of Molecular Biology, Cambridge, United Kingdom) for providing the ST2−/− mice. We also thank Dr Hiroshi Kiyonari (Laboratory for Animal Resources and Genetic Engineering, Center for Developmental Biology, Institute of Physical and Chemical Research, Kobe, Japan) for providing the IL-33−/− mice.
Author Contributions
G.H. and W.M. conceptualized, designed, and supervised the study. R.G., M.P., X.X., and L.D. performed most of the experiments, collected and analyzed most of the data, organized the figures, and co-wrote the manuscript. X.R., G.F., T.L., L.Z., Y.L., and P.T. performed some of the experiments, collected and analyzed some of the data, and co-wrote the manuscript. Y.Z., Y.W., Z.Z., J.G., and H.Z. participated in the discussions along the study and provided technique support. All the authors revised and approved the manuscript.
Availability of data and material
All data in this paper are available upon reasonable requests.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
The national guidelines for the care and use of animals have been followed, and that the study have been approved by the Institutional Animal Care and Use Committee of Fudan University.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Research Ethics
All animal protocols were approved by the institutional animal care and use committee of Fudan University (No. 201904002Z).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (82173461, 31670858, 82271248) and Innovative Research Team of High-level Local Universities in Shanghai.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
