Abstract
Human small intestinal crypts are the source of intestinal stem cells (ISCs) that are capable of undergoing self-renewal and differentiation to an epithelial layer. The development of methods to expand the ISCs has provided opportunities to model human intestinal epithelial disorders. Human crypt samples are usually obtained from either endoscopic or discarded surgical samples, and are thereby exposed to warm ischemia, which may impair their in vitro growth as three-dimensional culture as spheroids or enteroids. In this study we compared duodenal samples obtained from discarded surgical samples to those isolated from whole-body preserved cadaveric donors to generate in vitro cultures. We also examined the effect of storage solution (phosphate-buffered saline or University of Wisconsin [UW] solution) as well as multiple storage times on crypt isolation and growth in culture. We found that intestinal crypts were successfully isolated from cadaveric tissue stored for up to 144 h post-procurement and also were able to generate enteroids and spheroids in certain media conditions. Surgical samples stored in UW after procurement were sufficiently viable up to 24 h and also allowed the generation of enteroids and spheroids. We conclude that surgical samples stored for up to 24 h post-procurement in UW solution allowed for delayed crypt isolation and viable in vitro cultures. Furthermore, in situ, hypothermic preservation in cadaveric duodenal samples permitted crypt/ISC isolation, and successful culture of spheroids and enteroids from tissues held for up to 6 days post-procurement.
Introduction
In vitro intestinal epithelial cell (IEC) culture methods have significantly enhanced our understanding of epithelium biology and have served as a source for high-throughput screening for drug discovery and toxicology. In addition, these stem cell-based methods are being used to model monogenic, epigenetic, inflammatory, and infectious intestinal disorders in a dish, and may one day be used as novel cell or tissue replacement therapy to treat various intestinal diseases 1,2 .
Crypt base columnar cells (CBCCs) are the rapidly cycling intestinal stem cells (ISCs) continuously undergoing self-renewal and differentiation into the various epithelial lineages of the gut. Specifically, goblet, enteroendocrine, Paneth, Tuft, M cells, and enterocytes are six differentiated epithelial lineages that arise from ISCs 3 . Various subepithelial lymphoid, endothelial, mesenchymal, and other cells form an important niche contributing soluble factor, required for self-renewal and differentiation during homeostatic and injured conditions 3 . Within the in vitro setting, these soluble factors are added exogenously to support ISCs expansion and differentiation on various extracellular matrices, including Matrigel and collagen 4 –8 .
Canonical Wnt signaling provided by Wnt3a and R-spondin is crucial to ISC proliferation and self-renewal 4,6,7 . Moreover, exogenous epidermal growth factor (EGF) also promotes ISC proliferation, while the bone morphogenetic protein inhibitor, Noggin, enables the maintenance and passage of intestinal enteroids and spheroids in vitro 4,7,9 –11 . In addition, the use of glycogen synthase kinase-3 inhibitor (GSKi) increases the efficiency of enterosphere formation, and the Rho-associated protein kinase (ROCK) inhibitor (Y-27632), which attenuates anoikis, is an important additive that improves proliferation and stabilization of the culture 11,12 . The use of these exogenous factors when culturing crypts in vitro leads to the generation of enteroids and spheroids in vitro 2,3 .
Isolated human intestinal crypts containing ISCs can be reliably obtained from small intestinal samples. Tissue typically is derived from discarded samples from patients undergoing either surgical resection or endoscopic biopsies 1,2,13 . Although intestinal epithelium can be isolated from procured surgical tissue, it may be subjected to prolong warm ischemia, diminishing crypt quality and quantity, and impairing the generation and growth of spheroids and enteroids 14,15 . Furthermore, the availability and timely acquisition of samples are affected by institutional, surgical/endoscopic, and pathology practices, which may impact the length of time that it takes to transfer the specimens from the clinical to the research setting.
To the best of our knowledge, a comparison of intestinal samples procured from cadaveric organ donors and discarded surgical samples has not been previously described. Candidates for organ donations undergo organ preservation via full-body fluid replacement with infused hypothermic preservation fluids 16,17 . As a result, tissues have limited warm ischemic time and the cellular metabolic deterioration rate is significantly reduced, potentially improving the quality of isolated crypts 16 . Cadaveric samples can be shipped beyond local research facilities to areas that may be lacking direct access/supply of samples, and in most cases the entire duodenum can be made available.
In this study, we explored various storage times of cadaveric and surgical duodenal samples and evaluated the preservative effects of UW solution on the storage of these samples. In addition, we documented the results of crypt isolations from these tissues and measured the effects of different additives to the culture media on the generation of enteroids and spheroids.
Materials and Methods
Surgical and Cadaveric Intestinal Sample Procurement
Human surgical samples (n = 6) were obtained from discarded surgical specimens from pancreatico-duodenectomy following pathologic evaluation by the Department of Pathology staff at UCLA. Surgical samples were obtained from patients who did not undergo either radiation or chemotherapy, and only the healthy appearing section of the bowel was used for crypt isolation. Prior to crypt isolation, surgical samples were stored in either phosphate-buffered saline (PBS) or University of Wisconsin (UW) preservation solution for up to 6 days at 4°C .
Cadaveric samples (n = 5) were obtained from the Texas Organ Sharing Alliance through an agreement with the Scharp-Lacy Research Institute (Aliso Viejo, CA, USA) and received en bloc small bowel with pancreas and spleen 18 . Preservation was achieved by the introduction of chilled UW solution in sufficient volume (4 to 6 l of chilled solution) into the major vascular channels (the abdominal and thoracic aorta, portal vein, and pulmonary artery) to wash out the blood and achieve moderate cooling to 32°C (10) into the major vascular channels. The pancreas with attached duodenal section was then removed from the body and transferred to sterile back-tables where the organs were then placed in fresh sterile solutions, double-bagged, and buried in melting ice in transport containers for dispatch to the Scharp-Lacy Research Institute. Duodenal samples were then dissected from the head of the pancreas. The duodenum was then opened and washed/flushed with 1% chlorhexidine. The lumen was filled with UW preservation solution and multiple smaller segments of the duodenum were created by tying off ∼4 cm segments using polypropylene suture, and stored at 4°C in UW preservation solution for up to 6 days. Individual segments were used for crypt isolation at time intervals between 12 and 144 h post-procurement.
Intestinal Crypt Isolation
Both surgical and cadaveric samples underwent crypt isolation at 12, 24, 48, 120, and 144 h post-procurement. Surgical and cadaveric samples underwent crypt isolation using methods that had previously been optimized using surgical samples 1,13 . A method based on exposure to 8 mM EDTA (Cat# E6758-100G; Sigma, St. Louis, MO, USA) and dithiothreitol (Sigma) chelation was used for crypt isolation as described 1 . Crypt yield was determined by counting the number of crypts per gram tissue using an inverted light microscope. Isolated crypts were suspended in basic media (Advanced Dulbecco’s Modified Eagle Medium [ADMEM]/Ham’s F12 [Invitrogen, Carlsbad, CA, USA] with 2 mM GlutaMAX, 10 mM HEPES [Invitrogen], and 1× antibiotic-antimycotic [Invitrogen]).
Initial Intestinal Crypt Culture and Subculture
Surgical and cadaveric crypts were suspended within Matrigel (BD Biosciences, San Jose, CA, USA) at a concentration of 100 crypts per 25 µL of Matrigel. At time points greater than 24 h, the detection of full-sized crypt diminished and was variable. Therefore, we suspended to our best estimation ∼100 crypts per 25 µl, but solutions contained numerous single cells from and usual crypt fractions were used when intact crypts were not isolated. Crypts were then plated into 48-well Nunclon Delta-treated cell culture plates (Thermo Scientific, Waltham, MA, USA).
Crypts were treated with one of five types of media conditions. Common to all six conditions included the following: basic medium (see before), 1 mM N-acetylcysteine (Sigma), 1× N2 supplement (Invitrogen), 1× B27 supplement (Invitrogen), 100 ng/ml recombinant murine Noggin (PeproTech, Rocky Hill, NJ, USA), 50 ng/ml recombinant murine EGF (PeproTech), and 1 µg/ml recombinant human R-spondin-1 (R&D Systems, Minneapolis, MN, USA). This medium consisting of EGF, Noggin, and R-spondin was labeled ENR.
In addition to 10 µM ROCK inhibitor (Sigma), four out of five conditions included an additional combination of the following: (1) 50% intestinal subepithelial myofibroblast conditioned medium (ISEMF-CM); (2) 10 nM prostaglandin E2 (PGE2); (3) 10 nM PGE2 and 50% L-Wnt3A conditioned medium (Wnt3a-CM); (4) 5 µM GSKi (CHIR99021; Stemgent, Cambridge, MA, USA), and Wnt3a-CM.
Culture medium was replaced with fresh medium every 2 days until cultures were either fixed for histological analysis or subcultured. Growth and generation of spheroids and enteroids were assessed using inverted light microscopy.
Surgical and cadaveric crypt cultures at each time interval were subcultured after 7 days in initial culture. Generated spheroids and enteroids within the Matrigel were digested using TrypLE (Life Technologies, Carlsbad, CA, USA) at 37°C for 3 to 5 min. TrypLE was then quenched using 10% fetal bovine serum (FBS) in ADMEM/F12 and structures were mechanically split into small clusters of cells using a syringe. In a 1:2 or 1:3 split, cell cluster was resuspended in 25 µl of fresh Matrigel and treated with the media conditions as listed earlier.
ISEMF-CM was isolated from ISEMFs cultured for 7 days in DMEM/low glucose/GlutaMAX (Invitrogen), 10% FBS (Invitrogen), 1× antibiotic-antimycotic (Invitrogen), 0.25 U/ml insulin (Sigma), 20 ng/ml recombinant murine EGF, and 10 μg/ml transferrin (Sigma) 1,10 . Conditioned media were then collected at day 7 for culture use. L-Wnt3a-CM was prepared as previously described 19 .
Histologic Assessment
Portions of surgical and cadaveric duodenal samples were fixed in 10% formalin and embedded in paraffin. Serial 5 µm sections were cut and prepared for hematoxylin and eosin staining.
RNA Analysis
Messenger RNA (mRNA) was isolated from cadaveric and surgical cultures using RNeasy Mini Kit (Qiagen, Valencia, CA, USA). Reverse transcriptase polymerase chain reaction (RT-PCR) was performed to determine the expression levels of genes of interest, using Taqman Gene Expression Assays (Applied Biosystems, Carlsbad, CA, USA) for caudal type homeobox 2 (CDX2), mucin 2 (MUC2), defensin (DEFA5), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), as previously described 1,13 . RT-PCR reactions were performed on a Prism 7900 HT Sequence Detection System (Applied Biosystems). Cycle numbers were analyzed according to the comparative CT method, using GAPDH as the internal calibrator and human intestinal crypts as the reference tissue 1 .
Statistical Analysis
Two-tailed independent student’s t-tests were used to compare results, and associated p-values are reported with an alpha level set at 0.05.
Results
Histological Assessment of Cadaveric and Surgical Samples
We assessed the epithelial architecture of cadaveric and surgical samples using standard histology (Figure 1), and quantified the number of intact crypts per microscopic high-power field (HPF) (Figure 2). We observed that cadaveric samples had preservation of intestinal epithelium along the crypt and villus axis for up to 6 days (144 h) post-procurement (Figures 1A and 2). In contrast, surgical samples stored in PBS solution had a significant reduction in epithelial layer preservation and the number of crypts per HPF when stored beyond 12 h post-procurement (Figures 1C and 2). Specifically, surgical samples stored in PBS beyond 12 h resulted in massive epithelial cell lysis (Figure 1C, and data not shown). We tested the ability of UW solution to preserve the integrity of the epithelial layer of surgical samples at various time intervals. The epithelial layer and the number of intact crypts per HPF of surgical samples processed within 12 h of procurement were not influenced by the use of either UW or PBS storage solutions (Figures 1B, C and 2). However, in contrast to surgical samples stored in PBS solution, the use of UW preservation solution significantly improved the intestinal epithelial layer and the retention of crypt abundance beyond 12 h post-procurement (Figures 1B and 2). Moreover, with the exception of 48 and 120 h, there were no significant differences in the number of crypts per HPF when comparing cadaveric samples and surgical samples stored in UW solutions (Figure 2). Our findings suggest that in addition to using cadaveric samples, the use of UW solution has an important role in preserving the crypt–villus architecture of surgical samples beyond 12 h.

Histological assessment of cadaveric and surgical samples. Representative hematoxylin and eosin images of intestinal cross-sections demonstrating the crypt/villus architecture of (A) cadaveric and (B) surgical samples stored in UW solution; and (C) surgical samples stored in PBS solution for 12 (1), and 144 (2) h post-procurement.

Presence of crypts in intestinal samples from 12 to 144 h post-procurement. Graph demonstrates the presence of intestinal crypts in cadaveric and surgical intestine. At the time of procurement, surgical samples were placed in UW solution and PBS solution. † p < 0.05 when cadaveric samples were compared to surgical samples stored in UW solution. *p< 0.05 when cadaveric samples were compared to surgical samples stored in PBS solution. ‡ p < 0.05 when surgical samples stored in UW solution were compared to surgical samples stored in PBS solution.
Crypt Isolation from Cadaveric and Surgical Samples
Using methods standardized for surgical samples 13 , cadaveric crypts were successfully isolated from samples 12 to 144 h post-procurement (Figure 3). However, when comparing cadaveric to surgical samples, we found that the number of crypts per gram tissue isolated from cadaveric samples was highly variable, and resulted in significantly fewer crypts than those isolated from surgical samples that are routinely isolated in our laboratory <6 h post-procurement (mean ± SD: 2,044 ± 1,441 vs. 12,350 ± 1,520, p = 1.3 × 10−6; Figure 3).

Crypt isolation in cadaveric and surgical samples. Graph represents cadaveric and surgical crypts isolated per gram of mucosa. Cadaveric crypt isolation was performed from 12 to 144 h post-procurement. Surgical crypt isolation was performed <6 h post-procurement. *p < 0.05 when isolated cadaveric crypts were compared to the number of isolated surgical crypts. Error bars = standard error of the mean (SEM).
As the cadaveric sample post-procurement time increased beyond 12 h, the number of crypts remained significantly low. We found that starting at 24 h post-procurement, the isolated cadaveric crypts were increasingly fragmented with longer post-procurement time, losing their structural integrity during the isolation process, and resulted in a mixture of partially undamaged crypts and single cells. As a result, it was difficult to accurately assess the number of crypts isolated per gram beyond 24 h post-procurement (Figure 3).
Spheroids and Enteroids Generated from Crypts of Cadaveric and Surgical Samples Following Short-Term Procurement
We investigated the ability of cultured crypts isolated from either cadaveric or surgical samples stored in UW solutions and processed 12 h post-procurement to generate spheroids and enteroids in an in vitro setting after 7 days in culture. We found that ENR supported enteroidal structures growth of both cadaveric and surgical samples after 7 days in culture (Figures 4, 5 and Figure S1). In the other three media conditions (i.e., ISEMF-CM, PGE2, and GSKi/Wnt3a-CM), cadaveric and surgical samples generated both spheroid and enteroids structures after 7 days in culture (Figures 4, 5 and Figure S1). Spheroid structures generation in cadaveric cultures using ISEMF-CM were more abundant than crypts isolated from surgical cultures supported with the same media (p = 0.016) (Figure 4). The number of enteroids from crypts generated from both cadaveric and surgical samples were similar (Figures 4 and 5) (p = 0.37). This demonstrates that cadaveric crypts can be reliably cultured using various supportive media, similar to surgical samples procured at this earliest time point.

Total in vitro structures formed in 7 days from cultured crypts isolated from (A) cadaveric and (B) surgical samples stored in UW solutions and processed 12 h post-procurement. Analysis shows *p < 0.05 when structures formed from cadaveric crypts were compared to structures formed from surgical crypts in similar conditions. Error bars, SEM.
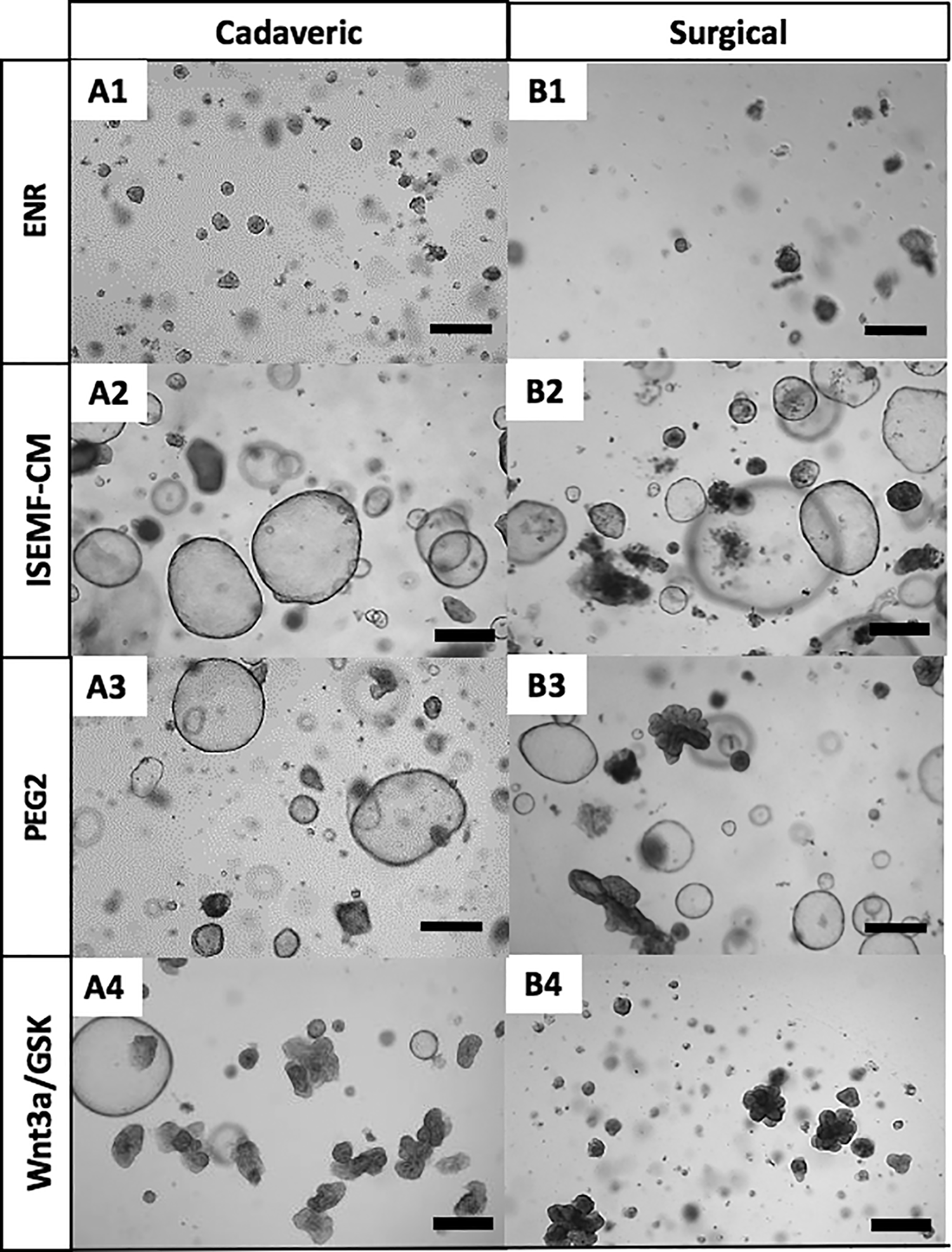
In vitro structures formed in 7 days from crypts isolated from (A) cadaveric and (B) surgical samples at 12 h post-procurement. Representative images of in vitro structures grown in various media for 7 days. Scale bar 500 µm.
Culturing of Crypts from Surgical Tissue Stored in UW or PBS Solution
We assessed the ability to culture crypts isolated from surgical tissue stored in either UW or PBS solution beyond the initial time of procurement. Crypts isolated from surgical samples stored in UW solution up to 24 h post-procurement were cultured, and generated spheroid and enteroidal structures when grown in GSKi/Wnt3a-CM or ISEMF-CM (Figure 6A1-2). In contrast, surgical tissues stored in PBS solution and procured in 24 h were incapable of generating any enterosphere structures with either media (Figure 6B1-2). However, crypts isolated from surgical samples 144 h post-procurement that were stored in UW, but not PBS solution were only able to generate a few spheroidal structures in ISEMF-CM (Figure 6C and D1-2). Overall, our findings suggest that beyond the initial procurement period, the storage of surgical samples in UW solution has an important role in preserving the ability to generate structures in vitro.

In vitro structures formed in 7 days from crypts isolated from surgical samples at 24 and 144 h post-procurement and stored in UW (A/C) and PBS (B/D) solution. Crypts were cultured either with (1) ISEMF-CM or (2) GSKi/Wnt3a-CM. Scale bar 500 µm.
Spheroids and Enteroids Generated from Cadaveric Samples Stored Between 12 and 144 h Post-Procurement and Subculturing
We assessed the ability to culture crypts isolated from cadaveric tissue beyond 12 h post-procurement in order to generate enterosphere structures. In vitro structures were reliably generated from crypts isolated from cadaveric samples 12 to 144 h post-procurement when grown in ISEMF-CM, GSKi/Wnt3a-CM, or PGE2, but not ENR alone (Figure 7 and Figure S1). However, we found that the ability to generate structures with GSKi/Wnt3a-CM decreased significantly after culturing crypts beyond 48 h post-procurement (Figure 7).

In vitro structures of cadaveric samples up to 144 h post-procurement when grown in different media.
Cell cultures generated from cadaveric crypts were subcultured and propagated similar to cell cultures generated from isolated surgical samples 13 . Here we found that cadaveric structures generated from cells/crypts isolated from tissues stored for even 144 h post-procurement could be propagated with remarkable reproducibility (Figure 8).

Passing of in vitro structures isolated from cadaveric samples procured at 12 and 144 h when grown in ISEMF-CM. (A–C) Cadaveric structures cultured from crypts at 12 h post-procurement, and passage 0, 1, and 2 at 7 days in culture. (D and E) cadaveric spheroids cultured from crypts at 144 h post-procurement, and passages 0, 1, and 2 at 7 days in culture. Scale bar 500 µm.
mRNA Assessment of In Vitro Enterosphere Structures
The phenotypic differentiation of cadaveric and surgical cultures was assessed by RT-PCR (Figure S2). Cadaveric cultures similarly expressed levels of CDX2, Mucin 2 (MUC2), and DEF5, which are indicative of small intestinal epithelium.
Discussion
In this study, we demonstrate that intestinal crypts can be isolated from human cadaveric intestinal samples several days post-procurement and cultured to form enterospheroidal structures in culture. Moreover, we determined that the use of UW solution on surgical samples isolated 1 day after procurement preserves the ability to generate three-dimensional epithelial structures in vitro.
In vitro models utilizing nontransformed epithelial cells are being broadly used to study ISCs and epithelial cell function during homeostatic and diseased states, and hold promise for stem cell-based transplantation 1,13,19,20 . Most in vitro intestinal epithelial models for spheroid or enteroid structures generated from crypts of the small or large bowel 1,13,10 . The majority of intestinal samples used to isolate crypts are obtained from either endoscopic biopsies or discarded bowel specimens from surgical patients 1,20 . However, the quality of these specimens may be variable given the uncontrollable exposure to long periods of warm ischemia that occur during the harvesting process. Therefore, exploring additional reliable sources and methods to sustain crypts and ISCs is an important goal of the field of stem cell therapy.
Warm intestinal ischemia results in the depletion of adenosine triphosphate (ATP) and other metabolic energy products due to the deprivation of oxygen 21 . During warm ischemic times, hypoxanthine accumulates from the depletion of ATP, leading to oxidative stress. Oxidative stress is associated with the generation of reactive oxygen species (ROS), neutrophil-induced bowel tissue damage, and the release of cytotoxic proteins into the extracellular fluid, leading to cellular death 21,22 . ROS not only result in direct cellular injury but also necrosis from the peroxidation of cellular membrane lipids 14,23 –25 . Lipid peroxidation can impair the function of cellular surface and internal membranes and their associated enzymes and receptors 22 .
Hypothermic preservation is a feature common to both preservation modalities. Organs are typically cooled by infusing chilled isotonic fluid by major vessels (the abdominal or thoracic aorta, portal vein, or pulmonary artery depending on the organ of interest), and washing out the blood while achieving significant cooling (10 to 15°C) before harvesting the organs 26 .
The benefit of hypothermic preservation is slowed metabolism and significantly decreased warm ischemia time 16,17 . Decreasing the temperature within the tissues by 10°C diminishes the metabolic rates by approximately a factor of 2, although it does not halt it entirely 16,17,23 . During hypothermic preservation, tissues experience acidosis due to the increased accumulation of intracellular lactic acid. Disruptions in osmoregulation result in decreased oncotic pressure, causing expansion of interstitial space, edema, and cellular swelling from the decreased activity of Na-K-ATPase (resulting from a decrease in ATP) leading to the accumulation of intracellular sodium and water 25 . The utilization of preservation solution ameliorates cold ischemic effects 16,17 .
UW solution was developed to mitigate the deleterious effects of cold ischemia 27 . Its components have been well characterized in enhancing hypothermic preservation. Ion ratios of Na+/K+ and Ca2+/Mg2+ in UW solution prevent expansion of the interstitial space, edema, and cellular swelling, by diminishing the passive diffusion of ions at low temperatures. Stabilization of osmoregulation is achieved by the addition of hydroxyethyl and raffinose, which elevate the intracellular osmotic pressure. Lactobiotine is a metabolite in UW that prevents cellular edema and acidosis, and is further obviated by the buffered solution in UW. Although hypothermic preservation decreases the generation of ROS, cold ischemia does result in the generation of some ROS through depletion of ATP. Glutathione adenosine and allopurinol serve to increase ATP synthesis and reduce oxidative cellular damage 27 .
As shown in this study, crypts could be used to grow epithelium using cadaveric duodenum samples up to 144 h post-procurement. We also found that the use of preservation solution, UW—compared to PBS in surgical samples—led to histological crypt preservation for up to 144 h. Storing surgical samples in PBS for 24 h resulted in necrotic degeneration (Figures 1 and 2). This result was in stark contrast to both cadaveric and surgical samples stored in UW, which showed preservation of intestinal epithelium and crypt base cells up to 144 h post-procurement. Although the crypt morphology of samples preserved in UW solution appeared viable in situ, we had poor recovery of intact crypt structures from the UW-preserved cadaveric samples. It should be noted that the methods used for crypt isolation were standardized procedures developed for surgical samples and were not specifically modified for cadaveric samples. To continue the use of UW-preserved cadaveric donors in future research projects, further development and improvements of crypt isolation techniques may be prudent.
UW storage of surgical samples for 24 h allowed isolation of viable crypts and the generation of both spheroids and enteroids, as seen after 7 days of culture (Figures 1A and 6). However, neither spheroids nor enteroids could be found after culturing crypts from surgical samples that were previously held for 144 h post-procurement in UW (Figure 1C). While hypothermic preservation was standardized for all cadaveric donors, none of the surgical samples underwent hypothermic preservation. This leads us to conclude that storage in preservation solution alone is insufficient to allow intact crypt isolation and culture beyond 24 h after procurement. These study results imply that the UW preservation solution should be introduced to the duodenal samples via vascular means, at a cellular level, in order to achieve the most successful in vitro generation of spheroids and enteroids.
We conclude that hypothermic preservation with the use of infused UW solution decreases cellular death of the intestinal crypts, thus permitting isolation and culture of crypts 12 to 144 h post-procurement. The decrease in the total number of crypts isolated in cadaveric samples when compared to surgical samples may be linked to prolonged exposure to preservation solution 23,24 . We observed that crypts isolated beyond 24 h began to slowly lose their structural integrity when processed for crypt isolation. By 144 h, crypt isolates consisted mainly of single-cell suspensions, which negated our attempts to quantify crypt units, and to standardize the number of crypt equivalent units to add to each well.
Previous studies have shown that cells exposed to preservation solutions for prolonged periods experience dysregulation of junctional proteins, resulting in loss of cell-to-cell adhesion and cytoskeleton disruption; this fact may have contributed to our findings 23,24 . Moreover, crypt isolation techniques are optimized for surgical samples and not cadaveric samples, suggesting that this technique may be too rigorous for the preserved cadaveric samples 1,2,13 . Although the presence of formed crypts decreased as we approached 144 h, isolated crypts/cells at later time points treated with CM supported spheroid and enteroid growth (Figure 7).
Previous studies also have demonstrated that single cells from CBCCs can generate intestinal enteroids 11 . We demonstrated that spheroids could be generated reliably from cadaveric crypts and from crypts dissociated into single cells at later time points, up to 144 h post-procurement. However, crypts dissociated into single cells from cadaveric samples beyond 48 h post-procurement showed only marginal generation of enteroids.
We have been able to demonstrate the generation of enteroids and spheroids from cadaveric samples using a variety of media conditions, and found that cadaveric cultures behaved similarly to surgical cultures 1,10 . Furthermore, spheroids and enteroids generated from cadaveric samples could be subsequently subcultured in a similar manner and with the same reproducibility as surgical samples from 12 to 144 h post-procurement. In general, cadaveric and surgical cultures expressed intestinal cell lineage on mRNA analysis. The use of GSKi/Wnt3a-CM promoted the generation of enteroids, which have more differentiated cell types, in both cadaveric and surgical samples 1,10 . When compared to surgical cultures, cadaveric cultures had lower expression of mRNA transcripts, indicative of advanced differentiation. We postulate that prolonged exposure to preservation solution decreases the ability to generate and express differentiated cell types/markers in vitro.
We conclude that hypothermic preservation of infused cadaveric intestine permits isolation of cells/crypts for up to 7 days after procurement. In vitro cadaveric cultures readily form spheroids and enteroids comparable to standard cultures from surgical samples. Although enteroid formation and growth decrease after 48 h, spheroids are reliably generated from cells/crypts up to 144 h post-procurement. Furthermore, cadaveric structures can be subcultured and amplified from samples initially cultured from 12 to 144 h post-procurement. Although the use of UW solution preserves intestinal architecture and the crypt base when assessed in situ, its use did not yield increased number of isolated crypt isolation beyond 24 h. This study demonstrates that the use of cadaveric intestine represents an excellent alternative source of crypts, which can be used for in vitro culture on intestinal epithelium.
Supplemental Material
Supplemental Material, Screen_Shot_2019-11-20_at_3.56.26_PM - Comparison of Surgical and Cadaveric Intestine as a Source of Crypt Culture in Humans
Supplemental Material, Screen_Shot_2019-11-20_at_3.56.26_PM for Comparison of Surgical and Cadaveric Intestine as a Source of Crypt Culture in Humans by Andrew Scott, Barbara Olack, Joshua D. Rouch, Hassan A. Khalil, Brent A. Kokubun, Nan Ye Lei, Jiafang Wang, Sergio Solorzano, Michael Lewis, James C.Y. Dunn, Matthias G. Stelzner, Joyce C. Niland and Martín G. Martín in Cell Transplantation
Supplemental Material
Supplemental Material, Screen_Shot_2019-11-20_at_3.56.51_PM - Comparison of Surgical and Cadaveric Intestine as a Source of Crypt Culture in Humans
Supplemental Material, Screen_Shot_2019-11-20_at_3.56.51_PM for Comparison of Surgical and Cadaveric Intestine as a Source of Crypt Culture in Humans by Andrew Scott, Barbara Olack, Joshua D. Rouch, Hassan A. Khalil, Brent A. Kokubun, Nan Ye Lei, Jiafang Wang, Sergio Solorzano, Michael Lewis, James C.Y. Dunn, Matthias G. Stelzner, Joyce C. Niland and Martín G. Martín in Cell Transplantation
Footnotes
Authors’ Contributions
BJO, JCN, AS, JC, and MGM conceived and designed the experiments; AS, JDR, HAK, BAK, SS, NYL, JW, and ML performed the experiments; AS, JDR, BJO, MGM, JCYD, and MGS analyzed the data; HAH, NYL, and SS contributed reagents/materials/analysis tools. AS wrote the paper; and MGM, BJO, AS, JDR, JC, JCYD, JCN, MGS, and ML did the critical review of the manuscript.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human Rights
All human tissues used in this study were obtained from de-identified subjects, and was approved by the UCLA Institutional Review Board (IRB #11-002504).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by National Institute of Diabetes and Digestive and Kidney Disorders (NIDDK) and National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health under grant numbers DK085535, P30 DK041301. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
