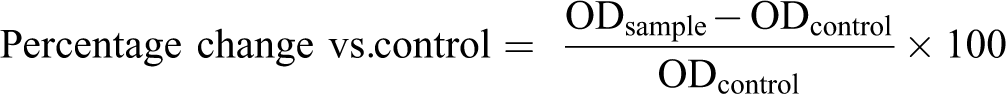Abstract
We studied the paracrine function of mesenchymal stem cells (MSCs) derived from various sources in response to pulsed focused ultrasound (pFUS). Human adipose tissue (AD), bone marrow (BM), and umbilical cord (UC) derived MSCs were exposed to pFUS at two intensities: 0.45 W/cm2 ISATA (310 kPa PNP) and 1.3 W/cm2 ISATA (540 kPa PNP). Following pFUS, the viability and proliferation of MSCs were assessed using a hemocytometer and confocal microscopy, and their secreted cytokine profile determined using a multiplex ELISA. Our findings showed that pFUS can stimulate the production of immunomodulatory, anti-inflammatory, and angiogenic cytokines from MSCs which was dependent on both the source of MSC being studied and the acoustic intensity employed. These important findings set the foundation for additional mechanistic and validation studies using this novel noninvasive and clinically translatable technology for modulating MSC biology.
Introduction
Mesenchymal stem cells (MSCs) are multipotent stem cells 1 that can be isolated from various tissues 2 –10 . Although bone marrow-derived MSCs (BM-MSCs) have traditionally been used as the main source of MSCs in clinical practice, MSCs derived from adipose tissue (AD-MSCs) and umbilical cord (UC-MSCs) have emerged as new and readily available sources with well-documented regenerative and immunomodulatory properties 11 –14 . While AD-MSCs can be easily isolated with high yield from adipose tissue obtained during routine liposuction/lipoplasty procedures 15 , UC-MSCs are retrieved from the umbilical cord which is considered a medical waste at the time of birth.
MSCs actively secrete cytokines and growth factors that act either on themselves (autocrine function) or neighboring cells (paracrine function) to modulate the immune system, inflammatory response, as well as stimulate neo-angiogenesis 16 . For instance, MSC-secreted cytokines have been implicated in the repair and regeneration of the central nervous system (CNS) 17 , heart 18 –21 , bone 16,22 , and other damaged tissues 23 . Given that MSCs have the ability to sense and respond to various stimuli 24 –26 , several groups have investigated preconditioning MSCs (i.e., intentionally exposing them to a controlled amount of stimulus for a defined period of time in order to produce the desired response) to enhance their secretion of trophic factors 16 ; these stimuli include hypoxia 27 , thermal shock induction 28 , pharmacologic treatment 29 , and proinflammatory (i.e., interferon-gamma [IFN-γ] or tumor necrosis factor-alpha [TNF-α]) cytokine exposure 30,31 . However, following their administration into patients, there is currently no way in which MSCs can be actively stimulated.
One approach to noninvasively stimulate MSCs, in a controlled and systematic way outside of the body, as well as inside the body following their administration, is to utilize sound waves 32 . Focused ultrasound (FUS) is a novel technology, which can focus sound waves at specific locations deep in the body with pinpoint accuracy but without the use of any incisions. Pulsed FUS (pFUS) is a variation of this technology that uses short duty cycles to minimize temperature elevations, thereby allowing the biomechanical effects of ultrasound to predominate 33 . We have recently shown that pFUS can stimulate pancreatic islets to increase insulin release 34 . Hence, we hypothesized that pFUS can also stimulate MSCs and modulate their paracrine function by changing their profile of secreted cytokines. We therefore examined the effect of pFUS on the viability and function (determined by their paracrine function) of MSCs derived from various sources (i.e., BM, AD, and UC-MSCs).
Materials and Methods
MSC Isolation and In Vitro Expansion
Human AD-MSCs and UC-MSCs were kindly donated from the University of Miami (from Drs Ricordi and Patel) 35,36 , and human BM-MSCs were donated from the laboratory of diagnostic research at the NIH (from Dr Frank) 37 . All MSCs were fully characterized as previously described 35 –37 . BM-MSCs and AD-MSCs were cultured in Mesenchymal Stem Cell Growth Medium (Lonza, NJ, USA), supplemented with 10% fetal bovine serum (FBS) with additional supplements (MSCGM hMSC SingleQuot Kit, Lonza, NJ, USA). UC-MSCs were cultured in low glucose Dulbecco’s modified Eagle medium (Fisher Scientific, Grand Island, NY, USA) supplemented with 10% XcytepLUS (ibiologics), 1% glutamax (Gibco, Grand Island, NY, USA), 1% nonessential amino acid solution (NEAA; Gibco), and 1% penicillin and streptomycin (Life Technologies, Grand Island, NY, USA). All cells were cultured in the incubator at 37 °C with 5% carbon dioxide (CO2), and the culture media were changed every 3 days.
MSC Stimulation with pFUS
pFUS was performed on MSCs as described previously 34 . For each pFUS treatment, experiments were performed using a six-well plate (Corning, USA) containing 105 MSCs/well. MSCs (BM-MSC, AD-MSC, and UC-MSC) were first cultured in well-plates for 24 h; the plates were then immersed in an autoclaved water bath and placed above the pFUS transducer (KB Aerotech Inc., Lewiston, PA, USA) at the transducer’s focal spot (i.e., 50 mm away from the transducer surface). For sound waves to cover all of MSCs cultured in each well-plate, each well was divided into 25 spots (5 × 5 mesh, 5.75 mm distance between each point). Culture plates were immobilized, while a 1 MHz piston transducer was attached to an Acoustic Intensity Measurement System (AIMS III, Onda, Sunnyvale, CA, USA) for precise positioning of the pFUS transducer to cover all 25 spots. The following pFUS parameters were fixed: 1 MHz frequency, 20% duty cycle, 100 Hz pulse repetition frequency, with a total duration time of 6 min (i.e., 14.4 s/spot). MSCs were then divided into three groups: Group 1: MSCs stimulated with low intensity pFUS (i.e., 0.45 W/cm2 ISATA; 310 kPa peak negative pressure [PNP]); Group 2: MSCs stimulated with high intensity pFUS (i.e., 1.3 W/cm2 ISATA; 540 kPa PNP); and Group 3: MSCs with no pFUS stimulation (controls). Each treatment was repeated in duplicate.
Analysis of MSC-Secreted Cytokines
Following pFUS stimulation, MSCs were incubated at 37 °C and 5% CO2 for 48 h, after which time their culture media was collected for multiplex immunoassay analysis (human multiplex ELISA; eBiosciences/Affymetrix/Fisher) to assess and measure the levels of secreted cytokines. In brief, beads were first added to a 96-well plate and washed (Biotek EL ×405). Samples were then added to the plate containing the mixed antibody-linked beads and incubated at room temperature for 1 h followed by overnight incubation at 4 °C on a plate shaker (500 rpm). A biotinylated detection antibody was then added, after which the plates were incubated at room temperature for 75 min on a plate shaker (500 rpm). Next, the samples were washed and streptavidin-phycoerythrin added followed by incubation of the plates for 30 min at room temperature on a plate shaker (500 rpm). The plates were then washed, and a reading buffer was added to the wells. Finally, a Luminex Flex 3D instrument was used to read the plates with a lower bound of 50 beads per sample per cytokine. Control assay beads (Radix Biosolutions) were added to wells. Multiplex ELISA assays were performed on all three sources of MSCs (i.e., BM-MSC, AD-MSC, and UC-MSC), which were sampled twice, and the average cytokine value was taken from two separate readings. The percentage change in cytokine expression from pFUS-stimulated MSCs relative to control (i.e., nonstimulated) MSCs was then calculated (Eq. 1):
where ODsample is the optical density (absorbance) of MSCs stimulated with pFUS and ODcontrol is the optical density of control MSCs. Data were compiled as a heat map with upregulation represented as a red color gradient and downregulation represented as a green color gradient. We then categorized secreted cytokines into three subgroups: immunomodulatory, anti-inflammatory, and angiogenic cytokines.
Determination of MSCs’ Morphology and Viability
Following pFUS stimulation, MSCs were incubated at 37 °C and 5% CO2 for 48 h and then harvested and counted using a hemocytometer 38 . Cell numbers were compared with the cell number at time point 0, and the results expressed as the fold change versus control. Cell morphology was also observed under a confocal microscope (Zeiss LSM710).
Statistical Analysis
All experimental data are expressed as the mean ± SEM. Statistical analysis of all quantitative data was performed using one-way analysis of variance with post hoc Tukey test (Astatsa.com; Online Web Statistical Calculators, USA) with any differences considered statistically significant when P < 0.05.
Results
Analysis of MSC-Secreted Cytokines
BM-MSCs
Stimulation of BM-MSCs with low-intensity and high-intensity pFUS resulted in a 15% ± 20% and 5% ± 10% increase in cytokine secretion, respectively, when compared with control BM-MSCs (Fig. 1A, P < 0.05). Stimulation of BM-MSCs with low-intensity pFUS upregulated the expression of a subset of immunomodulatory (IL31, SCF, RANTES, IFNG, MIP1B, IFNA, TNFB, GROA, IL1A, IL12P40, IL15, IL18, MCP3, ICAM1, VCAM1, IL22, and ENA78), anti-inflammatory (FASL, IL1B, TGFB, IL1RA, TGFB, IL9, BDNF, TRAIL, IL10, and IFNB), and angiogenic (VEGFG, VEGF, FGFB, IL2, and EOTAXIN) cytokines, while also downregulating the expression of the angiogenic cytokine PDGFBB when compared with the control BM-MSCs (Fig. 1B, P < 0.05). Stimulation with high-intensity pFUS caused upregulation of immunomodulatory (IL31, TNFA, MCP3, LEPTIN, and CD40 L), anti-inflammatory (FASL, MIP1A, IL1B, IL6, IL8, IL9, BDNF, IFNB, and LIF), and angiogenic (VEGFG, VEGF, TGFA, FGFB, and PAI1) cytokines, while also downregulating select immunomodulatory (IL23), anti-inflammatory cytokine (TGFB) and angiogenic (IL2, and IP10) cytokines when compared with control BM-MSCs (Fig. 1B, P < 0.05).
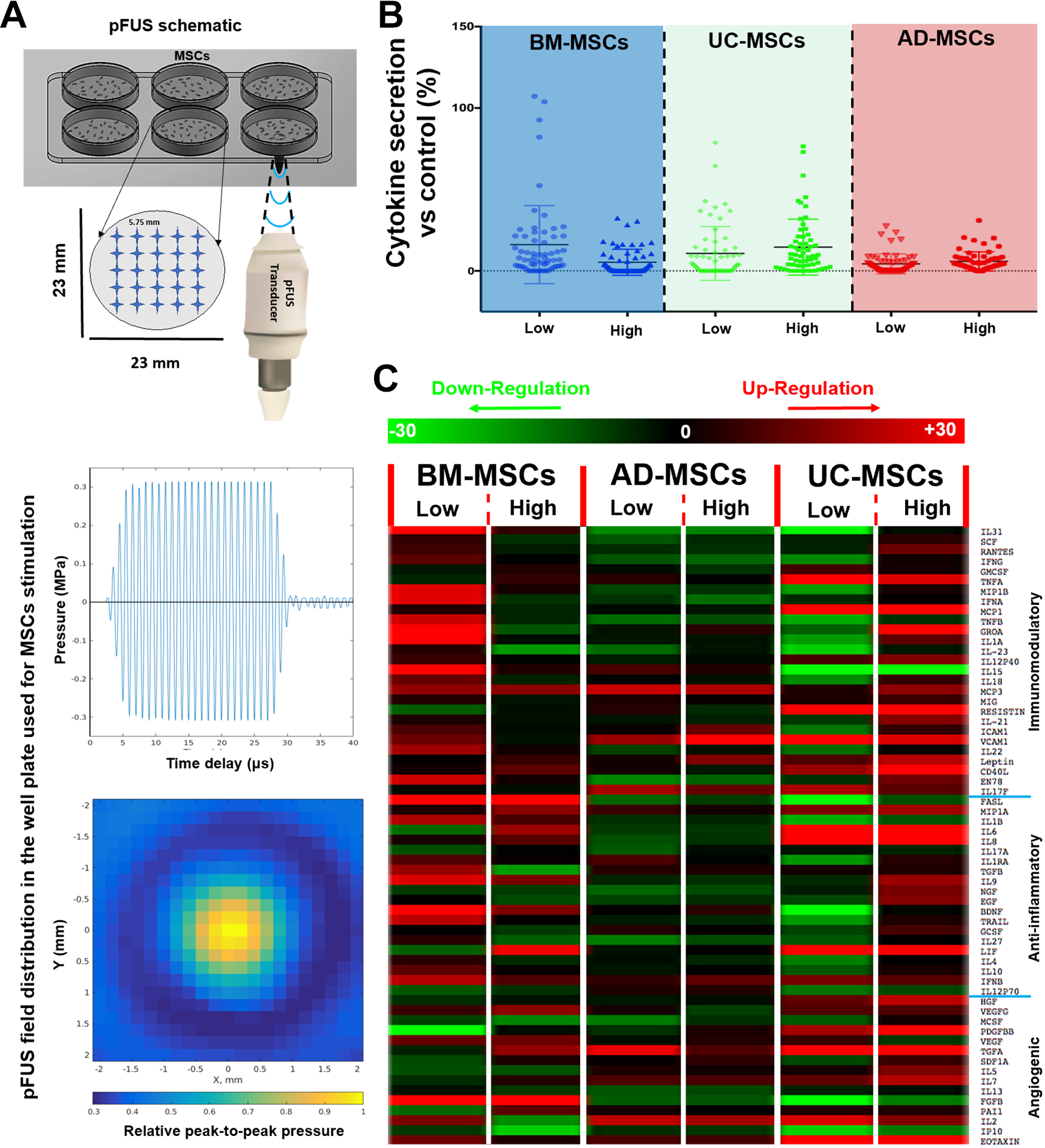
Analysis of MSC-secreted paracrine cytokines. (A) Schematic and field distribution of pFUS used in our study for MSCs stimulation: MSCs were first cultured in the well plates; the plate was then immersed in an autoclaved water and placed above the pFUS transducer at the transducer’s focal spot. The transmitted ultrasound waves were produced by a function generator, amplified through a power amplifier at a constant gain, and emitted from a focused piston transducer. In order for sound waves to cover whole MSCs cultured in well-plate, each well was graded into 25 spots (5 × 5 mesh, 5.75 mm distance between each point). (B) Stimulation of all three types of MSCs (i.e., BM-MSCs, AD-MSCs, and UC-MSCs) with low-intensity or high-intensity pFUS resulted in changes in cytokine secretion compared with control samples and
AD-MSCs
Stimulation of AD-MSCs with low-intensity and high-intensity pFUS resulted in a 3% ± 5% and 5% ± 7% increase in cytokine secretion, respectively, when compared with control AD-MSCs (Fig. 1A, P < 0.05). Stimulation of AD-MSCs with low-intensity pFUS upregulated the expression of a subset of immunomodulatory (IL15, MCP3, VCAM1, and IL17F), anti-inflammatory (MIP1A, IL1RA, and IFNB), and angiogenic (TGFA, IL7, IL2, and EOTAXIN) cytokines, while also downregulating the expression of the immunomodulatory cytokine IL31 when compared with control AD-MSCs (Fig. 1B, P < 0.05). Stimulation of AD-MSCs with high-dose pFUS also caused upregulation of immunomodulatory (MCP3, ICAM1, VCAM1, LEPTIN, and IL17F), an anti-inflammatory (IFNB), and angiogenic (TGFA, SDF1A, IL7, IL2, and EOTAXIN) cytokines when compared with control AD-MSCs (Fig. 1B, P < 0.05).
UC-MSCs
Stimulation of UC-MSCs with low-intensity and high-intensity pFUS resulted in a 10% ± 15% and 15% ± 17% increase in the cytokine secretion, respectively, when compared with control UC-MSCs (Fig. 1A, P < 0.05). Stimulation of UC-MSCs with low-intensity pFUS upregulated the expression of a subset of immunomodulatory (GMCSF, TNFA, MCP1, IL12P40, RESISTIN, VCAM1, LEPTIN, CD40 L, IL17F), anti-inflammatory (MIP1A, IL6, IL8, LIF, IFNB), and angiogenic (HGF, VEGFG, PDGFBB, VEGF, TGFA, IL7, IL2, EOTAXIN) cytokines, while also downregulating the expression of immunomodulatory (IL31, MIP1B, TNFB, IL1A, IL23, IL15, IL18), anti-inflammatory (FASL, IL1B, IL1RA, BDNF, TRAIL), and the angiogenic cytokine IP10 when compared with control UC-MSCs (Fig. 1B, P < 0.05). Stimulation of UC-MSCs with high-dose pFUS caused upregulation of a subset of immunomodulatory (SCF, RANTES, TNFA, MCP1, GROA, IL1A, IL12P40, IL18, MCP3, MIG, RESISTIN, IL21, ICAM1, VCAM1, LEPTIN, CD40 L, EN78, and IL17F), anti-inflammatory (MIP1A, IL6, IL8, IL9, NGF, EGF, GCSF, LIF, and IFNB), and angiogenic (HGF, VEGFG, PDGFBB, TGFA, SDF1A, IL5, IL7, IL2, and EOTAXIN) cytokines. High-dose pFUS also caused downregulation of the immunomodulatory cytokine IL15 when compared with control UC-MSCs (Fig. 1B, P < 0.05).
Determination of MSCs’ Morphology and Viability
Stimulation of all three types of MSCs (i.e., BM-MSCs, AD-MSCs, and UC-MSCs) with pFUS, at both low and high intensities, did not significantly change the morphology and viability of MSCs compared towith their control (Fig. 2; BM-MSCs: low intensity = 1.01 ± 1.00 fold change vs control, high intensity = 1.02 ± 1.05, control = 1.00 ± 0.50; AD-MSCs: low intensity = 0.85 ± 1.13, high intensity = 0.90 ± 0.77, control = 1.00 ± 0.50; UC-MSCs: low intensity = 0.96 ± 0.60, high intensity = 0.91 ± 1.02, control = 1.00 ± 0.50; P > 0.05).
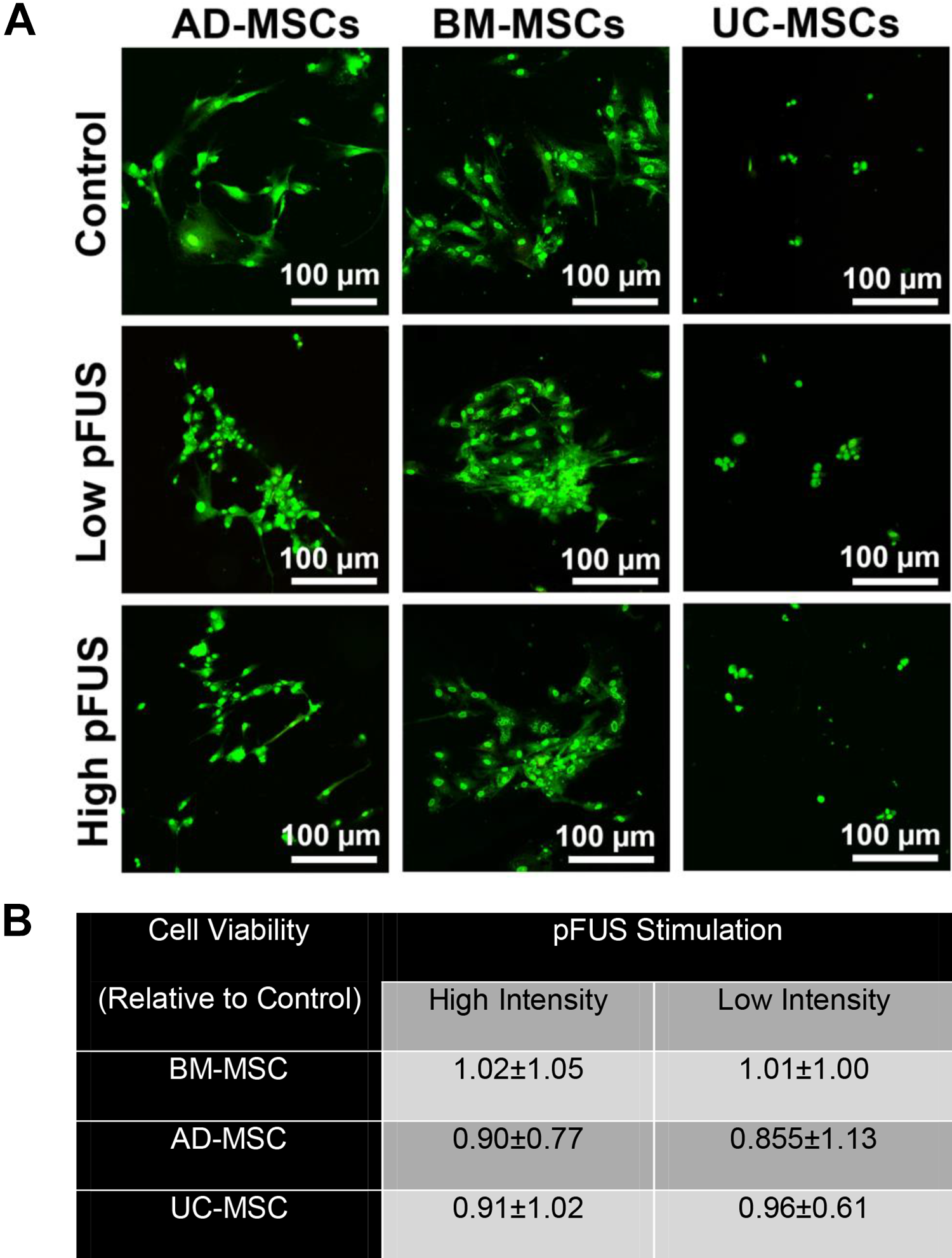
Determination of MSC morphology and viability: stimulation of all three types of MSCs (i.e., BM-MSCs, AD-MSCs, and UC-MSCs) with pFUS in both low and high intensity did not significantly change the
Discussion
MSCs are a promising regenerative cellular therapy which have been shown to have a significant benefit in multiple preclinical models 16 –23 . In addition to BM-MSCs, AD-MSCs and UC-MSCs are now being used in clinical trials to treat multiple conditions 39,40 . In this study, we investigated (1) whether pFUS (i.e., sound waves) can safely be used to biomechanically stimulate MSCs and if this is dependent on the acoustic intensity employed and (2) whether different sources of MSCs respond differently to pFUS, as determined by their cytokine profile.
Our results show that pFUS can be used in vitro, at low and high intensities, with no adverse effect on MSC morphology or viability. The effect of acoustic intensity on cytokines is dependent on the source of MSCs with BM-MSCs showing increased secretion at lower intensities, UC-MSCs showing increased secretion at higher intensities, and AD-MSCs demonstrating the least amount of sensitivity to sound waves at both high and low intensities. Finally, our results show MSCs respond to pFUS in a source-dependent manner, with each source producing a distinct cytokine profile (i.e., the highest level of a cytokine produced by BM-MSCs was IL15, for AD-MSCs was TGF-α, and for UC-MSCs was LIF).
Given that pFUS can produce a different profile of cytokines depending on the source of MSC, this will become important for choosing a specific MSC for a particular disease indication, especially if pFUS is used for preconditioning MSCs. In terms of the cytokine produced at the highest level for each MSC, IL15 has been shown to induce the differentiation and proliferation of T, B, and natural killer cells and induces maturation of dendritic cells, thereby highlighting an important immunomodulatory function 41,42 ; TGF-α has been shown to initiate multiple cell proliferation events that play a role in wound healing as well as promoting angiogenesis 43 ; LIF has been shown to promote growth and cell differentiation as well as modulate embryonic stem cell self-renewal and differentiation 44 –46 .
Taken together, it is clear that pFUS can be used to stimulate MSCs. While the present study did not investigate the mechanisms underlying this effect, possible pathways include the mitogen-activated protein kinase 47 , focal adhesion kinase activates-extracellular signal-regulated kinase 1/2 48 , and stromal cell-derived factor-1/C-X-C chemokine receptor type 4 49 . These pathways have already been shown to be activated following stimulation of stem cells with low-intensity pulsed ultrasound (LIPUS) 47 –49 . Future work will aim to investigate these mechanisms systemically and the effects of different acoustic parameters on these pathways, in order to better determine how to effectively modulate the function of MSCs. Similar to other studies that have compared the secretory profile of MSCs in response to proinflammatory cytokines, our data also show that different sources of MSCs respond differently to the same stimulus (i.e., sound waves) 50,51 . Future studies will aim to better understand the underlying molecular biology governing these changes as well as other regenerative outputs of MSCs (i.e., contents of their extracellular vesicles).
Ultrasound also has a well-documented influence on MSC differentiation. However, there are some inconsistencies, regarding how it affects MSCs in normal culture conditions. While Kusuyama et al found that LIPUS enhances stemness, in part by upregulating the stem cell factor Nanog 52 , Lai et al found that LIPUS pushes MSCs toward an osteogenic fate 53 , whereas Lee et al found continuous LIPUS (cLIPUS) to push MSCs to a chondrogenic fate 54 . These discrepancies may arise due to different ultrasound settings, culture conditions, or cell source. What has consistently been demonstrated is that when MSCs are already induced toward a certain fate, LIPUS enhances differentiation toward that lineage. For MSCs cultured in osteogenic induction media, LIPUS enhances the expression of osteogenic markers 55 . When MSCs are cultured in a chondrogenic medium, cLIPUS and LIPUS enhance the expression of chondrogenic genes and the production of glycosaminoglycans 56 . Finally, a few studies have shown that cLIPUS 57 and LIPUS 58 enhance the differentiation of MSCs into neural fates, increasing the secretion of neurotrophic factors as well as the expression of neural markers and calcium channels 57 . While changes in the MSC secretome were evaluated in our study, the phenotype stability of MSCs was not investigated. However, previous studies 59 –61 have shown that the MSC phenotype is a function of ultrasound intensity. For example, a low-intensity ultrasound (< 0.2 W/cm 2 ), applied as pulsed LIPUS or cLIPUS wave, has been documented to enhance the chondrocyte phenotype 59 , improve cartilage repair 60 , and induce hBM-MSC chondrogenesis in vitro 61 and in vivo 62 , notably in the absence of exogenous chondroinductive biochemical factors 54 . There is therefore a possibility of changes in phenotype and differentiation potential in MSCs using our pFUS parameters in both low (0.45 W/cm 2 ) and high (1.3 W/cm 2 ) intensities; however, further studies will be required to evaluate this phenomenon.
Finally, we will aim to better understand the ability of translating this approach of using pFUS from the in vitro setting (to precondition MSCs) to in vivo setting where we can stimulate MSCs after they have been given into living subjects. Once the pFUS parameters are optimized and validated, clinical applications of pFUS will only require that the derated pFUS parameters match the optimized determined pFUS parameters.
In summary, our study showed that pFUS can stimulate MSCs, and the response profile is dependent on the intensity of pFUS as well as the source of MSC. These important findings should set the foundation for additional mechanistic and validation studies using this noninvasive and translatable technology in regenerative medicine.
Footnotes
Ethical Approval
Stanford University does not require ethical approval for the use of MSCs in research.
Statement of Human and Animal Rights
Based on Stanford’s IRB, the research using MSCs does not involve human subjects as defined in 45 CFR 46.102(f) or 21 CFR 50.3(g).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the NIDDK (R01DK119293) and the Akiko Yamazaki and Jerry Yang Faculty Scholar Fund in Pediatric Translational Medicine and the Stanford Maternal and Child Health Research Institute.