Abstract
Maintaining the efficacy of anti-CD19 chimeric antigen receptor modified (CAR) T-cell therapy in patients with B-cell acute lymphoblastic leukemia (B-ALL) relapse after allogeneic hematopoietic stem cell transplant (allo-HSCT) is an urgent problem. In this study, we aimed to compare the efficacy of donor hematopoietic stem cell infusion (DSI) therapy and donor lymphocyte infusion (DLI) therapy as a maintenance therapy after R/R B-ALL patients achieved CR in anti-CD19-CAR T-cell therapy but relapsed after allo-HSCT. In total, 22 B-ALL patients who relapsed after allo-HSCT received anti-CD19-CAR T-cell therapy. Patients who responded to CAR T-cell therapy received DSI or DLI as maintenance therapy. We compared the clinical responses, acute graft versus host disease (aGVHD), expansion of CAR-T-cells, and adverse events between the two groups. In our study, 19 patients received DSI/DLI as maintenance therapy. After DSI/DLI therapy, progression-free survival and overall survival were higher in the DSI group than in the DLI group at 365 days. The grades I and II of aGVHD was observed in four patients (36.4%) in the DSI group. Only one patient developed grade II aGVHD in the DLI group. The peaks of CAR T-cells in the DSI group were higher than those in the DLI group. IL-6 and TNF-α levels increased again in nine of 11 patients after DSI but not in the DLI group. Our findings indicate that for B-ALL patients who relapse after allo-HSCT, DSI is a feasible maintenance therapy if CR is obtained with CAR-T-cell therapy.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplant (allo-HSCT) for B-cell acute lymphoblastic leukemia (B-ALL) is an effective treatment and has improved the long-term survival considerably1,2. However, some patients still relapse, with a very poor prognosis 1 . Refractory B-ALL and its progression are the major causes of death in these patients 3 . Salvage chemotherapy, a second allo-HSCT, and donor lymphocyte infusion (DLI) are considered salvage treatments for patients with B-ALL who relapse after allo-HSCT. However, all these salvage treatments have low complete response (CR) rates and poor results4–7.
Anti-CD19 chimeric antigen receptor-modified (anti-CD19-CAR) T-cell immunotherapy has shown satisfactory effects in patients with relapsed/refractory (R/R) B-ALL8–10. However, the recurrence rate after anti-CD19-CAR T-cell therapy is approximately 34% 11 . Bridging allo-HSCT after anti-CD19-CAR T-cell therapy in patients with R/R B-ALL has been reported to improve progression-free survival (PFS) without increasing the risk of severe acute graft versus host disease (aGVHD) 12 . Donor-derived anti-CD19-CAR T-cell therapy has also shown a high CR rate and promising results in B-ALL patients who relapse after allo-HSCT, without serious aGVHD in most studies13–15. It is thus a promising, safe, and effective option for B-ALL patients who relapse after allo-HSCT and might be superior to DLI 16 .
However, B-ALL patients who relapse after allo-HSCT still experience relapse after obtaining CR from anti-CD19-CAR T-cell therapy. Therefore, maintaining the efficacy of CAR-T therapy in these patients after allo-HSCT and avoiding relapse is an urgent problem that must be addressed. In the relevant literature, however, it is unclear how to prevent the recurrence of disease in these patients after CR from anti-CD19-CAR T-cell therapy. In this study, we compared the efficacy of donor hematopoietic stem cell infusion (DSI) therapy and DLI therapy as a maintenance therapy after R/R B-ALL patients achieved CR in anti-CD19-CAR T-cell therapy but relapsed after allo-HSCT.
Patients and Methods
Study Oversight
The study protocol was approved by the Tianjin First Center Hospital (Tianjin, China; Approval Nos 2015002X and 2018N105KY). All patients provided written informed consent for the collection, analysis, and publication of their anonymized outcome data.
Patient Characteristics
Twenty-two B-ALL patients who relapsed after allo-HSCT between January 2019 and August 2020 were enrolled in our study. All patients showed high CD19 expression on their B-ALL cells upon flow cytometry (FCM) analysis. They were enrolled in a clinical trial of anti-CD19 chimeric antigen receptor-modified (anti-CD19-CAR) T-cell therapy (ChiCTR16009862 and ChiCTR1800019622). None of these patients had been received blinatumomab, anti-CD19-CAR T-cell therapy or allo-HSCT before enrollment. They all provided informed consent before enrollment. From the date of anti-CD19-CAR T-cell infusion, follow-up was carried out up to the cutoff date or the date of death. The cutoff date was November 30, 2021.
Anti-CD19-CAR T-Cell Therapy
All the donors of the 22 patients with B-ALL provided their peripheral blood mononuclear cells (PBMCs) by hemapheresis for this anti-CD19-CAR T-cell therapy. CD3+ T-cells were isolated by Ficoll density gradient centrifugation and elected by CD3 microbeads (Miltenyi Biotec, Inc., Cambridge, MA, USA), then stimulated by anti-CD3/anti-CD28 mAb-coated Human T-Expander beads (Cat. no. 11141D; Thermo Fisher Scientific, Inc., Waltham, MA, USA) and cultured in T-cell medium X-Vivo 15 (Lonza Group, Ltd., Basel, Switzerland) supplemented with 250 IU/mL interleukin-2 (IL-2; Proluekin; Novartis International AG, Basel, Switzerland). All CD3+ T-cells (3 × 106) of the 22 donors were transduced with lentiviral vector encoding humanized CD19 CAR constructs (10 µg, lenti-CD19-2rd-CAR, 4-1BB costimulatory molecule; Shanghai Genbase Biotechnology Co., Ltd. Shanghai, China) and cultured in media containing recombinant human IL-2 (250 IU/mL). On the 12th to 14th day of cultivation, transduction efficiencies of anti-CD19-CAR were analyzed using FCM (BD Biosciences, San Jose, CA, USA). Lymphodepletion chemotherapy comprised fludarabine (30 mg/m2) and cyclophosphamide (400 mg/m2) from day 4 to day 2. All donor-derived anti-CD19-CAR T-cells were infused on day 0 (1 × 106 cells/kg) in patients with B-ALL.
Maintenance Therapy With Donor Hematopoietic Stem Cell Infusion (DSI) Therapy and Donor Lymphocyte Infusion (DLI) Therapy
Approximately 60 days after anti-CD19-CAR T-cell infusion, when the adverse events (AEs) and the subsequent aGVHD disappeared, patients who obtained CR or CR with incomplete count recovery (Cri) and had previously preserved frozen stem cells, received DSI (DSI group) as maintenance therapy. The other patients who reached CR/CRi received DLI (DLI group) as maintenance therapy. The cells in DLI therapy were not previously frozen, but were collected by hemapheresis without mobilization by granulocyte colony stimulating factor (G-CSF) in every DLI maintenance therapy. All DSI/DLI was the same donor used for the original hematopoietic stem cell transplant. The interval time between the two DSI/DLI therapies was generally 1 month. If the patients develop severe aGVHD, DSI/DLI maintenance therapy would be discontinued. Moreover, one patient who reached CR/Cri received a second allo-HSCT following DLI therapy (Fig. 1). One patient who did not reach CR/CRi after anti-CD19-CAR T-cell therapy received DSI as a salvage therapy. Two other patients who did not reach CR/Cri died because of disease progression within a short time.

Design process of clinical trials and grouping methods in our study.
Clinical Response Criteria and Donor Chimerism Analysis
The therapy response in this study was evaluated at 14 days, 28 days, and monthly thereafter. The detection methods included bone marrow (BM) morphology and BM flow cytometry. Disease status was defined as CR, CRi, and no remission (NR). In this study, from the date of anti-CD19-CAR T-cell infusion, follow-up was performed until the patient died. We then evaluated progression-free survival (PFS) and overall survival (OS) after anti-CD19-CAR T-cell therapy.
The changes of donor chimerism in BM were analyzed using fluorescence-labeled multiple polymerase chain reaction (PCR) amplification of short-tandem repeat (STR) on day 14 after anti-CD19-CAR T-cell infusion. Then, it was analyzed once a month after anti-CD19-CAR T-cell therapy.
Observation of aGVHD After Anti-CD19-CAR T-Cell Therapy and DSI/DLI Therapy
The occurrence and extent of aGVHD were observed from the day of anti-CD19-CAR T-cell infusion to the date of disappearance of aGVHD or death. GVHD was classified using the Glucksberg and Seattle classical scales17,18.
Adverse Events (AEs) of Anti-CD19-CAR T-Cell Therapy and DSI/DLI Therapy
AEs were observed for 28 days following anti-CD19-CAR T-cell infusion and for 100 days following DSI/DLI. The cytokine release syndrome (CRS) grade was determined according to the National Cancer Institute Common Terminology Criteria for AE v4.03 19 . Neurotoxicity syndrome was determined according to immune effector cell-associated neurotoxic syndrome (ICANS) 20 .
The proportions of anti-CD19-CAR T-cell in the peripheral blood CD3+ T-cells were determined on days 0, 4, 7, 14, 21, 28, and 56 after CAR-T-cell infusion using FCM, and on days 28, 42, and 56 after DSI/DLI therapy. Anti-CD19-CAR gene expression was detected simultaneously using quantitative polymerase chain reaction (qPCR).
Cytokine levels in peripheral blood, includinginterleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), were measured on days 0, 4, 7, 14, 21, and 28 after CAR-T-cell infusion and on days 28, 42, and 56 following DSI/DLI therapy using respective enzyme-linked immunosorbent assays.
Follow-Up
From the date of anti-CD19-CAR T-cell infusion, follow-up was performed until the cutoff date or the date of death. Progression-free survival (PFS) and overall survival (OS) were calculated from the date of anti-CD19-CAR T-cell infusion.
Statistical Analysis
Data are expressed as mean ± SE. CRS and ICANS were grouped and compared using the Mann–Whitney rank test. Nonnormal distribution data are expressed as median and interquartile range (IQR). The Pearson correlation coefficient was used to evaluate the correlation between different factors. The PFS and OS probabilities were estimated using the Kaplan–Meier method and were compared using the log-rank test. All statistical analyses were performed using SPSS (version 17.0). Statistical significance was set at P < .05.
Results
Patient Characteristics
Overall, 22 patients with B-ALL who relapsed after allo-HSCT followed by anti-CD19-CAR T-cell therapy were included in this study. The detailed characteristics of the patients are shown in Table 1. The median time from allo-HSCT to relapse was 13 months (IQR 7–15 months). The median proportion of leukemia cells was 44.7% (IQR 32.8%–58.4%) in BM and 37.2% (IQR 25.8%–45.5%) in the peripheral blood (PB) when patients were enrolled. The median percentage of donor BM chimerism at enrollment was 55.9% (IQR 48.2%–71.3%). Patients had no GVHD or central nervous system disease at enrollment. Moreover, patients had not previously received blinatumomab or CD19-CAR T-cell therapy.
Patients Baseline Characteristics.

aGVHD: acute graft versus host disease; B-ALL: B-cell acute lymphoblastic leukemia; BM: bone marrow; DLI: Donor lymphocyte infusion; DSI: Donor hematopoietic stem cell infusion; GVHD: graft versus host disease; Haplo-HSCT: Haploid donor transplantation; MMUDT: HLA-mismatched unrelated donor transplantation; MSDT: HLA-matched sibling donor transplantation; MUDT: HLA-matched unrelated donor transplantation; PB: peripheral blood; TKI: tyrosine kinase inhibitor.
Clinical Responses and Donor Chimerism Analysis After Anti-CD19-CART-Cell Therapy and Maintenance Therapy With DSI/DLI
In the first evaluation of therapy response on day 14 after anti-CD19-CAR T-cell infusion, 19 patients (19/22, 86.4%) achieved CR/CRi. Of these, six (6/19, 31.6%) achieved CR and 13 patients (13/19, 68.4%) achieved CRi. Seventeen patients (17/19, 89.5%) achieved minimal residual disease (MRD)-negative responses, whereas two patients were evaluated for CR/CRi with MRD-positive response (PtDSI5,11). The other three patients were evaluated as NR owing to the progression of leukemia.
After 14 days of anti-CD19-CAR T-cell infusion, the donor chimerism in BM increased from 56.3 ± 22.9 to 99.9 ± 0.2% in the 19 patients who achieved CR/CRi. The donor chimerism in a patient evaluated as NR with extramedullary leukemia was 99.0%, whereas it was 10.7% and 29.2% in the other two patients who were evaluated as NR. Before or after anti-CD19-CAR T-cell therapy, there was no difference in donor chimerism in the DSI and DLI groups (Pbefore = .056 and Pafter = .828).
In maintenance therapy with DSI/DLI following anti-CD19-CAR T-cell therapy, the donor chimerism in BM was analyzed once a month. It remained almost above 99.9% in all the patients in the DSI/DLI group who maintained CR/CRi.
Maintenance Therapy With DSI/DLI Following the Anti-CD19-CART-Cell Therapy
Of the patients achieving CR/CRi, 11 patients (Pt DSI 1-11) who had previously preserved frozen stem cells received DSI, whereas the other eight patients (Pt DLI 1-8) received unfrozen DLI maintenance therapy. Only one patient (Pt DLI 7) received a second allo-HSCT after DLI. In the DSI group, six patients received one DSI treatment, whereas five received two DSI treatments. In the DLI group, three patients received one DLI treatment, whereas four patients received DLI twice and one patient received DLI thrice. As all patients received DSI/DLI for different times, we only compared the changes in various indicators after the first DSI/DLI therapy.
In DSI/DLI maintenance therapy, the median of donor CD34+ cells infused in these patients was 1.3 (IQR 1.2–1.4) × 105 cells/kg and that of CD3+ T-cells was 2.2 (IQR 1.9–2.3) × 107 cells/kg in DSI therapy. In the DLI group, the median of donor CD3+ T-cells infused was 2.4 (IQR 2.2–2.7) × 107 cells/kg. There was no difference in the number of CD3+ T-cell infusions between the two groups (P = .351). The median percentage of CD19+ cells in the donor product in the DSI group was 0.02% ± 0.03%, whereas it was 2.20% ± 0.71% in the DLI group.
One patient who did not achieve CR/CRi with the anti-CD19-CAR T-cell therapy received DSI once (Pt3). The donor CD34+ cells and CD3+ cells infused was 1.4 × 105 cells/kg and 1.9 × 107 cells/kg, respectively.
aGVHD After Anti-CD19-CAR T-Cell Therapy and After DSI/DLI Therapy
After anti-CD19-CAR T-cell therapy, aGVHD was observed in six patients (6/11, 54.6%) from 19 to 48 days before DSI, and it was observed in four patients (4/8, 50.00%) before DLI. Only one patient (Pt DSI 5) developed grade III aGVHD, whereas no patient developed grade-III -IV aGVHD in the DLI group. There was no difference in the grade of aGVHD between the two groups (P = .732).
In the DSI group, aGVHD was observed in four patients (4/11, 36.4%) from 32 to 56 days after the first DSI. These four patients (PtDSI1, 2, 4, and 6) developed grade-I and grade-II aGVHD. Only one patient (PtDLI3) developed grade-II aGVHD after 52 days of the first DLI. There was no difference in aGVHD between the two groups after DSI/DLI therapy (P=0.636). None of the patients in the DSI and DLI groups developed grade-III and grade-IV aGVHD. Pt 3 did not develop any grade of aGVHD after DSI. None of the patients died because of aGVHD in our study.
Expansion of Anti-CD19-CAR T-Cell
The proportion of anti-CD19-CAR T-cell was detected using FCM at 0, 4, 7, 14, 21, 28, and 56 days after anti-CD19-CART-cell infusion and at 28, 42, and 56 days after the first DSI/DLI therapy (Fig. 2A). The median peak of theanti-CD19-CAR T-cell in CD3+ T-cells in peripheral blood was34.9% ± 14.0% on day 8.3 ± 2.7 of anti-CD19-CART-cell therapy in all 22 patients. In the process of anti-CD19-CAR T-cell therapy, there was no difference in the expansion peaks of anti-CD19-CART-cell in the DSI and DLI groups (P = .124) (Fig. 2B). From 28 to 56 days after DSI therapy, the expansion of anti-CD19-CART-cell increased again in nine of 11 patients (Fig. 2A). The expansion peaks of anti-CD19-CART-cell in the DSI group were higher than those in the DLI group after DSI/DLI therapy (P < .0001) (Fig. 2C).
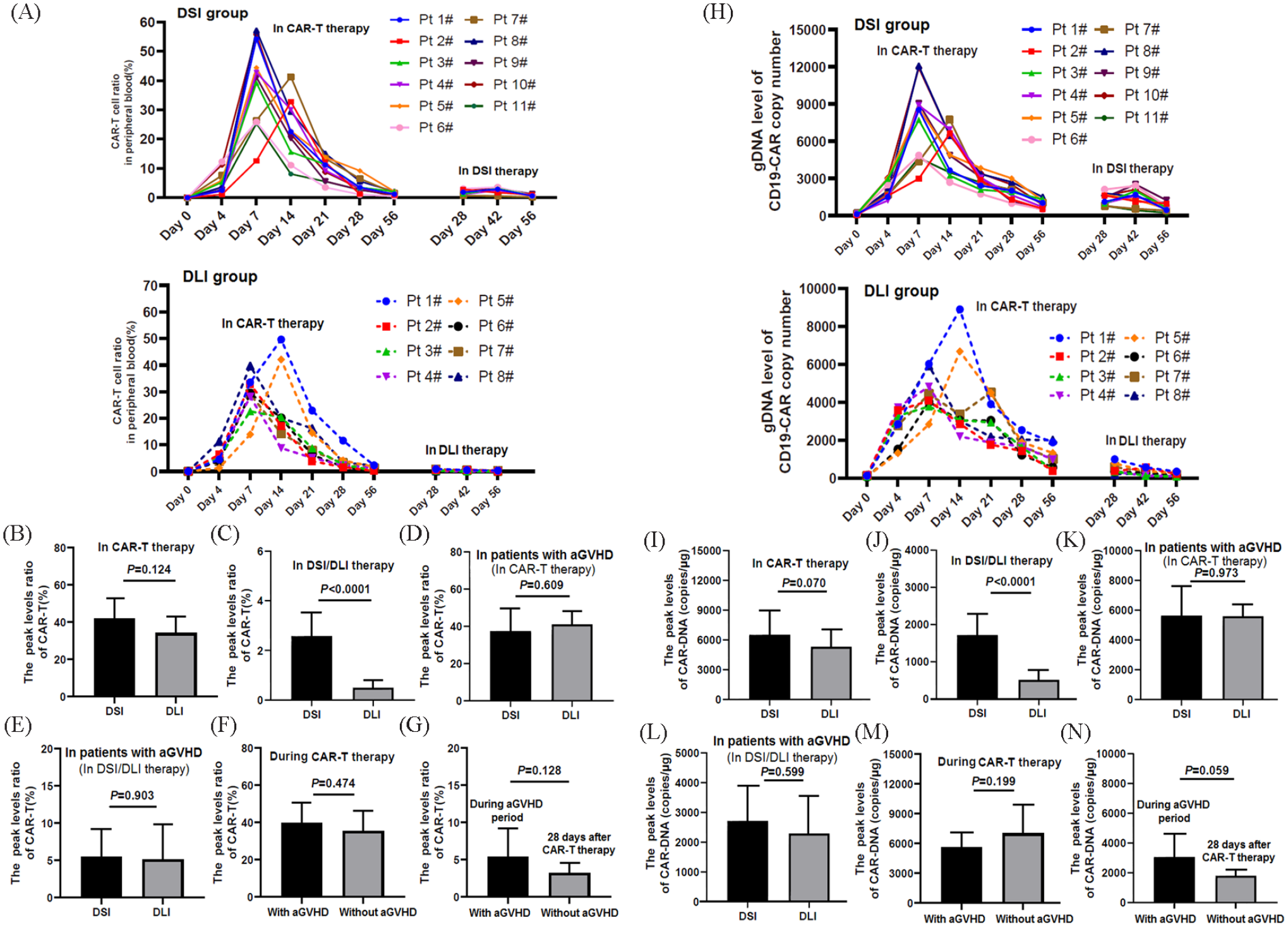
Expansion of anti-CD19-CAR T-cell and anti-CD19-CAR DNA changes upon anti-CD19-CAR T-cell therapy and DSI/DLI therapy. (A) The proportion of anti-CD19-CAR T-cell upon anti-CD19-CART-cell therapy and DSI/DLI therapy. (B) No difference in the expansion peaks of anti-CD19-CART-cell in the two groups with anti-CD19-CAR T-cell therapy. (C) The peaks of the anti-CD19-CART-cell in the DSI group were higher than those in the DLI group. (D and E) In patients with aGVHD after CAR-T-cell therapy, there was no difference in the expansion peaks of CAR-T-cells in the period of CAR-T-cell therapy and in the period of aGVHD between the two groups. (F) There was no difference in the average expansion peaks of the anti-CD19-CART-cell in the period of CAR-T-cell therapy between the patients with and without aGVHD before DSI/DLI therapy. (G) There was no difference in the average expansion peaks of the anti-CD19-CART-cell at 28 days after CAR-T-cell therapy between patients with and without aGVHD. (H) The average level peaks of anti-CD19-CAR DNA in patients with anti-CD19-CAR-T-cell therapy and DSI/DLI therapy. (I) There was no difference in the average level peaks of anti-CD19-CAR DNA in the two groups in anti-CD19-CAR T-cell therapy. (J) In DSI/DLI therapy, the average anti-CD19-CAR DNA peaks increased again in the DSI group but not in the DLI group. (K and L) In patients with aGVHD after CAR-T-cell therapy, there was no difference in the average level peaks of anti-CD19-CAR DNA in the period of CAR-T-cell therapy and in the period of aGVHD between the two groups. (M) There was no difference in the average level peaks of anti-CD19-CAR DNA in the period of CAR-T-cell therapy between patients with and without aGVHD before DSI/DLI therapy. (N) There was no difference in the average level peaks of anti-CD19-CAR DNA at 28 days after CAR-T-cell therapy between patients with and without aGVHD.
In all patients with aGVHD after CAR-T-cell therapy or DSI/DLI therapy, there was no difference in the expansion peaks of anti-CD19-CART-cell in the period of CAR-T-cell therapy and in the period of DSI/DLI therapy between the DSI and DLI groups (PCAR-T = .609 and PDSI/DLI = .903) (Fig. 2D, E).
In the period of CAR-T-cell therapy, there was no difference in the expansion peaks of the anti-CD19-CART-cell between patients with and without aGVHD (P = .474) (Fig. 2F). The mean peak of anti-CD19-CART-cell in patients with aGVHD in the period of aGVHD before DSI/DLI therapy was 5.4% ± 3.9%, and was not higher than that the mean peak of anti-CD19-CART-cells in patients without aGVHD at 28 days after CAR T-cell infusion (3.2% ± 1.4%; P = .128) (Fig. 2G).
Anti-CD19-CAR DNA Changes in Anti-CD19-CAR T-Cell Therapy and DSI/DLI Therapy
In the two groups of patients, the copies of anti-CD19-CAR DNA were detected at 0, 4, 7, 14, 21, 28, and 56 days after CAR-T-cell infusion and at 28, 42, and 56 days after the first DSI/DLI therapy (Fig. 2H). In the process of anti-CD19-CAR T-cell therapy, there was no difference in the median peak of anti-CD19-CAR DNA in the DSI and DLI groups (P = .070) (Fig. 2I). From 28 to 56 days after the first DSI/DLI therapy, the median anti-CD19-CAR DNA peaks in the DSI group increased again after DSI therapy, but not in the DLI group (P < .0001) (Fig. 2J).
In all patients with aGVHD after CAR-T-cell therapy or DSI/DLI therapy, there was no difference in the mean peak of anti-CD19-CAR DNA in the period of CAR-T-cell therapy and in the period of DSI/DLI therapy between the two groups (PCAR-T=0.973 and PDSI/DLI = .599) (Fig. 2K, L). In anti-CD19-CART-cell therapy and in DSI/DLI therapy, the mean peak of anti-CD19-CAR DNA showed the same trend as that of the mean peak of anti-CD19-CART-cells between patients with and without aGVHD (P = .199 and P = .059) (Fig. 2M, N).
Cytokine Levels in Anti-CD19-CAR T-Cell Therapy and DSI/DLI
In the anti-CD19-CAR T-cell therapy, the cytokines reached their peaks at 7 to 10 days after anti-CD19-CAR T-cell infusion, and then declined from 14 to 18 days after infusion (Fig. 3A). There was no difference in the peaks of IL-6 and TNF-α in the two groups in anti-CD19-CART-cell therapy and in the period of aGVHD before DSI/DLI therapy (Fig. 3B, C). However, following DSI/DLI therapy, the IL-6 and TNF-α levels increased again in nine of the 11 patients in the DSI group. All four patients who developed aGVHD after DSI therapy were included in these nine patients. However, the levels of the two cytokines did not increase again in the DLI group after DLI (Fig. 3D).

The level of cytokines, cytokine release syndrome (CRS), and immune effector cell-associated neurotoxic syndrome (ICANS) in the therapy. (A) The level of IL-6 and TNF-α in the anti-CD19-CAR T-cell therapy and DSI/DLI therapy. (B) There was no difference in the peaks of IL-6 and TNF-α in the DSI/DLI groups in CAR-T-cell therapy. (C) There was no difference in the peaks of cytokines in patients with aGVHD before DSI/DLI therapy. (D) IL-6 and TNF-α levels increased again in nine of 11 patients in the DSI group after DSI but not in the DLI group. (E and F) There were no differences in the grades of CRS and ICANS between the two groups in anti-CD19-CAR T-cell therapy.
Observation of AEs Upon Anti-CD19-CAR T-Cell Therapy and DSI/DLI Therapy
In anti-CD19-CAR T-cell therapy, clinical symptoms were similar to those reported in our previous studies 21 . These AEs resolved 14–18 days after anti-CD19-CAR T-cell infusion. There was no difference in the incidence of AEs between the two groups during this period (Table 2). In DSI/DLI therapy, at 28 to 56 days post DSI/DLI treatment, AEs reappeared in the two groups. There was no difference in the incidence of AEs between the two groups during this period as well (Table 2). The duration of AEs in the DSI/DLI treatment ranged from 7 to 15 days.
The Notable Adverse Events (AEs) in DSI/DLI Groups.

CAR-T cell: chimeric antigen receptor modified (CAR) T-cell; DLI: donor lymphocyte infusion; DSI: donor hematopoietic stem cell infusion.
Eighteen patients who obtained CR/CRi (18/22, 81.8%) had grade 3-4 hematological toxicity after anti-CD19-CAR T-cell infusion. The other four patients only had grade 2 hematological toxicities. The hematological toxicity in all patients was recovered prior to their subsequent DSI/DLI therapy.
In anti-CD19-CAR T-cell therapy, 17 (17/22, 77.3%) patients were diagnosed with grade 0–2 of CRS, whereas five (5/22, 22.7%) patients were diagnosed with grade 3-4 of CRS. Only one patient in the DSI group and two patients in the DLI group developed grade-1 ICANS during this period. There were no differences in the grades of CRS and ICANS between the two groups receiving anti-CD19-CAR T-cell therapy (Fig. 3E, F). No CRS or ICANS-related deaths were observed in our study.
Antipyretic drugs and methylprednisolone were administered to overcome AEs. After anti-CD19-CAR T-cell therapy or DSI/DLI therapy, five patients received JAK1/JAK2 kinase inhibitor (Ruxolitinib) to treat grade II-III aGVHD. Only four patients (PtDSI 3,5 and Pt DLI 2,4) who developed grade 3-4 CRS received to cilizumab after anti-CD19-CAR T-cell therapy. None of the patients received tocilizumab after DSI/DLI therapy.
Survival After the Anti-CD19-CAR T-Cell Therapy and DSI/DLI Therapy
By November 30, 2021, 13 patients survived without leukemia. Anti-CD19-CAR T-cell therapy and DSI/DLI therapy, second allo-HSCT, response to anti-CD19-CAR T-cell therapy, occurrence of aGVHD, PFS and OS, and cause of death are shown in Fig. 4A. The rates of PFS and OS at 180 days were 90.91% and 90.91% in the DSI group, respectively, and those at 365 days were 63.64% and 63.64% in the DSI group, respectively. The PFS and OS rates were 62.50% and 50.50% at 180 days and 12.50% and 12.50% at 365 days in the DLI group, respectively. Although the rates of PFS and OS in the DSI group were higher at 180 days, there was no difference in the rate of PFS or OS between the two groups at 180 days (PPFS = .075 and POS = .057). The PFS and OS rates in the DSI group were higher than those in the DLI group at 365 days (PPFS = .010 and POS = .009; Fig. 4B, C). Moreover, Pt1, 2, and 3 did not respond to anti-CD19-CAR T-cell therapy and died of leukemia. PtDSI 11 and PtDLI 6 died of disease recurrence with negative CD19 expression, whereas PtDLI 5 and 8 died of disease recurrence with positive CD19 expression. PtDSI 2 died of cytomegalovirus infection at 720 days post anti-CD19-CAR T-cell infusion and 620 days after the second DSI therapy. PtDLI 4 died of sudden cardiac death at 83 days after anti-CD19-CAR T-cell infusion and 22 days after DLI therapy without aGVHD.
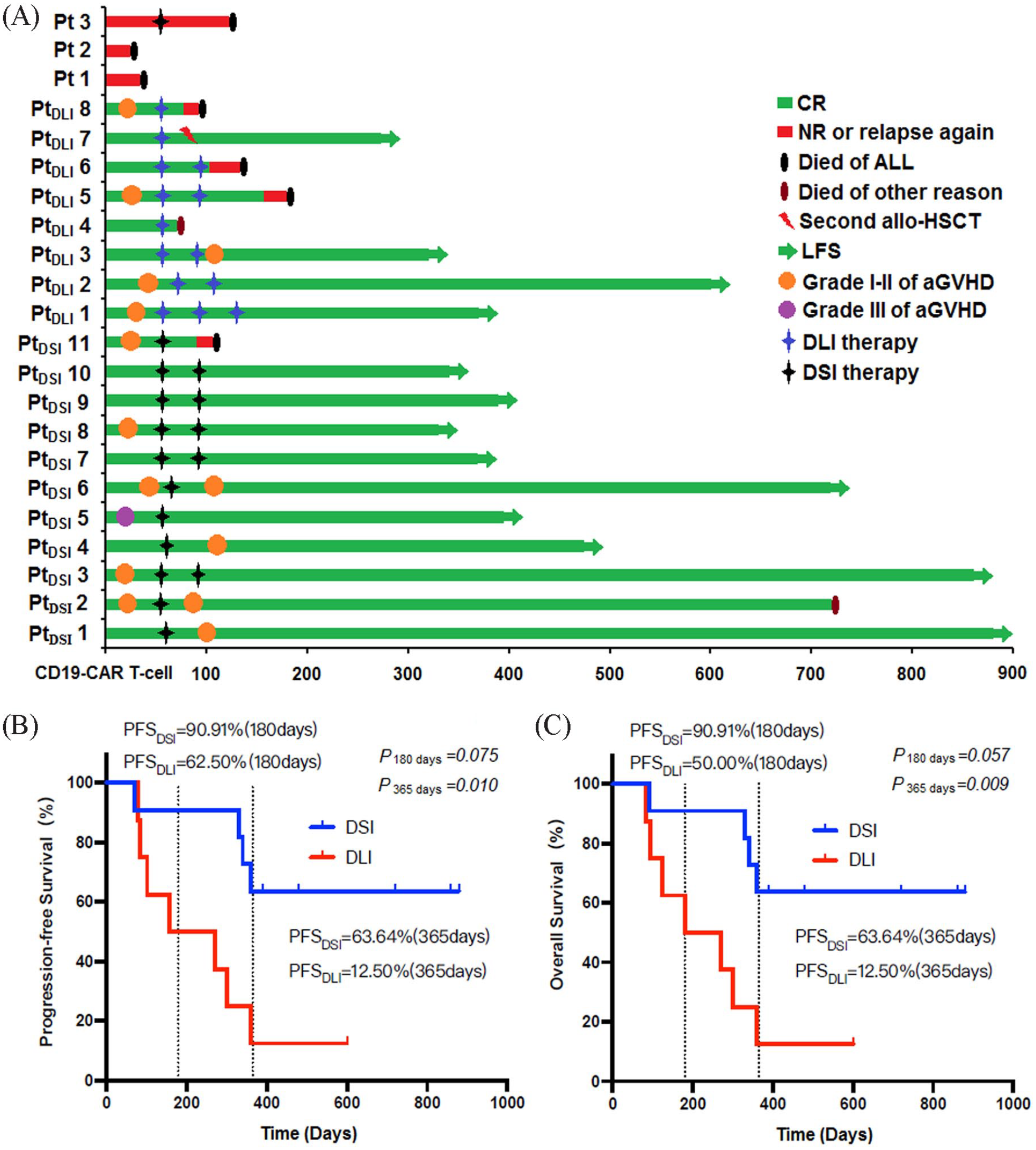
Survival observation for anti-CD19-CAR T-cell therapy and DSI/DLI therapy. (A) All treatment processes, occurrence of aGVHD, leukemia-free survival (LFS), progression-free survival (PFS) and overall survival (OS), cause of death. (B and C) The rates of PFS and OS in the DSI group were higher at 180 days, but there was no difference in PFS or OS between the two groups at 180 days. The rates of PFS and OS in the DSI group were higher than those in the DLI group at 365 days.
Discussion
Relapse after allo-HSCT remains a major issue in B-ALL treatment failure. Patients with higher risk B-ALL are still at a high rate of recurrence after allo-HSCT, with a short survival1,22,23. Owing to the poor efficacy of salvage chemotherapy, some studies have selected DLI therapy for patients with B-ALL who relapsed after allo-HSCT24–27. This could induce durable remission by enhancing the graft versus leukemia (GVL) effect 28 . The efficacy of DLI varies among different types of leukemia. DLI therapy was more effective in chronic myeloid leukemia (CML) relapse after allo-HSCT than in ALL relapse after allo-HSCT, with CR rates of 70% to 80% and less than 40%, respectively29,30. Moreover, DLI has been found more potent in acute myelocytic leukemia (AML) than in ALL 31 . However, the GVL effect of DLI therapy usually leads to different levels of aGVHD. Infusion of large numbers of donor T-cells might result in serious aGVHD or even therapy-related mortality32,24. Compared with DLI therapy, few studies have assessed DSI therapy after allo-HSCT. Furthermore, despite severe aGVHD, the prognosis of DLI therapy remains unsatisfactory, especially in patients with B-ALL who relapsed after allo-HSCT.
Donor anti-CD19-CAR T-cell therapy is an alternative salvage therapy for patients with B-ALL who relapse after allo-HSCT. Compared with DLI therapy and other conventional therapies, anti-CD19-CAR T-cell therapy is relatively safe and effective. Allogeneic anti-CD19-CAR T-cell could clear leukemia cells directly, they also exhibit GVL effects without serious aGVHD in most studies14,15,33–35. However, maintaining the efficacy of anti-CD19-CAR T-cell therapy after allo-HSCT remains an urgent problem. Our previous study showed that DSI and DLI could be used as maintenance treatments after anti-CD19-CAR T-cell therapy for patients with B-ALL who relapsed after allo-HSCT. DSI induced an increased proportion of anti-CD19-CAR T-cell and an increased level of anti-CD19-CAR DNA expression with mild aGVHD. This might lead to further clearance of minimal residual disease (MRD) and longer PFS 36 . Notably, our DSI or DLI therapy was initiated when the patients with B-ALL who relapsed after allo-HSCT had obtained a CR from their anti-CD19-CAR T-cell therapy without relapse.
DLI therapy in patients with complete donor chimerism without MRD was defined as prophylactic DLI therapy (pro-DLI), which could obviously reduce the relapse rate37–39. Therefore, early DLI therapy after allo-HSCT may be a more reasonable and effective therapy. In our study, 19 B-ALL patients who relapsed after allo-HSCT, obtained CR/CRi from their anti-CD19-CAR T-cell therapy. DSI or DLI therapy was administered 60 days after anti-CD19-CAR T-cell therapy to prevent their disease progression. After 1–3 times of DSI or DLI therapy, the PFS and OS were higher in the DSI group than in the DLI group at 365 days. Thus, further studies with increased number of cases are needed to confirm these observations. However, DSI maintenance therapy has the potential to present better survival in patients who relapsed after allo-HSCT and obtained CR from anti-CD19-CAR T-cell therapy.
We observed the side effects of the two types of maintenance treatments and found no difference in the grade of aGVHD in the DSI and DLI groups during the anti-CD19-CAR T-cell therapy and the DSI/DLI therapy. After DSI therapy, the expansion of anti-CD19-CART-cell increased again in nine of the 11 patients in DSI group. The peaks of the anti-CD19-CART-cell were higher than those in DLI group simultaneously. This suggests that DSI therapy might facilitate the re-amplification of anti-CD19-CART-cell. The levels of IL-6 and TNF-α also increased again in DSI group after DSI.
In conclusion, progression-free survival and overall survival were higher in the DSI group than in the DLI group at 365 days. For patients with B-ALL who relapsed after allo-HSCT, DSI is a feasible maintenance therapy if CR is obtained with CAR-T-cell therapy. However, it is not clear why it is more effective in the DSI group. The possible difference between the DSI group and DLI group was that the re-amplification of anti-CD19-CAR T-cells was higher in the DSI group. The re-amplification of CAR-T-cells led to the elimination of remaining ALL cells, which might be one of the reasons for the better efficacy in the DSI group. Whether the reason for the re-amplification of anti-CD19-CAR T-cell in the DSI group was the re-appearance of B lymphocytes due to the implantation of stem cells in peripheral blood, which led to the clearance of normal B lymphocytes and the re-amplification of CAR-T-cells, requires further studies.
Footnotes
Author Contributions
LQ performed the study, analyzed the data, designed the figures, and wrote the paper. LCC, WJ, and JEL performed clinical works. MN provided materials and technical supports for cell therapy. LMJ and LCC did the statistical analysis. DQ and ZRL supervised the study, designed the clinical trial, and analyzed the data. LQ wrote the paper. All authors read and approved the final manuscript.
Ethical Approval
This study was approved by the Medical Ethics Committee of the Tianjin First Center Hospital (Tianjin, China).
This Clinical trial is registered at ![]() as ChiCTR-ONN-16009862 and http://www.chictr.org.cn/index.aspx as ChiCTR1800019622.
as ChiCTR-ONN-16009862 and http://www.chictr.org.cn/index.aspx as ChiCTR1800019622.
Statement of Human Rights
All procedures in this study were conducted in accordance with the ethics committee of the Tianjin First Center Hospital (Tianjin, China). (Approved no. of ethic committee: 2015002X and 2018N105KY.) This article does not contain any studies with animal subjects.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by the National Natural Science Foundation of China (81900186) and the Tianjin Municipal Science and Technology Commission Grant (21JCQNJC00070).
