Abstract
For ischemic cardiomyopathy (ICM) with limited therapeutic options, the induction of arteriogenesis has the potential to improve cardiac function through major restoration of blood flow. We hypothesized that transplantation of a Notch signaling-modified mesenchymal stem cell (SB623 cell) patch would induce angiogenesis and arteriogenesis in ischemic lesions, leading to improvement of left ventricular (LV) function in a rat ICM model. Two weeks after the induction of ischemia, SB623 cell patch transplantation into ICM rats (SB group, n = 10) or a sham operation (no-treatment group, n = 10) was performed. The LV ejection fraction was significantly improved at 6 weeks after SB623 cell patch transplantation (P < 0.001). Histological findings revealed that the number of von Willebrand factor (vWF)-positive capillary vessels (P < 0.01) and alpha smooth muscle actin (αSMA)- and vWF-positive arterioles with a diameter greater than 20 µm (P = 0.002) was significantly increased in the SB group, suggesting the induction of angiogenesis and arteriogenesis. Moreover, rat cardiomyocytes treated with SB623 cell patch transplantation showed upregulation of ephrin-B2 (P = 0.03) and EphB4 (P = 0.01) gene expression, indicating arteriogenesis induction. In conclusion, SB623 cell patch transplantation improved LV function by inducing angiogenesis and arteriogenesis in a rat ICM model.
Introduction
Patients with ischemic cardiomyopathy (ICM) have a poor prognosis, even if treated with maximum surgical intervention, such as coronary artery bypass grafting (CABG), mitral valve surgery, or left ventricular (LV) reconstruction1–3. Cellular therapy is under investigation for use as a supplement or alternative to those surgical strategies, especially for patients who are not eligible for mechanical circulatory support or heart transplantation4,5. Although the Thrombolysis in Myocardial Infarction (TIMI) trial and other studies involving patients who underwent CABG have suggested that blood flow restoration is associated with improved left ventricular ejection fraction (LVEF), leading to functional recovery and better survival, the beneficial effects of CABG were only observed in a limited number of patients6–10. A possible explanation for the limited therapeutic effect is the inability of CABG to form a microvascular network, which is a major microcirculation component. In contrast, cellular therapy, which induces angiogenesis and improves microcirculation, is expected to enhance therapeutic effects in patients with ICM. However, no clinical trial has clearly shown the beneficial effects of cellular therapy with regard to survival, and further enhancement of the therapeutic effects is required4,5.
Arteriogenesis has been widely investigated to improve the effects of cellular therapy for ischemic disorders and the induction of mature large-caliber arteries11–13. Arteriogenesis has shown the potential to dramatically restore blood flow; thus, it is expected to strengthen the therapeutic effects of angiogenesis14,15. Previously, we demonstrated that Notch signaling-modified human mesenchymal stem cell (hMSC) (SB623 cell) transplantation improved tissue perfusion via arteriogenesis in a hindlimb ischemia model 16 . In the study, arteriogenesis was successfully induced by the upregulation of various arteriogenic pathways, including Notch signaling. Furthermore, we demonstrated the efficacy and safety of cell patch transplantation, indicating that it is a promising delivery method for promoting angiogenesis in patients with heart failure17–20. For this study, we hypothesized that SB623 cell patch transplantation improves LV function by inducing angiogenesis and arteriogenesis in a rat ICM model.
Materials and Methods
SB623 Cell Patch Preparation
Notch signaling-modified hMSC, SB623 cells were produced by transient transfection of a plasmid encoding the human Notch-1 intracellular domain gene sequence into hMSCs obtained from the bone marrow of a healthy human adult donor (SanBio Inc, Mountain View, CA, USA). Bone marrow aspirate for the production of hMSCs used in this study was prepared by Lonza Walkersville Inc (Walkersville, MD, USA), and informed consent was obtained by that company. Changes in Notch pathway profiling and responses of selected Notch target genes were ascertained in previous studies21–23. Transient transfection with the plasmid encoding the Notch-1 intracellular domain was confirmed by quantitative polymerase chain reaction of the plasmid DNA and Western blotting to determine the expressed recombinant Notch-1 intracellular domain protein. Cell patches consisting of 106 SB623 cells were prepared using temperature-responsive culture dishes (UpCell; CellSeed, Koto, Tokyo, Japan) 24 . Briefly, 106 SB623 cells/well were plated in a 24-well UpCell dish and then cultured at 37°C and 5% CO2 overnight. Before transplantation, the cells in each well were spontaneously detached at room temperature as a cell patch.
Animal Care
All animal protocols were approved by the Animal Experimentation Committee of Osaka University (approval number: 04522) and were performed in accordance with the Guidelines for Animal Experiments of Osaka University.
ICM Model
A rat ICM model was established by ligating the proximal site of the left anterior descending coronary artery in athymic nude rats (F344/NJcl-rnu/rnu, 8 weeks old, female; CLEA Japan, Meguro, Tokyo, Japan) 25 . Two weeks following ischemic induction, SB623 cell patch transplantation (n = 10) or a sham operation (n = 10) was performed. In the cell patch transplantation group, an SB623 cell patch was placed on the ischemic area of the heart. A left thoracotomy approach, conducted under mechanical ventilation, was used for ischemic induction followed by transplantation or a sham operation.
Evaluation of Cardiac Function
Cardiac function was evaluated using echocardiography (Sonos 7500; Philips, Andover, MA, USA) at 2, 4, and 6 weeks after cell patch transplantation. The left ventricular end-diastolic diameter (LVDd) and left ventricular end-systolic diameter (LVDs) were measured under 3% isoflurane anesthesia, and LVEF was calculated as follows: LVEF (%) = (LVDd 3 − LVDs 3 ) / LVDd 3 × 100.
Histological Analysis
The rats were euthanized with CO2 6 weeks after cell transplantation. Hearts were dissected, formalin-fixed, paraffin-embedded, and cut into 5-µm sections using a microtome for histological analysis. To assess capillary vessels, ischemic muscle sections were stained with a sheep polyclonal anti-von Willebrand factor (vWF) antibody (AB7356, 1:50; Millipore, Billerica, MA, USA). To assess arteriogenesis, samples were stained with sheep polyclonal anti-vWF and rabbit polyclonal anti-alpha smooth muscle actin (αSMA) (MO851, 1:50; Dako, Carpinteria, CA, USA) antibodies, and then, arterioles greater than 20 µm were counted 26 . Furthermore, the sections were stained using sirius red. Fibrosis was quantified based on the collagen volume fraction using the Metamorph software package (Molecular Devices, Sunnyvale, CA, USA). Histological measurements were performed in five randomly selected fields for each tissue section. The images obtained were examined using optical microscopy (Keyence, Osaka, Osaka, Japan).
Real-Time Quantitative Polymerase Chain Reaction
Gene expression in the heart samples was measured using real-time quantitative polymerase chain reaction (RT-PCR). Each heart was harvested 5 days after cell transplantation or sham operation and then immersed in RNAlater (Invitrogen, Carlsbad, CA, USA). Total RNA was isolated using an RNeasy Kit (Qiagen, Hilden, Germany), and reverse transcription was performed using an Omniscript Reverse Transcriptase kit (Qiagen). RT-PCR was performed using the TaqMan Gene Expression Assay Master Mix (Applied Biosystems, Foster City, CA, USA) on the 7500 Fast Real-Time PCR System (Applied Biosystems). The primers used in this study were as follows: ephrin-B2 (assay ID: Rn01756899_m1), EphB4 (assay ID: Rn01481051_m1), fibroblast growth factor (FGF)-2 (assay ID: Rn00570809_m1), glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (assay ID: Rn01775763_g1), hepatocyte growth factor (HGF) (assay ID: Rn00566673_m1), monocyte chemotactic protein (MCP)-1 (assay ID: Rn00580555_m1), placental growth factor (PIGF) (assay ID: Rn01472372_m1), and vascular endothelial growth factor (VEGF) (assay ID: Rn01511601_m1).
Statistical Analysis
All continuous variables are summarized as the mean ± standard deviation and were compared using Student’s t test. All P values were two-sided, and statistical significance was set at P < 0.05. All statistical analyses were performed using JMP Pro 14 (SAS Institute, Cary, NC, USA).
Data Availability
The datasets of this study are available from the corresponding author upon reasonable request.
Results
SB623 Cell Patch Transplantation Improved Cardiac Function
The effects of SB623 cell patch transplantation on cardiac function in ICM model rats were assessed. Two weeks after ischemic induction, the SB623 cell patch transplantation or sham operation was performed. Before transplantation, LVEF (no-treatment group vs SB group; 40.4 ± 4.5% vs 37.6 ± 8.0%, P = 0.38), LVDs (6.1 ± 0.3 mm vs 6.1 ± 0.5 mm, P = 0.98), and LVDd (7.3 ± 0.2 mm vs 7.3 ± 0.4 mm, P = 0.64) were not significantly different between the two groups. Two weeks after SB623 cell patch transplantation, LVEF in the SB group was significantly improved (39.4 ± 4.0% vs 47.7 ± 6.1%, P = 0.004), and the improvement was maintained at week 4 (37.8 ± 4.7% vs 51.2 ± 6.2%, P < 0.001) and week 6 (33.7 ± 5.3% vs 52.3 ± 6.5%, P < 0.001) (Fig. 1A). Similarly, LVDs was significantly smaller in the SB group at week 2 (6.4 ± 0.5 mm vs 5.9 ± 0.5 mm, P = 0.04), week 4 (6.7 ± 0.6 mm vs 5.8 ± 0.8 mm, P = 0.02), and week 6 (7.3 ± 0.6 mm vs 5.6 ± 0.7 mm, P < 0.001) after transplantation (Fig. 1B). In contrast, LVDd was not statistically different between the two groups at week 2 (7.7 ± 0.5 mm vs 7.4 ± 0.5 mm, P = 0.18) or week 4 (7.9 ± 0.6 mm vs 7.6 ± 0.7 mm, P = 0.37) after transplantation, though it was significantly decreased in the SB group at week 6 (8.5 ± 0.5 mm vs 7.3 ± 0.7 mm, P < 0.001) (Fig. 1C), indicating attenuation of LV remodeling.

Evaluation of left ventricular systolic function and remodeling by echocardiography. (A) Comparison of LVEF. (B) Comparison of LVDs. (C) Comparison of LVDd. LVEF: left ventricular ejection fraction; LVDd: left ventricular end-diastolic diameter; LVDs: left ventricular end-systolic diameter. *P < 0.05; **P < 0.01.
Induction of Angiogenesis and Arteriogenesis
Capillary and arteriole numbers were analyzed on myocardium tissue cross-sections 6 weeks after SB623 cell patch transplantation. Compared with that in the no-treatment group, the number of capillary vessels in the peri-infarct myocardium stained with anti-vWF was significantly increased in the SB group (246.4 ± 22.3/mm2 vs 513.6 ± 97.3/mm2, P < 0.001) (Fig. 2A). Representative images of capillary staining are shown in Fig. 2B, C. Similarly, in the peri-infarct zone, the number of mature arterioles defined as anti-αSMA and anti-vWF positive with a diameter greater than 20 μm was also significantly increased in the SB group (6.0 ± 2.8/mm2 vs 24.8 ± 7.7/mm2, P = 0.002) (Fig. 2D). Representative images of arteriole staining are shown in Fig. 2E, F.
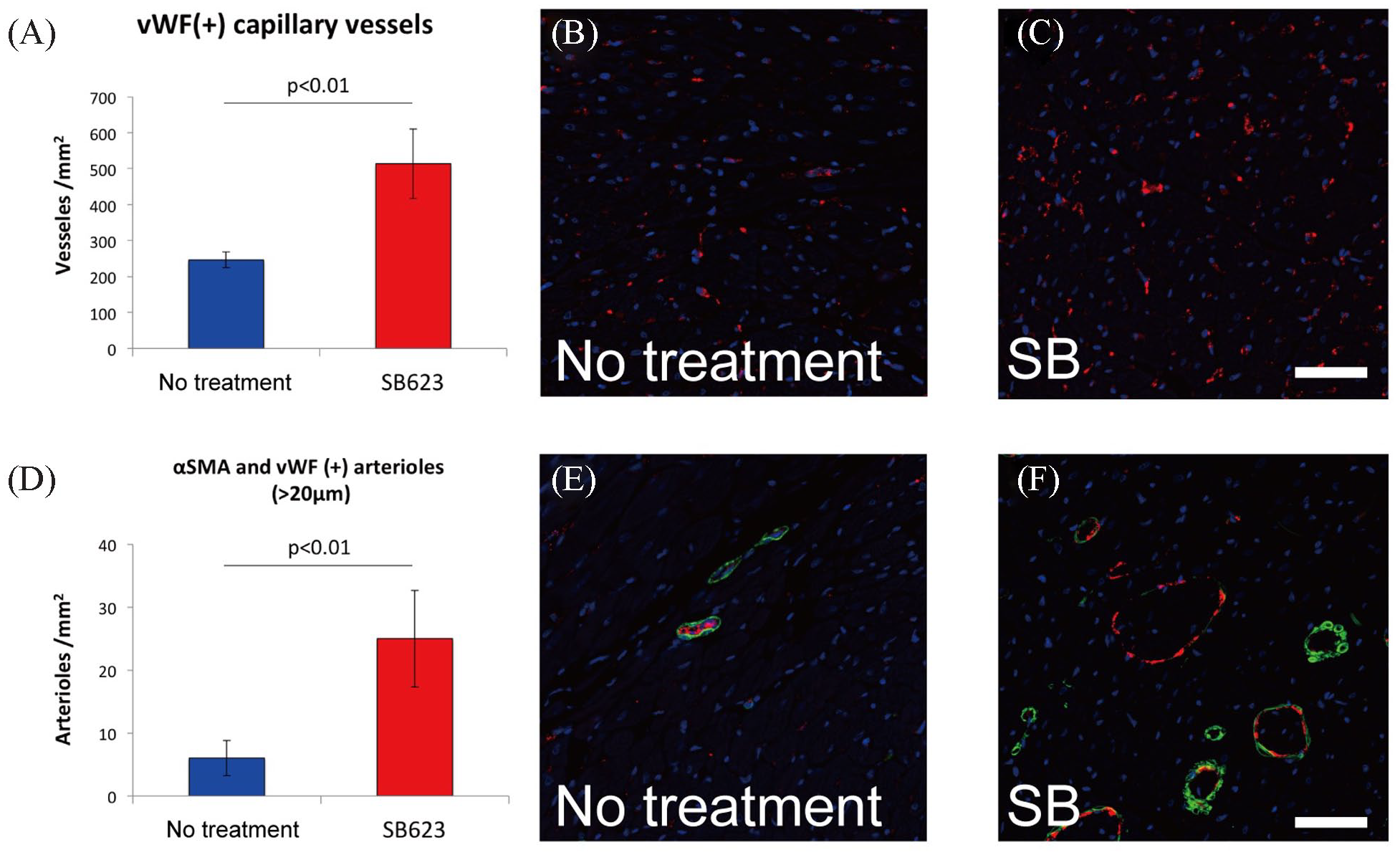
Histological assessment of angiogenesis and arteriogenesis. (A) Density of capillary vessels. (B, C) Immunostaining of capillary vessels with anti-vWF (red) and nuclei (blue). (D) Density of arterioles with diameter greater than 20 μm. (E, F) Immunostaining for arterioles with anti-vWF (red), anti-αSMA (green), and nuclei (blue). vWF: von Willebrand factor; αSMA: alpha smooth muscle actin. Scale bars indicate 50 μm.
Attenuation of Fibrosis and Hypertrophy in Myocardium Treated with Cell Patch
Cardiac structure and interstitial fibrosis accumulation were analyzed 6 weeks after cell patch transplantation (Fig. 3A, B). Compared with the no-treatment group, myocardium fibrosis was significantly decreased in the SB group (25.4 ± 5.0% vs 16.4 ± 6.2%, P = 0.04) (Fig. 3C). Furthermore, the myocardium cells in the remote areas were significantly smaller in the SB group (19.7 ± 2.5 μm vs 15.4 ± 2.1 μm, P = 0.02) (Fig. 3D), suggesting inhibition of cellular hypertrophy.

Histological analysis of fibrosis and cellular hypertrophy. (A) Sirius red staining of the untreated whole heart. (B) Sirius red staining of the whole heart treated with the SB623 cell patch. (C) Interstitial fibrosis quantification. (D) Cellular hypertrophy quantification. Scale bars indicate 1,000 μm.
Upregulation of Angiogenic and Arteriogenic Cytokines
Gene expression of angiogenic and arteriogenic cytokines in the myocardium 5 days after SB623 cell patch transplantation was evaluated by RT-PCR. As for cytokines associated with both angiogenesis and arteriogenesis, the gene expression level of FGF-2 was significantly higher (P < 0.01) and that of PIGF showed a trend to be higher (P = 0.07) in the SB group, whereas that of VEGF was not significantly different (P = 0.92) compared with the no-treatment group (Fig. 4A–C). The gene expression of MCP-1, which specifically induces arteriogenesis, was not significantly higher (P = 0.18) in the SB group (Fig. 4D), nor was that of HGF (P = 0.17), a well-known angiogenic cytokine (Fig. 4E).

Quantitative polymerase chain reaction analysis of cardiomyocytes treated with the SB623 cell patch. Gene expressions of FGF-2 (A), PIGF (B), and VEGF (C), cytokines known to induce angiogenesis and arteriogenesis; MCP-1 (D), an arteriogenic cytokine; HGF (E), an angiogenic cytokine; and ephrin-B2 (F) and EphB4 (G), markers of arteriogenesis. FGF-2: fibroblast growth factor 2; PIGF: placental growth factor; VEGF: vascular endothelial growth factor; MCP-1: monocyte chemotactic protein 1; HGF: hepatocyte growth factor.
Upregulation of Arteriogenic Markers
It has been reported that the most important processes during arteriogenesis include activation of ephrin-B2 and EphB4 27 . To confirm the arteriogenic responses in the present model, gene expression of ephrin-B2 and EphB4 in rat heart muscle samples 5 days after SB623 cell patch transplantation was examined by RT-PCR. In the SB group, ephrin-B2 (P = 0.03) and EphB4 (P = 0.01) expression was significantly upregulated compared with that in the no-treatment group (Fig. 4F, G), indicating the induction of arteriogenesis in the ischemic myocardium.
Discussion
In this study, we examined the effects of Notch signaling-modified hMSC patch transplantation in a rat ICM model. SB623 cell patch transplantation improved LVEF and attenuated LV remodeling. Histologically, the myocardium treated with the SB623 cell patch showed induction of angiogenesis and arteriogenesis. Furthermore, the myocardium treated with the SB623 cell patch showed higher gene expression levels of FGF-2, ephrin-B2, and EphB4.
In this study, we showed the simultaneous induction of angiogenesis and arteriogenesis by transplantation of the SB623 cell patch resulted in improved LVEF and attenuated LV remodeling in a rat ICM model. While angiogenesis provides beneficial effects for an ischemic heart via the improvement of microcirculation, arteriogenesis enhances these effects by inducing a several-fold increase in blood flow. Between these two modes of new vessel formation, the induction mechanism is clearly different, as angiogenesis is induced by hypoxia and arteriogenesis by shear stress or inflammation 14 . Although simultaneous induction of angiogenesis and arteriogenesis is an attractive therapeutic strategy for the treatment of ICM, few reports have noted the achievement of both, probably because of their different induction mechanisms. We believe that the paracrine effects of SB623 cells play the most important role in the induction of arteriogenesis. Previously, we reported that SB623 cells secreted several kinds of angiogenic factors16,28. In this study, the gene expression level of FGF was significantly higher in the myocardium treated with SB623 cells. FGF induces Notch ligand, delta-like 1 (DLL1), and subsequent Notch signal upregulation, which seems to be a primary mechanism of arteriogenesis induction caused by SB623 cell transplantation 29 .
In this study, the myocardium treated with the SB623 cell patch showed upregulation of ephrin-B2 and EphB4. During arteriogenesis, activation of Notch signaling followed by activation of ephrin-B2 and EphB4 are the most important processes27,30. Ephrin-B2 expression mediates a sequence of vascular responses, including arterial remodeling, branching, and outward growth 29 . Furthermore, EphB4 expression was reported to lead to venous neovascularization, which may have an important role as a drainage system for tissue perfusion 31 . Although arteriogenesis is a process composed of highly complicated morphogenetic events mediated by various factors, the present in vivo findings indicated that the transplanted SB623 cells activated two of those essential factors.
The paracrine effects caused by SB623 cell patch transplantation may play the most important role in the simultaneous induction of angiogenesis and arteriogenesis. The present examination of the gene expression of angiogenic and arteriogenic cytokines in the myocardium after SB623 cell patch transplantation showed upregulation of FGF-2 and a trend for PIGF upregulation. Although cytokine therapy using arteriogenic factors, such as granulocyte colony-stimulating factor (G-CSF) and MCP-1, is an attractive option for the treatment of ischemic disorders, other investigators found that the use of G-CSF did not provide explicit cardiac benefits in patients with coronary artery disease, and MCP-1 therapy was associated with the risk of plaque progression in a mouse hindlimb ischemia model32,33. Similarly, cytokine therapy utilizing VEGF, FGF-2, or PIGF was reported to be associated with the induction of both angiogenesis and arteriogenesis but has not clearly shown benefit in patients with ICM 34 . From the viewpoint that the mechanism of arteriogenesis is more complex than that of angiogenesis, Kinnaird et al. 35 insisted that delivery of a broad spectrum of cytokines increases the possibility of induction of arteriogenesis as compared with delivery of only one or two cytokines. In their study, to deliver a broad spectrum of cytokines, MSCs were transplanted into a mouse hindlimb ischemia model, and the results showed promotion of arteriogenesis through paracrine effects. Similar to their concept, we examined the induction of arteriogenesis by SB623 cell patch transplantation in this study. Although upregulation of only FGF-2 and PIGF was found 5 days after SB623 cell patch transplantation in this study, cellular therapy utilizing SB623 cells is suggested to have greater potential to induce arteriogenesis in the treatment of ICM.
In this study, the effects of SB623 cells on ICM were investigated. In our previous in vitro study, SB623 cells were found to secrete higher concentrations of angiogenic and arteriogenic cytokines, including MCP-1 and VEGF, as compared with hMSCs from the same donors 28 . Furthermore, we previously reported that paracrine factors secreted by SB623 cells showed upregulation of the Notch signaling pathway, a key pathway of arteriogenesis, as well as several other pathways potentially leading to arteriogenesis in endothelial cells 16 . Together with the present results, it is suggested that modification of intracellular signaling, which has key roles in arteriogenesis, is an attractive strategy to enhance the effect of cellular therapy for ischemic disorders.
This study had some limitations. We did not examine the upregulation of angiogenetic or arteriogenic cytokines at several time points, which may explain why the upregulation of key angiogenic factors, such as VEGF, were not observed in this study. Although the main goal of this study was to examine the therapeutic efficacy of SB623 cells in ICM via angiogenesis or arteriogenesis, further studies are required to investigate the exact therapeutic mechanism of SB623 cell patch transplantation.
Conclusion
In conclusion, SB623 cell patch transplantation improved LV function by inducing angiogenesis and arteriogenesis in a rat ICM model. These findings suggest that our strategy has the potential to simultaneously induce angiogenesis and arteriogenesis in the ischemic myocardium, leading to an enhanced therapeutic effect in patients with ICM.
Footnotes
Acknowledgements
The authors thank Akima Harada and Kaori Ikuma for their technical assistance.
Author Contributions
Shusaku Maeda, Takuji Kawamura, Dai Chida, Kazuo Shimamura, Koichi Toda, Yoshiki Sawa, and Shigeru Miyagawa conceived and designed the experiments and contributed to the discussion. Shusaku Maeda and Akima Harada performed the experiments and analyzed data. Shusaku Maeda and Takuji Kawamura drafted the article. Shusaku Maeda and Dai Chida contributed to the materials. Yoshiki Sawa and Shigeru Miyagawa supervised the project. All authors have reviewed the final version of the article.
Ethical Approval
This study was approved by the Animal Experimentation Committee of Osaka University (approval number: 04522).
Statement of Human and Animal Rights
All animal protocols were performed in accordance with the Guidelines for Animal Experiments of Osaka University (approval number: 04522).
Statement of Informed Consent
There were no human subjects in this study and informed consent was not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dai Chida is an employee of SanBio Inc. There are no other relationships with any industry to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research project was provided by SanBio Inc.
