Abstract
Optic neuropathies refer to a group of ocular disorders with abnormalities or dysfunction of the optic nerve, sharing a common pathophysiology of retinal ganglion cell (RGC) death and axonal loss. RGCs, as the retinal neurons in the central nervous system, show limited capacity in regeneration or recovery upon diseases or after injuries. Critically, there is still no effective clinical treatment to cure most types of optic neuropathies. Recently, stem cell therapy was proposed as a potential treatment strategy for optic neuropathies. Adult stem cells, including mesenchymal stem cells and hematopoietic stem cells, have been applied in clinical trials based on their neuroprotective properties. In this article, the applications of adult stem cells on different types of optic neuropathies and the related mechanisms will be reviewed. Research updates on the strategies to enhance the neuroprotective effects of human adult stem cells will be summarized. This review article aims to enlighten the research scientists on the diversified functions of adult stem cells and consideration of adult stem cells as a potential treatment for optic neuropathies in future clinical practices.
Keywords
Introduction
Optic neuropathies refer to a group of disorders with abnormalities and dysfunction of the optic nerve. Apart from the primary optic nerve disorders, some optic neuropathy cases are associated with systemic disorders, such as demyelinating inflammatory optic neuritis (ON), which are associated with multiple sclerosis or neuromyelitis optic neuropathy. Other types of inflammatory ON can be associated with systemic autoimmune disorders, such as systemic lupus erythematosus or sarcoidosis1,2. Ischemic optic neuropathy is suggested to be associated with other conditions, including hypertension and diabetes. Tumors involving the optic nerves can co-exist with the tumors in the central nervous system. Different types of optic neuropathies can lead to various severities of visual impairment and even blindness. Acute and rapid visual deterioration is typical for demyelinating, inflammatory, ischemic, and traumatic causes, while a gradual course indicates the compressive, toxic/nutritional, or hereditary causes. Central scotoma and centrocecal scotoma are usually found in hereditary optic neuropathy. Altitudinal defects present in ischemic optic neuropathy and hemianopia can be associated with compressive optic neuropathy. Glaucoma, the main type of irreversible optic neuropathy, begins without apparent symptoms in the early stages but with initial gradual deterioration of peripheral vision 3 . The classification and characteristics of optic neuropathies were summarized in Table 1.
Characteristics of Representative Optic Neuropathies and Current Adult Stem Cell Applications.
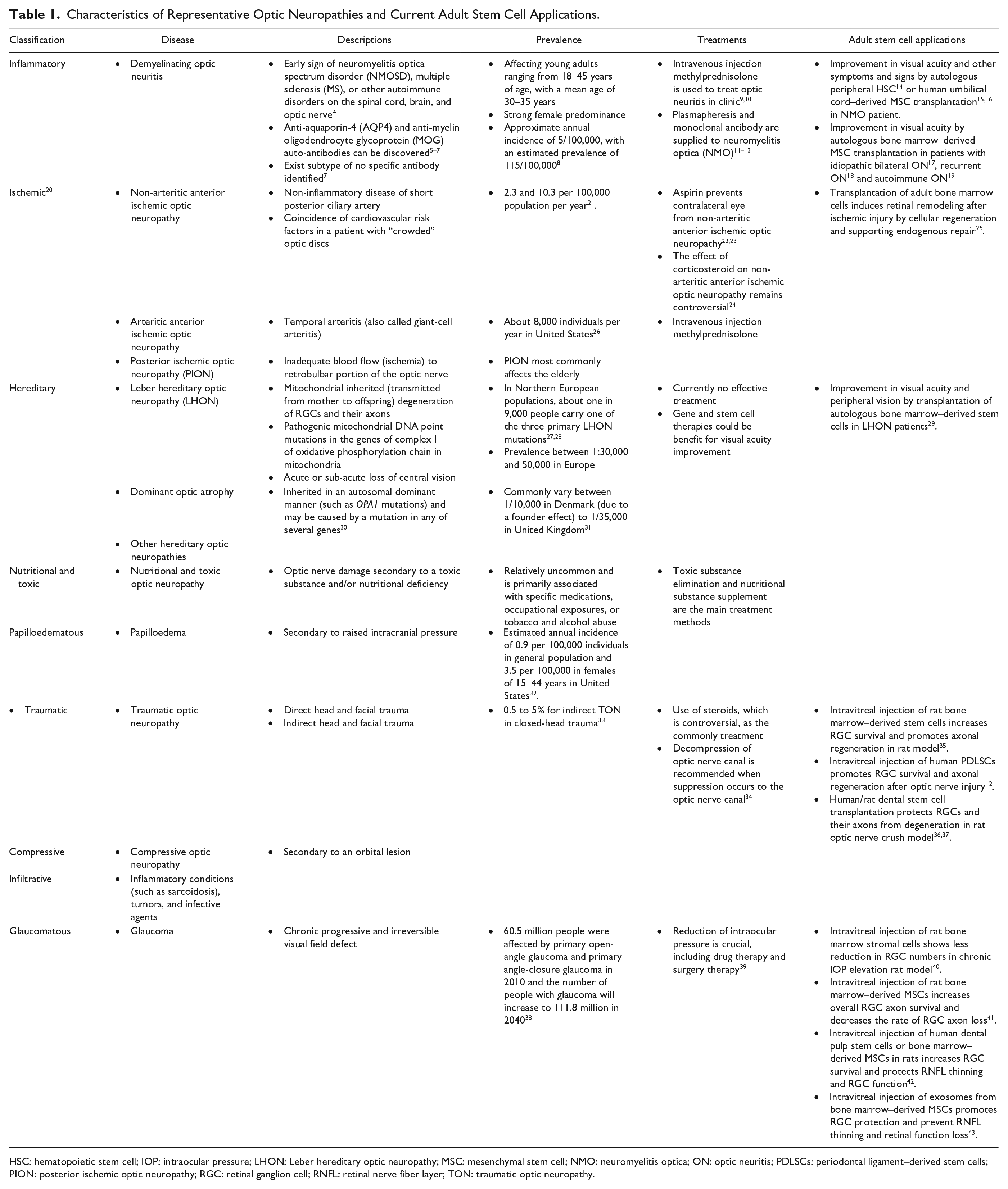
HSC: hematopoietic stem cell; IOP: intraocular pressure; LHON: Leber hereditary optic neuropathy; MSC: mesenchymal stem cell; NMO: neuromyelitis optica; ON: optic neuritis; PDLSCs: periodontal ligament–derived stem cells; PION: posterior ischemic optic neuropathy; RGC: retinal ganglion cell; RNFL: retinal nerve fiber layer; TON: traumatic optic neuropathy.
Optic neuropathies can also be classified as acquired or inherited 44 . Acquired optic neuropathies affect people with wide spectrum of ages, but commonly occur in adults. The morphological changes involve not only the optic nerve head but also the entire optic nerve and blood vessels, and even co-existing with the intracranial problems. Their etiologies are diverse, including vascular, inflammatory, compressive, infiltrative, traumatic, toxic, and nutritional optic neuropathies 3 . In contrast, hereditary optic neuropathies belong to those with underlying heritable etiology. Leber’s hereditary optic neuropathy (LHON) and autosomal dominant optic atrophy (ADOA) are the most common hereditary optic neuropathies mostly affecting the children and teenagers 45 . Optic nerve damage in LHON and ADOA are usually observed in earlier life, and the optic nerve is permanently affected as the results of gene mutations 46 . Glaucoma is also considered as a complex genetic disease with the interplay between multiple associated genes and the environmental factors 47 . Glaucoma is characterized by the optic nerve head cupping and visual field defects 47 , and intraocular pressure (IOP) elevation is a reversible risk factor 39 .
In spite of different etiologies for different types of optic neuropathies, retinal ganglion cell (RGC) death and the axonal loss are the commonly shared pathophysiological changes for all types of optic neuropathies. RGCs collectively transmit the image-forming and non-image forming visual information from the retina to the lateral geniculate nucleus and superior colliculus in the brain; therefore, they are the important treatment target in plenty of the optic neuropathy research. RGC death in optic neuropathies is complex and complicated 48 , and RGC protection and regeneration are the major goals in the design of treatment regimes.
Clinically, a variety of medications and surgical procedures have been applied to different optic neuropathies in multiple clinical trials or routine practices, targeting the pathological conditions in different types of optic neuropathies. However, the visual defect of a significant number of patients with optic neuropathies still cannot be fully recovered under current clinical interventions. Completely rescuing RGCs from degeneration, activating their regeneration endogenously, and replenishing them exogenously are the major challenges for the clinicians and researchers. Besides, RGCs are vulnerable to stresses and injuries, and they have limited regenerative capacity. Partial functional recovery is possible; yet, the vision loss is inevitable and irreversible.
Basically, mammalian retina has low intrinsic regenerative capacity. Lack of trophic support, scar formation at the injury site, and the myelin-associated inhibitors further contribute to RGC degeneration and failure in axonal regeneration upon diseases and after injuries 49 . Many research studies strived through variable ways to alleviate RGC death and axonal loss, including supplying exogenous neurotrophic factors, electrical stimulation, inhibiting the apoptosis mediators, counteracting glial scar formation and the myelin-associated inhibitors, as well as gene and cell therapies. Exogenously supplying brain-derived neurotrophic factor (BDNF) and ciliary neurotrophic factor (CNTF) can promote RGC survival in rodents after optic nerve injury50,36. Moreover, glial cell line–derived neurotrophic factor (GDNF), fibroblast growth factor (FGF), transforming growth factor-β (TGF-β), and insulin-like growth factors (IGFs) can also enhance RGC survival in retinal degeneration models, such as ischemic/reperfusion model 51 . However, multiple injections of neurotrophic factors are needed to demonstrate a prominent effect of RGC protection. Trans-corneal electrical stimulation for 1 month shows increase in RGC survival with preservation of RGC function after hypertensive injury in mouse retina 52 . Inhibiting the apoptotic pathway by caspase-3 inhibitors, such as z-DEVE-fmk, has been reported to promote RGC survival 53 . By targeting the myelin-associated growth-inhibitory molecules, the inhibitor of myelin-associated glycoprotein can promote neurite outgrowth from dorsal root ganglia neurons 54 . Gene therapy mediated through the adeno-associated viruses (AAVs) on different target genes, such as ectopic expression of BDNF 55 and CNTF as well as knockdown of RhoA 50 , can enhance RGC survival and axonal regeneration in rodents. The disease types, tissue tropism of AAVs, the gene targets, the vector carrying capacity, potential genome integration, immune responses, as well as the treatment safety and toxicity should reasonably be considered before the clinical application of gene therapy 56 . It is also warranted to develop novel and effective treatments for optic neuropathies. Recently, stem cell therapy was proposed as a potential treatment for optic neuropathies. Adult stem cells, including mesenchymal stem cells (MSCs) and hematopoietic stem cells (HSCs), have been applied in clinical trials based on their neuroprotective properties. In this article, the applications of adult stem cells on different types of optic neuropathies and the related mechanisms will be reviewed. Research updates on the strategies to enhance the neuroprotective effects of human adult stem cells will be summarized. In addition, the potential of cell replacement therapy by adult stem cells for optic neuropathies will also be discussed.
An Overview and the Neuroprotective Effects of Adult Stem Cells
Stem cells are defined as the immature cells with the capability of self-renewal and differentiation into mature cells. They are essential for tissue growth, development, and homeostasis. According to the developmental stages, stem cells can be sub-classified into embryonic stem cells (ESCs) and adult stem cells. ESCs, derived from the inner cell mass of a blastocyst prior to gastrulation, are regarded as the gold standard of pluripotent stem cells, which are capable to differentiate into all cells of the three germ layers: ectoderm, mesoderm, and endoderm. Adult stem cells, referring to the stem cells originated from fully developed tissues, have been identified in multiple organs and tissues, including the umbilical cord, bone marrow, teeth, adipose tissue, brain, eye, intestine, and salivary glands 57 . Adult stem cells function to maintain the specificity of adult tissues through homeostatic cell replacement and tissue regeneration 58 . Although they are presumed quiescent within the adult tissues, they can divide infrequently to produce a stem cell clone and a transiently amplifying cell. The transiently amplifying cells can undergo limited cell divisions before terminally differentiating into mature functional tissue cells. Adult stem cell differentiation is lineage restricted, and different types of adult stem cells have their specific functions in different tissues and organs. This review article focused on the research of HSCs, MSCs, and retinal stem cells in the treatment of optic neuropathies.
Hematopoietic stem cells (HSCs) can differentiate into all cell lineages in mature blood (erythroid, myeloid, and lymphoid) and sustain the blood production for the entire human life 59 . HSCs for transplantation in many blood-related diseases are mainly obtained from adult bone marrow, umbilical cord blood, and mobilized peripheral blood. HSCs can be sorted with the positive selection by CD34, CD45, and CD133 as well as the negative selection by CD31, CD105, and CD146 60 . Activation of HSCs is driven by the PI3K/AKT/mTORC1 signaling pathway, whereas their re-entry into quiescence depends on the MEK/ERK signaling pathway 61 .
Mesenchymal stem cells (MSCs) are the multipotent stromal cells with the capability differentiating into mesodermal lineages, including adipocytes, chondrocytes, and osteocytes 62 . Although MSCs were originally identified in bone marrow, they can also be found in different tissues, including adipose tissue, umbilical cord, and teeth 63 . Human MSCs can be sorted with the positive selection by CD29, CD44, CD73, CD90, CD105, CD146, and STRO-1 as well as the negative selection by CD31, CD34, CD45, CD49f, and CD133 64 . Apart from the expression of specific cell surface markers, MSCs are also defined to be growing in adherence to the plastic surface while maintained in standard culture conditions and are capable to be in vitro induced into mesenchymal lineages with appropriate medium as recommended by the International Society of Cellular Therapy 65 .
Retinal stem or progenitor cells (RPCs) are the neural stem cells generating all seven types of mature retinal cells: cone/rod photoreceptors, bipolar cells, horizontal cells, amacrine cells, RGCs, and Müller glia. RPCs are originated from neuroectoderm during development 66 and have been reported to be resided in the retina and the pigmented ciliary margin in zebrafish, chick, and rodent 67 . Moreover, Müller glia and retinal pigment epithelium have also been suggested demonstrating the RPC properties through de-differentiation upon injury 68 . The expressions of PAX6, VSX2, RAX, SOX2, Nestin, and c-Kit can be found in RPCs, but negative for SSEA4 69 .
Endogenous regeneration by stem cells upon diseases or after injuries in human retina, referring to the progenitor cells localized in the ciliary margin zone of the retina and the Müller glia, is very rare and largely unknown. Besides, the collection of RPCs from a living patient could possibly further damage the retina, which is not ethical. Therefore, treatments using adult stem cells for optic neuropathies mainly rely on the application of autologous/heterologous stem cells from other tissues, including HSCs and MSCs.
Adult stem cells, in addition to the capability differentiating into lineage-specific mature cells, also possess the neuroprotective properties, especially for HSCs and MSCs. The neuroprotective effect is mainly mediated by the paracrine bystander effect, including modulating the plasticity of the host damaged tissues, secreting the survival-promoting and neurotrophic factors, restoring the release of synaptic transmitters, integrating into the existing neural and synaptic networks, and rebuilding the afferent and efferent connections 70 . Moreover, MSCs also demonstrate the immunomodulatory properties 71 , allowing autologous/heterologous transplantation without additional immunosuppression treatment. MSCs have been found to facilitate cell repair via immunomodulation72,73. In addition, organelle or material transfer, such as mitochondrial transfer, from MSCs into the targeted cells through tunneling nanotubes can also rescue cells from damage and apoptosis74,75. Importantly, the neuroprotective properties are naturally acquired by adult stem cells without the need of reprogramming or genetic modification, and the usage of adult stem cells has no moral objection or ethical controversies. The risk of teratoma formation is very limited after adult stem cell transplantation 76 . Despite the accessibility and naturality of adult stem cells, the retina is comparatively resistant to transplant integration with limited long-term xenograft survival. Application of erythropoietin or chondroitinase ABC can facilitate the transplanted cell migration into the retina 77 . Adult stem cell treatment should be promising for optic neuropathies.
Application of Adult Stem Cells for Different Types of Optic Neuropathies
Application of Adult Stem Cells for Demyelinating Optic Neuritis
Demyelinating ON is one of the most common causes of unilateral or bilateral painful visual loss in young adults 4 . The incidence of ON was estimated as five per hundred thousand individuals annually, with a prevalence of 115 per hundred thousand individuals in the United States 8 . Demyelinating ON can be an early sign of neuromyelitis optica spectrum disorder (NMOSD), multiple sclerosis, or other autoimmune disorders on the spinal cord, brain, and optic nerve 4 . The anti-aquaporin-4 (AQP4) and anti-myelin oligodendrocyte glycoprotein (MOG) auto-antibodies are discovered in some demyelinating ON patients, but targeting different tissues under different pathogenesis5,6. The pathophysiological mechanism of AQP4 antibody-related ON could be related to the binding of AQP4 antibody (AQP4-IgG or AQP4-Ab) to the AQP4 water channel on the surface of astrocytes, resulting in astrocyte cytotoxicity along with secondary oligodendrocyte damage and demyelination 5 . On the other hand, the pathophysiological mechanism of the MOG antibody–related ON is caused by the antibodies directed against MOG on the oligodendrocytes 6 . There is also a group of ON patients without identification of specific auto-antibody. However, no targeted treatment for demyelinating ON is clinically available. High dose of intravenous methylprednisolone (IVMP), aiming to reduce the inflammation at the optic nerve, can promote visual function improvement in the patients with ON 78 . In addition, the plasma exchange treatments, oral steroids, and immunosuppressive and immunomodulatory drugs can also be applied to prevent further relapse 79 . Nevertheless, high dose IVMP and the enhanced immunosuppressive treatment will extensively suppress the normal immune function, leading to severe side effects and increasing the risk of serious infection. Application of adult stem cells, especially MSCs, in ON should help with the immunomodulation, anti-inflammation, and enhancing RGC survival18,80,81.
In the experimental MOG-induced autoimmune encephalomyelitis model resembling multiple sclerosis, tail vein injection of human bone marrow–derived MSCs has been shown to alleviate RNFL thinning and RGC death as well as protect the pattern electroretinography response with reducing the HIF-1 signaling activation and upregulating the Abca1 expression 80 . For the clinical study of MSC therapy on ON and NMOSD (Table 2), the clinical trial (NCT02249676) on 12 neuromyelitis optica (NMO) patients, one recurrent ON patient, and two patients with recurrent longitudinally extensive transverse myelitis received autologous MSC transplantation, showed that increase in RNFL thickness and optic nerve diameters with visual function improvement could be observed at 1 year after transplantation 18 . Another clinical trial (NCT01920867) on a 54-year-old female subject with autoimmune ON reported improvements in visual acuity, visual field, and RNFL thickness after autologous bone marrow–derived MSC transplantation 19 . Moreover, five Chinese NMO patients treated with human umbilical cord–derived MSCs showed improvement in symptoms, and the signs were improved in four study subjects with reduction in relapse frequencies, magnetic resonance imaging (MRI) characteristics, and the severity of lesions 15 . Although few adverse events reported, peripheral blood B lymphocytes were inhibited, and T lymphocytes increased after treatment 15 . In addition, a clinical trial (NCT01920867) on a 27-year-old female subject with idiopathic bilaternal ON reported that the visual acuity of the eyes receiving retrobulbar, subtenon, and intravitreal stem cell injections improved from 20/800 to 20/100 while those receiving intra-optic nerve injection of stem cells and vitrectomy improved from 20/4,000 to 20/40 at 6 months after autologous bone marrow–derived MSC injection 17 . The visual acuity improvement remained stable even after the 12-month follow-up period. However, the transplanted bone marrow–derived MSCs in patients with NMO exhibit the decreased proliferation capacity and increased cell death rate 82 . Furthermore, there are 2 clinical trials applying the autologous bone marrow–derived MSCs, including the SCOTS2 (NCT03011541) in the United States for the NMO treatment, and the Treatment of Optic Neuropathies Using Autologous Bone Marrow-Derived Stem Cells (NCT02638714) in Jordan for the safety and efficacy on functional restoration in ON.
Registered Clinical Trials of Adult Stem Cell Therapy for Optic Neuropathies.

Information obtained from http://clinicaltrials.gov/. NMO: neuromyelitis optica.
For the clinical study on HSCs, a study on 13 NMOSD patients with the infusion of mobilized autologous peripheral blood stem cells, one patient with coexistent systemic lupus erythematous died due to the complications of active lupus; yet, the mental (from 37.6 to 65), physical (from 28 to 56), and total quality of life (from 34.2 to 62) were improved in 11 patients after a 5-year follow-up period 16 . Critically, among the 11 patients with sero-positive on baseline AQP4-IgG, nine patients became sero-negative after stem cell transplantation. Moreover, in a 23-year-old Chinese female NMO subject who received autologous peripheral HSC transplantation, the visual acuity of the right eye increased from 0.02 to 0.04 at 6 months after transplantation 14 . In spite of the remaining atrophic optic nerve, other symptoms were improved, including the muscle power. In contrast, a study on 21 opticospinal MS patients with intravenous transplantation of autologous peripheral blood stem cells reported no significant improvement after transplantation even though the expanded disability status scale score was decreased 83 . Besides, failure in preventing the relapse of NMO was also reported in one patient with autologous HSC transplantation, which could be due to the inadequate reset of immune tolerance or sustained autoimmunity to aquaporin-4 auto-antigen 84 . There are also two clinical trials on autologous HSCs for NMO withdrawn or terminated without any data released: The Autologous Transplant To End NMO Spectrum Disorder (ATTEND; NCT03829566) in the United States and the Autologous Hematopoietic Stem Cell Transplant in Neuromyelitis Optica (SCT-NMO; NCT01339455) in Canada.
Application of Adult Stem Cells for Non-Arteritic Anterior Ischemic Optic Neuropathy
Non-arteritic anterior ischemic optic neuropathy (NAION) belongs to one of the most common acute optic neuropathies with an incidence of 2.3 to 10.3 per hundred thousand individuals per year in the United States 21 . The pathophysiology of NAION is presumed as the acute hypoperfusion of short posterior ciliary arteries, resulting in insufficient blood flow to the optic nerve head and anterior optic nerve segment85,86. The ischemic insult becomes worsen by the optic disc edema compressing the axon and capillaries and leads to a compartment syndrome 86 . Along with the ischemia, the cytotoxic factor and cytokine release would lead to further damages on the optic nerve 87 , and eventually, optic nerve atrophy would develop 86 . Different therapeutic strategies for NAION have been investigated, including intravitreal, peri-ocular, systemic, and surgical therapies 88 , to reduce the related vascular risk factors, improve the perfusion of the optic nerve by vasodilators, relieve the optic nerve head pressure, inhibit cytotoxic factors by the molecular targeted therapy, and promote optic nerve axonal regeneration. Application of adult stem cells in NAION should be able to improve the vascular circulation and perfusion and nourish the RGCs with the neurotrophic factors.
In a mouse model of NAION, intravenous (1 × 106 cells in 0.2 ml) or intravitreal injection (2 × 105 cells in 3 μl) of adult mouse bone marrow–derived stem cells at 1 month showed stem cell migration to various retinal layers and differentiation into endothelium, immune cells, astrocytes, and neural cells, indicating that the transplanted adult bone marrow cells can contribute to the retinal remodeling after ischemia by promoting cell regeneration and sustaining endogenous repair 89 . Early neural and sustained astrocyte differentiation from adult bone marrow–derived stem cells could be regulated through the Flt-1-mediated vascular endothelial growth factor (VEGF) signaling 89 . In addition, both BDNF and CNTF can enhance the neural differentiation of adult bone marrow–derived stem cells in the retina of AION mice 90 . In a rat model of AION, intravitreal injection of human Wharton’s jelly MSC-derived conditional medium can inhibit RGC apoptosis and microglia activation and preserve visual function 81 . Notably, the transplantation of human Wharton’s jelly MSCs in normal rats would cause severe inflammation and impair the retinal structure and function as compared with the phosphate buffered saline (PBS)-treated rats 81 . The effect of xeno-transplantation of human MSCs cannot be ignored.
Currently, there are two clinical studies using adult stem cells for NAION (Table 2). The Stem Cell Ophthalmology Treatment Study (SCOTS; NCT01920867), conducted in the United States, is a non-randomized study based on the combinations (retrobulbar, subtenon, intravitreal, and intravenous) of autologous bone marrow–derived stem cell injection. In SCOTS, 10 NAION patients received autologous bone marrow–derived stem cell treatment 91 . About 80% patients showed improvement in Snellen binocular vision within 6 months, and 20% remained stable. This study showed an average of 22.74% vision improvement in Snellen and maximum 83.3% improvement in logMAR visual acuity, with an average logMAR visual acuity gain of 0.364 in the treated eyes.
Another Phase II clinical trial for acute NAION in Spain (NCT03173638) aimed to assess the safety and inflammatory reaction of intravitreal allogenic bone marrow–derived MSC injection in the acute phase of NAION based on the neuroprotective properties of MSCs to reduce the progressive axonal degeneration in NAION. The outcome of the study has not been reported yet.
Application of Adult Stem Cells for Traumatic Optic Neuropathy
Traumatic optic neuropathy (TON) refers to the optic nerve injured directly or indirectly or by a blast. In 1998, the World Health Organization estimated trauma causing 1.6 million binocular blindness, 2.3 million people with both eyes low vision, and 19 million monocular blindness 92 . The injuries from traffic accidents, sports, and recreational activities account for 78.2% cases in India 93 . Direct optic nerve injury includes penetrating trauma and impinging injury by bone fragments or orbital hemorrhage. Indirect injury commonly occurs during head or globe injury when the optic nerve could be sheared and avulsed by the rotational forces 94 . The energy can be transmitted through the skull to the optic nerve and retina, causing rapid changes in IOP and inducing RGC damage 94 . Following the direct mechanical disruption on the axons of the optic nerve, the enzymatic processes could be activated to degrade the cytoskeleton, leading to the delayed impairment of the axonal structure and function 95 . In the TON treatment trial (TONTT) study, daily intravenous administration of erythropoietin (stimulating erythrocyte production) or methylprednisolone for three consecutive days significantly improves the best corrected visual acuity and color vision in the TON patients 94 . Adult stem cells, with the secretion of neurotrophic factors and exosomes, can nourish the microenvironment in TON so as to promote RGC survival and axonal regeneration.
To mimic the clinical situation of TON, the experimental model of optic nerve injury by crushing the optic nerve in rodents is adopted. Intravitreal injection of rat bone marrow–derived stem cells (5 × 106 cells in 5 μl) can increase RGC survival by 1.6 folds and promote axonal regeneration at 2 weeks after injection as compared with those with saline or dead cell injection 35 . Similarly, our group demonstrated that intravitreal injection of human periodontal ligament–derived stem cells (PDLSCs) can enhance RGC survival and axonal regeneration after optic nerve injury in rats, possibly through direct cell–cell interaction as well as neurotrophic factor secretion by PDLSCs 36 . Higher expression of FGF-2 and IL-1β in the transplanted retina can explain the neuroprotective effect of MSCs 96 . Human or rat dental stem cells can secrete nerve growth factor (NGF) and BDNF to promote RGC survival and axonal regeneration in the rat optic nerve crush model through the TrK receptor36,37. Our group also found that retinal injury can enhance the secretion of BDNF from human PDLSCs 36 . In addition, intravitreal injection of rat MSCs and human Wharton’s jelly MSCs as well as its extracellular vesicles can enhance axonal regeneration and synaptic reconnection in superior colliculus, but fail to preserve the visual behavior in rodents with optic nerve injury 97 , indicating that MSCs can promote long-distance axonal regeneration. Yet, the neuroprotective mechanisms of MSCs still require further investigations. Besides, there is still no clinical trial on human stem cells for TON.
Application of Adult Stem Cells for Leber’s Hereditary Optic Neuropathy
LHON belongs to one of the mitochondrial disorders with an incidence of 1 per 100,000 individuals 45 . LHON is maternally inherited due to the point mutations in mitochondrial DNA, leading to the reduction in mitochondrial ATP production and the elevation in oxygen-free radicals. The oxidative stress in turn leads to the dysfunction and apoptosis of RGCs 98 . For the treatment, creatine and dietary antioxidant supplements, including coenzyme Q10 and L-carnitine, have been suggested to enhance mitochondrial bioenergetics and to ameliorate the neurodegenerative process 99 . Application of adult stem cells in LHON might be able to transfer healthy mitochondria to the diseased RGCs through cell–cell contract-mediated cellular material transfer so as to enhance RGC survival.
LHON mouse model has been established 100 ; yet, no stem cell transplantation study has been tested in this mouse model. Alternatively, patient-specific LHON model can also be generated from induced pluripotent stem cells (iPSCs) 101 . Repairing mitochondrial dysfunction via mitophagy activation could be a possible treatment strategy for LHON 102 . For the LHON clinical trial on human adult stem cells (Table 2), SCOTS (NCT01920867) recruited five LHON patients, who received the transplantation of autologous bone marrow–derived stem cells 29 . The visual acuity and peripheral vision persistently increased, improving from finger counting to 20/100 and hand motion to 20/200. However, the thicknesses of macula and optic nerve head are not correlated with the vision improvement. Although adverse or serious adverse event was not found in this study, the sample size was relatively too small to draw the conclusion.
Application of Adult Stem Cells for Glaucoma
Glaucoma, characterized by the optic disc cupping and progressive RGC degeneration 39 , is a leading cause of irreversible visual impairment and blindness in the world. It was estimated that 60.5 million individuals suffered from glaucoma in 2010, and the glaucoma patients will increase to 111.8 million in 204038. The risk factors include aging, gender, ethnicity, hypertension, smoking, myopia, and diabetes 103 . Lamina cribrosa is considered as the primary site of injury. The imbalanced IOP level imposes mechanical stress to RGCs and leads to RGC death 104 . Although the pathogenesis of glaucoma is yet to be elucidated, various theories have been proposed, including the vascular, biochemical, and biomechanical theories 104 . For the biomechanics, the elevated IOP, together with the concept of the decreased intracranial pressure and the high trans-lamina cribrosa pressure difference, induces lamina cribrosa pore distortion, nerve fibers compression, and RGC death 105 . For the vascular theory, the decreased ocular perfusion caused by the elevated IOP can lead to intraneural ischemia, which would lead to RGC damage owing to inadequate supply of oxygen and nutrients as well as the activation of astrocytes and Müller glia. The elevated endothelin-1 and deranged nitric oxide-cyclic GMP signaling could also be involved in the vascular dysregulation 106 . The biochemical theory suggests that the involvement of reactive oxygen species (ROS), excitatory amino acids, nitric oxide, caspases, matrix metalloproteinases (MMPs), and tumor necrosis factor-α could lead to RGC apoptosis and neuroinflammation in glaucomatous optic neuropathy107,108. Current clinical treatments mainly focus on lowering the IOP, and they can effectively delay the disease progression in majority of the glaucoma patients. The development of IOP-independent strategies to enhance RGC survival is warranted 108 . Application of adult stem cells in glaucoma should be able to improve the vascular circulation for better nutrient supply, exert the anti-inflammatory and anti-oxidative effects, and enhance RGC survival by the paracrine effect of adult stem cells.
In the chronic IOP elevation rat model by ligating the episcleral veins, RGC reduction was ameliorated in rats with intravitreal injection of rat bone marrow stromal cells (4 × 104cells in 5 μl) 40 . The transplanted bone marrow stromal cells were present along the inner limiting membrane or integrated into the nerve fiber and GC layer, but rarely expressed the neural lineage markers. Increased expression of bFGF and CNTF in bone marrow stromal cell treatment could explain the RGC protective effect. In the laser-induced IOP elevation rat model, intravitreal injection of rat bone marrow–derived MSCs (5 × 106 cells in 5 μl) increases RGC axonal survival and decreases axonal loss at 4 weeks after transplantation 41 . Comparatively, intravenous transplantation of bone marrow–derived MSCs showed no effect on optic nerve damage rescue. Moreover, intravitreal injection of human bone marrow–derived MSCs or dental pulp stem cells (1.5 × 105 cells in 5 µl) in rats before bi-weekly intra-cameral injection of TGF-β1 can increase RGC survival and protect retinal nerve fiber layer thinning and RGC function at 1 month after transplantation, which was not observed in adipose-derived stem cells 42 . Mouse bone marrow–derived MSC-promoted RGC survival in acute IOP elevation mice model could be mediated through the miR-21/PDCD4 axis with the suppression of caspase-8-mediated apoptosis, microglia activation, and inflammatory mediator production 109 . Apart from stem cell transplantation, intravitreal injection of small extracellular vesicles collected from bone marrow–derived MSCs can also promote RGC protection and prevent retinal nerve fiber layer degenerative thinning and the retinal function loss in the microbeads and laser photocoagulation-induced IOP elevation rat model 43 . The RGC protection mechanism of MSC-derived small extracellular vesicles could be mediated through miRNAs that, with the knockdown of Argonaute-2, the protective effects of MSC-derived exosomes on RGC soma and axons after optic nerve injury would be diminished 110 .
Up to now, there are four clinical trials studying adult stem cells in glaucoma (Table 2): SCOTS (NCT01920867) and SCOTS2 (NCT03011541) in the United States on autologous bone marrow–derived stem cells; the Effectiveness and Safety of Adipose-Derived Regenerative Cells for Treatment of Glaucomatous Neurodegeneration (NCT02144103) in Russia on subtenon injection of autologous adipose–derived regenerative cells for primary open-angle glaucoma; and the Intravitreal Mesenchymal Stem Cell Transplantation in Advanced Glaucoma (NCT02330978) in Brazil on intravitreal autologous bone marrow–derived MSC transplantation for the worst eye of legal bilaterally blinded glaucoma patients. The two advanced glaucoma patients in NCT02330978 trial did not show improvements on visual acuity, visual field, and electroretinographic (ERG) responses after transplantation. Yet, retinal detachment with proliferative vitreoretinopathy was found in one patient at 15 days after MSC transplantation 111 . The strategy of MSC transplantation in glaucoma patients should be a concern, and modifications may be needed. The safety of intravitreal MSC transplantation requires further investigations. The results of other three clinical trials have not been released yet.
Enhancement of the Neuroprotective Effects for Adult Stem Cell Application
Although adult stem cell transplantation has been proven to alleviate RGC death and promote axonal regeneration in different RGC degeneration models, the neuroprotective effect of adult stem cells is still modest. The regenerative properties can be influenced by the intrinsic and extrinsic factors, such as age effect 112 and cigarette smoke component 113 . To boost up the treatment effect of adult stem cells for optic neuropathies, priming or preconditioning of adult stem cells is a prospective strategy to enhance adult stem cell properties before transplantation. These include improving the grafting ability of adult stem cells, increasing stem cell survival in host tissues, and promoting the paracrine effects and migration ability of stem cells. Recently, there are growing research interests to explore different attempts to modulate the properties of adult stem cells (Fig. 1).

Enhancement of adult stem cell properties. Priming or preconditioning of adult stem cells can enhance the properties of adult stem cells with hypoxia, electrical stimulation, laser irradiation or by chemicals, growth factors, cytokines or herbal medicine/molecules. CREB: cAMP response element-binding protein; CXCR: chemokine receptor; ERK: extracellular signal-regulated kinase; HIF: hypoxia inducible factor; IDO: indoleamine 2,3-dioxygenase; JNK: c-Jun N-terminal kinase; MMPs: matrix metalloproteinases; NRF-2: nuclear factor erythroid 2-related factor 2; PEG2: prostaglandin E2; ROS: reactive oxygen species; SOX9: sex determining region Y-box transcription factor 9; STAT3: signal transducer of activators of transcription 3.
Hypoxia
Hypoxic condition (2% oxygen) has been demonstrated to increase the proliferation rate and stemness of adipose-derived stem cells without altering cell morphology 114 , which promotes DNA synthesis and reduces the expression of MMPs 114 . Human bone marrow–derived stem cells and umbilical blood CD133+ cells, when exposed to 1.5% oxygen for 24 h, could increase total colony numbers with hypoxia inducible factor (HIF)-1α upregulation 115 . The increase in stem cell survival after hypoxia treatment is associated with the expression of HIF-1α through the increased phosphorylation of AKT and p38 MAPK 116 . Low oxygen–activated HIF-1α delays cell senescence with macrophage inhibitory factor activation and p53-mediated pathway inhibition 117 . In addition, canine adipose tissue–derived MSCs preconditioned with deferoxamine (a hypoxia-mimetic agent) exhibit an increased secretion of anti-inflammatory prostaglandin E2 and tumor necrosis factor-α–stimulated gene-6, while decrease the M1 macrophage but increase the M2 macrophage marker expression 118 . Furthermore, hypoxic preconditioning prior to transplantation can suppress MSC apoptosis and increase the migration abilities with higher CXCR4 expression, resulting in promoting neuronal regeneration and improving neural function after transplantation in rat model of middle cerebral artery occlusion 119 .
Chemicals, Growth Factors, and Cytokines
FGF can promote the proliferation of MSCs from different sources120,121. Preconditioning human umbilical cord blood–derived MSCs with both interferon-γ (IFN-γ) and interleukin-1β (IL-1β) can induce regulatory T-lymphocyte differentiation and inhibit Th1 T-lymphocyte differentiation with increasing prostaglandin E2 secretion and indoleamine 2,3-dioxygenase (IDO) and cyclooxygenase 2 (COX2) gene expression 122 . Melatonin preconditioning can increase MMP-9 and MMP-13 expression in bone marrow–derived MSCs 123 , and improves transplanted bone marrow–derived MSC survival and engraftment, which promotes renal regeneration in chronic kidney disease rat model 123 . Moreover, preconditioning with hydrogen peroxide (H2O2) also reported to enhance the proliferation, clonogenicity, adhesion, and migration of decidua basalis–derived MSCs and reduce IL-1β expression 124 . H2O2 preconditioning can increase superoxide dismutase, NQO1, catalase, and heme oxygenase 1 expression in bone marrow–derived MSCs via nuclear factor erythroid 2-related factor 2 pathway, resulting in decrease in intracellular ROS levels, increase in the anti-oxidative stress ability, and promotion of bone marrow–derived MSC survival 125 . In addition, preconditioning human cardiac stem cells with cobalt-protoporphyrin (heme oxegenase-1 inducer) can result in greater improvement in left ventricular remodeling by protecting human cardiac stem cells from apoptosis via ERK/NRF-2 signaling pathway activation 126 . Thrombin preconditioning can accelerate human umbilical cord blood–derived MSC-derived extracellular vesicles biogenesis and enrich the cargo contents by protease-activated receptor-mediated signaling pathways 127 , whereas less degradation of exosomes could be achieved by kartogenin preconditioning on bone marrow–derived MSCs 128 . Furthermore, treatment of rat MSCs with valproic acid and lithium can stimulate cell migration through the HDAC/CXCR4 and GSK-3β/MMP-9 pathways, respectively 129 .
Herbal Medicine and Molecules
We recently demonstrated that curcumin treatment (5 μM) on human bone marrow–derived MSCs can enhance MMP13-mediated osteogenic differentiation, reduce MMP1 expression, and upregulate immunomodulatory gene IDO1 expression although it would inhibit MSC proliferation and migration in high concentration (10 μM or above) 130 . Treatment of a Chinese medicinal herbal formula, Danggui Buxue Tang (a mixture containing Astragali radix and Angelicae sinensis radix), can increase the number of bone marrow stromal cells and promote cell adhesion and migration via focal adhesion and PI3K/Akt signaling pathway activation 131 . Moreover, treatment of Chinese medicinal herbal formula Du-Huo-Ji-Sheng-Tang and its active constituent Ligusticum chuanxiong hort can delay human MSC aging process by decreasing cell senescence and increasing its osteogenic activity 132 . Treatment of Salvia miltiorrhiza Bunge attenuates apoptosis and improves cell viability of MSCs, leading to the recovery of the infarcted brain region and behavior in rat model of middle cerebral artery occlusion 133 . In addition, treatment of icariin (the active component of epimdium koreanum or epimdium berberidaceae maxim) promotes the survival of adipose-derived MSCs against oxidative stress, reduces cell apoptosis, and attenuates intracellular ROS through the PI3K/Akt-STAT3 signaling pathway 134 . Treatment of cajanine, isolated from the extracts of pigeon pea (Cajanus cajan L. Millsp.), can promote bone marrow–derived MSC proliferation by activating the cell cycle signal transduction pathway and accelerating their osteogenic differentiation 135 . Similarly, treatment of polydatin (isolated from the bark of Picea sitchensis or Polygonum cuspidatum) can enhance the proliferation of bone marrow–derived MSCs and the alkaline phosphatase activity via the BMP-Wnt/β-catenin signaling pathways 136 .
Laser Irradiation
Low-level laser irradiation (LLLI; 660 nm, 30 mW) with 1 J/cm2energy density can enhance human PDLSC proliferation 137 . Moreover, LLLI (660 nm, 70 mW) with the energy density of 4 J/cm2can improve the bone healing process of adipose-derived stem cells in rats with critical-sized calvarial defects 138 . Furthermore, the proliferation, adipogenic differentiation ability, and VEGF, TGF-β, and platelet-derived growth factor (PDGF) secretion of adipose-derived stem cells can be enhanced by 4 J/cm2 GaAIAs low-level red laser (650 nm, 523 mW) 139 .
Electrical Stimulation
Porcine bone marrow–derived MSCs preconditioned with nanosecond pulsed electric fields (10 ns at 20 kV/cm and 100 ns at 10 kV/cm) enhances the chondrogenic differentiation with an increase in matrix deposition through the activation of JNK/CREB-STAT3 signaling pathway, and transplantation of preconditioned MSCs also enhances cartilage regeneration in rats suffered from articular cartilage defects 140 . Moreover, in vitro electrical preconditioning of human neural progenitor cells with a conductive polymer scaffold by a +1 V to −1 V square wave at 1 kHz for 1 hour can enhance stroke recovery in rat model of distal middle cerebral artery occlusion through VEGF-A pathway 141 . In addition, epidural stimulation of the motor cortex by electrical current for 1 month in normal rats can increase the proliferation and migration of progenitor cells in the subventricular zone 142 . Furthermore, in the rat ischemic stroke model, electrical stimulation (100 μA, 100 Hz) for 2 weeks after injection of rat bone marrow–derived MSCs (62.5 × 106 cells/ml) showed amelioration in neurological severity score and reduction in the infarction areas 143 . Longer migration distance and wider migration area are also observed for the transplanted bone marrow–derived MSCs with electrical stimulation.
Replacement Therapy for Optic Neuropathies by Adult Stem Cells
Cell replacement therapy conceptually aims to generate RGCs from stem cells to replace the diseased RGCs so as to rescue the visual function loss. New RGCs could be produced from different stem cell sources. Majority of research studies currently focus on the generation of RGCs from pluripotent stem cells, such as ESCs and iPSCs. Nevertheless, adult stem cells have also shown possessing the ability differentiating into retinal/neuronal lineage. The patients’ stem cell–derived RGCs can serve as an in vitro platform carrying the patients’ genome to study human disease pathology and mechanism as well as for drug screening and development 101 .
Retinal Differentiation of Induced Pluripotent Stem Cells
Pluripotent stem cells, including ESCs and iPSCs, are the most promising sources of stem cells for cell replacement therapies due to their powerful differentiation capacities. The ESC therapy must be allogenic and is still under ethical controversial. In contrast, iPSCs can be generated from patients’ somatic cells with the ability to differentiate into the cells of three germ layers, including retinal cells 144 . Importantly, iPSCs can also be used to generate retinal organoids 145 . Not only from the normal subjects, RGCs can also be generated from the iPSCs of the patients with Leber hereditary optic neuropathy 146 and autosomal dominant optic atrophy 147 . Understanding the disease pathology and mechanism by the patients’ iPSC-derived RGCs could be one of the important strategies in future optic neuropathy research.
Retinal Differentiation of Müller Glia
Retinal Müller glia are believed acting like RPCs when responding to the retinal injury 148 . Human Müller cell line (MIO-M1) demonstrated the photoreceptor differentiation ability through the regulation of canonical Wnt signaling in vitro149–151. Moreover, transplantation of human Müller cell line (MIO-M1; 3 × 104 cells in 3 μl) in the laser-induced chronic IOP elevation rat model showed the capability to differentiate toward a neuronal phenotype rather than glial at 2 weeks after transplantation 77 . Although previous studies reported that adult stem cells directly injected into the retina could not spontaneously differentiate into retinal cells in the transplanted animal eyes, recent findings suggest that adult stem cells could also stimulate Müller glia re-entering the cell cycle and dedifferentiating to the RPC stage in vivo. The transplanted HSCs have been shown to fuse with Müller glia in the mouse model of N-methyl-N-nitrosourea-induced progressive photoreceptor degeneration that the Wnt signaling pathway is activated in the hybrid cells and drives the de-differentiated hybrid cells toward the intermediate photoreceptor progenitor fate150,152. Not only the transplanted cells, the endogenously mobilized bone marrow cells could also fuse with Müller glia in the retina through the SDF1/CXCR4 axis regulation in the N-methyl-D-aspartate-induced retinal damage mouse model, which the hybrid cells could further differentiate toward a neuronal fate, including RGCs and amacrine cells. 153 Critically, the hybrid cells can also promote the regeneration of other damaged retinal cells. Retinal damage is critical for the hybrid cell formation in vivo, and retinal Müller glia can spontaneously fuse with the transplanted or endogenously mobilized HSCs. The hybrid cells can proliferate and commit to differentiation toward the mature retinal cells through the activation of the signaling pathways150,152,153. This discovery makes an advancement in the in vivo cell replacement therapy for retinal damage. Yet, the conversion efficiency, regenerated cell survival, and functional integration and rescue still need further in-depth investigations.
Retinal Differentiation of Mesenchymal Stem Cells
Retinal differentiation is not limited to stem cells from retinal lineage since adult stem cells can give rise to an entirely distinct lineage of mature cells out of its physiological differentiation process. MSCs, which normally differentiate into chondrocytes, adipocytes or osteoblasts, can be induced into neural cells 62 . Upon bone marrow transplantation, genetic markers from donor’s hematopoietic cells could be found in multiple tissues, including brain cortex and cerebellum 154 . Moreover, intravitreally injected adult rat hippocampus-derived neural stem cells (5 × 105 cells in 5 μl) in the transient retinal ischemia-reperfusion adult rat model can integrate into the host retina and express neuronal differentiation marker Map2ab at 4 weeks after transplantation 155 . In addition, subretinal transplantation of adult hippocampus-derived neural progenitor cells in transient retinal ischemia model could display the neuron-like morphologies with fibers extending to RNFL at 4 to 5 weeks after transplantation, but they rarely co-express the retinal phenotypic markers 156 . The plasticity of adult stem cells could be interpreted by five possible mechanisms: cell fusion, transdifferentiation, dedifferentiation, heterogeneous stem cell populations, and pluripotency 58 . Cell fusion postulates that stem cells fuse with the host cells and acquire a mature phenotype of the embedded tissue. Transdifferentiation refers to the direct lineage conversion with the activation of dormant differentiation program to alter cell lineage specificity, whereas dedifferentiation hypothesizes that tissue-specific stem cells dedifferentiate spontaneously into basal multipotent cell state and re-differentiate into another cell lineage. Heterogeneity in stem cell population indicates a mixed population of stem cells found within the tissue origin, which might explain some events of transdifferentiation and dedifferentiation. Pluripotency is believed that pluripotent stem cells exist in adult tissues as a rare sub-population within the stem cell niches, and these pluripotent stem cells have been found in bone marrow–derived MSCs, spermatogonial stem cells, and the remnants of migrating neural crest 157 . Unlike iPSC reprogramming, retinal/neuronal differentiation of adult stem cells can bypass the complicated dedifferentiation process so as to reduce the risk of teratoma formation by the undifferentiated iPSCs upon transplantation 158 . Once the retinal/neuronal differentiation protocols of adult stem cells could be optimized, the in vitro neuron production could overcome the difficulty to collect retinal/neural progenitor cells from the patients.
Retinal differentiation of MSCs from different sources has been reported (Table 3). Human umbilical cord blood–derived MSCs and amniotic epithelial stem cells have been shown to be induced into cone/rod photoreceptor-like cells by inhibition of microRNA (miRNA)-203149. Mouse bone marrow–derived MSCs treated with BDNF, NGF, and bFGF can also express the photoreceptor and neural markers after 14-day induction, which can be enhanced by supplementation of human recombinant Wnt1152. Apart from photoreceptors, retinal pigment epithelial (RPE)-like cells have been reported to be generated from human adipose-derived stem cells 159 , and bone marrow–derived stem cells 160 . Moreover, rat bone marrow–derived MSCs can also be directed to RGC-like cells by co-culturing with postnatal day 1-3 rat retinal cells 161 . Coherent to other studies, we previously showed that human PDLSCs can be induced into retinal lineage with the treatment of noggin, Dkk-1, IGF-1, and bFGF with photoreceptor marker expression and glutamate-evoked calcium response 162 . Based on the noggin, Dkk-1, IGF-1, and bFGF treatment, with the addition of BDNF, CNTF, NGF, and Shh, can direct human PDLSCs to RGC lineage after 24-day induction with neuronal and RGC marker expression, synapse formation, the glutamate-evoked calcium response, and spontaneous electrical activities 163 . The neuronal differentiation of human PDLSCs could be mediated by miRNA-132 164 . In addition to PDLSCs, we demonstrated that human adipose-derived stem cells can also be directed to retinal cells, and Notch signaling activator JAG1 can enhance the expression of RGC and RPC markers 165 . Furthermore, we revealed that spermatogonial stem cells possess the capability to differentiate toward the RGC lineage upon the 3-dimensional organoid culture 166 . Notably, apart from the pluripotent spermatogonial stem cells, pluripotent stem cells with neural crest origin could be isolated and purified from human PDLSCs by connexin-43 167 . These pluripotent adult stem cells should improve the retinal cell differentiation form human adult stem cell sources. Therefore, transplantation of adult stem cell–derived retinal cells should be a manageable treatment against retinal diseases in future 69 .
Retinal Differentiation of Adult Stem Cells.

BDNF: brain-derived neurotrophic factor; bFGF: basic fibroblast growth factor; CNTF: ciliary neurotrophic factor; Dkk-1: dickkopf-related protein 1; DMEM: Dulbecco’s modified Eagle medium; EGF: epidermal growth factor; FBS: IGF-1: insulin-like growth factor 1; ITS: insulin-transferin-selenium; ME: mercaptoethanol; MEM: minimal esstinal medium; MIO-M1: Human Müller cell line; NEAA: non-essential amino acids; NGF: nerve growth factor; Shh: sonic hedgehog.
Challenges and Prospects
The therapeutic potentials of adult stem cells on optic neuropathies have been intensively investigated (Fig. 2). The neuroprotective effect of HSCs and MSCs for RGC protection is convincing; yet, there are concerns and challenges for the adult stem cell treatments: (1) A standard guideline should be established to standardize the purity and condition of the transplanted adult stem cells as well as the modes of MSC transplantation since severe bilateral visual loss was reported in three age-related macular degeneration patients with intravitreal autologous adipose tissue–derived “stem cells” injections 169 , and retinal detachment with proliferative vitreoretinopathy was found in one patient at 15 days after MSC transplantation in a clinical trial study 111 . (2) Preconditioning could be a promising strategy to enhance the neuroprotective effects of adult stem cells; yet, studies in this area are still limited. Other preconditioning techniques and strategies as well as their mechanisms warrant further exploration and optimization. (3) Intravitreal transplantation of bone marrow–derived MSCs could induce reactive gliosis by activating the JAK/STAT3 and MAPK cascades in Müller glia, which could be a main obstacle for successful transplantation in RGC replacement and axonal regeneration 170 . (4) Stem cell–derived retinal cell integration and connection with the existing network in the host retina are the major challenges for the transplantation of exogenous stem cell–derived retinal cells. The connectivity of stem cell–derived RGCs could be hindered by the internal limiting membrane 171 . Activation of endogenous retinal stem cells or Müller glia to replace the diseased cells could be a possible alternative strategy to minimize these concerns 172 . (5) The majority of retinal disease modeling by stem cells currently relies on iPSCs 101 . Whether adult stem cells could similarly resemble the models of human diseases remains to be determined. These questions are critical for future clinical application of adult stem cell therapy, which creates the research opportunities on adult stem cells for RGC protection and regeneration. Furthermore, functional modulation of other cells in the optic nerve, including astrocytes, oligodendrocytes, and microglia, should also be considered in the treatments for optic neuropathies. Further studies are needed to optimize the application of adult stem cells in the treatment regime for optic neuropathy.

Research progress of adult stem cells in optic neuropathies. Adult stem cells can be conveniently isolated from different tissues and organs, including adipose tissue, bone marrow, and periodontal ligament. Cell replacement therapy (based on the differentiation of adult stem cells into retinal ganglion cells [RGCs]) and the neuroprotective effects of adult stem cells (based on the neurotrophic factor and exosome secretion) have been investigated to alleviate the loss of RGCs and their axons and promote RGC survival and axonal regeneration. RNFL: retinal nerve fiber layer. Parts of the figure were created by Figdraw (www.figdraw.com).
Footnotes
Acknowledgements
We are grateful to Mr. Nikky Yuk-Ki Lai and Miss Samantha Antonio Leong from the Chinese University of Hong Kong for their assistance.
Author Contributions
S.T. and T.K.N.: Conception and design. S.T. and T.K.N.: Financial support. S.T., Y.Y., Q.Y., X.L.Y., and T.K.N.: Collection and/or assembly of data. Q.Y., X.L.Y., and T.K.N.: Data analysis and interpretation. S.T., Y.Y., Q.Y., X.L.Y., and T.K.N.: Manuscript writing. S.T., Y.Y., L.P.C., and T.K.N.: Critical revision of manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Natural Science Foundation of China (project code: 81800822 to. S.T.), the Joint Regional Basic Science and Applied Basic Science Research Fund of Guangdong Province (project code: 2019A1515110685 to T.K.N.), internal grant from the Joint Shantou International Eye Center of Shantou University and the Chinese University of Hong Kong (project code: 18-001 to T.K.N.), China, and internal research grant from the Hong Kong Polytechnic University (project code: ZVS7 and BD82 to S.T.), Hong Kong.
