Abstract
The main objective of this study is to test the potential of specific populations of mouse bone marrow-derived stem cells (BMSCs) to differentiate into the neuronal cell lineage. Bone marrow of 33 mice was aspirated under general anesthesia. The collected marrows were analyzed for cell counts, compositions, and percentages of different stem cell types. We used the Midi MACS magnetic separator to purify specific populations of stem cells from the aspirated bone marrow. Cells were analyzed using flow cytometry. We assessed the presence of stem cell antigen-1 (Sca-1+) and prominin-1+ cells in the cellular fraction that was depleted of lineage-committed cells (lineage-). Both purified and nonpurified cells were cultured ex vivo using specific growth media with factors that drive the cells to differentiate into the neuroglial cell types. Cells were then analyzed by flow cytometry for expression of specific neuronal markers. Our results showed that there was an increase of Sca-1+ and prominin-1+ cells in the lineage- fraction over the unpurified BM. After lineage depletion, the percentages of Sca-1+ and prominin-1+ cells increased from 4.9% and 2.6%, up to 76.1% and 59%, respectively. Unpurified mouse BM differentiated into fibroblasts, whereas Sca-1+ cells were able to generate astrocytes. Interestingly, purified prominin-1+ cells were able to generate neuronal cells. Purification of adult bone marrow-derived stem cells enhances their potentiality for differentiating into specific neuronal cell lineages.
Introduction
The nonhematopoietic part of the bone marrow (BM) consists of various heterogeneous populations of cells. Accordingly, there has been much effort directed towards separating the different cellular populations of the BM. Most striking are primitive multipotent mesenchymal stem cells or marrow stromal cells (MSCs), because they are easily expanded in culture and differentiate into multiple tissue lineages in vitro, including myoblasts, hepatocytes, and even neural tissue (12–14,30). Therefore, there has been much interest in their clinical potential for tissue repair and gene therapy (3,6,34). The first step in stem cell therapy is to obtain the required amount of specific population of stem cells. They may be isolated and cultured in vitro in order to increase their number and, if necessary, can be differentiated into the de-sired cell types. They can be isolated from various sources, including bone marrow (26,35), peripheral blood (9), umbilical cord (4), Wharton jelly (40), amniotic fluids (37), and deciduous teeth (22). Although MSCs represent a very small fraction in the bone marrow, 0.001–0.01% of the total nucleated cell (26), MSCs can be expanded under defined culture conditions.
The identification and selection of stem cells within a given tissue/organ largely relies on the presence of specific cell surface markers such as CD146, CD34, STRO-1 antigen, Thy-1, nestin, Sca-1, and CD133 (25, 26,28,29,31,38,44). SC isolation has been achieved mainly by following protocols that utilize flow cytometry-based sorting of stem cells (11) or positive and negative selections using magnetic beads tagged with antibodies targeting specific markers on the surface of stem cells (17). The most promising stem cell markers are stem cell antigen (Sca-1, Ly-6A/E) and prominin-1 (CD133, AC133), which were found to be expressed on stem cells in the BM, where the positive cells were shown to have a higher potential to differentiate into multiple tissue lineages (19,21,23,33)). Prominin-1 is also expressed on neural stem cells (20), the apical surfaces of neuroepithelial cells, and the brain ependymal layer of the adult mouse (41). Additionally, 10–15% of the neural cells in the subventricular zone of postnatal mice were shown to be prominin-1+ (41).
In the present study, we isolated specific populations of bone marrow- derived stem cells (BMSCs), which are highly enriched in the population characterized by low levels of the lineage markers found on mature hematopoietic cells (CD5, CD45R, CD11b, Anti-GR-1, and Ter-119), and by high levels of Sca-1 and prominin-1 antigens. The major aim of this work was to isolate and investigate the ability of specific populations of mouse BMSCs to differentiate in vitro toward neuronal-like cells in response to a treatment involving low-affinity nerve growth factor and spinal cord extract as physiological inducers.
Materials and Methods
Preparation and Aspiration of Bone Marrow
Animal experiments were approved by University Animal Ethics Committee and conducted according to Jordan University of Sciences and Technology research standards.
Bone marrow samples were collected from 33 Balb/c mice, 6–8 weeks old, weighing 100–150 g. The mice were anesthetized using halothane anesthesia (HIKMA Pharmaceuticals, Jordan). The femur bone was exposed and a small hole at an angle of 60° toward the knee was opened using a special drill (Wuyi Dongda Electric Appliance Co, China). The BM was aspirated using a 1-ml syringe with 26-gauge needle by flushing the shaft by cold buffer under aseptic conditions. The buffer consisted of phosphate-buffered saline (PBS) (Miltenyi Biotec, GmbH, Germany), 0.5% bovine serum albumin (BSA) (Life Technologies, Scotland), and 2 mM EDTA (Sigma). Flushing and aspiration was repeated five times. The collected BM was stored in a 15-ml conical tube on ice. The incision of the muscles and skin was sutured. Then the animals were put in the cage, and returned to the animal house where surveillance was made.
Processing the Cells
A 30-μm nylon mesh filter (Miltenyi Biotec, Germany) was wetted with 1–2 ml of cold buffer. The collected BM cell suspension passed through the filter to remove cell aggregates and bone fragments. The filtered cell suspensions were washed by adding buffer up to 14 ml and centrifuged at 3000 x g for 10 min at 4–8°C using a refrigerated centrifuge (Beckman Coulter, US). The supernatant was decanted and the cell pellet was resuspended in 1 ml buffer. The cells were then counted using a counting chamber and the results were recorded.
Selection of Stem Cells by MACS® Magnetic Labeling and Separation
Specific types of mouse BMSCs were purified using depletion or positive selection strategies. For depletion of unwanted cells, Lineage Cell Depletion Kit (Miltenyi Biotec) was used according to the manufacturer's protocols. Briefly, we used a specific magnetic labeling method for the depletion of mature hematopoietic cells such as T cells, B cells, monocytes, granulocytes, and erythrocytes from the BM. This method allows the negative purification of multiple types of cells, leaving only progenitor cells of known and unknown types (lineage negative) to be used in further experiments. The kit contains 1 ml cocktail of biotin-conjugated monoclonal antibodies against CD5, CD45R, CD11b, anti-GR-1, and Ter-119 and 2 ml anti-biotin-conjugated microbeads. Cells were first centrifuged as above, and the supernatant was removed. Cells were resuspended in 40 μl buffer per 107 cells. Then, 10 μl of the cocktail of biotin-conjugated monoclonal antibodies was added per 107 cells. The cells were mixed well and incubated for 10 min at 4–8°C. Then 30 μl of buffer per 107 cells was added, followed by adding 20 μl of microbeads per 107 cells. The mixture was well mixed and incubated for an additional 15 min at 4–8°C. The cells were then washed by adding 1–2 ml of buffer per 107 cells and centrifuged as above. The supernatant was removed, and cells were finally resuspended in 500 μl of buffer for up to 108 cells. For the magnetic separation of cells, the LS MACS column was placed in the Midi MACS® magnetic separator (Miltenyi Biotec). The column was prepared by rinsing it with 3 ml of buffer. The labeled cells were loaded onto the column, and were allowed to pass through. The unlabeled cells (lineage negative) passed through the column and were collected in a fresh 15-ml tube, whereas the labeled (lineage-positive) cells were trapped by the magnet. Next, the column was washed three times, each with 3 ml buffer. To collect the lineage-positive cells (to be used as a control), the column was removed from the magnet and placed into a fresh 15-ml tube. Lineage-positive cells were collected in a fresh tube by adding 3 ml of buffer and pressing the cells abruptly with the plunger of the LS column. Both cell populations were centrifuged as above, the supernatants were removed, and the cells resuspended in the appropriate media as described below. Unpurified BM, lineage-negative, and lineage-positive cells were assessed for the percentages of Sca-1+ and prominin-1+ cells.
For magnetic purification using a positive selection system for purifying specific cell types, similar steps to the Lineage Cell Depletion Kit were repeated using anti-Sca-1 and anti-prominin-1 microbead kits (Miltenyi Biotec) according to the manufacturer's instructions. We separately used the Sca-1 kit to positively purify Sca-1+ cells and the prominin-1 kit to purify prominin-1+ cells. Cells in the unpurified BM, lineage negative, lineage positive, purified Sca-1+, and purified prominin-1+ cell fractions were analyzed by flow cytometry and using fluorescent antibodies against specific cell surface markers.
Cells were analyzed using fluorescein-5-isothiocyanate (FITC) or phycoerythrin (PE)-conjugated monoclonal antibodies (Sca-1 and prominin-1), and flow cytometer (BD BioSciences Discovery Labware, USA).
Ex Vivo Culture and Differentiation of BMSCs
Purified stem cells, as well as nonpurified bone marrow mononuclear cells, were cultured as described below in nonhematopoietic expansion (NHE) media (Miltenyi Biotec) supplemented with 100 U/ml penicillin, 100 mg/ml streptomycin, and 100 mg/ml fungisone (GiBCO 15140, Invitrogen). Cells were cultured in NHE media alone, NHE supplemented with low-affinity nerve growth factor (LNGF) (Miltenyi Biotec), or in NHE supplemented with mouse spinal extract (SpE) (10,16) to allow differentiation of stem cells into neural-like cells. SpE was freshly prepared by collecting mouse spinal cord, resuspending it in 10 ml NHE, and homogenization using GentleMACS Dissociator (Miltenyi Biotec) with C tubes. This procedure allows rapid homogenization of the neuronal tissue and obtaining single cell suspensions. The homogenized extract was filtered to remove clumps and large tissue fragments, and then centrifuged as described above. The filtered extract was incubated in a 37°C incubator with 5% CO2 for 48 h. Then, media containing the extract was centrifuged at 300 x g at 4°C for 20 min. The supernatant was removed and filtered with 0.2 μm and aliquoted into 0.5-ml aliquots, and was stored at −20°C to be used on the stem cells every 3 days.
Immunocytochemistry
Cultured mBMSCs were fixed with 4% paraformaldehyde, incubated with primary monoclonal antibodies for glial fibrillary acidic protein (GFAP), neurofilaments, or S100 (Dako, Japan) overnight at 4°C, incubated with secondary antibody for 1 h, followed by exposure to avidin-biotin complex for 1 h (25°C). DAB served as chromagen.
Results
The collected bone marrow was analyzed for cell counts, compositions, and percentages of different types. Specific stem cell types were purified according to specific surface marker expression. The average of BMNC counts that were harvested from mice BM was 283 million cells with a standard deviation of 225 million.
Purification of Stem Cells and Assessment of Their Percentages in Mice BMs
Flow cytometry was used to determine the percentages and phenotype of BMSCs, and the cell-surface markers were analyzed. Variations in the percentage of stem cells and progenitors in BM samples ware apparent. The results showed that the cells expressed the putative markers for BMSCs, such as Sca-1 and prominin-1 in the unpurified BM. After lineage depletion, the percentages of Sca-1+ and prominin-1+ cells increased in some samples from 4.9% and 2.6%, up to 76.1%, and 59%, respectively (Fig. 1). Alternatively, Sca-1 microbeads were used to purify Sca-1+ stem cells (magnetic purification, positive selection). The results showed there was enrichment of Sca-1+ cells from 0.64% in the unpurified BM up to 23.59% in the Sca-1-enriched fraction. Interestingly, prominin-1+ cells were also enriched through Sca-1 purification from 1.64% in unpurified BM to 41.14% in the Sca-1-positive fraction (Fig. 2). Furthermore, purification of prominin-1+ cells (magnetic purification, positive selection) from mouse BM was performed using anti-prominin-1 microbeads. Prominin-1 purification results revealed that the percentage of prominin-1+ cells increased from 1.58% in unpurified BM to 74.72% in the prominin-1+ fraction, whereas Sca-1 expression was decreased from 0.32% in unpurified BM to 0.16% in the prominin-1+ fraction (Fig. 3). The observations of the present study showed that almost similar purities were obtained from both purification of prominin-1+ cells using prominin-1 microbeads and lineage depletion strategies (Fig. 4); the percentages of prominin-1+ cells increased from 0.85% in the BM, up to 74.3% and 70.7%, respectively.

Depletion of lineage-committed (lineage positive) cells from mouse BM and assessment of Sca1+ and prominin1+ cells. The figure shows percentages of two stem cell populations according to their surface marker expression before and after depletion of lineage-committee cells. The left panels show percentages of cells that express the corresponding marker in mouse BM. The right panels show percentages of cells expressing the same marker after depletion of lineage-committed cells using magnetic purification.
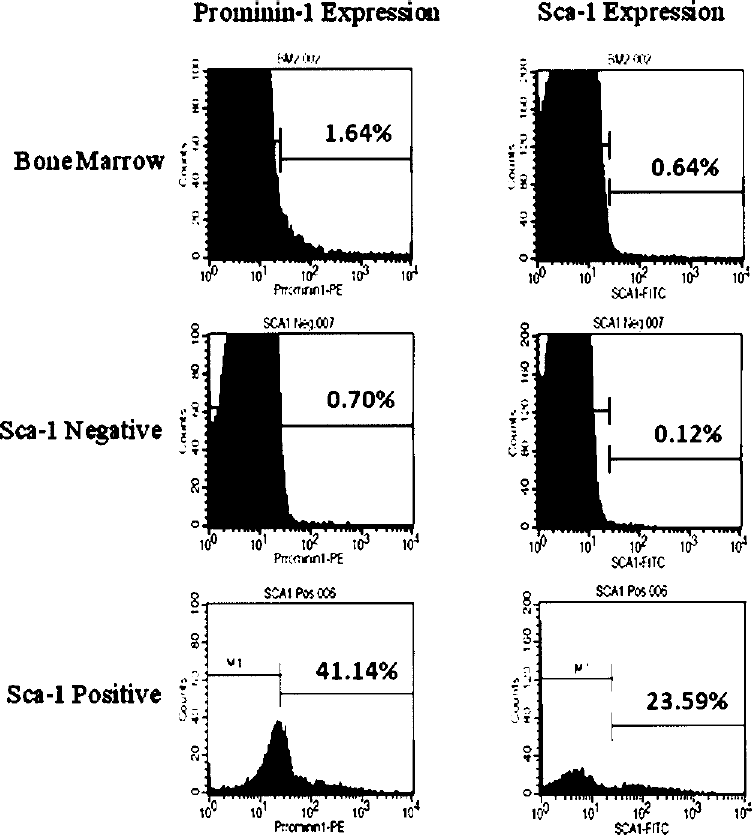
Purification of Sca-1+ cells from mouse BM. Histograms in the right panels show percentages of Sca-1+ cells in mice BM and in cellular fractions purified by Sca-1 microbeads, whereas histograms in the left panels show percentages of prominin-1+ cells in BM cells and in populations purified by Sca-1 microbeads.

Magnetic purification of prominin-1+ cells from mouse BM. Upper row shows expression of prominin-1, Sca-1, and both together in unseparated mouse BM. The left panels show expression of prominin-1, the middle panels show expression of Sca-1, and the right panels show expression of both together in percentages of cells expressing both markers.

Purification of prominin-1+ cells either by depletion of lineage-committed cells or by direct purification of prominin-1+ cells.
Ex Vivo Cultures of Sca-1+ Cells, Prominin-1+ Cells, and BM Cells
During the first 14 days in culture, cells in all three types of cells showed similar properties in terms of cell proliferation, differentiation, and morphology. However, starting in the third week, we noticed different behaviors of cells in different types. There were more dividing cells in unpurified BM culture, compared with Sca-1+ and prominin-1+ cultures.
Mouse BMSCs differentiated into different types of neuronal-like cell lineages according to the type of purified cells present in the fifth week. Primarily, cell bodies became increasingly spherical and refractile, exhibiting a typical neuronal perikaryal appearance. Processes elaborated, displaying primary and secondary branches, growth cone-like terminal expansions, and putative filopodial extensions. Purified Sca-1+ cell culture showed cells with multiple cellular protrusions resembling astrocytes (Fig. 5). Purified prominin-1+ cells were able to generate neurons and astrocytes (Figs. 6, 7, 8, 9, and 10). BMSC-derived neurons displayed distinct neuronal morphologies ranging from simple unipolar and bipolar to large, extensively branched multipolar cells (Figs. 7, 8, 9, and 10). In contrast, when we cultured unpurified mouse BM MNCs in the same media, the cells were driven to differentiate mainly into fibroblasts. Cells grown in unsupplemented NHE media all died after 15–20 days in culture.

SCA1+ clusters of MSC-derived neurons.

Prominin-1+ MSC-derived bipolar neuron.

Morphology of prominin-1+ MSC-derived unipolar neuron.

Morphology of prominin-1+ MSC-derived multipolar neuron.

Morphology of prominin-1+ MSC-derived bipolar neuron.

Morphology of prominin-1+ MSC-derived neuron displays condensed cell bodies and highly branched processes.
To further characterize neuronal differentiation we stained cells for the neural markers glial fibrillary acidic protein (GFAP), neurofilament, and S100. Progressive transition of BMSCs to a neuronal phenotype coincided with increased expression of all three neuronal markers (Figs. 11, 12, and 13). These results show the different potentialities of Sca-1+ cells and prominin-1+ cells to generate various types of neuronal cell types. However, these observations were based on the morphology of cells in culture, and further assessment of cellular characterization is required before drawing final conclusions.

GFAP expression in prominin-1+ cells fraction.

S100 expression in prominin-1+ cells fraction.

Neurofilaments expression in prominin-1+ cell fraction.
Discussion
Recently, stem cells have been widely characterized in different aspects. It is broadly agreed that stem cells are going to be an important part of cell-based therapies, in which a relatively high number of healthy cells are needed. Development of rapid, simple, inexpensive, and, maybe more importantly, specific separation techniques will be the main technological targets in this direction. Magnetic separation has been considered as quite easy, effective, and a very rapid technique to isolate unaltered and viable cells (15,22,24,32)).
This study presents unique strategies for obtaining specific populations of stem cells from mouse BM, and assessing their potentials for differentiation into cells of neuronal lineages. BMs of mice were aspirated, and specific cells were purified using different strategies.
Using the depletion methods yielded increased percentages of Sca-1+ stem cells in the lineage-negative fraction over unpurified BM fraction, where the remaining population in the lineage-negative fraction contained enriched populations of Sca-1+ and prominin-1+ cells. On the other hand, our results showed that, using prominin-1 magnetic beads, cells expressing prominin-1 were enriched, which included two populations: prominin-1+/Sca-1- cells (common), and prominin-1+/Sca-1+ cells (rare). To compare the efficiency of purifying Sca-1+ cells, we compared two methods: lineage depletion and direct purification of Sca-1 cells. Our results showed that the methods yielded similar results in terms of recovery and purity of Sca-1+ cells. However, using the depletion strategy, we were able to get other populations of stem cells in the BM that express other markers, including Sca-1, and other unidentified cell populations.
In the present study, different populations of stem cells were isolated from mouse bone marrow, by positive and negative selections using magnetic separation, and expanded in vitro successfully. Several reports have shown that BMSCs have the potential to differentiate into neuronal phenotypes (either neurons or glia) in vitro (1,18,27,33,43). Our results confirmed the data reported by these authors. The observation of the present study clearly demonstrates that BMSCs can be differentiated into neuronal-like phenotype in vitro after 30 days of culture in nonhematopoietic expansion (NHE) media supplemented with low-affinity nerve growth factor (LNGF) or mouse spinal extract. In this treatment, besides the morphological evidence, we have also demonstrated that the neuronal-like phenotypes derived from BMSCs express cell surface markers that are specific to neural cells such as GFAP, S100, and neurofilaments, associating with remarkable morphological modifications similar to other previous reports (5,7,8,36,39,42).
In our study, remarkable fraction of treated BMSCs showed immunoreactivity for neural markers, suggesting that only a subset of BMSCs can differentiate into cells with a neural phenotype. We can therefore assume that this fraction of BMSCs may be constituted by tissue-specific progenitor cells with restricted differentiational potential, and are able to give rise to cells characteristically found in other tissues under specific experimental conditions. Actually, morphological heterogeneity of BMSCs has been demonstrated by our data, consistent with a previous report (2). On the other hand, our findings may indicate the possibility that a population of pluripotent stem cells resides in adult bone marrow.
In conclusion, this study confirmed that BM may include a population of pluripotent stem cells that are capable of differentiating into neural cell lineages when cultured under appropriate conditions. The neurodifferentiational manner of BMSCs showed considerable plasticity, depending on the protocols of inducement. Nonhematopoietic expansion (NHE) media supplemented with low-affinity nerve growth factor (LNGF) or mouse spinal extract may enhance BMSCs to differentiate into neurons, astrocytes, and glial cells.
Footnotes
Acknowledgments
The authors would like to thank the National Center for Diabetes, Endocrinology and Genetics for generously allowing us to use their research facility, Rahaf Al-Zoubi for assistance with cell processing, Ahed El-Khatib for his assistance in immunohistochemistry of mouse cells, Feras Twal and Mohammad Ghneim for their assistance in mouse cell cultures, and Al-Amal Hospital for allowing us to use their microscope.
