Abstract
Coronary artery bypass grafting remains the gold standard in the therapy of advanced-stage patients. But the vein grafts are prone to restenosis or failure. Pentoxifylline (PTX) is a methylxanthine derivative with a function of inhibiting cell proliferation. We thus applied PTX locally to the vein grafts to study its effect on the inhibition of graft restenosis using a rat vein graft model. Morphometric results showed a significant decrease in the thickness of vein grafts intimal and medial at day 28 after the bypass operation. Results from Western blot and immunohistochemistry showed that PTX also significantly reduced the proliferating cell nuclear antigen (PCNA), alpha-smooth muscle actin (α-SMA) expression, and phosphorylation of p38 in vein grafts. These results firstly discovered the positive role of PTX in preventing the vein grafts restenosis and the mechanism may be inhibition of vascular smooth muscle cells (VSMCs) proliferation via the p38MAPK pathway.
Introduction
Coronary artery disease (CAD) is a leading disease with increasing morbidity and mortality in the world 1 . Coronary artery bypass grafting (CABG), the method aimed to bypass the occluded coronary arteries, remains the gold standard in the therapy of advanced-stage patients 2 . The saphenous vein is the most commonly used (up to 50%) conduit for CABG, which is reasoned by its convenience of harvesting, sufficient length, and its character of rarely affected by competitive coronary blood flow3,4. But the grafted saphenous vein is prone to restenosis with a patency rate of 65% to 80% in 5 years after operation5,6. Therefore, how to attenuate the restenosis of vein grafts is under challenge.
Intimal hyperplasia (IH) plays a causal role in the restenosis of vein graft. The proliferation of endothelial cells (ECs) and vascular smooth muscle cells (VSMCs), also the migration of VSMCs to intimal, plays a vital role for IH6,7. After sudden exposure to the high-pressure and pulsatile arterial system, the endothelium is vulnerable to be damaged 8 . The vasodilation after vein grafting and endothelial injury will activate the release of inflammatory factors, leading to the proliferation and remodeling of both ECs and VSMCs9,10. Thus, inhibiting the proliferation of ECs and VSMCs to alleviate the IH process is promising for attenuating vein grafts restenosis.
Pentoxifylline (PTX) is a methylxanthine derivative, with properties similar to theobromine, caffeine, and theophylline. Initially, it was used to treat the intermittent claudication. Later studies discovered its role of inhibiting tumor necrosis factor-α (TNF-α)11,12. More notable is that recent researches found the role of inhibiting cell proliferation, such as glia and breast cancer cells12–14. These remind us of the potential function of PTX in the inhibition of vein grafts restenosis.
According to these findings, we hypothesize that PTX may prevent vein grafts restenosis by inhibiting cell proliferation. Thus, we investigated the role of PTX on vein graft restenosis through local delivery of PTX to the vein grafts. The results show that the PTX significantly decreased the intimal and medial thickness of vein grafts. In addition, PTX inhibited cell proliferation in vein graft and decreased the phosphorylated-p38 (p-p38) level. These results first discovered the role of PTX in preventing vein grafts restenosis and lightened the potential mechanism.
Methods
The details for experiments have been shown in previous studies15–17. Sprague Dawley (SD) rats (male, aged 10–12 weeks, body weight 275–325 g) were purchased from Anhui Lab Animal Research Center and were randomly assigned to sham group (sham operated without bypass), control group (bypass only), vehicle gel treatment group (bypass + pluronic-F127 gel), and PTX treatment groups (bypass + pluronic-F127 gel + PTX) (n = 10 per group).
Perivascular Drug Delivery Constructs
The Pluronic F127 gel (Sigma, St. Louis, MO, USA) was used as vehicle gel for local delivery of PTX; 0.3 mg PTX (Aladdin, Shanghai, China) was dissolved with 150 μl distilled water, then resuspended into 300 μl of pluronic-F127 gel (30%, w/v) for local delivery to the vein grafts (PTX group). An equal amount of pluronic-F127 gel was smeared to grafts in the vehicle gel group (gel group). The pluronic-F127 gel was taken out from a 4°C environment until it was used, which aims to maintain its liquid form.
Rat Autologous Vein Graft Model
SD rats were anesthetized with 10% chloral hydrate (300 mg/kg) first and administered heparin (200 U/kg) via caudal vein injection (Qilu Pharmaceutical Co., Ltd, Jinan, Shandong, China). The left jugular vein was harvested for use as a carotid interposition graft. The bypass was performed to be an anastomotic cuff model, as previously described15,17. Specifically, 2 mm cuffs were cut from a 20 G arterial puncture needle (BD Company, Franklin Lakes, NJ, USA). The carotid artery was isolated, the proximal and distal ends of the artery were then placed; suture traction lines and hemoclips were used to block blood flow before a cut in the middle. Both ends of the arteries were then pulled through the cuff. Then the arteries were everted and secured to the cuff with 6–0 silk suture (Ethicon, Cincinnati, OH, USA). The cuffs were then inserted into and secured to the vein with a 6–0 silk suture.
Harvest of Implanted Grafts
Rats were euthanized at day 28 after bypass operation to harvest vein grafts and were cut into two sections. One segment was immediately fixed by 4% formaldehyde after perfusion with heparinized saline for hematoxylin and eosin (H&E) staining, while the other was placed at –80°C for Western blot analysis. Animals were sacrificed by sodium pentobarbital overdose.
Morphometric Analysis
After fixation in 4% formaldehyde and processing in 70% ethanol, specimens harvested at day 28 were paraffin embedded; 3-μm sections were taken throughout the graft, excluding regions immediately adjacent to the anastomotic cuffs. Sections were stained with hematoxylin kit (Gefan Biological, Shanghai, China), and staining sections were observed by an Olympus-IX73 microscope (Olympus, Tokyo, Japan). At least three equally spaced sections of the bypass grafts were analyzed for each specimen. Standard morphometric measurements were recorded, including intimal thickness and medial thickness. These data were then calculated by ImageJ (National Institutes of Health, Bethesda, MD, USA).
Western Blot Assay of PCNA, α-SMA, p38, and p-p38 Protein Expression Analysis
Total protein was isolated from grafted veins. After electrophoresis and electrotransfer, membranes were blocked with 5% skimmed milk and were incubated at 4°C with primary antibodies overnight (with anti-proliferating cell nuclear antigen (PCNA) monoclonal antibody 13110 Cell Signaling, 1:1000 dilution; anti-alpha-smooth muscle actin (α-SMA) antibody ab134047, Abcam, 1:3000 dilution; anti-α-SMA antibody, ab7817, 1:3000 dilution; anti-p38 antibody, ab170099, Abcam, 1:3000 dilution; anti-phosphorylated p38-antibody, 4511, Cell Signaling, 1:1000 dilution). After being incubated with a secondary antibody, the bands were processed by Odyssey v1.2 software (Li-COR Biosciences, Lincoln, NE, China). Gray values were measured based on the internal reference of glyceraldehyde-3-phosphate dehydrogenase 2 (GAPDH-2).
Immunohistochemical Analysis
The paraffin-embedded tissue was sliced into 3 μm sections. The antigen was recovered with citrate buffer for 20 min, followed by deactivated endogenous peroxidase with 3% H2O2 for 20 min and sealing with 5% bovine serum albumin for 10 min. The sections were then treated overnight at 4°C with anti-PCNA monoclonal antibody (13110, Cell Signaling, 1:50 dilution), anti-α-SMA antibody (ab134047, Abcam, 1:500 dilution), anti-p38 antibody (ab119871, Abcam, 1:100 dilution), and anti-phosphorylated p38-antibody (4511, Cell Signaling, 1:100 dilution). Horseradish peroxidase conjugated secondary antibody was added to the slices after wash with phosphate-buffered saline (PBS). The diaminobenzidine tetrahydrochloride (DAB) was used to visualize. All images were acquired by an Olympus-IX73 microscope (Olympus, Tokyo, Japan). At least three sections per graft were analyzed for all markers.
Statistical Analysis
Data were presented as mean ± standard error (SEM) and were processed by SPSS v.20.0 software (IBM, Chicago, USA). Because data followed a normal distribution, comparisons among multiple groups were analyzed by analysis of variance (ANOVA). A P value < 0.05 was considered statistically significant.
Results
As shown in Fig. 1A, the vein grafts were fully filled and beat well after the operation, which means successful transplantation of the jugular vein. The incision was closed after the operation. The incision status and rats’ activity were checked every day after the operation, and one, two, three rats for sham, control, and PTX group died because of incision infection or vein graft hemorrhage. In the control group at day 28, the vein grafts were still fully filled and were slightly stiff. And the grafts were adhesive with surrounding tissues. In comparison, grafts in the PTX group were easy to separate from surrounding tissues (Fig. 1B).
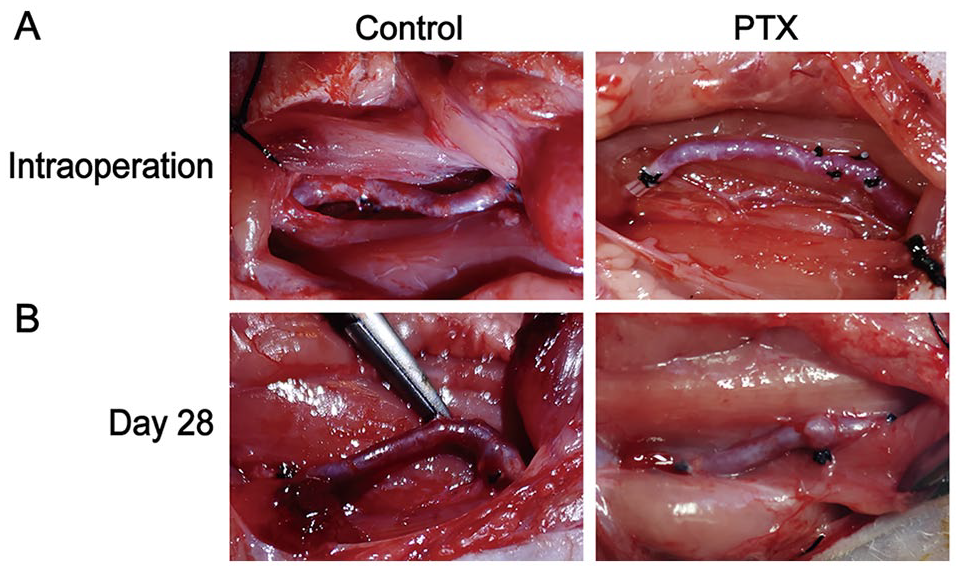
Photographs of vein grafts. (A) Intraoperative photographs for control and PTX groups, grafts were fully filled and beat well. (B) Postoperative photograph at day 28, the vein grafts were still fully filled and were adhesive with surrounding tissues in control group. PTX: pentoxifylline.
PTX Alleviates the Hyperplasia of Graft Intimal and Medial
From H&E staining, we can see that the vein grafts of the control group were obviously thickened (Fig. 2A). Quantification analysis of the intimal and medial thickness was performed by at least three sections for each bypass graft. In view of the graft thickness at day 28, the intimal and medial for the vehicle gel group were not significantly different from the control group (Fig. 2B, Table 1), which means that the application of Pluronic F-127 gel as the vehicle of PTX will not affect the pathophysiology changes of vein grafts.

PTX reduced the intimal and medial thickness of vein grafts. (A) Micrographs of H&E stained sections of vein grafts at 4× (first line), 10× (second line), and 20× (third line) objective lens. (B) The effects of PTX on intimal, medial thickness, and I/M ratio. H&E: hematoxylin and eosin; PTX: pentoxifylline. *P < 0.05, **P < 0.01, ***P < 0.0001.
Thickness of Intimal, Medial, and I/M for Sham, Control, Vehicle Gel, and PTX Groups.

The values of thickness are presented as mean ± SEM. PTX: pentoxifylline.
Compare with control group.
Compare with sham group.
*P < 0.05, **P < 0.01, ***P < 0.0001, #P < .05, ##P < 0.01, ###P < 0.0001.
In specific, the thickness of grafts intimal and thickness of medial for control group were significantly higher than the sham group (intimal: 59.52 ± 5.23 vs 2.35 ± 0.16, P < 0.001; medial: 134.89 ± 15.48 vs 23.60 ± 1.78, P < 0.001). These mean significant hyperplasia of intimal and medial for vein grafts at day 28. For the PTX group, the intimal thickness of vein grafts at day 28 was significantly reduced when compared with the control group (4.17 ± 0.60 vs 59.52 ± 5.23, P < 0.001). Similarly, the medial thickness for the PTX group was significantly decreased when compared with the control group (88.34 ± 10.46 vs 134.89 ± 15.48, P = 0.01). This means that the PTX inhibited the hyperplasia of intimal and medial in vein grafts. The medial of PTX group was still thicker than the sham group (88.34 ± 10.46 vs 23.60 ± 1.78, P < 0.001). The intimal of PTX group was also thicker than the sham group but was not significant (4.17 ± 0.60 vs 2.35 ± 0.16, P = 0.0734). We also measured the value of intimal-to-medial, which showed a significant decrease for the PTX group than the control group (0.05 ± 0.01 vs 0.48 ± 0.06, P < 0.001). Even it was slightly smaller than the sham group (0.05 ± 0.01 vs 0.10 ± 0.01, P = 0.345).
PTX Decreased Cell Proliferation of Vein Grafts Through p38MAPK Pathway
To evaluate the cell proliferation, we measured the PCNA and α-SMA (a VSMCs specific maker) level of vein grafts by immunohistochemical and Western blot. The immunoreactivity to anti-PCNA in vein grafts of control and gel groups were higher than in the sham group. With the local delivery of PTX, the PCNA expression of grafts at day 28 was reduced (Fig. 3A). The Western blot confirmed the efficacy of PTX (Fig. 3B and C), which means a significant inhibition of PTX to the cell proliferation in vein grafts. Also, the immunohistochemical and Western blot results for the control and gel groups showed an increment of α-SMA when compared with the veins in the sham group. And the α-SMA level decreased after the intervention of PTX (Fig. 3A–C), which means that the local delivery of PTX inhibited the proliferation of SMCs in the vein graft.

Effects of PTX on PCNA, α-SMA, p38, and p-p38 level. (A) The immunohistochemical section of PCNA, α-SMA, p38, and p-p38 in vein grafts at day 28 for sham, control, gel, and PTX groups. (B) Expression PCNA, α-SMA, p38, and p-p38 in vein grafts at day 28 with Western Blot detection. (C) Histograms of PCNA, α-SMA, p38, and p-p38 levels in vein grafts at day 28 calculated from Western Blot. PTX: pentoxifylline. *P < 0.05, **P < 0.01, ***P < 0.0001.
To investigate the mechanism underlying the inhibition role of PTX to vein graft restenosis, we measured the p38 and p-p38 levels. As shown in Fig. 3A, the p38 levels seemed to be the same between the sham, control, gel, and PTX groups. This tendency was also observed in Western blot results (Fig. 3A and C). While, the p-p38 level of grafts was obviously higher for the control and gel groups than the veins for the sham group. The p-p38 level of grafts for the PTX group was higher than the sham group, but more notably was that the p-p38 level for the PTX group was significantly decreased when compared with the sham group (Fig. 3A–C). These results indicated that PTX may inhibit the restenosis of vein graft through decrease the phosphorylation of p38.
Discussion
Restenosis, or failure of vein grafts, challenges patient’s health and medical advances in the world 1 . Many researchers tried to better understand the underlying mechanisms of the pathological changes and to identify new approaches to avoid them18,19. Our research here showed that local delivery of PTX to the vein grafts significantly reduced intimal and in medial thickness at day 28 after the operation. PCNA and α-SMA results indicate an inhibition role of PTX in cell proliferation. Moreover, the PTX also reduced the p-p38 level in vein grafts.
The remodel of grafted vein including the intimal, medial, and adventitia results in restenosis of the vein grafts and failure in long term, but the mechanism is still puzzling. Previous studies have shown that the process of restenosis concerns thrombosis, neointimal formation, intimal, and medial hyperplasia in early stage and later atherosclerosis 4 . After vein transplanted to artery system, the increased pressure, changed stress, and shear force will destroy the endothelia, accompanied with inflammatory cytokines release (such as endothelium-derived growth factors, interleukin 8, and monocyte chemotactic protein 1), interacts with platelets, and so on, which directs to VSMC proliferation and migration to intimal20–22. In this study, the pathology results also showed the thickening of graft intimal and medial after 28 days of bypass, which is consistent with previous studies15,17.
PTX is one derivative of methylxanthine. Initially, it was used to treat the intermittent claudication. Recent researchers found that it can also inhibit cell proliferation, such as promoting the lysis of cancer cells. Thus, it has been introduced to cancer therapy12,13,23. With the delivery of PTX locally to the vein grafts here, we found that both the intimal and medial thickness decreased. These results indicate the role of PTX in the inhibition of vein graft restenosis. Through measurements of PCNA and α-SMA, we further confirmed that the PTX attenuates the restenosis of vein grafts by inhibiting VSMCs proliferation. Moreover, we found here that the application of PTX also inhibited the activation of the p38MAPK pathway, which is consistent with Gregory’s work on astrocytes that PTX induced the p38MAKP phosphorylation 13 . These introduce that the PTX attenuates vein graft restenosis by inhibiting VSMCs proliferation via the p38MAPK pathway. p38MAPK signaling pathway is induced by stress stimuli and proinflammatory cytokines, and TNF-α is kind of a activator of p3813, 24 . Also, previous studies have shown a positive relationship between TNF-α and vein graft restenosis16,25. Thus, PTX may reduce the TNF-α expression to suppress the p38MAPK pathway for attenuation of restenosis.
Pluronic F127 is a non-ionic triblock copolymer of poly (ethylene oxide)-b-poly (propylene oxide)-b-poly (ethylene oxide) (PEO-PPO-PEO) with very good biocompatibility26,27. And it is a thermo-responsive polymer, which can form the hydrogel in-situ at the body temperature. These properties make it widely used for drug delivery to treat diseases. For example, Wang et al.28,29 loaded the traditional Chinese drug Cortex moutan with Pluronic F127 to fight against atopic dermatitis. As a drug delivery system, it showed a better release of docetaxel than free docetaxel in the cancer treatment 30 . Also, our previous studies have shown that the Pluronic F127 can be used to locally deliver drugs for anti-vein graft restenosis15,31. This research proved its role again.
The pharmacokinetic properties of PTX have been extensively researched. In both animals and humans, it can be rapidly absorbed from the stomach and intestine32,33. In dogs, the maximal plasma concentration can be obtained within 1 h after oral dosing. Humans have also experienced similar findings34,35. In our investigation, the PTX was loaded with Pluronic F127 gel and administered locally to the periphery of grafting. We believe that this system will have no effect on PTX release or diffusion into vein graft tissue even in intimal layer.
Conclusion
In the present study, we found that local delivery of PTX can attenuate vein grafts restenosis in the rat carotid bypass model. This effect appears to be mediated by decrease in VSMCs proliferation through inhibiting the p38MAPK pathway activation. Thus, PTX is a promising candidate for the prevention of restenosis and failure of vein grafts.
Footnotes
Author Contributions
Yangming-Fan and Jianjun-Ge designed the study, Yangming-Fan accomplished the experiments and data analysis, and wrote the paper. Jianjun-Ge reviewed the manuscript. All authors have read and approved the final manuscript.
Ethical Approval
This study was approved by the Animal Care and Use Committee of First Affiliated Hospital to University of Science and Technology of China (number 2020—N [A]—208).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Animal Care and Use Committee of First Affiliated Hospital to University of Science and Technology of China (number 2020—N [A]—208) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Natural Science Foundation of China (grant numbers NSFC 81470530 to J.G.) and the Major Science and Technology Projects of Anhui (18030801132 to J.G.).
