Abstract
Keywords
Introduction
Non-neoplastic B-cell predominant lymphoid proliferations (NBPLP) constitute a heterogeneous group of disorders that lack the sufficient morphological and molecular hallmarks for a definitive diagnosis of B-cell lymphoma. 1 It is a reactive phenomenon related to infections, medications, allergens, neoplasms, and more. It may harbor aberrant immunohistochemical patterns such as the absence of antigens normally expressed on B or T cells or the presence of immunoglobulin gene rearrangement. 1 It can be seen in the lower female genital tract, terminal ileum, skin, tonsillar etc.2–4
NBPLP can present atypical pathologic characteristics and often render a diagnosis of atypical lymphoid hyperplasia. Atypical lymphoid hyperplasia (AtLP) is a condition in which it is impossible to differentiate the benign and malignant nature of a lymphoid infiltrate. 1 AtLP has morphological features that mimicked malignant lymphoma and can be confusing, easily leading to a wrong judgment of malignant lymphoma when the IGH rearrangement is also positive. Recent literature has evaluated lymphoproliferative lesions in the orbit and gastrointestinal tract using resume scoring systems and stepwise evaluation methods. This has yielded some relatively practical differential diagnostic methods for reactive lesions, atypical lesions, and lymphoma.5,6 However, these methods have limitations in terms of location. Therefore, we aimed to retrospectively review cases of NBPLP with atypical presentation to better understand the specific patterns of this disease and to avoid a diagnostic pitfall in lymphoma.
Materials and methods
Case selection
A search of the anatomic pathology database at the Department of Pathology, Ruijin Hospital, Shanghai Jiaotong University School of Medicine, was conducted for the period from 2016 to 2021.18 patients diagnosed with AtLP were identified. Histological and immunohistochemical diagnoses were independently reviewed by two hematopathologists (Hong-Mei Yi and Chao-Fu Wang)based on the 4th edition of the World Health Organization Classification for Lymphoid Neoplasms .7–9 Patients with immunodeficiency disorder or a history of lymphoma were excluded from this study. Medical histories were collected from the electronic medical record system or through telephone consultations, and informed consent was obtained from each patient.
Immunohistochemistry (IHC) and Epstein-Barr Virus Encoded RNA (EBER) in situ hybridization analysis
All biopsies had IHC analysis that included a combination of CD20, CD79α, CD3, CD5, Bcl-6, CD10, Bcl-2, MUM-1, CD21, CD23, C-MYC, Cyclin D1, and Ki67.10% formalin-fixed, paraffin-embedded sections of 4 μm were deparaffinized in xylene and hydrated in a series of graded alcohols. An automatic immunohistochemical staining machine (Dako Omnis) and commercially available antibodies (all obtained from DAKO, Glostrup, Denmark, prediluted) were performed on the paraffin section according to the instruction manual. Moreover, Epstein-Barr Virus in situ Hybridization kits (Dako, Carpinteria, CA, US) were utilized to detect Epstein-Barr Virus Encoded RNA (EBER) in the paraffin sections.
B-cell receptor and T-cell receptor (BCR/TCR) rearrangement analysis
83.3% (15/18) and 22.2% (4/18) patients underwent B-cell receptor (BCR) or T-cell receptor (TCR) gene rearrangement detection respectively, utilizing the PCR technique. B cell clonality was detected by analyzing the immunoglobulin heavy chain (IGH) and Kappa light chain (IGK) genes, fluorescently labeled primers from InVivoScribe Technologies were utilized to conduct PCR amplification. These primers target both the conserved V-framework (FR1, FR2, or FR3) region and the conserved J-region of the IGH gene, following the BIOMED-2 concerted action protocol. 10 T cell clonality was evaluated by testing for TCRβ (TCRB), TCRδ (TCRD), and TCRγ (TCRG). A Gene Clonality Assay Kit (InVivoScribe Technologies, San Diego, CA) was applied to the extracted DNA samples and visualized by capillary electrophoresis on an ABI 3500 Genetic Analyzer with subsequent analysis using GeneMapper v5.0 software. The peaks were classified as clonal peaks upon the detection of one or more distinct peaks within the expected size range. Peaks exceeding a threshold height of three times the amplitude of the third highest peak were designated as positive. Results were considered valid if PCR products of the control gene were observed at 400 base pairs.
Results
Clinical characteristics
Clinical features of 18 patients with NBPLP.

NA: Not Available, ANED: alive with no evidence of disease.
Histopathology, immunophenotype, and molecular features
All patients exhibited benign presentations, with no lymphomas diagnosed. We devised a histopathological classification system based on proliferative patterns and cellular composition shown in Figure 1: • Type A: Characterized by predominantly follicular proliferation with plasma cell infiltration. • Type B: Marked by diffuse proliferation of large cells accompanied by plasma cell infiltration. • Type C: Defined by diffuse proliferation of small cells, lymphoepithelial lesions, and plasma cell infiltration. • Type D: Consists of diffuse proliferation of heterogeneous cell populations, including large cells, small lymphoid cells, plasma cells, and eosinophils. Four histologic types.

Among the observed pathological changes, Type A (6/11) was most frequently identified in gastrointestinal samples, followed by Types B (3/11) and C (2/11). In pharynx and lingual samples, we detected changes consistent with Types A and C. Notably, unique to our findings, only Type D pathological changes were observed in the bladder, external urethral orifices, and cervix.
The immunophenotype in type A is characterized by CD21 staining revealing an enlarged follicular dendritic mesh. The mantle zone staining for IgD appears relatively more intact, without aberrant expression of specific markers (Figure 2). In type B, the majority of large cells are immunoblasts exhibiting variable levels of CD30 expression. 2/3 of type B samples tested positive for EBER. One case each from a rectal sample and a pharynx sample presented as type C. Type C exhibited CD20+ small B lymphocytes with varying degrees of small lymphocyte, plasma cell, and eosinophil infiltration. Type D predominantly showed B lymphocyte or T lymphocyte proliferation, including mature forms such as immunoblasts, small lymphocytes, and plasma cells. The enlarged B or T lymphocytes in type C expressed CD30. A cervical sample case ((1/3) demonstrated a small focal area of EBER positivity (Figure 3). Case 5 (a). There are many irregular hyperplasia follicular structures under the rectal mucosa. (b). Visible pale and dark zones and starry phenomena in the germinal center. (c). Centroblasts, dendritic cells, small lymphocytes, and tingible body macrophages can be seen in the germinal center. (d). Immunostaining with CD20 highlights the irregular hyperplasia of the follicular structures. (e). Staining with CD3 reveals the presence of numerous T lymphocytes in the interfollicular regions. (f). The germinal center express CD10, (g). but does not express BCL-2, (h). Additionally, the proliferation marker Ki-67 is highly expressed in the follicular areas but low in the interfollicular areas. Staining with CD21 outlines a complete follicular dendritic cell network (image not shown). (i). Molecular analysis indicates a positive IGH clonal rearrangement in this case. Case 17 (a). Dense lymphoid cell infiltration is visible under the cervical mucosa. (b). The infiltrate consists of a mixture of large cells, small lymphocytes, plasma cells, neutrophils, and eosinophils, with accompanying interstitial vascular proliferation. (c). Large cells have hyperchromatic and irregular nuclei with 1-3 nucleoli, the mitotic figures are not difficult to find. (d). Immunostaining with CD20 indicates that the large cells are predominantly B lymphocytes. (e). Staining with CD3 reveals a significant number of T lymphocytes present as well. (f). Some large cells expressed CD30 in the focal region. (g). Additionally, the proliferation marker Ki-67 is highly expressed in this area. (h). Scattered EBER positive cells are observed. (i). Molecular analysis confirms a positive IGH clonal rearrangement in this case.

Pathological features of 18 patients with NBPLP.

AtLP: Atypical lymphoid hyperplasia; DLBCL: Diffuse large B cell lymphoma; MALT: Extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue.
Normal: No abnormal immunophenotype seen in the hair growth centre and interfollicular area.
Non-neoplastic B-cell predominant lymphoid proliferations (NBPLP):histological subtypes and differential diagnosis.
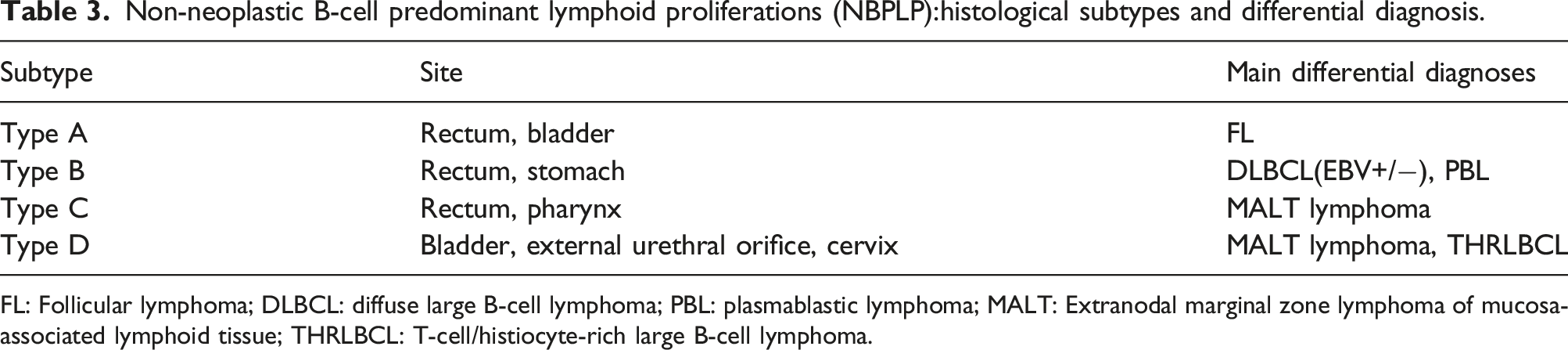
FL: Follicular lymphoma; DLBCL: diffuse large B-cell lymphoma; PBL: plasmablastic lymphoma; MALT: Extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue; THRLBCL: T-cell/histiocyte-rich large B-cell lymphoma.
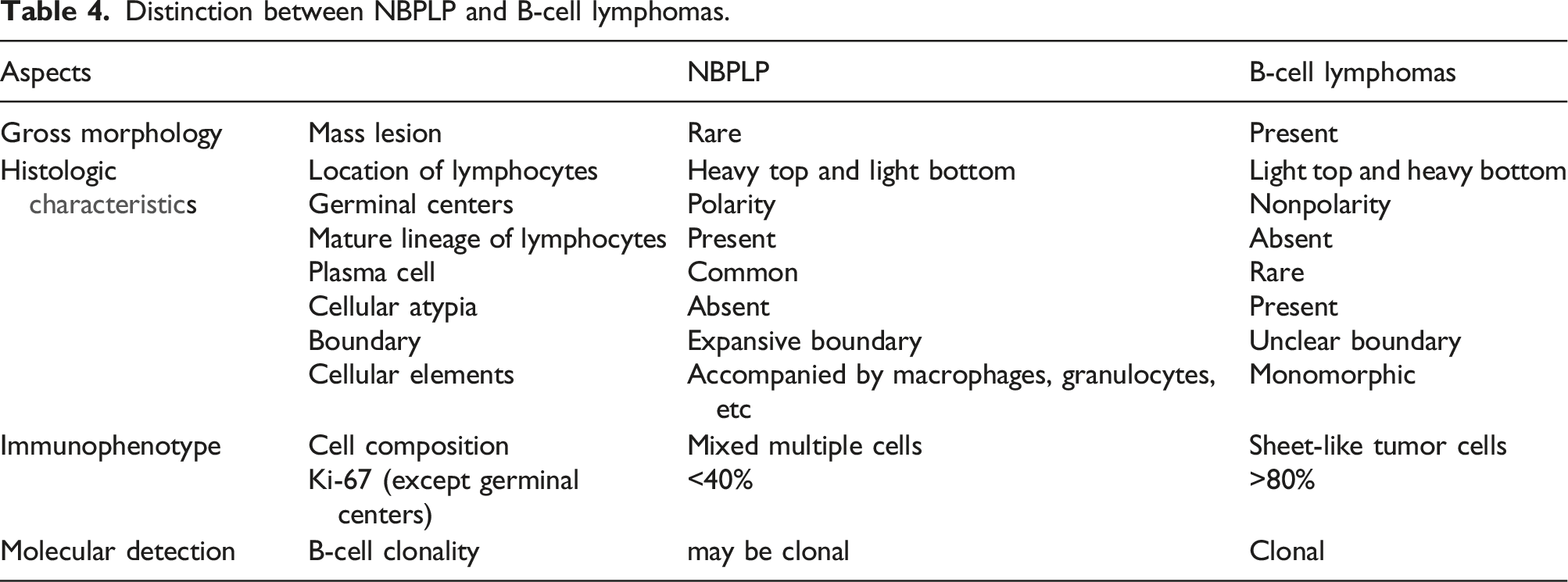
Distinction between NBPLP and B-cell lymphomas.
Discussion
Endoscopic biopsies are commonly obtained from hollow organs exposed to the external environment in clinical routine due to the benefit of minimally invasiveness. Mucosa-associated lymphoid tissue hyperplasia with atypical morphology with or without clonal IGH gene rearrangement often mimics lymphoma. This phenomenon often occurs in the aforementioned type of biopsies based on our experience. Theoretically, the mucosa of the hollow organs exposed to the external environment as the primary barrier easily experiences infections and inflammation that can give rise to an alteration in the morphology at a high frequency. Therefore, the understanding of the differential criteria between the atypical presentation and lymphoma plays a significant role in avoiding overdiagnosis and overtreatment.
A comprehensive evaluation of the clinical history, microscopic morphology, immunophenotype, and molecular analysis assist in distinguishing the malignant or benign behavior of the atypical presentation. In the study cases presented, the Atypical Lymphoid Proliferation (AtLP) was determined to be benign based on a long follow-up period that showed no signs of malignancy. Endoscopically, the AtLP of the rectum was commonly present as polyps, ulcers, or submucosa bulge. Occasionally, it may present as a mass, which can be mistaken for a malignant growth. In contrast, the lymphoma of the gastrointestinal tract commonly presents as an infiltrative tumor. Partial of it presents as nodular, multiple polyps, or ulcer.11,12 In non-gastrointestinal cases, AtLP lesions are usually small and exhibit limited growth, which can aid in differentiation from lymphoma, which is characterized by destructive growth.
Good et al 13 discussed six key points for distinguishing between atypical lymphocyte proliferation and lymphoma, considering morphological features, immunophenotypes, and molecular correlations. We classified the morphology into types A, B, C, and D based on the growth pattern of lymphoid tissue and the composition of infiltrating cells to aid in differential diagnosis. Type A growth resembles florid follicular hyperplasia and mainly present in the intestine, requiring differentiation with follicular lymphoma (FL). 14 Noticeably, AtLP is commonly presented as florid follicular hyperplasia which is limited to the mucosa or submucosa layer, with the presence of mantle zone and a starry-sky pattern. In contrast, typical FL often lacks a mantle zone and starry-sky pattern, showing back-to-back, fused follicles expressing CD10, BCL6, and BCL2. Additionally, 90% of the FL are positive for IGH rearrangement. 15 These points can be useful for differential diagnosis. However, Case 5 in our studies presented exuberant hyperplasia of lymph follicles without immunophenotypic abnormality but positive IGH rearrangement and was finally diagnosed as AtLP (Figure 2). A study that determined 5 cases with reactive lymphoid hyperplasia with positive IGH rearrangement were finally diagnosed as normal in follow-up biopsy. 2 This suggested that positive IGH rearrangement does not necessarily render a positive diagnosis of lymphoma.
Type B is primarily characterized by the proliferation of large cells. 2/3 of the presented type B cases were EBER positive, with varying levels of CD30 expression among the large cells. Therefore, it required differential with EBV + diffuse large B-cell lymphoma (DLBCL) and plasmablastic lymphoma (PBL). For EBV + DLBCL, it usually manifests as sheets of immunoblast-like cells, infiltration of Reed-Sternberg-like cells, increased reactive lymphocytes, plasma cells, and tissue infiltration as the background. The immunophenotype of EBV + DLBCL includes MUM-1+, BCL6+, CD30+, CD10-and CD15-. The IG gene rearrangement is often positive. In contrast, CD21 staining for our type B cases showed the presence of follicular dendritic mesh, but negative IG gene rearrangement does not support the diagnosis of EBV + DLBCL. PBL usually occurs in immunosuppressive individuals, presenting as atypical diffuse infiltration growth of immunoblasts or plasmoblasts. The immunophenotype of PBL is usually CD138+, CD38+, or CD20-, which serves as a strong differential key. 16
Type C growth patterns are primarily characterized by the proliferation of small cells, which can be challenging to distinguish from small B-cell lymphomas, particularly Mucosa-Associated Lymphoid Tissue (MALT) lymphomas that occur in the gastrointestinal tract. In the past, two cases of Type C (one in the colon, one in the pharynx) were misdiagnosed as MALT lymphoma before molecular results were available. The occurrence of MALT lymphoma is usually related to C. jejuni infection or immunoproliferative small intestinal disease (IPSID). Chronic inflammation and infection can lead to the proliferation of monoclonal B cells, 17 presenting as destructive growth of small B cells in the marginal zone, accompanied by lymphoepithelial lesions. The immunophenotype of MALT lymphoma includes the expression of B cell markers, CD21 which reveals an expanded and distorted follicular dendritic meshwork. Kappa and lambda show the restrictive expression of the light chain. Fluorescent in situ hybridization (FISH) for t (14;18)/BCL2 gene rearrangement is useful for diagnosing MALT lymphoma. For IGH gene rearrangement in MALT lymphoma, partial reactive biopsy samples also can be tested positive. This might be due to the temporary pseudoclonality of B cells under a generalized proinflammatory state. Our cases demonstrated follicular structure and increased plasma cells in the lesions, suggesting the existence of polyclonal. No distinctive immunophenotypes were observed, and negative IGH gene rearrangements indicated a lack of evidence for diagnosing MALT lymphoma. A 22-months follow-up also showed no signs of lymphoma in the patients. Some studies have suggested that IRTA1 might be a specific marker for marginal zone lymphoma. 18 Immunophenotyping for IRTA1 could assist in more accurate diagnosis. Additionally, gastrointestinal lymphoproliferation with persistent clinical symptoms or submucosal tumors seen on gastroscopy requires long-term follow-up to detect neoplastic transformation at an early stage. One study suggested that localized lymphoid hyperplasia of the rectum might progress to ulcerative colitis and MALT lymphoma during a median follow-up of approximately 3 years. 19 Further research with a longer follow-up period and larger sample size is needed to improve the diagnostic accuracy of lymphomas.
Type D growth patterns are typically associated with the urinary tract and the mucosa of the cervix. This category includes three cervical specimens attributed to HPV infection, which did not exhibit any noticeable mass formation upon macroscopic examination. Both the macroscopic and microscopic characteristics of these cases were found to be consistent. Microscopically, many large lymphoid (immunoblast/centroblast-like) cells were distributed in small patches. The stroma was mixed with some small lymphocytes, plasma cells, and eosinophils. All the cases tested demonstrated positivity for IGH gene rearrangement, which could potentially lead to misdiagnosis as MALT lymphoma or T-cell/histiocyte-rich large B-cell lymphoma. Microscopically, lymphoma is often characterized by deep tissue infiltration. In contrast, reactive changes usually present as localized shallow mucosa changes and more T lymphocytes as the background.
Expression levels of CD30 vary among the large cells, indicating the presence of activated immunoblasts characteristic of reactive changes. It also accompanies more mature plasma cells. In tissue biopsies, “pseudoclonality” of the IGH gene rearrangement is frequently observed. Studies have shown that repeated PCR testing and the use of high-resolution GeneScan techniques can reduce the impact of pseudoclonality, leading to more precise diagnostic outcomes. Compared with lymphoma, lymphoma-like lesion (LLL) usually occurs in women during the reproductive period. Microscopic examination reveals that LLL involving the superficial layers and the mixed existence of multiple cells (B cells, T cells, and polyclonal plasma cells) can be seen. It rarely has a presentation of B-cell predominance. IgH clonal rearrangement can be positive in in cases of LLL.3,20 However, a positive molecular result does not necessarily lead to the diagnosis of a malignant tumor. A comprehensive evaluation that includes the location of the lesion and clinical history is necessary for accurate diagnosis.
The following features may indicate non-lymphoma lesions and assist in distinguishing the malignancy of a proliferative extra lymphatic tissue: (1) The size of the lesion is relatively limited, it can expand but is not invasive; (2) Generally, the presence of polymorphous large cells, medium-sized cells, small lymphocytes, and plasma cells mixed infiltrate, suggesting a benign pathological process. (3) Proliferative and polar germinal centers with active apoptosis are usually present, and there is no abnormal immunophenotype. CD38 can demonstrate the presence of plasma cells within the germinal center. And scattered CD30-positive activated large cells may also be observed. The ki-67 index can be very high and may be accompanied by a small number of large cells infected with the EB virus. Clonal IGH or TCR rearrangements can also occur in benign lesions.2,3
For EBV + cases, the mucosal tissue exposed to the external environment is easily stimulated by external antigens. This antigenic stimulation can lead to the proliferation of B or T cells. These cases typically consist of a heterogeneous mixture of cell types, including small lymphocytes, activated blast cells, and plasma cells, as observed microscopically. The patients have no clinical history of autoimmune diseases, immunodeficiency, or associated therapies. Among the 4 cases complicated by EBV infection, LLL occurred in the rectum (type B), stomach (type B), pharynx (type C), and cervix (type D), respectively. It is composed predominantly of B cells, the positive rate of EBV + cells varying from 10%–70%. EBV infection may lead to immunoblastic proliferation or structural destruction. Multiple factors, such as immune status, age, race, etc., determine the clinical course.
The pathogenesis of EBV-associated lymphoproliferative diseases is related to the lysis cycle of EBV infection, the immune response, and the release of inflammatory factors. 21 The architecture of EBV-associated reactive lymphoid proliferations is usually preserved, whereas lymphoma typically shows destructive and diffuse cell positivity.20,22 Additionally, clonal B-cell lymphocyte aggregation has been observed in patients with autoimmune or infectious diseases. 23 It is also important to identify specific lymphoproliferative diseases associated with EBV, such as Epstein-Barr virus (EBV)-positive mucocutaneous ulcer (EBVMCU) and EBV + diffuse large B-cell lymphoma (DLBCL). Over 90% of EBVMCUs are associated with the use of immunosuppressants. The morphology of EBVMCU includes ulcer formation and the infiltration of plasma cells, lymphocytes, and eosinophils, along with a significant number of large EBER + transformed cells. EBV + DLBCL typically occurs in older individuals and is associated with immunosenescence. The diagnostic criteria included >80% of the EBER + tumor cells. When atypical cells express EBER, it is necessary to consider the clinical history and microscopic morphology to differentiate between these two diseases. 24 Nevertheless, due to the small sample size and the fact that most specimens were small biopsies, the components available for evaluation were limited. Therefore, more clinical data and case collections are needed in the future to further explore the prognostic value of such diseases.
Conclusions
Organs exposed to the external environment are often stimulated by various antigens. This can lead to florid reactive lymphoid hyperplasia and positive gene rearrangement, which may result in the misdiagnosis of malignant disease. To improve differential diagnosis, this study classified non-specific borderline proliferative lesions (NBPLP) into four subtypes based on morphology. We combined clinical, pathological, molecular, and follow-up findings to draw more definitive conclusions.
Positive IHG gene rearrangement is commonly associated with the diagnosis of small B-cell lymphomas, such as MALT lymphoma, However, it can also indicate a non-neoplastic lesion. Morphological features, such as swelling boundaries, follicle colonization, the presence of plasma cells, and heterotypic lymphocytes, as well as the relationship with surrounding glands, assist in differential diagnosis. Additionally, abnormal immunophenotypic expression in the follicular region—for example, BCL-2 in the germinal center, CD5, or CyclinD1 in the nested region—and the integrity of the follicular dendritic network (stained with CD21 and CD23) provide valuable clues for differentiation. In summary, a comprehensive evaluation of clinical history, histopathology immunophenotype, and molecular alterations is essential for a definitive diagnosis.
Supplemental Material
Supplemental Material - Non-neoplastic B-cell predominant lymphoid proliferations at the organs exposed to external environment mimicking lymphoma: A potential diagnostic pitfall
Supplemental Material for Non-neoplastic B-cell predominant lymphoid proliferations at the organs exposed to external environment mimicking lymphoma: A potential diagnostic pitfall by Yue Fan, Benjamin Ka Seng Thong, Ouyang Binshen, Xia Shen, Hongmei Yi, and Chaofu Wang in International Journal of Immunopathology and Pharmacology.
Footnotes
Author contributions
YF and BJ performed the histological examination of these diseases and was a major contributor in writing the manuscript. OY performed histological examination of these disease and immunohistochemical stainging. BM was responsible for picture collection and history collection. XS was carried out the molecular assays and interpretation of the results. HY constructed the idea of the article and revised the article. CW participated in the design of the study and provides financial support. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by grants from the Science and Technology Commission of Shanghai Municipality (21ZR1440100).
Ethical statement
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Data availability statement
The original contributions presented in this study are included in the article/supplementary material, further inquiries can be directed to the corresponding authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
