Abstract
Spinal cord injury (SCI) causes a dysfunction of sympathetic nervous system innervation that affects the immune system, leading to immunosuppression syndrome (ISS) and contributing to patient degeneration and increased risk of several infections. A possible therapeutic strategy that could avoid further patient deterioration is the supplementation with Vitamin E or trace elements, such as Zinc, Selenium, and Copper, which individually promotes T-cell differentiation and proliferative responses. For this reason, the aim of the present study was to evaluate whether Vitamin E, Zinc, Selenium, and Copper supplementation preserves the number of T-lymphocytes and improves their proliferative function after traumatic SCI. Sprague–Dawley female rats were subjected to moderate SCI and then randomly allocated into three groups: (1) SCI + supplements; (2) SCI + vehicle (olive oil and phosphate-buffered saline); and (3) sham-operated rats. In all rats, the intervention was initiated 15 min after SCI and then administered daily until the end of study. Locomotor recovery was assessed at 7 and 15 days after SCI. At 15 days after supplementation, the quantification of the number of T-cells and its proliferation function were examined. Our results showed that the SCI + supplements group presented a significant improvement in motor recovery at 7 and 15 days after SCI. In addition, this group showed a better T-cell number and proliferation rate than that observed in the group with SCI + vehicle. Our findings suggest that Vitamin E, Zinc, Selenium, and Copper supplementation could be part of a therapy for patients suffering from acute SCI, helping to preserve T-cell function, avoiding complications, and promoting a better motor recovery. All procedures were approved by the Animal Bioethics and Welfare Committee (Approval No. 201870; CSNBTBIBAJ 090812960).
Keywords
Introduction
Traumatic spinal cord injury (SCI) is defined as a pathological process that results in temporary or permanent motor, sensitive, and/or autonomic dysfunction 1 .
After SCI, several important functions are affected such that homeostasis is lost. Impaired immune system function is among the altered functions, as there is a loss or dysfunction of the innervation of lymphatic and endocrine tissues after SCI. This alteration continues to change over time from the acute to the chronic stages, originating an immunosuppression syndrome (ISS), which is clinically relevant as SCI patients have a high susceptibility to several infections2–7.
After injury, there is a disruption in the innervation of the sympathetic nervous system (SNS) and hypothalamic-pituitary-adrenal (HPA) axis, thus leading to an excessive response to sensory stimuli in the autonomic nervous system (ANS)8–12, and an important atrophy of secondary lymphatic organs10,13.
The disconnection in the descending sympathetic modulation through visceral and somatic sympathetic preganglionic neurons (SPNs) leads to an uncontrolled release of norepinephrine (NE) and glucocorticoids (GCs) from post-ganglionic nerves and the adrenal gland 14 . This unregulated stimulation of GC or β-2 adrenergic receptors produces immunosuppression10,12.
Most immune cells, such as B- and T-lymphocytes, express adrenergic and GC receptors that are overstimulated after SCI, thus promoting a decrease in their activation and, finally, apoptosis of these cells. The latter could occur to such an extent that it can lead to leukopenia, thus perpetuating long-term immunosuppression10,11,15,16.
Despite these observations, the conventional treatment for SCI continues to be methylprednisolone (MP), an immunosuppressive steroid that could exacerbate ISS and impair the beneficial effects of T-cells in SCI recovery3,17–21. Several studies published in the last decades have overwhelmingly reported the ineffectiveness and potential adverse effects of MP17,22,23, a strategy that, compared with non-steroidal drugs, does not contribute to a better neurological recovery. In fact, MP increases the risk of adverse events in patients with SCI 24 .
Currently, there is extensive research studying other therapeutic strategies that could prevent further deterioration of SCI patients. In this sense, the use of supplements such as vitamins or trace elements could be an interesting therapeutic option, especially to improve the immune system function. It has been reported that supplementation with Vitamin E, a potent antioxidant, and trace elements, such as Zinc (Zn), Selenium (Se), and Copper (Cu), improves the immune response. For example, Vitamin E increases lymphocyte proliferation in response to mitogens 25 ; Zn enhances phagocytic activity and antigen presentation to T-lymphocytes26,27; Se and Cu increase T-cell differentiation and proliferative responses, superoxide dismutase, and IL-2 levels28–32. In view of these beneficial effects, it is relevant to evaluate the effect of such supplements to improve the immune system function after SCI. In the present investigation, we aimed to evaluate whether Vitamin E, Zn, Se, and Cu supplementation preserves the number and proliferative function of T-lymphocytes in an experimental model of acute SCI.
Materials and Methods
Animals
Adult female Sprague–Dawley rats (14–15 weeks old, 230–260 g) were supplied by the Animal Breeding Center of the faculty of health sciences of the Universidad Anáhuac México. Efforts were made to minimize the number of animals used and their suffering.
Ethics Statement
All procedures were in accordance with the National Institutes of Health (US) Guide for the Care and Use of Laboratory Animals and the Mexican Official Norm on Principles of Laboratory Animal Care (NOM 062-ZOO-1999). All experiments were designed and reported according to the ARRIVE guidelines. To perform euthanasia, animals were previously anesthetized by an intramuscular injection of a mixture of ketamine (50 mg/kg) and xylazine (10 mg/kg).
Study Design
To evaluate the effect of Vitamin E, Zn, Se, and Cu supplementation on the number of T-cells and their proliferative function, 21 animals were allocated into three groups (n = 7 per group; rats were randomly assigned to each group by using the GraphPad QuickCalcs: http://www.graphpad.com/quickcalcs/): (1) SCI + supplements; (2) SCI + vehicle [olive oil and phosphate-buffered saline (PBS)]; and (3) sham-operated rats. The sample size for this experiment was calculated using an α of 0.05 and β of 0.20. Fifteen minutes after SCI (the frame time to finish surgical procedures and prepare supplementation), group 1 received the supplements and, groups 2 and 3, the vehicle intraperitoneally. To verify the homogeneity of injury among the animals, locomotion recovery was evaluated at 7 and 15 days after SCI. At the end of the study (15 days after SCI), the animals were euthanized, and the cervical lymph nodes were removed for quantification of the number of T-cells and its proliferative function. Three independent experiments with the above-mentioned characteristics (the same number of animals, follow-up, and evaluations) were performed. As the variability among the three experiments was quite low (variance values below 0.09), we randomly choose one of them to report the results. All rats were placed on a 12–12 light–dark cycle. Food and water were provided ad libitum during the study.
Spinal Cord Injury
Rats were anesthetized by intramuscular injection of ketamine (80 mg/kg; PISA Laboratories, Mexico City, Mexico) and xylazine (12.5 mg/kg; Bayer Laboratories, Mexico City, Mexico). The spinal cord was exposed 1 h after induction of anesthesia, and the animals underwent moderate SCI at T9 level by direct contusion of the exposed spinal cord using the IH-0400 impactor at 200 Kdyn. This device has been shown to inflict a well-calibrated contusive lesion to the spinal cord33,34. After injury, the aponeurotic plane and the skin were sutured separately.
Animal Care
Animals were age- and weight-matched in each experiment and housed in pairs in a light- and temperature-controlled room. To minimize stress, animals were handled daily at least once a day for the 7-day period prior to the surgical procedure. Sterile bedding and filtered water were replaced daily. Bladder expression was manually assisted twice a day until automatic voiding was regained. During the first week after contusion, the animals received enrofloxacin (Marvel, Mexico City, Mexico) in drinking water at a dose of approximately 64 mg/kg/d. All rats were carefully monitored for evidence of postoperative complications. Animals with signs of infection were excluded from the study.
Supplementation
After the SCI, animals in Group 1 (SCI plus supplements) received a daily dose (until euthanasia was performed) of the supplements intraperitoneally: Vitamin E (Alpha Tocopherol Acetate) 50 mg/kg diluted in 3.4 mL of olive oil35–37 and Zn 3 mg/kg 38 , Cu 6.5 mg/kg 32 and Se 0.2 mg/kg39,40 diluted in 1 ml of PBS. In the case of groups 2 and 3, the rats received only the vehicle (PBS and olive oil per separate).
Assessment of Functional Recovery
Motor functional recovery was assessed using the Basso, Beattie, and Bresnahan (BBB) open-field test of locomotor ability 41 . The recovery was scored on the BBB Locomotor Rating Scale of 0 (complete paralysis) to 21 (complete mobility). Animals were examined weekly for a period of 2 weeks by observers blinded to the treatment received by each rat.
Active Immunization for T-Cell Proliferation Assay and Cell Count
After the SCI, animals were immunized subcutaneously in the cervical area with 150 mg of Complete Freund’s Adjuvant (CFA), emulsified (contained 1 mg/ml of Mycobacterium tuberculosis: F5506, Sigma-Aldrich) with 150 µl of PBS. Fifteen days later, the animals were euthanized with pentobarbital sodium (200 mg/kg intraperitoneally) and, under sterile conditions, cells were pooled from the excised cervical lymph nodes after mechanical dissociation. The cells were suspended on LYMPHOPREP (Sigma-Aldrich®), a density gradient medium suitable for the isolation of lymphocytes 42 , on 1:1 concentration, and the samples were centrifuged at 800 × g for 20 min at room temperature (15°C–25°C). The cellular precipitate was suspended on 1 ml of RPMI-1640 medium (GIBCO, New York, USA) supplemented with 10% fetal bovine serum (GIBCO).
Lymphocyte Count
T-cells (cells between 6 and 10 microns) were quantified using a Neubauer counting chamber. The cells were obtained from cervical lymph nodes. After mechanical dissociation and before the proliferation assay, we took 100 µl of Trypan Blue–treated cell suspension, and T-cells were counted. The number of viable cells per group was obtained.
T-Cell Proliferation Assay
For proliferation assessment, cells (300,000 cells per well) from excised cervical lymph nodes were pooled (n = 7 per group). The cells were then placed in quintuplicate on a 96-well microtiter microplate during 72 h in antigen-free medium (5 wells) or concanavalin-A (ConA; 10 mg/ml; Sigma, St. Louis, MO, USA; five wells) at 37°C and 5% CO2 for each studied rat. They were subsequently washed with RPMI 1640 and labeled with 1 mM (5 ml/well with an equal volume of fetal bovine serum) carboxyfluorescein diester amine (CFSE) (Molecular Probes). The CFSE-labeled cells divided and, as a result, the total number of carboxyfluorescein-labeled molecules decreased by half with each mitosis and thus allowed us to measure cell division. The final concentration of CFSE was of 1 µM and 5 µl/well. Cells were incubated for 24 h at 37°C and evaluated by flow cytometry based on the light scattering characteristics (size/granularity), using unstained cells as controls. Mean fluorescence intensity data were obtained from fluorescence histograms to evaluate the fractions of T-cells that have completed certain number of divisions. Ten thousand events were collected for each sample on a FACSCalibur flow cytometer (BD Biosciences, Mountain View, CA, USA) and analyzed using CellQuest Pro software (BD Biosciences). We took into consideration the mean fluorescence intensity to evaluate the fractions of T-cells that have completed certain number of divisions (Software FlowJo®). The stimulation index (SI) was then calculated by dividing the average proliferation percentage in the experimental wells (ConA) by the average proliferation percentage in the controls (free antigen medium).
Statistical Analysis
Data were analyzed with GraphPad Prism 8.0 software and presented as mean and standard deviation (SD). Kruskal–Wallis test with post hoc Mann–Whitney U test or Dunn’s test were used to assess motor recovery and proliferative response, while one-way analysis of variance (ANOVA) and Tukey–Kramer multiple comparisons test were used to determine cell viability.
Results
Beneficial Effect of the Supplement-Therapy on Motor Recovery
To verify the homogeneity of the injury among the animals, motor recovery was evaluated using the BBB scale 7 and 15 days after SCI. Surprisingly, after 7 days, the group of SCI plus supplement therapy presented a significantly better locomotor recovery (1.64 ± 1.10 SD) compared with the group without the supplement (0.21 ± 0.26 SD; P = 0.0006, Mann–Whitney U test; Fig. 1).

Motor recovery at 7 and 15 days after supplementation. Bars represent the mean and SD of seven rats per group. Animals treated with the supplements had a significantly better motor recovery compared with those without supplements. The sham group was used as a control. Day 7: *P = 0.0006; **P = 0.0025; ***P = 0.0013, Kruskal–Wallis followed by Mann–Whitney U test. Day 15: *P = 0.0002; **P < 0.0008, Mann–Whitney U test. This is one representative of three experiments performed.
Fifteen days after SCI, we evaluated motor recovery function to further analyze the effect of supplementation at the end of study. Rats treated with the supplements presented a significant increase in the BBB score (4.68 ± 1.64 SD) compared with the group without supplements (1 ± 0.53 SD). A score of 4 on the BBB scale represents movement of all three joints (hip, knee, and ankle), while a score of 1 represents limited movement of one joint (P = 0.0002, Mann–Whitney U test; Fig. 1).
Preserved Mononuclear Cell Number Induced by Supplementation
Fifteen days after SCI, we evaluated the number of T-cells obtained from lymph nodes. After treating with Trypan Blue, we determined cell viability and assessed the number of T-cells. Animals without supplementation presented a significant lower number of T-cells (1,400,000 ± 113,652 cells; mean ± SD) compared with the group receiving supplementation therapy (2,983,333 ± 555,778 cells; P < 0.0001). Similar numbers (to the ones observed in supplemented animals) of T-cells were obtained from the sham group (3,366,000 ± 383,152; one-way ANOVA and Tukey–Kramer multiple comparisons test; Fig. 2).
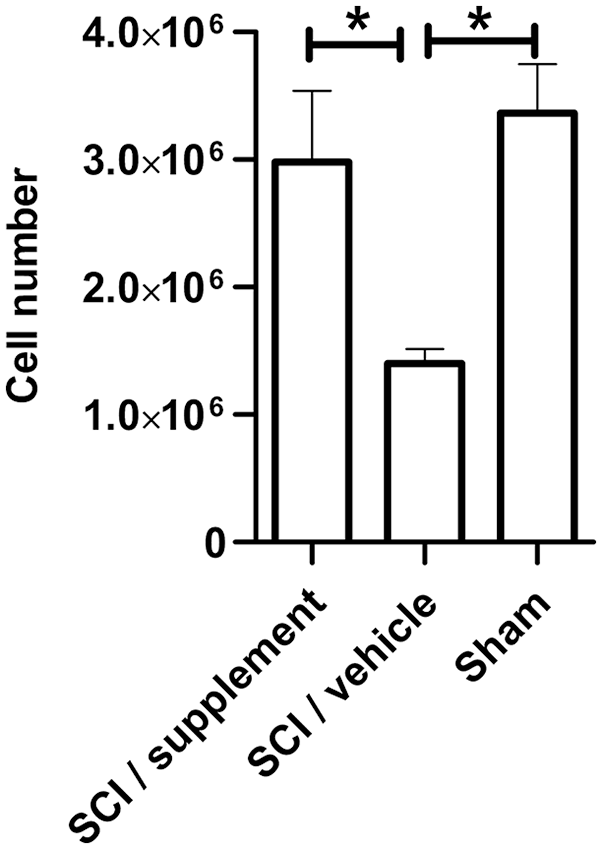
Total T-cell count after supplementation. The bar graph represents the mean and SD in each group. Animals treated with the supplements had a statistically significant increase in T-cell count compared with the non-supplemented group. The sham group was used as control (*P < 0.0001, one-way, ANOVA and Tukey–Kramer multiple comparisons test). This is one representative of three experiments performed. ANOVA: analysis of variance; SCI: spinal cord injury.
Supplementation Preserved the Proliferation of T-Cells
Based on the results mentioned above, we determined the SI of T-cells exposed to Concanavalin A (an unspecific activator of T-lymphocytes) in all groups, using T-cell free antigen as a control. We observed that the SCI plus supplements group significantly improved T-cell proliferation (SI = 2.2) compared with the group without supplements (SI = 0.7) (P < 0.0001, one-way ANOVA and Tukey–Kramer multiple comparisons test). A similar T-cell proliferation rate was observed between the sham group (SI = 2.7) and the supplemented group, with no significant differences (Fig. 3).
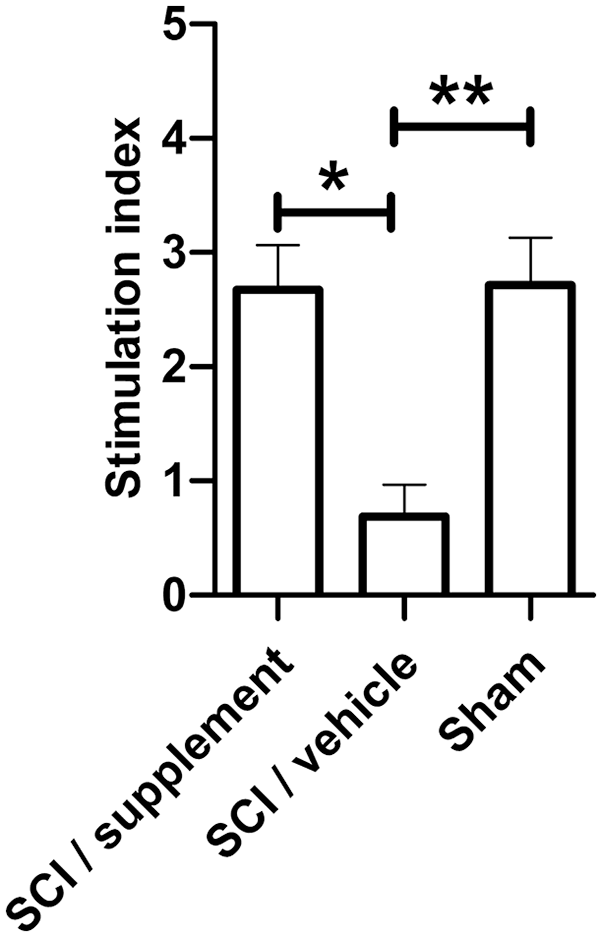
T-cell proliferation after supplementation. The bar graph represents the mean and SD in each group. The SCI plus supplement group had a statistically significant increase in the T-cell stimulation index compared with the non-supplemented group. The sham group was used as a control and there were no differences between the sham and supplementation groups (*P < 0.05; **P < 0.002, one-way ANOVA and Tukey-Kramer multiple comparisons test). This is one representative of three experiments performed. ANOVA: analysis of variance; SCI: spinal cord injury.
Discussion
After SCI, a significant number of patients develop ISS due to noradrenergic and GC overproduction, which decreases the number of immune cells in the blood and impairs T-cell activation, thus generating an immunosuppressive phenomenon. This alteration increases the susceptibility to develop various pathologies that compromise the stability and life of SCI patients16,43,44.
Several studies have elucidated that, in addition to the important role of T-cells in response to any infectious disease, they also participate in processes related to neuroprotection and neuroregeneration after injury18,19,45. Nevertheless, these cells decrease after SCI because of ISS, as reported by Riegger and coworkers 3 . In the first 24 h, T-lymphocytes and other cells such as monocytes and B-lymphocytes decrease dramatically up to 60%, and this suppression continues until day 14. This correlates with the release of adrenocorticotropic hormone (ACTH), GCs, and NE, leading to splenic atrophy, leukocyte apoptosis, and immune depletion with decreased T- and B-cells after SCI3,9,46.
In addition, the administration of MP—a GC approved for the treatment of SCI—amplifies immunosuppression and, at higher doses, induces kidney damage without having any proven benefit in improving motor or sensory function 24 . That is why several studies are still being carried out aimed at finding the best therapeutic strategy. Therefore, our goal in the present study was to determine whether Vitamin E, Zn, Se, and Cu supplementation preserves the number and proliferative function of T-lymphocytes—the main cells in the immune response—after acute SCI. These micronutrients are involved in the protection of immune cells through antioxidant mechanisms 47 .
In addition to their antioxidant effects, these supplements can improve the activation and proliferation of T-cells25,48,49. The proliferative effect that we observed in supplemented animals could be explained by the production of IL-2, which could be induced by the components of the supplement 26 . This cytokine is the main inducer of T-lymphocyte activation and proliferation. Supplementation helped to optimize T-lymphocyte response; this was observed in both the SI and lymphocyte count, which were similar between the supplemented and sham groups. These results suggest that the immunosuppression phenomenon induced by SCI could be circumvented by supplementation with trace elements and Vitamin E. In addition, the use of this therapeutic approach as an adjunctive treatment together with or even instead of MP could be contemplated. In any case, it will be important to first explore this supplementation strategy alone and the synergy when combining it with other therapies, as well as its pharmacokinetic, pharmacodynamic, and safety aspects, among others.
The beneficial effects of the supplementation therapy could be explained by the individual action of each one of the supplements. For example, different studies have reported an increase in lymphocyte proliferation after a supplementation period of 8 weeks with Vitamin E in old rodents. These same authors conducted other studies in which supplementation with Vitamin E using doses of 50 and 200 mg/kg increased the mitogenic responsiveness of T-lymphocytes by increasing the IL-2 production capacity from naïve T-cells, thereby promoting communication between immune cells46,50,51. In the present work, the recovery of T-cell-proliferative function was observed in a shorter time—after 2 weeks of supplementation—compared with previous investigations (8 weeks). This difference in time response could be due to the age-associated deterioration of T-cell-mediated immune response. Our study was performed in young animals, while the previously mentioned studies were done in old ones. Due to the age-related decline in T-cell-mediated immune response, lymphocytes from old rodents might need more stimulation time compared with those from young animals 50 .
On the contrary, Zn supplementation can restore thymulin activity, which is involved in the differentiation, maturation, and enhancement of T-lymphocyte activities, thus increasing the number of T-cells and improving phagocytic activity and antigenic presentation to T-lymphocytes49,52,53. The supplementation with Se, an immunostimulant, can boost cellular immunity, improve T-lymphocyte count 54 , and modify the expression of high-affinity IL2-R30,55,56. Finally, Cu is capable of enhancing the proliferation, differentiation, and activity of T-lymphocytes and improving the production of IL-246–56.
Together, the above observations support the results of our present investigation and suggest that a combined strategy of these supplements could improve T-cell function in SCI patients. This is the first time that this supplementation strategy has been used after acute SCI. The present results reveal that the supplementation strategy was able to maintain the number and functional capacity of T-lymphocytes with values quite similar to those presented by non-injured animals. This is a relevant finding as it implies that this supplementation therapy could prevent infectious complications, one of the main problems in SCI patients.
On the contrary, to verify the homogeneity of the injury among SCI animals, the BBB scale was performed. We noticed that supplemented animals also presented a better motor recovery compared with the non-supplemented ones. This is also an important finding, as it means that the supplementation therapy itself could enhance neuroprotection after SCI in addition to improving T-cell function restoration. This topic should be deeply investigated in future studies. By now, it is important to highlight that the motor recovery observed in supplemented animals could be the result of the antioxidant effect exerted by the supplements. In line with this, it has been reported that Vitamin E is a fat-soluble antioxidant that stops the production of peroxyl radicals and the oxidation of polyunsaturated fatty acids (PUFAs). In addition, Vitamin E improves bladder and motor recovery in SCI models57,58.
Zinc is capable of up- or down-regulating the immune response as, in addition to enhancing the activity of T-lymphocytes, it has anti-inflammatory effects. This alternative function results from the inhibition of the inducible nitric oxide synthase, decreasing the inflammatory effects caused by nitric oxide and, therefore, acute neurodegeneration secondary to neural injury 59 . Zinc has also a direct modulatory effect on oxidative indicators such as glutathione peroxidase and malondialdehyde, thus further promoting antioxidant effects. Finally, research on the topic has reported clinical improvement and significant motor neuron preservation after SCI and Zn therapy 60 . Thus, Zn is considered a predictor of clinical recovery after SCI as its levels are directly proportional to neurological impairment, especially in the first 24 h61,62.
Similarly, and through the action of different enzymes (glutathione peroxidases, thioredoxin reductases, and methionine sulfoxide reductase63,64), Se plays an important role in the homeostasis of the central nervous system 65 . These enzymes participate as anti-inflammatory factors, regulating redox reactions that are essential during the secondary phase of SCI66,67. Furthermore, previous studies have proven that Se supplementation induces faster improvement in bladder control 68 .
Last, Cu is used in various physiological mechanisms as a coenzyme to preserve homeostasis 69 , protecting tissues against lipid peroxidation and oxidative reactions in several degenerative diseases. In a similar way to Vitamin E and Zn, it has been shown that the administration of copper sulfate in rats with SCI induces better neurological recovery, as well as a reduction in malondialdehyde and myeloperoxidase and an increase in superoxide dismutase and glutathione 70 .
The hypotheses of the neuroprotective effect induced by antioxidants administered immediately after injury may be supported by previous studies in which antioxidant therapies combined with other drugs have shown increased protection of spinal cord tissue associated with enhanced motor recovery by preventing neural and glial apoptosis71,72. This topic should be investigated in detail in future studies.
According to our results and previous studies on the subject, supplementation with trace elements and vitamin E could be part of a therapy for patients with acute SCI, helping to re-establish T-cell function, avoiding complications, and promoting better motor recovery. Establishing the therapeutic window for this promising therapy should be the next step for research.
This study is the first approach to the evaluation of the immunological and neuroprotective effects induced by this supplementation therapy. The function of other cells, including B-lymphocytes, neutrophils, natural killer cells, and even the effect on a specific response or morphological recovery should be the aim of future investigations.
Conclusion
Supplementation with trace elements and vitamin E maintains the number and function of T-lymphocytes after acute SCI. This therapeutic strategy also promotes motor recovery, which is probably due to its antioxidant effect; this is a topic that should be deeply studied in the future.
Footnotes
Author Contributions
AI, FH-A, CB: concept and design of study.
FH-A, EG, RR-B, AF-R: acquisition of data or analysis and interpretation of data.
AI, EG, CB: drafting the article or revising it critically for important intellectual content.
AI, CB, EG: final approval of the version to be published.
Availability of Data and Materials
Research materials related to this manuscript will be provided upon request to the authors.
Ethical Approval
The Animal Bioethics and Welfare Committee of the Universidad Anáhuac México approved all animal procedures (Registry number: 201870; CSNBTBIBAJ 090812960).
Statement of Human and Animal Rights
All procedures were in accordance with the National Institutes of Health (US) Guide for the Care and Use of Laboratory Animals and the Mexican Official Norm on Principles of Laboratory Animal Care (NOM 062-ZOO-1999).
Statement of Informed Consent
The authors declare that they do not have a direct financial relation with the commercial identities mentioned in the paper that might lead to a conflict of interests for any of the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Universidad Anáhuac México Campus Norte.
