Abstract
While chimeric antigen receptor (CAR)-T-cell therapy has demonstrated remarkable effectiveness in the treatment of B-cell lymphomas and leukemias, research on T-cell malignancies is still limited. Here, we reported a patient with hepatosplenic γδ T-cell lymphoma refractory to multiple lines of chemotherapy, who eventually achieved first complete remission with flow cytometry-confirmed minimal residual disease negativity after human leukocyte antigen (HLA) fully-mismatched sibling-derived CD7 CAR-T therapy. However, given the allogeneic nature, CAR-T cells dropped rapidly after a peak of 83.4% of circulating T-cells. Cytokine release syndrome, cytopenia, and infections occurred but were manageable after treatments. After the consolidative haploidentical hematopoietic stem cell transplantation (HSCT), the patient remained in remission at the end of the follow-up (13 months post-CAR-T infusion). This is the first case of relapsed/refractory hepatosplenic γδ T-cell lymphoma who achieved lasting CR after HLA fully-mismatched sibling-derived CD7 CAR-T therapy bridging to haploidentical HSCT.
Keywords
Introduction
T-cell malignancies, which can comprise 28 different entities, are aggressive with unsatisfying treatments and poor prognoses 1 . Hepatosplenic T-cell lymphoma (HSTCL), which accounts for 5% or less of peripheral T-cell lymphomas, has a disappointing response to standard anthracycline-containing induction regimens 2 . The 5-year survival of those patients is less than 10% without stem cell transplantation 3 . Current studies fail to draw conclusions about beneficial treatments for this disease since standard anthracycline-involved induction regimens and CHOP regimens consisting of cyclophosphamide, doxorubicin, vincristine, and prednisone did not achieve satisfactory outcomes2,4. Clinical management of T-cell lymphoma remains a challenge.
These years, chimeric antigen receptor (CAR) T-cell therapies targeting CD19 have shown great potential for treating relapsed or refractory B-cell malignancies5,6. But for T-cell leukemias and lymphomas, there are still specific obstacles. Firstly, malignant T-cells share similar spectra of surface markers with normal T-cells, which results in not only difficulties in the separation of normal and malignant T-cells after leukapheresis but also the fratricide between CAR-T cells7,8. To address these problems, different solutions have been proposed. Peihua Lu et al 9 used CD7 CAR-T cells derived from naturally selected CD7− circulating T-cells, while other teams have also tested the feasibility of HLA-matched including sibling-derived and donor-derived 10 or haploidentical 11 CAR-T cells. We and other groups have used universal12,13 CAR-T cells, distinguished by their accessibility and affordability, in the cases of T-cell malignancies. They mostly involved gene editing of both the targeted marker and proteins mediating alloreactivity, e.g., HLA and T cell receptor (TCR) 13 , in order to extend CAR-T persistence and avoid graft-versus-host disease (GvHD).
Despite the impressive success achieved before, unknown gene editing-associated genotoxicity because of multilocus DNA cleavage as well as off-target mutations inducing DNA modifications of unintended sites might generate multiple chromosomal aberrations with potential toxicity14–16. Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)/Cas9 was also found to have the capability of inducing extensive chromosome rearrangement known as chromothripsis which was linked to the development of congenital disorders and tumors in humans 17 . When Cas9 was used in treatment, TP53-mutant cells showed a greater susceptibility to selection, which might confer tumorigenic potential 18 . To mitigate the potential hazards of unfavorable genetic alterations, there is an urgent need for applicable universal CAR-T cell products without gene editing.
Here, we present the first instance of hepatosplenic γδ T-cell lymphoma to receive HLA-fully-mismatched sibling-derived CD7 CAR-T without extensive gene editing, who achieved complete remission (CR) lasting after consolidative allogeneic hematopoietic stem cell transplantation (allo-HSCT). This case marks a significant milestone in the investigation of generic CAR-T cells that do not require gene editing.
Methods
In addition to the conventional CAR construct, we utilized IntraBlock technology to create a CD7-binding domain fused with an endoplasmic reticulum retention signal sequence (KDEL), which confined the translated CD7 proteins to the endoplasmic reticulum of CAR-T cells19,20. Consequently, CD7 molecules were retained within the cells rather than being expressed on the cell membrane.
The evaluation of primary disease and treatment response was based on the Lugano classification 21 and the international prognostic index (IPI) 22 . The severity of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) was assessed using the revised criteria by Lee et al 23 , while other adverse events such as cytopenia as well as infections were graded based on the Common Terminology Criteria for Adverse Events, version 5.0. Acute graft-versus-host disease (aGvHD) was graded based on the modified Glucksberg staging system 24 .
Case Report
The patient, a young male, was admitted to a local hospital in September 2021 for intermittent fever with pancytopenia followed by left upper quadrant pain which was once considered a splenic infarction. However, it turns out to be HSTCL with γδ phenotype after diagnostic splenectomy. Further work-up confirmed a stage IV disease with an IPI of 2 (low-intermediate risk). He was refractory to multiple lines of chemotherapy including ECHOP (etoposide, cyclophosphamide, liposomal doxorubicin, vindesine, and dexamethasone), DICE (dexamethasone, ifosfamide, cisplatin, and etoposide), and hyper-CVAD (cyclophosphamide, vindesine, liposomal doxorubicin, and dexamethasone) incorporated with selinexor. When he came to our hospital, bone marrow aspiration revealed infiltration by lymphoma cells, with a percentage of 30.5% according to morphology or 59.18% as per flow cytometry. No nodal or extranodal lesion was identified by positron emission tomography/computed tomography (PET/CT). Given the presence of circulating malignant T-cells in peripheral blood, we decided to use allogeneic CAR-T cells, and in consideration of the aggressiveness of γδ HSTCL, his sister, although having HLA alleles completely different from his, was chosen to provide the peripheral blood mononuclear cells (PBMCs) urgently. He thus received non-HLA-matched CD7 CAR-T cells without further gene editing at a dose of 4.0 × 108 after a lymphodepleting regimen comprised of fludarabine (30 mg/m2/day, day −6 to −2), cyclophosphamide (300 mg/m2/day, day −6 to −2), and etoposide (100 mg/day, day −6 to −2) (FCE) for 5 days.
The day after infusion, the body temperature of this patient rose rapidly to a peak of 40.5℃ which could not be relieved by antibiotics (Fig. 1A). With elevated cytokines, especially interleukin (IL)-6 and IL-10, as well as C-reactive protein (CRP), he was considered as suffering from Grade 2 CRS resulting in hyperpyrexia with an average temperature of approximately 39℃ for 7 days, which was relieved after tocilizumab treatment 8 days after infusion. The serum cytokines were also found to decline gradually and return to baseline levels 14 days after infusion (Fig. 1B). Transaminitis was considered to be FCE-related since it had already begun before CAR-T infusion (Fig. 1C). The patient did not suffer from ICANS during the treatment.

Hematology results of the patient from infusion to day 56. (A) Levels of CRP were measured by the left Y-axis, while levels of temperature were measured by the right Y-axis. (B) Levels of serum cytokines. (C) Levels of liver enzyme including alanine transaminase (ALT) and glutamic transaminase (AST). (D) Levels of white blood cells (WBCs). (E) Levels of neutrophils (NEU). (F) Levels of hemoglobin (Hb). (G) Levels of platelet (PLT). (H) Levels of lymphocytes (Lym). (I) Levels of absolute numbers of CD3+ T-cells by flow cytometry. (J) Levels of EBV and HCMV DNA copies.
Since the lymphodepletion before infusion, cytopenia took place with white blood cell (WBC) count declining to 0.42 × 109/l at minimum on day 5 after infusion, while the neutrophils (NEU) count was as low as 0.14 × 109/l. WBC counts increased up to baseline 38 days after infusion with granulocyte colony-stimulating factor treatment, but the recovery of neutrophils was not satisfying, which declined to neutrophil deficiency 17 days post-infusion despite a short period of elevation during 39–47 days after infusion (Fig. 1D, E). Grade 3 anemia was observed at the minimum level of 48 g/l of hemoglobin (Hb) 33 days after infusion as well as grade 4 thrombocytopenia at the minimum level of 5 × 109/l 31 days post-infusion, which were both improved gradually with blood component transfusion (Fig. 1F, G). No bleeding event was observed.
The number of lymphocytes decreased to 0.03 × 109/l within 8 days post-infusion and remained consistently low at less than 0.1 × 109/l for about 7 days (Fig. 1H). Approximately 14 days after infusion, CD3+ T-cells were found to rise in volatility along with the rapid decline of CAR-T cells which dropped to below 0.1% 16 days post-infusion (Fig. 1I). Within the first month after infusion, the patient suffered from virus activation including both Human Cytomegalovirus (HCMV) and Epstein-Barr virus (EBV) (Fig. 1J) exactly during the decline of CD3+ cells, which finally led to viral cystitis manifested as painful urination. Despite the negativity of viral DNA after receiving conventional antiviral treatment, the patient experienced recurrences of viral infections thereafter treated with antiviral drugs. The detailed adverse events are shown in Table 1, with a brief timeline of the case presented in Fig. 2A.
Detailed Adverse Events of the Patient.

CRS: cytokine release syndrome; EBV: Epstein-Barr virus. The grading of CRS was based on the American Society for Transplantation and Cellular Therapy Consensus, and the grading of other adverse events was based on the Common Terminology Criteria for Adverse Events 5.0.
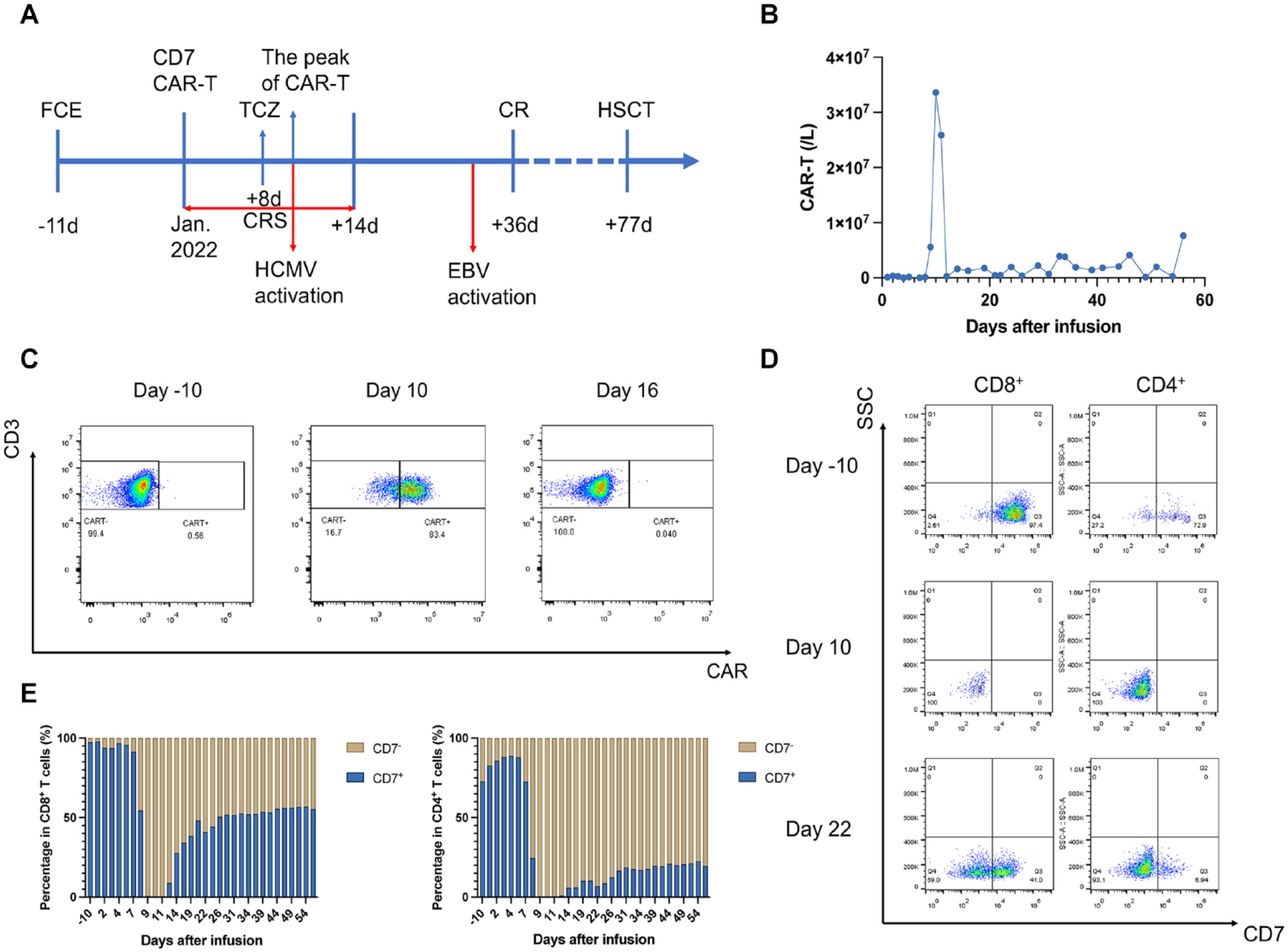
The efficacy of CD7 CAR-T cells. (A) Timeline of the key clinical events of this patient. (B) The absolute number of CAR-T cells in peripheral blood by flow cytometry. (C) The ratio of CAR− T-cells to CAR+ T-cells in CD3+ T-cells in peripheral blood on day −10, day 10, and day 16. (D) The ratio of CD7− T-cells to CD7+ T-cells was measured in CD8+ T-cells and CD4+ T-cells respectively on day −10, day 10 and day 16. (E) The trend of the ratio of CD7− T-cells to CD7+ T-cells in CD8+ T-cells and CD4+ T-cells from day 0 to day 56 post-infusion.
The CAR-T cells demonstrated robust expansion and peaked on day 10 at 3.36 × 107 cells/l (Fig. 2B,C), coinciding with a significant decrease in CD7+ T-cells which reached their lowest levels on day 9 accompanied by an increase in CD7− T-cells (Fig. 2D). Interestingly, CD4+CD7− T-cells seem to predominate the CD7− subset (Fig. 2D, E). During subsequent monitoring, the percentage of CAR-T cells in circulating T-cells dropped, resulting from the fully-mismatched nature, rapidly below 0.1% from day 16 on, along with the gradual reconstitution of the CD7+ T subset. Although CD7+ T-cells made up a large majority of T-cells before CAR-T infusion, CD7− T-cells took over the reins even after the reappearance of CD7+ T-cells, especially in the subset of CD4+ cells (Fig. 2E).
Despite the short persistence of CAR-T cells, the patient achieved morphological CR with minimal residual disease (MRD) negativity in bone marrow 28 days later. The PET/CT observed no extramedullary disease either.
Due to the short persistence of CAR-T cells, we promptly administered allogeneic HSCT to the patient as a consolidation treatment. After a 9-day pretreatment of Ara-C+BUCY+ME-CCNU+ATG, the patient received haploidentical stem cell transplantation at a total dose of 3.34 × 106/kg CD34+ cells from his mother aged 50 years. GVHD prophylaxis consisted of mycophenolate mofetil (MMF) and CsA with short-course methotrexate (MTX). He had neutrophil and platelet engraftment 12 days after HSCT. Grade I aGvHD occurred 9 days later shown as rash without diarrhea or abnormal liver function, which was resolved by low dose glucocorticoid. Five months later, he experienced GvHD shown as diarrhea, which was resolved by mesenchymal stem cell infusion. At a follow-up of nearly 9 months, the patient remained in a status of MRD-negative CR.
Discussion
At present, CAR-T cell therapy, which has made significant improvements in clinical application for B-cell malignancies, is still under exploration in T-cell lymphomas and leukemias since the fratricide of CAR-T cells and T-cell aplasia resulted in severe infections post-treatment. Since difficulty in distinguishing normal cells from malignant cells in manufacturing autologous CAR-T cells, allogeneic CAR-T cells were preferred. To deal with these problems, researchers have tried many approaches.
Previous research has demonstrated the efficacy and safety of universal CAR-T cells with modifications such as CD7 and TCR knockdown by gene editing in order to mitigate the risk of fratricide and GvHD 13 . To the best of our knowledge, this is the first case to report HLA fully-mismatched CD7 CAR-T cells without CD7 or TCR knockdown for the treatment of HSTCL.
Since the short duration of CAR-T cells, the prolonged T-cell aplasia, which was considered one of the major obstacles of CD7 CAR-T therapy, was circumvented. According to previous studies, B-cell aplasia was a recognized method for assessing the persistence of CAR-T cells and was reported to be a predictor of durable remissions25,26. However, B-cell aplasia was also found to be associated with infectious diseases caused by bacteria, viruses, and fungi, which can be life-threatening, even with immunoglobulin infusion 27 . Additionally, T-cell aplasia could cause severe infections that were even more challenging to deal with. This patient experienced virus infections 10 days after infusion, which recurred during subsequent treatments with antiviral drugs and even led to the development of viral cystitis. Previous research also indicated the obvious decline of T-cells for more than 1 month after CD7 CAR-T cells infusion with a 25% of infection rate including viral activations and fungal infections because of which a patient died eventually 19 . Due to the brief lifespan of this fully-mismatched CAR-T, T-cells were rapidly reconstituted, which in conjunction with the subsequent HSCT contributed to alleviating T-cell aplasia so that the patient could be shielded from extended and lethal infections.
Although CD7+ immunocytes were monitored to decrease, there was a compensatory increase in CD7− T-cells which demonstrated the ability to respond to virus activations 28 , consistent with previous studies 29 . An increase in CD7− cells has been observed in association with CAR-T cells, which subsequently experienced a rapid decline in numbers as CD7− cells became the predominant type of T-cells, showing that endogenous T-cells played a crucial role in allo-rejection, confirmed by a previous study 13 . Despite the reconstruction of CD7+ T-cells, CD7− T-cells maintained a high level, which was almost equivalent to that of CD7+ positive cells in CD8+ T-cells and even surpassed CD7+ T-cells in CD4+ T-cells, which might result in the short duration of CAR-T cells.
As the overall incidence of GvHD in allogeneic CAR-T for 132 patients with B-cell malignancies was 14% as reported 30 , gene-editing technology was used to create universal CAR-T cells to avoid GvHD, especially for HLA-mismatched CAR-T cells. Our case, however, applied non-gene editing and fully-HLA-mismatched CAR-T cells to the patient without GvHD-associated toxicities, showing its potential in dealing with relapse or refractory hematological malignancies, particularly for patients who have limited access to PBMCs. Despite the short duration of CAR-T cells due to the presence of allo-rejection-related antigens, the strong efficacy of this therapy was demonstrated via the morphological CR with MRD negativity at a follow-up of 13 months post-infusion. With the in-depth research on gene editing, related side effects were gradually getting attention. Off-target effects of CRISPR/Cas9 (14) as well as severe chromosomal variation were detected by researchers. Aneuploidy including a loss of chromosome 14 in 9% of primary human T-cells and a gain in 1.4% of cells, and chromosomal truncations for chromosome 7 in 9.9% of cells were detected 31 , indicating risks of this technology, which could be avoided in our CAR-T therapy.
In conclusion, our study suggested the benefit and potential of the non-HLA-matched sibling-derived CAR-T cells without gene editing. The short persistence of CAR-T cells somewhat reduces the impact of T-cell aplasia and also increases the risk of relapse, and the concerns over relapse can be dispelled by transplantation. This may provide a new direction for future treatment but still need exploration and research.
Footnotes
Acknowledgements
We would like to thank the patient and families for entrusting us with their care. We also thank all physicians, nurses, and other patient care providers involved in the study.
Author Contributions
X.X., C.Z., and M.Z. wrote the manuscript. P.X. and R.H. collected data. P.X., J.Y., and J.S. participated in the clinical treatments of the patient. J.C. and H.X. revised the manuscript. G.W., H.H., and Y.H. designed the study and participated in the treatments of the patient. A.H.C. was responsible for the product.
Ethical Approval
The study protocol was approved by the First Affiliated Hospital, School of Medicine, Zhejiang University Institutional Review Board (ClinicalTrials.gov, NCT04599556).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
The patient and family gave informed consent to participate in the study before taking part.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the National Natural Science Foundation of China (82270234 and 82130003) and Key Project of Science and Technology Department of Zhejiang Province (2021C03010 and 2023C0306).
