Abstract
To explore the correlation between T lymphocytes and clinical severity in patients of COVID-19. A total of 183 COVID-19 patients were recruited in Shenzhen Third People’s Hospital from January 11 to February 16, 2020. According to the clinical classification criteria, they were divided into severe group (46 cases) and non-severe (137cases). T lymphocyte counts, lymphocyte subpopulation, IL-6 levels, and clinical outcomes were compared between the two groups. Compared with the non-severe group, the lymphocyte count, T lymphocyte count, T lymphocyte percentage, CD4+ T lymphocyte count, CD4+ T lymphocyte percentage, CD8+ T lymphocyte count, and CD8+ T lymphocyte percentage were lower in the severe group (p < 0.05). Compared with the non-severe group, IL-6 were higher in the severe group (p < 0.05). Compared with admission, the T lymphocyte count, CD4+ T lymphocyte count, and CD8+ T lymphocyte count were significantly increased upon discharge in severe patients, non-severe patients and all patients. Multivariate Logsitic regression analysis showed CD4+ T lymphocyte count (OR −0.011; 95% CI −0.041 to −0.001; p = 0.011), CD8+ T lymphocyte count (OR −0.14; 95% CI −0.048 to −0.003; p = 0.013) were closely correlated with the clinical severity in patients of COVID-19. Multivariate Logsitic regression analysis also showed CD4+ T lymphocyte count (OR −0.012; 95% CI −3.177 to 0.261; p = 0.021), CD8+ T lymphocyte count (OR −0.019; 95% CI −5.852 to 0.115; p = 0.004) were independent predictors of disease progressing to the composite endpoint. Subgroup analysis for critically ill patients: The T lymphocyte count, CD4+ T lymphocyte count, and CD8+ T lymphocyte count remained low in the death patients. The T lymphocyte count, CD4+ T lymphocyte count, and CD8+ T lymphocyte count recovered soon in the discharged patients. In the event of COVID-19 infection, the T-lymphoid system is the primary activated immune system. The T lymphocytes, CD4+ T lymphocytes, CD8+ T lymphocytes continued to be low may be significantly related to the deterioration of the disease, and may indicate a poor prognosis.
Introduction
The new coronavirus pneumonia (named COVID-19 by the World Health Organization) was currently a major public health event that seriously affects human health. Some studies thought that the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) invades the human body by the same way as SARS-CoV. The binding of spike proteins on the surface of the virus to converting enzyme 2 (ACE2) on the surface of respiratory epithelial cells mediates the virus entry into the cells. Then it entered the cell and stimulated the immune system to release inflammatory cytokines, caused a series of pathological changes. 1 As early as 2005, Huang et al. 2 found that cytokines play an important role in acute respiratory distress syndrome caused by SARS-Cov. Elevated levels of serum pro-inflammatory cytokines were observed in COVID-19 patients, which were also related to lung injury. 3 However, little is known about the role of T lymphocytes in the immune response and disease progression in COVID-19 patients. COVID-19 is reported to be more likely to occur in elderly men with respiratory complications and impaired immune function. 4 Therefore, this single-center, retrospective study was designed to analyze the differences of T lymphocytes between different clinical types in COVID-19, and to find early predictors of disease development and prognosis.
Objects and methods
A total of confirmed 417 COVID-19 patients were admitted to the Shenzhen Third People’s Hospital, the only COVID-19 admission hospital in Shenzhen, from January 10 to February 17, 2020. Among them, 183 patients were older than 18 years and had T lymphocyte subsets detected within 48 h of admission. According to the clinical classification criteria, they were divided into severe group (46 cases) and non-severe (137cases). Twelve patients were severe on admission. Thirty-four patients progressed to severe, 14 patients progressed to critical illness during the study period and three patients died. The primary outcome was a composite of death, or progression to critically ill stage during the study period. The follow-up time ended on March 20, 2020.
The inclusion criteria are described as follow according to World Health Organization interim guidance: The severity of COVID-19 was judged according to the sixth Revised Trial Version of the Novel Coronavirus Pneumonia Diagnosis and Treatment Guidance. 5 Those who met one of the following conditions were defined as severe-type: (1) Respiratory distress with the respiratory rate over 30 per min; (2) Oxygen saturation ⩽93% in the resting state; (3) Arterial blood oxygen partial pressure (PaO2)/oxygen concentration (FiO2) ⩽300 mmHg. Those who met one of the following conditions were defined as critical-type: (1) Respiratory failure occurs, and artificial airway needs to be established to perform invasive mechanical ventilation; (2) Shock occurs; (3) ICU monitoring treatment is required for the combined failure of other organs.
Ethics approval Ethical approval for this study was obtained from Ethics Committee of Shenzhen Third People’s Hospital (IRB ID: 2020-044). Written informed consent was waived by the Ethics Commission of the designated hospital for emerging infectious disease.
Clinical information
Data including demographic data, medical history, symptoms, signs, and laboratory findings were collected from patients’ medical record. Laboratory results included blood routine, biochemical indicators, T lymphocyte subsets, infection-related biomarkers, inflammatory cytokines, immunoglobulins, and complement proteins. Laboratory findings were collected within 48 h of admission. The critically ill patients were divided into seven stages according to the course of admission, when they switched to severe, 1 week after they switched to severe, 2 week after they switched to severe, 3 week after they switched to severe, 4 week after they switched to severe and the end point of the observation. The T lymphocyte subsets of each stage were collected. The observation endpoint was based on the patient’s death or discharge or the last indicator.
Statistical methods
We used SPSS22.0 statistical software for statistical analysis. We described the categorical variables as frequency rates and percentages, and continuous variables as mean and standard deviation (SD), median and interquartile range (IQR) values. Independent group t tests were used for the comparison of means for continuous variables that were normally distributed; conversely, the Mann–Whitney U test was used for continuous variables not normally distributed. Comparison of admission and discharge T lymphocytes used paired t test. Proportions for categorical variables were compared using the χ2 test. The association between the clinical and laboratory characteristics and the composite endpoint was examined by logistic regression. The univariate analysis was first conducted for each of the risk factors by age–sex adjusted, then a stepwise selection-based multivariable analysis was conducted to select the variables with statistical significance. Odds ratio (OR) and 95% confidence interval (CI) were provided to indicate the effect. Calculate the area under curves (AUC) of receiver operating characteristic (ROC) to predict and analyze the severity of the disease. Two-sided p-values of less than 0.05 were considered statistically significant.
Results
Demographic and clinical characteristics of COVID-19
A total of 183 COVID-19 patients were included in this study, 46 (25.1%) of whom were clinically diagnosed as severe infection. In total, the median age was 49 years (IQR, 36–63; range, 20–81 years), and 103 (56.3%) were men. Compared with the non-severe group, the severe group was older (median age 63.5 years; IQR (53.25–67.50) vs 44 years IQR (33–58); p < 0.0001). The proportion of men in the severe group was higher than that in the non-severe group (78.3% vs 48.9%, p = 0.0005). Compared with the non-severe group, the proportion of complicated with basic diseases (60.87% vs 29.93%, p = 0.0002), hypertension (28.26% vs 13.14%, p = 0.0180), diabetes (15.22% vs 2.92%, p = 0.0061), coronary heart disease (10.87% vs 2.92%, p = 0.0457) was higher in the severe group. The proportion of severe group combined ARDS (47.83% vs 0%, p < 0.0001) was higher than that of non-severe group. The proportion of bilateral lung inflammation in the severe group (100% vs 84.67%, p = 0.0024) was higher than that in the non-severe group. The median time from onset to admission in the severe group (5 days IQR (3.75–8) vs 3 days IQR) was higher than that in the non-severe group (Table 1).
Baseline characteristics of patients with COVID-19.

Data are median (IQR), n (%), or n/N (%), where N is the total number of patients with available data. p Values comparing severe and non-severe cases are from χ² test or Fisher’s exact test.
p < 0.05, **p < 0.01, compared with non-severe group.
Biochemical indicators and immune indicators
Compared with the non-severe group, the neutrophil count (3.827 ± 1.818 vs 2.821 ± 1.352 × 109/L, p < 0.0001), the neutrophil percentage (68.96 ± 15.24 vs 56.84 ± 11.84%, p < 0.0001), and the neutrophil-to-lymphocyte ratio (5.0 ± 4.567 vs 2. 458 ± 4.055, p = 0.0005) were higher in the severe group. The lymphocyte count (0.9837 ± 0.3822 vs 1.442 ± 0.5245 × 109/L, p < 0.0001) and the lymphocyte percentage (20.78 ± 9.921 vs 31.51 ± 10.33%, p < 0.0001) in the severe group were lower than those in the non-severe group. Compared with the non-severe group, the albumin (39.88 ± 6.673 vs 43.26 ± 2.997 g/L, p < 0.0001), PaO2: FiO2 ratio (305.1 ± 102.1 vs 445.9 ± 112.3, p < 0.0001) were lower in the severe group. The creatinine (80.87 ± 30.44 vs 64.80 ± 16.09 umol/L, p < 0.0001), LDH (534.6 ± 332.3 vs 271.1 ± 130.0 U/L, p < 0.0001) in the severe group were higher than those in the non-severe group. The D-dimer (1.235 ± 2.937 vs 0.4359 ± 0.2399 ug/mL, p = 0.0018) and fibrinogen (4.807 ± 1.626 vs 4.098 ± 1.247 g/L, p = 0.0024) in the severe group were higher than those in the non-svsere group. Compared with the non-severe group, the IL-6 (40.23 ± 32.94 vs 12.66 ± 13.72 pg/mL, p < 0.0001), ESR (47.54 ± 25.49 vs 30.43 ± 21.58 mm/h, p < 0.0001), CRP (46.57 ± 35.00 vs 15.30 ± 17.10 mg/L, p < 0.0001) were higher in the severe group. Compared with the non-severe group, the T lymphocyte count (519.9 ± 298.4 vs 1040 ± 402.8/µL, p < 0.0001), T lymphocyte percentage (54.21 ± 11.36 vs 67.39 ± 9.809%, p < 0.0001), CD4+ T lymphocyte count (297.5 ± 174.5 vs 582.0 ± 248.8/µL, p < 0.0001), CD4+ T lymphocyte percentage (31.57 ± 10.09 vs 37.73 ± 8.060%, p < 0.0001), CD8+ T lymphocyte count (184.6 ± 123.0 vs 393.0 ± 182.4/uL, p < 0.0001), CD8+ T lymphocyte percentage (19.11 ± 6.727 vs 25) .61 ± 8.002%, p < 0.0001) were lower in the severe group. There was no difference in Immuno-globulin A, Immunoglobulin G, Immunoglobulin M, C3, C4 between the severe group and the non-severe group (Table 2).
Laboratory findings of patients with COVID-19.

Data are mean (SD). p Values comparing severe and non-severe cases are from T test, or Mann–Whitney U test.
The severe group n = 32, the non-severe group n = 85.
p < 0.05, **p < 0.01, compared with non-severe group.
Multivariate Logsitic regression analysis showed CD4+ T lymphocyte count (OR −0.011; 95% CI −0.041 to −0.001; p = 0.011), CD8+ T lymphocyte count (OR −0.140; 95% CI −0.048 to −0.003; p = 0.013), IL-6 (OR 0.037; 95% CI −0.006 to 0.120; p = 0.032), LDH (OR 0.004; 95% CI 0.001 to 0.013; p = 0.008) and Bilateral lung inflammation (OR 17.139; 95% CI 13.867 to 19.719; p = 0.004) were related to the clinical severity in patients of COVID-19 (Table 3).
Multivariable analysis of risk factors associated with the severe COVID-19 in all 183 patients.

p < 0.05. **p < 0.01.
Multivariate Logsitic regression analysis showed T lymphocyte count (OR 0.009; 95% CI 0.004 to 1.216; p = 0.004), CD4+ T lymphocyte count (OR −0.015; 95% CI −1.883 to −0.008; p = 0.001),
CD8+ T lymphocyte count (OR −0.018; 95% CI −2.443 to −0.011; p = 0.001) and IL-6 (OR 0.047; 95% CI −0.008 to 1.808; p = 0.005) could predicte of disease progressing to severe in 171 non-severe COVID-19 patients on admission (Table 4).
Multivariable analysis of predictor of disease progressing to severe in 171 non-severe COVID-19 patients on admission.

p < 0.05. **p < 0.01.
Multivariate Logsitic regression analysis showed CD4+ T lymphocyte count (OR −0.012; 95% CI −3.177 to 0.261; p = 0.021),CD8+ T lymphocyte count (OR −0.019; 95% CI −5.852 to 0.115; p = 0.004),IL-6 (OR 0.049; 95% CI −0.242 to 12.051; p = 0.005) and respiratory rate(OR 0.253; 95% CI −1.926 to 146.212; p = 0.023) were independent predictors of disease progressing to the composite endpoint(Table 5).
Multivariable analysis of predictor of disease progressing to the composite endpoint in all 183 COVID-19 patients.

p < 0.05. **p < 0.01.
We calculate the AUC values of ROC for the severity of the disease. The AUC values of ROC curve for T lymphocyte count, CD4+ T lymphocyte count, CD8+ T lymphocyte count, IL-6, LDH, and bilateral lung inflammation to predict COVID-19 severity were 0.866, 0.843, 0.851, 0.824, 0.783, 0.577 (Figure 1).
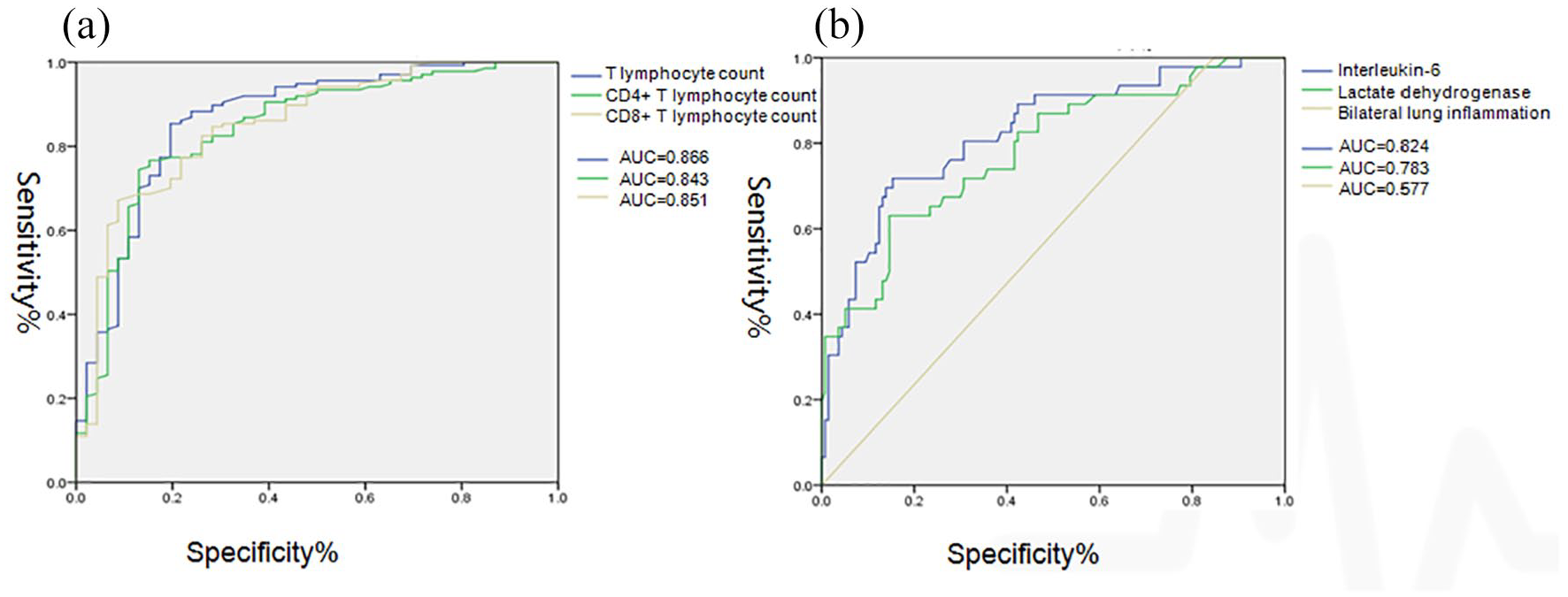
ROC curve for predicting the severity of COVID-19.
Compared with admission, the T lymphocyte count, CD4+ T lymphocyte count and CD8+ T lymphocyte count were significantly increased upon discharge in severe patients, non-severe patients and all patients (Figure 2).
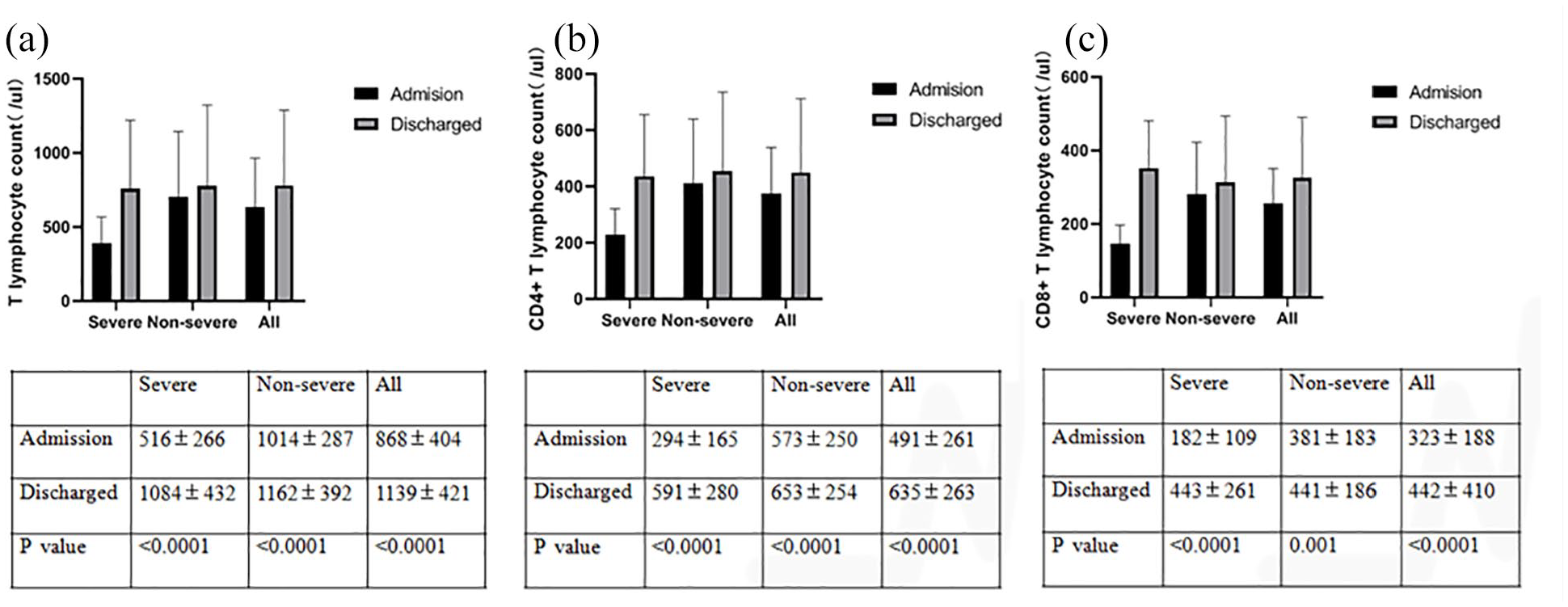
The difference between of T lymphocyte count, CD4+ T lymphocyte count, CD8+ T lymphocyte between admission and discharge in COVID-19 patients.
Subgroup analysis for critical patients: The T lymphocyte count, CD4+ T lymphocyte count and CD8+ T lymphocyte count remained low in the death patients. The T lymphocyte count, CD4+ T lymphocyte count and CD8+ T lymphocyte count recovered soon in the discharged patients (most of them were 2 week after they switched to severe). The T-lymphocyte count, CD4+ T-lymphocyte count and CD8+ T-lymphocyte count of patients still in hospital recovered slowly (Tables 6–8).
T lymphocyte counts of critical COVID-19 patients in different periods.

The first stage = on admission; the second stage = when the patients became severe; the third stage = 1 week after the patients switched to severe; the fourth stage = 2 week after the patients switched to severe; the fifth stage = 3 week after the patients switched to severe; the sixth stage = 4 week after the patients switched to severe; the seventh stage = the observation endpoint, the observation endpoint was based on the patient’s death or discharge or the last indicator.
Case 1, case 2, case 4, case 7, case 8, case 9, case 10, case 11 were severe on admission.
CD4+ T lymphocyte counts of critical COVID-19 patients in different periods.

The first stage = on admission; the second stage = when the patients became severe; the third stage = 1 week after the patients switched to severe; the fourth stage = 2 week after the patients switched to severe; the fifth stage = 3 week after the patients switched to severe; the sixth stage = 4 week after the patients switched to severe; the seventh stage = the observation endpoint, the observation endpoint was based on the patient’s death or discharge or the last indicator.
Case 1, case 2, case 4, case 7, case 8, case 9, case 10, case 11 were severe on admission.
CD8+ T lymphocyte counts of critical COVID-19 patients in different periods.

The first stage = on admission; the second stage = when the patients became severe; the third stage = 1 week after the patients switched to severe; the fourth stage = 2 week after the patients switched to severe; the fifth stage = 3 week after the patients switched to severe; the sixth stage = 4 week after the patients switched to severe; the seventh stage = the observation endpoint, the observation endpoint was based on the patient’s death or discharge or the last indicator.
Case 1, case 2, case 4, case 7, case 8, case 9, case 10, case 11 were severe on admission.
Discussion
We retrospectively analyzed the immune response of 183 patients with COVID-19 in the Third People’s Hospital of Shenzhen. In this study, most of the COVID-19 patients had a decrease in lymphocytes, and the reduction in severe patients was more obvious, which was consistent with the early research reports. 4 Compared with the non-severe group, the T-lymphocyte count, CD4+ T-lymphocyte count, CD8+ T-lymphocyte count decreased more significantly in the severe group. T lymphocytes display a very important role in the body’s specific immune response, mainly composed of CD4+ and CD8+ cell groups. CD4+ T cells are inducible helper cells and are responsible for the elimination and control of multiple infections by assisting other cells to conduct immune modulation. CD8+ lymphocytes are effector cells of the body’s immune response. Under normal conditions, lymphocyte subsets are in coordination and balance with each other to resist pathogen infection. Different virus infection may trigger adaptive immune responses in different types of T cells. Previous studies have shown that during SARS-COV infection, CD8+ T cells account for approximately 80% of the total inflammatory cells in lung interstitial lesion, which is beneficial to remove virus from infected cells. 6 Depletion of CD4+ T cells causes a decrease in neutralizing antibody levels, leading to a strong immune-mediated interstitial pneumonia that delays the clearance of MERs-CoV. Therefore, both CD8+ and CD4+ T cells play an important immunomodulatory role in the body’s response to different types of coronavirus infection. In support of this point, compared with admission, the T lymphocyte count, CD4+ T lymphocyte count and CD8+ T lymphocyte count were significantly increased upon discharge in severe patients, non-severe patients and all patients after regular treatment. Another implication of our research is that the dynamic change of T lymphocyte could be viewed as a predictor for prognosis of critically ill COVID-19 patients, because of the relative lower level of T lymphocyte counts in the death patients. In line with our finding, Qin et al. 7 found that the decrease of T lymphocytes in COVID-19 patients, especially the decrease of CD4+ T lymphocytes is more common in critically ill patients, but the number of CD8+ T lymphocytes and B cells had no significant changes. Chen et al. 9 found T lymphocytes, CD4+ T and CD8+ T cells decreased in nearly all the patients, and were markedly lower in severe cases. Wang et al. 8 found CD4+ T cells, CD8+ T cells, B cells decreased in COVID-19 patients, and severe cases had a lower level than mild cases, especially CD8+ T cells and CD4+/CD8+ ratio. 9 To sum up, in the future treatment of COVID-19, more attention should be paid to the dynamic changes in the number of T lymphocytes and their subsets during the course of the disease to adjust the treatment regimen.
SARS-COV-2 may be the same as SARS-CoV, which may mainly affect lymphocytes, especially T lymphocytes, caused the decreases of T lymphocytes, CD4+ T lymphocytes, CD8+ T lymphocytes. The significant decrease in T lymphocytes indicates that SARS-COV-2 consumes many immune cells and suppresses the body’s cellular immune function. The dysfunction of T lymphocytes may be an important factor leading to the deterioration of the patient’s condition. 10 The first COVID-19 pathological anatomy found a dramatically decrease in blood lymphocytes with over-activation. In particular, CCR4+, CCR6+, and Th17 cells with high proinflammatory effects in CD4+ T cells were increased and T lymphocytes with high cytotoxicity in CD8+ T cells were over-activated. 11 The Th17 cells were a subset of T cells found in recent years, they can secrete a large amount of IL-17 to play an inflammatory role. Stimulated by IL-6 and other cytokines, the transformation from primitive T lymphocytes to Th17 cells were mediated by non-receptor tyrosine kinase and STAT3 signaling dependent pathway. 12 In our study, the increased level of IL-6 in severe patients is more obvious than that in non-severe patients. The same trend was also observed in neutrophil count, Neutrophil-to-lymphocyte ratio, CRP, and other inflammation indicators. COVID-19 may be the same as SARS-CoV. SARS-CoV infection can induce up-regulation of pro-inflammatory cytokines (TNF, IL-6) and up-regulation of inflammatory chemokines (CCL3, CCL5, CCL2, and CXCL10). 13 As a whole, up-regulated cytokines such as IL-6 stimulated the differentiation of primitive T lymphocytes into Th17 cells with high proinflammatory effects. The increase of Th17 cells led to excessive secretion of cytokines, forming a cascade waterfall effect. In order to recruit more immune cells, the vascular permeability of the lung lesion was increased, which makes the pathogens more easily to enter the blood vessel in the lesion. At the same time, increased inflammation level also accelerates the extravasation of the fluid in the blood vessels, thereby destroying the tissue, and ultimately, the inflammation in the lung tissue is out of control. This abnormally elevated cytokines and over-activated immune cells cause diffuse damage to pulmonary capillary endothelial cells and alveolar epithelial cells, and a large amount of exudate accumulates to block the airway, eventually leading to the occurrence of ARDS. 14 At the same time, severe lung injury combined with damage to the body’s immune function and systemic cytokine storms caused by cytokines in the circulatory system will further cause dysfunction of the whole body’s organs, which was likely to cause a sudden exacerbation of the disease, even death. 15 Therefore, it was important for the early detection and timely treatment to stop the constant increase in inflammation levels in severe patients. Patients with ARDS tendencies and persistently elevated cytokines could try to use glucocorticoids as early as possible. Patients with reduced T lymphocytes and impaired immune function could try to use immunoglobulins to enhance their anti-infective capacity. 4 Carrying out dynamic monitoring of cytokines and other indicators may help early detection of the occurrence of cytokine storms and determine the necessity for CRRT. The IL-17 inhibitor secukinumab and the drugs that directly target IL-6, tocilizumab and stolimumab, may have effects on COVID-19 patients.11,16,17
However, our study had some limitations. First, this was a retrospective, small sample study of admitted patients. Secondly, we only tested T lymphocytes, not other lymphocytes, NK cells, etc. only IL-6 was tested, other cytokines and chemokines were not tested. Third, COVID-19 patients with bacterial infections may affect the immune response.
Conclusion
Our research showed some new information about the immune response of COVID-19 patients, COVID-19 may mainly induce the T lymphatic system immune response. The T lymphocytes, CD4+ T lymphocytes, CD8+ T lymphocytes continued to be low may be significantly related to the deterioration of the disease, and may indicate a poor prognosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shenzhen Third People’s Hospital.
Ethics approval
Ethics approval Ethical approval for this study was obtained from Ethics Committee of Shenzhen Third People’s Hospital (IRB ID:2020-044).
Informed consent
Written informed consent was waived by the Ethics Commission of the designated hospital for emerging infectious disease.
Trial registration
Name of trial redistry: Association of T lymphocytes level and clinical severity in patients of COVID-19 :A single-center retrospective data.
Trial registration number:ChiCTR2000030986.
Chinese Clinical Trial Registry
