Abstract
Loss of photoreceptor cells is a primary feature of inherited retinal degenerative disorders including age-related macular degeneration and retinitis pigmentosa. To restore vision in affected patients, photoreceptor cell replacement will be required. The ideal donor cells for this application are induced pluripotent stem cells (iPSCs) because they can be derived from and transplanted into the same patient obviating the need for long-term immunosuppression. A major limitation for retinal cell replacement therapy is donor cell loss associated with simple methods of cell delivery such as subretinal injections of bolus cell suspensions. Transplantation with supportive biomaterials can help maintain cellular integrity, increase cell survival, and encourage proper cellular alignment and improve integration with the host retina. Using a pig model of retinal degeneration, we recently demonstrated that polycaprolactone (PCL) scaffolds fabricated with two photon lithography have excellent local and systemic tolerability. In this study, we describe rapid photopolymerization-mediated production of PCL-based bioabsorbable scaffolds, a technique for loading iPSC-derived retinal progenitor cells onto the scaffold, methods of surgical transplantation in an immunocompromised rat model and tolerability of the subretinal grafts at 1, 3, and 6 months of follow-up (n = 150). We observed no local or systemic toxicity, nor did we observe any tumor formation despite extensive clinical evaluation, clinical chemistry, hematology, gross tissue examination and detailed histopathology. Demonstrating the local and systemic compatibility of biodegradable scaffolds carrying human iPSC-derived retinal progenitor cells is an important step toward clinical safety trials of this approach in humans.
Keywords
Introduction
Loss of photoreceptor cells is a pathophysiologic feature shared between the common condition age-related macular degeneration and much rarer Mendelian retinal degenerative disorders, such as retinitis pigmentosa, Stargardt disease, and Usher syndrome. Unlike lower vertebrates (eg, salamander), which can rebuild their entire retina via activation of an endogenous progenitor cell population 1 , the human retina has little intrinsic ability to regenerate following injury. The most promising treatment strategy for restoring vision to patients blinded by one of these disorders is stem cell-mediated photoreceptor cell replacement2–16. Unlike stem cell-derived retinal pigmented epithelial (RPE) cells, which have been successfully transplanted in a number of human clinical trials17–22, photoreceptor cells are light-detecting neurons that must extend their axons into the host retina and form synaptic connections with host bipolar cells to restore visual function.
A variety of different donor cell types have been evaluated for retinal cell transplantation23,24. For example, fate-restricted retinal progenitor and photoreceptor precursor cells can be obtained from fetal donors and transplanted directly into patients25–29. These cell types have been shown to be at a developmental stage that is suitable for photoreceptor cell replacement, but their fetal origin is associated with significant practical and ethical limitations. In contrast to these immediately transplantable fetal cells, embryonic and induced pluripotent stem cells both require many weeks of in vitro differentiation prior to transplantation. However, the latter cell types both have the tremendous advantage of unlimited self-renewal and have become the strategy of choice for cell replacement. Patient-derived induced pluripotent stem cells (iPSCs) have the additional advantage that they can be derived from and transplanted into the same patient, thus creating an immunologic match, and avoiding the need for lifelong immunosuppression.
Although several studies have illustrated the potential for stem cell-derived photoreceptor cell replacement to restore vision7,13,30–33, a major limitation to this therapeutic application has been the loss of donor cells following subretinal delivery. For instance, up to 95% of cells delivered as a subretinal bolus cell suspension are lost through a combination of efflux from the injection site and apoptosis triggered by the loss of adhesion and the shear stresses that occur during dissociation of mature cell cultures and injection through small-bore cannulas34–36. To overcome these limitations, we and others have explored the use of biodegradable polymers as cell support scaffolds34–42. These scaffolds are designed to provide donor cells with the structural support required to prevent efflux and mitigate apoptosis34–42. In addition, scaffolds can be designed to promote proper photoreceptor cell alignment, packing density, maturation and synaptic integration following transplantation37,38,41,42.
We recently evaluated local and systemic biocompatibility of a polycaprolactone (PCL) cell support scaffold following subretinal transplantation in a pig model of advanced retinal degeneration 37 . In addition to demonstrating that PCL scaffolds and their degradation products are well tolerated, we confirmed that the subretinal transplantation strategy and instrumentation that will be used clinically were both safe and effective. That is, we were able to successfully place and maintain 3D scaffolds in the subretinal space of a pig model of advanced retinitis pigmentosa without serious adverse events. The next step toward developing a clinical autologous photoreceptor cell replacement strategy is to demonstrate that PCL-supported patient-derived retinal progenitor cells (ie, retinal cell grafts) are non-tumorigenic and do not induce local or systemic toxicity following subretinal transplantation. In choosing a recipient model system for these experiments we had two main considerations: (1) the host should be immunocompromised and in turn conducive to receiving a xenograft, and (2) the host should have eyes large enough to have a 3D retinal cell graft placed in the subretinal space and yet small enough to be housed in large numbers for an extended period of time. The latter feature is essential for one to be able to test grafts generated from multiple different patients for up to 6 months following placement. The Rowett Nude rats (RNU) rat is a readily available immunocompromised animal that at just 2 months of age has eyes large enough to receive a 1-mm subretinal graft of the same thickness intended for human clinical use. In addition, the baseline health status, which includes hematology and clinical chemistry parameters as well as strain specific lesions, has been thoroughly documented43–47. While this animal is not suitable for evaluating treatment efficacy (ie, it does not suffer from an inherited retinal degeneration), the above features make it ideally suited for use in the local and systemic toxicity studies reported later in this article.
In this study, we describe fabrication of a retinal cell graft (DTVRF-hiPSC-ARG), methods for subretinal transplantation in an immunodeficient RNU rat model, as well as comprehensive local and systemic tolerability analyses following subretinal delivery with 1, 3, and 6 months of follow-up. We demonstrate that DTVRF-hiPSC-ARGs do not induce tumor formation or cause local or systemic toxicity following subretinal transplantation.
Materials and Methods
Ethics Statement
Approval to obtain dermal fibroblast cells and generate iPSCs from patients with inherited retinal degeneration and normal controls was obtained under informed consent. This study was reviewed and approved by the University of Iowa Institutional Review Board (IRB# 200202022). All rat experiments were conducted with the approval of the University of Iowa Animal Care and Use Committee (animal welfare assurance #8051317) and were consistent with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
Fabrication of PCL Retinal Progenitor Cell Delivery Scaffolds
PCL-acrylate was fabricated by Creative PEGWorks as follows: PCL triol (Mn ~300 g/mol) was functionalized by mixing it with potassium carbonate and acryloyl chloride at a 1:3:3 ratio. The PCL triol was dissolved in methylene chloride (1:2 v/v) and then mixed with potassium carbonate. Acryloyl chloride was added dropwise to the PCL triol-potassium carbonate mixture. The reaction mixture was filtered to remove solids. Sodium bicarbonate was added to the mixture at a 1:1 ratio and run through a separatory funnel. Excess solvent was removed from the functionalized PCL by rotary evaporation and the final product stored in dry, airtight containers at −20°C. The degree of functionalization and final molecular weight were measured for each batch using nuclear magnetic resonance ( 1 H NMR) and gel permeation chromatography, respectively. One gram of functionalized PCL (Creative PEGWorks, Chapel Hill, NC, USA; Cat#: PCL-AC-300-50g) and 100 µl of photoinitiator solution (0.1 g Irgacure® 2959 in 1 ml of sterile ethanol, Advanced BioMatrix, Inc., Carlsbad, CA, USA) were combined and used for polymerization.
Retinal progenitor cell scaffolds were generated via photopolymerization using UV light (approximately 100 lumens/cm2) and a photomask containing 75 µm black spots spaced at 25 µm in a hexagonal pattern. Following fabrication, scaffolds were removed from the polymerization chamber and placed onto a 6-well Millicell insert (EMD Millipore; Cat#: MCMP06H48, Burlington, MA, USA) where a vacuum allowed active rinsing of the scaffolds. The scaffold was rinsed 10 times with 1 ml of sterile phosphate buffer saline (PBS) (Gibco/Thermo Fisher Scientific, Waltham, MA, USA) and then submerged in sterile PBS overnight. Following the final rinse, scaffolds were placed into a 12-well plate, submerged in sterile PBS, and double packaged in an instant sealing sterilization pouch to be terminally sterilized via gamma irradiation. The scaffolds remained in the sealed sterilization pouch until ready for use, at which time they were opened in a laminar flow hood to maintain aseptic processing. Scaffolds were fabricated 2 to 3 weeks prior to being seeded.
Generation and Differentiation of Patient-Derived iPSCs
In this study, four independent iPSC lines [a normal control and three patients (P1 and P2 were diagnosed with wolfram syndrome [ie, WFS1-associated disease affects retinal ganglion cells and patients retain normal photoreceptor cells throughout their life] and P3 was diagnosed with enhanced S cone syndrome [non-progressive disease in which the patient has an abundance of blue cones and few rod cells])] were generated and differentiated into transplantable retinal progenitor cells using our previously published protocols48,49. Briefly, each iPSC line was generated via reprogramming of dermal fibroblast using the CytoTune 2 Sendai viral reprogramming kit (Gibco/Thermo Fisher Scientific, Waltham, MA, USA). Following clonal expansion, karyotyping and scorecard analysis at passage 10, iPSCs were differentiated into 3D retinal organoids 48 . At 70 ± 5 days, retinal organoids were harvested, dissociated into individual retinal progenitor cells, and plated onto PCL scaffolds fabricated and sterilized as described above at a density of 40,000 cells per 1-mm circular graft (0.79 mm2). From this point forward, DTVRF-hiPSC-ARG refers to these PCL scaffolds seeded with human iPSC-derived retinal progenitor cells. For immunocytochemistry, grafts were fixed with 4% paraformaldehyde and stained with the following primary antibodies overnight: OTX2 (R&D Systems; Cat#: AF1979, Minneapolis, MN, USA), Recoverin (EMD Millipore; Cat#: AB5585, Burlington, MA, USA), SOX2 (R&D Systems; Cat#: MAB2018), NRL (R&D Systems; Cat#: AF2945), NRL (R&D Systems; Cat#: AF2945), ARR3 (Lifespan Biosciences; Cat#: LS-C368677, Seattle, WA, USA), NR2E3 (R&D Systems; Cat#: PP-H7223-00), Opsin, red/green (EMD Millipore; Cat#: AB5405), Rhodopsin (EMD Millipore; Cat#: MAB5316). The following secondary antibodies (Thermo Fisher Scientific, Waltham, MA, USA) were incubated for 1 hour: donkey anti-goat 488 (Thermo Fisher Scientific; Cat#: A11055, Waltham, MA, USA), and donkey anti-rabbit 488 (Thermo Fisher Scientific; Cat#: R37118, Waltham, MA, USA), donkey anti-mouse 488 (Cat#: A21202), donkey anti-sheep 647 (Cat#: A21448), donkey anit-rabbit 647 (Cat#: A31573), donkey anti-goat 647 (Cat#: A21447). Cell nuclei were counterstained using DAPI (Thermo Fisher Scientific; Cat#: 62248) or SYTOX Orange (Thermo Fisher Scientific; Cat#: S11368, Waltham, MA, USA).
Subretinal Transplantation and Study Design
To assess both local and systemic tolerability of DTVRF-hiPSC-ARG, T-cell deficient homozygous Rowett Nude rats (RNU-/-; Cr:NIH-RNU; Charles River, Wilmington, MA, USA, hereafter referred to as RNU) were utilized. Rats were anesthetized with isoflurane and eyes were dilated with 1–2 drops of 1% tropicamide (Alcon Laboratories, Fort Worth, TX, USA). All rat surgeries were performed by a fellowship-trained retina surgeon (I.C.H.). Under an operating microscope (Leica M620; Leica Microsystems, Inc., Wetzlar, Germany), subretinal transplantation of grafts was performed using a modified version of previously described methods50,51. In brief, a limited conjunctival peritomy was created to expose the temporal sclera (Fig. 1A). A 15° sideport blade (Beckon Dickinson and Company, Waltham, MA, USA) was used to create a transscleral, transretinal incision into the vitreous cavity (Fig. 1B). Any prolapsed vitreous was trimmed with Vannas scissors and balanced salt solution was then dripped into the incision site with a 30-gauge anterior chamber cannula (Beaver-Visitec International, Waltham, MA, USA) to create a localized retinal detachment (Fig. 1C). Grafts were transported to the surgical suite in a 40-µm cell strainer contained within a 6-well culture plate (Corning Incorporated, Corning, NY, USA) (Fig. 1D) containing 1× Hanks’ Balanced Salt solution (Thermo Fischer Scientific, Waltham, MA, USA). Each individual graft was elevated from the transport solution using the strainer and grasped at its edge with McPherson tying forceps (Miltex Inc., York, PA, USA), which were then used to transplant the polymer into the subretinal space (Fig. 1E). A 30-gauge anterior chamber cannula was then inserted subretinally to push the polymer away from the incision site. The conjunctiva was then reflected over the scleral wound, which was left to close by secondary intention.

Surgical placement of DTVRF-hiPSC-ARG into the subretinal space of athymic nude RNU rats. (A–C) The surgical procedures required to prepare the RNU rat eye for subretinal placement of DTVRF-hiPSC-ARG. (D) The surgeon retrieving a 1-mm DTVRF-hiPSC-ARG from the transport container immediately prior to subretinal transplantation. (E) Surgical placement of a 1-mm DTVRF-hiPSC-ARG through a transscleral incision into the subretinal space of the RNU rat. (F) Image depicting DTVRF-hiPSC-ARG immediately following placement into the subretinal space of an RNU rat. RNU: Rowett Nude.
Fundus Imaging and Optical Coherence Tomography (OCT)
Rats were anesthetized with ketamine/xylazine mixture [40–100 mg/kg ketamine (Clipper Distributors, Saint Joseph, MO, USA), 5–13 mg/kg xylazine (Akorn Pharmaceuticals, Lake Forest, IL, USA)], and eyes were dilated with 1 to 2 drops of Tropicamide (Alcon Laboratories, Fort Worth, TX, USA). Prior to sacrifice for biodistribution studies or histologic evaluation (see the next section), eyes were examined by a retina specialist (I.C.H.) under an operating microscope to record clinical findings, including presence of iris synechiae (suggestive of anterior chamber inflammation), cataract formation, vitreous haze or opacities (consistent with posterior chamber inflammation), retinal reattachment, chorioretinal atrophy, and subretinal fibrosis. Fundus photographs were obtained in vivo using the Micron III fundus camera (Phoenix Technology Group, Pleasanton, CA, USA). OCT imaging was performed using the Bioptigen Envisu R2200 (Bioptigen, Inc., Morrisville, NC, USA) with a slit-lamp-based mount. A limbal suture was placed at the corresponding clock hour of the transplantation site for reference when embedding tissues for sectioning.
Clinical Chemistry, Hematology, and Histologic Evaluation
RNU rats were sacrificed 1, 3, and 6 months following subretinal transplantation via CO2 inhalation followed by cervical dislocation. As indicated in Table 1, 10 rats (five male and five female) were used for each of the three posttransplant survival time points evaluated in this study. To allow for possible differences between patient-derived donor cells, four different patient iPSC lines and resulting retinal progenitor cells were used in this study for a total of 120 rats receiving iPSC-derived cells on scaffolds. Scaffold-only-injected animals were used as controls (N = 30, five male and dive female for each of the three survival time points, that is, 1, 3, and 6 months postsurgery). To evaluate systemic effects, blood was collected at sacrifice for hematology (Table 2) and clinical chemistry analyses (Table 3). Gross pathology was performed at necropsy, and organ weights and photos were collected. Finally, the organs listed in Table 4 were collected, embedded in paraffin, sectioned, stained with hematoxylin and eosin (H&E), and evaluated by a board-certified veterinary pathologist.
DTVRF-hiPSC-ARG Study Design.

3D Scaffold (vehicle control): animals received a single 1-mm cell delivery scaffold, which are used to construct DTVRF-hiPSC-ARG, via subretinal transplantation without any human induced pluripotent stem cells-derived retinal progenitor cells loaded onto it. DTVRF-hiPSC-ARG: animals received a single 1-mm retinal cell graft via subretinal transplantation that contained induced pluripotent stem cells-derived photoreceptor precursor cells generated from four independent individuals, one with no disease and three with rod-selective photoreceptor cell disease (ie, each of these individuals have normal cone cells).
List of Hematology Tests Performed at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

List of Clinical Chemistry Tests Performed at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

HDL: high-density lipoprotein; LDL: low-density lipoprotein.
List of Tissues Weighed and Evaluated at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

Results
DTVRF-hiPSC-ARG Fabrication
PCL triol (Mn ~ 300 g/mol) was functionalized via mixing with potassium carbonate and acryloyl chloride at a ratio of 1:3:3 as described in section “Materials and Methods.” To generate PCL-cell-delivery scaffolds, functionalized PCL was injected into a custom polymerization mold and exposed to UV light at an intensity approximately 100 lumens/cm2. The polymerization chamber consists of two glass slides separated by two 50-µm spacers placed approximately 2.5 cm apart (Fig. 2A). The upper slide was covered by a photomask containing 75-µm black spots spaced at 25-µm intervals with adjacent spots aligned in a hexagonal pattern (Fig. 2B). The photomask was designed to block UV light and prevent polymerization, thereby creating pores in the resulting polymerized scaffold. Following polymerization, the chamber was disassembled, and the polymerized scaffold was washed to remove residual unpolymerized prepolymer. Scaffolds were subsequently sterilized via gamma irradiation (25,000 Gray for approximately 18 h) before being returned to a cGMP compliant BioSpherix cell culture system for seeding with patient iPSC-derived retinal progenitor cells (iPSC-RPCs). As determined via both confocal microscopy (Fig. 2C) and scanning electron microscopy (Fig. 2D), the resulting scaffolds were 50-µm thick and contained full-thickness pores that were 60 µm in diameter.

Fabrication of polycaprolactone cell delivery scaffolds via ultraviolet polymerization. (A) The scaffold fabrication chamber. (B) Phase micrograph of the photomask used to create pores in the polycaprolactone cell delivery scaffold. This photomask can be seen as a gray square denoted by blue brackets on the upper layer of the fabrication chamber shown in panel A. Confocal (C) and scanning electron (D) micrographs of the polycaprolactone scaffold following fabrication. Scale bar = 100 µm (B), 50 µm (C), and 1 mm (D).
Following sterilization, PCL scaffolds were coated with Laminin 521 and seeded with patient iPSC-derived retinal progenitor cells harvested from retinal organoids differentiated for approximately 80 days as described in section “Materials and Methods.” At this point in time, retinal organoids largely contain retinal progenitor cells that express markers such as SOX2 and CHX10 (Fig. 3A, B). If differentiated for an additional 70 days, these cells give rise to both NR2E3-, NRL-, and rhodopsin-positive rod photoreceptor cells and ARR3- and blue opsin-positive cone photoreceptor cells (Fig. 3C–E). As shown in Fig. 3F, 14 days following seeding (ie, ~94 days post-differentiation) scaffolds were densely packed with iPSC-RPCs. At this time point, approximately 70% of the cells expressed the photoreceptor precursor cell markers OTX2 (Fig. 3G, H) and Recoverin (Fig. 3I, J). Although some retinal progenitor cells did adhere to the surface of the polymer, most of the cells were located within the 60-µm vertical pores of the scaffold (Fig. 3J).
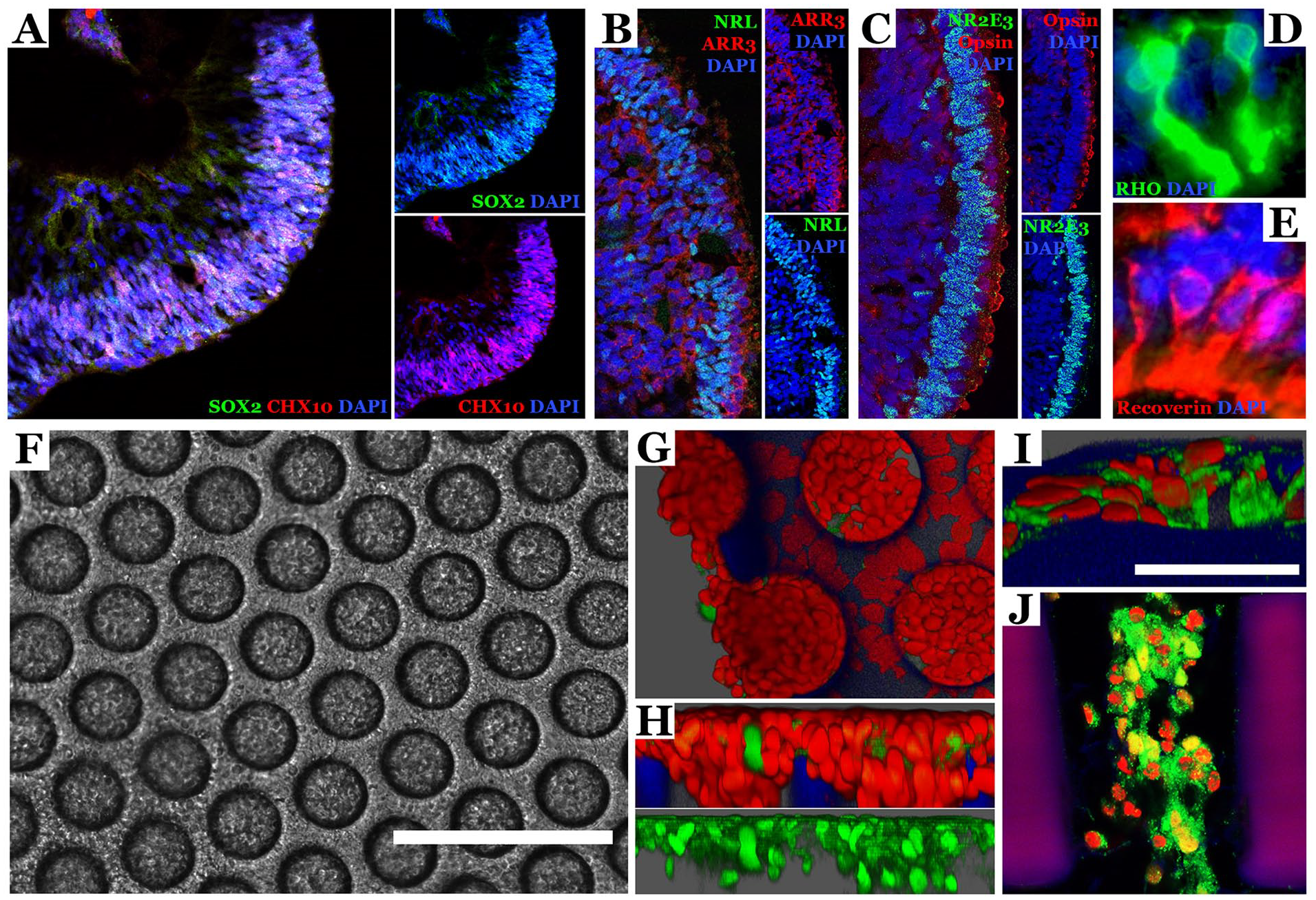
Generation of DTVRF-hiPSC-ARGs via seeding of PCL scaffolds with patient iPSC-derived retinal progenitor cells. (A–E) Immunocytochemical staining of hiPSC-derived retinal organoids at differentiation days 80 (A—time of dissociation and scaffold seeding) and 150 (B–E). Antibodies targeting the retinal progenitor cell markers SOX2 and CHX10, the rod photoreceptor cell markers NR2E3, NRL, and rhodopsin, and the cone photoreceptor cell markers ARR3 and blue cone opsin. DAPI was used as a nuclear counterstain. (F) Phase micrograph of DTVRF-hiPSC-ARG 2 weeks following seeding. Note the uniform seeding of pores across the entire scaffold. (G–J) Immunocytochemical staining of DTVRF-hiPSC-ARG 2 weeks following seeding. Antibodies targeting OTX2 (G and H—green) and Recoverin (I and J—green) were used to confirm the presence of photoreceptor precursor cells. Red = Sytox™ nuclear stain. Scale bar = 200 µm (F) and 50 µm (I). The scaffold material autofluoresces blue-magenta under these conditions. PCL: polycaprolactone; iPSC: induced pluripotent stem cells; hiPSC: human induced pluripotent stem cell.
Subretinal Transplantation and Analysis of DTVRF-hiPSC-ARG Tolerability
To evaluate local and systemic compatibility of DTVRF-hiPSC-ARG, 1-mm grafts were transplanted into the subretinal space of immunodeficient RNU rats as described in section “Materials and Methods.” As shown in Table 1, 10 rats (five male and five female) were sacrificed at each of three posttransplant survival times (1, 3, and 6 months, N = 30 per patient for a total of 120 rats). Scaffold only transplanted animals were used as vehicle controls (N = 30, that is, five male and five female, each sacrificed at 1, 3, and 6 months postsurgery). Prior to sacrifice, a clinical ophthalmic exam was performed, and clinical findings were recorded. Unfortunately, RNU rats are prone to developing prolapsed eyelashes and corneal clouding with age, and as a result a clear view of the fundus was not possible for the majority of the animals 3 and 6 months following transplant. However, in cases where the cornea was clear, no evidence of iris synechiae (suggestive of anterior chamber inflammation), vitreous haze or opacities (consistent with posterior chamber inflammation), retinal detachment, or chorioretinal atrophy was identified. As shown by both fundus photography (Fig. 4A) and OCT (Fig. 4B), 1 month following transplant, the host retina had reattached, and no evidence of abnormal donor cell proliferation, tumor formation, or toxicity to adjacent retinal tissue was detected. To augment these clinical data an additional small cohort of rats (n = 3) was sacrificed 6 months following transplant and the eyes were harvested and subjected to histological examination in addition to the animals subjected to detailed histopathologic study as part of the original study design.

Local tolerability of DTVRF-hiPSC-ARG. Fundus photo (A) and optical coherence tomogram (B) 1 month following subretinal placement of DTVRF-hiPSC-ARG in an RNU rat. (C–E) H&E staining of RNU rat eye at 6 months following subretinal placement. Note the intact photoreceptor cell outer nuclear layer, inner segments, and outer segments adjacent to the polymer (D). Scale bar = 100 µm. White arrows denote full-thickness pores in the PCL scaffold. Black arrowheads denote the RPE cell layer. Black * denotes photoreceptor inner and outer segments. Blue dashed box denotes polymer scaffold. RNU: Rowett Nude; PCL: polycaprolactone; RPE: retinal pigmented epithelial; CC: choriocapillaris; GCL: ganglion cell layer; IPL: inner plexiform layer; INL: inner nuclear layer; OPL: outer plexiform layer; ONL: outer nuclear layer; IS/OS: photoreceptor cell inner and outer segment layer; IS: inner segments; OS: outer segments; S: scaffold.
As shown in Fig. 4C, while the outer nuclear layer (ONL) immediately above the implant (marked by blue dashed square) was disrupted as one would expect with transplantation into a non-diseased model, the host retina adjacent to the implant was normal. Specifically, the outer nuclear layer, photoreceptor cell inner and outer segments (Fig. 4C black *, D), RPE cell layer (Fig. 4C, arrowheads) and choroid remained intact. In fact, the host RPE cell layer and choroid were preserved immediately beneath the implant (Fig. 4C, E). Finally, cells were detected both on top of the scaffold and within the vertical pores, which as depicted in Fig. 2 is precisely where they were seeded (Fig. 4E, arrows). Collectively, these data demonstrate that 3D human iPSC-derived retinal progenitor cell grafts (ie, DTVRF-hiPSC-ARG) are well tolerated locally.
To evaluate systemic tolerability, blood was collected at the time of sacrifice for hematology and clinical chemistry. Organs were removed, carefully examined, weighed, fixed, and paraffin embedded. All tissues, including the head with eyes, were subsequently sectioned, H&E stained and subjected to detailed histopathology by a veterinarian pathologist. While sporadic differences were detected in several of the hematology (Table 5, significant differences highlighted) and clinical chemistry (Table 6) parameters, no evidence of DTVRF-hiPSC-ARG induced toxicity was detected at 1-, 3-, or 6-month post-subretinal transplant. Likewise, no evidence of tumor formation or DTVRF-hiPSC-ARG induced toxicity was detected in any animal via gross pathology (Table 7). Similarly, as shown in Table 8, while the thymus of 1-month female and the pancreas of 3-month female animals that received P3 were significantly heavier than those of vehicle control animals at the same posttransplant time point, no donor cell proliferation, tumor formation, or other histopathologic abnormalities were seen in these organs. In fact, no evidence of tumorigenicity, tissue necrosis, or toxicity was detected in any of the animals studied with detailed histopathology (Table 9). Collectively, these data demonstrate that DTVRF-hiPSC-ARG is well tolerated systemically for up to 6 months following transplant.
CBC Analysis at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

CBC: complete blood count, RBC: red blood cell, HGB: hemoglobin, HCT: hematocrit, MCV: mean corpuscular volume, MCH: mean corpuscular hemoglobin, MCHC: mean corpuscular hemoglobin concentration, RDW-CV: red cell distribution width, PLT: platelet, WBC: white blood cells, RET: reticulocyte, NEUT: neutrophil, LYMPH: lymphocyte, MONO: monocyte, EO: eosinophil, BASO: basophil.
*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Clinical Chemistry Analysis at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Cell Grafts in Immunocompromised Rowett Nude Rats.

HDL: high-density lipoprotein; LDL: low-density lipoprotein.
*P < 0.05, **P < 0.01, ***P < 0.001.
Summary of Histopathology Findings (Gross) at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

(—): not observed in any animals; N/A: not applicable.
Animal Organ Weights at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

Summary of Histopathology Findings (Microscopic) at 1, 3, and 6 Months Following Subretinal Transplantation of Engineered Human Retinal Progenitor Cell Grafts in Immunocompromised Rowett Nude Rats.

(—): not observed in any animals; N/A: not applicable.
Discussion
There is growing evidence that pluripotent stem cell-derived retinal progenitor cells can restore retinal function following subretinal transplantation2–15, and as a result, the field is rapidly progressing toward human clinical trials. However, before initiating such human trials, it is essential to demonstrate that the proposed treatment has a favorable safety profile in preclinical studies. A concern often associated with the use of pluripotent stem cells for clinical human cell replacement therapy is the potential for residual undifferentiated stem cells within the transplant to form tumors known as teratomas that contain cells of all three embryonic germ layers. Although teratomas are benign and the likelihood of undifferentiated cells leaving the eye and giving rise to tumors in extraocular tissues is very low, the possibility of forming a mass within the eye is a real concern 2 . Fortunately, retinal differentiation protocols have evolved to the point where contaminating pluripotent stem cells is rare48,52–59. In addition, stringent release testing strategies designed to detect cells with tumorigenic potential prior to transplantation have been developed48,60. Also, one of the advantages of performing the first in human clinical trials in the eye is that the transplant can be safely and inexpensively observed with biomicroscopy and optical coherence tomography at frequent intervals following transplantation. In the unlikely event that a tumor did form within the eye, it could be easily destroyed with laser photocoagulation as is done routinely for the small retinal hemangioblastomas associated with von Hippel Lindau disease61,62. Additional safety concerns with polymer-supported retinal cell grafts are the interaction between the host tissue, the scaffold, and the polymeric degradation products. Specifically, it is important to demonstrate that the transplanted polymer does not induce local tissue damage and that as it degrades, its degradation products do not injure the adjacent retina or other organs as the degradation products find their way into the systemic circulation. This is especially important given that polymers that undergo rapid bulk degradation, such as poly lactic-co-glycolic acid (PLGA), have been shown to significantly lower local pH and cause widespread retinal injury when delivered in sufficient quantities 63 .
When selecting an animal model for evaluating the safety of a new 3D polymer-supported retinal cell graft, several considerations were made. First, the animal needed to have an eye that is large enough to receive a human equivalent dose of patient iPSC-derived retinal progenitor cells. Second, the animal needed to be immunocompromised to prevent acute xenograft rejection following transplantation. In previous polymer-scaffold-only studies, we utilized the Pro23His rhodopsin transgenic pig model of retinal degeneration to evaluate local and systemic tolerability 37 . Pig eyes are similar in size to humans and are thus ideal for translational research, especially for refining surgical techniques and instrument design 37 . Unfortunately, pigs are expensive to house in large numbers and difficult to immunosuppress for extended periods of time, which makes them less than ideal for evaluating the systemic tolerability of a human cell product. Although many mouse models of inherited retinal degeneration exist, including those that have been crossed onto immunocompromised backgrounds 64 , the mouse eye is too small to reliably place polymer-supported photoreceptor cell grafts into the subretinal space 64 . Unlike pigs, rats are inexpensive to house in sufficient numbers to allow for testing of multiple experimental parameters at once; and unlike mice, rats have eyes that are large enough to enable subretinal transplantation of a biodegradable retinal cell graft. Rat models of retinal degeneration have been used as recipients of full-thickness retinal sheets, polymer support RPE cell grafts, and light-sensing photovoltaic arrays6,50,51,65. In addition to being of sufficient size to receive a 3D subretinal graft, the rat also has a sufficient volume of blood to allow for a comprehensive hematology and clinical chemistry analysis in each animal following transplantation. For these reasons, the athymic nude RNU rat, which lacks functional T cells, was chosen for the studies described above.
Although the RNU rat can receive human cells without xenograft rejection, this animal is prone to developing conjunctivitis and corneal opacification 66 . In this study, we found that corneal clouding was independent of treatment. It occurred in sham, vehicle and treatment groups and was most often associated with prolapsed eyelashes and cage bedding embedded beneath the eyelid. Unfortunately, corneal opacification prevented in vivo clinical evaluation beyond 3 months of follow-up. In addition to the corneal clouding disadvantage, the RNU rat has a normal retina and as such is not useful for evaluating treatment efficacy.
For studies designed to evaluate treatment efficacy, immunocompromised retinal degenerative rats would be ideal. To that end, in 2014 Seiler and colleagues 46 reported production of an immunocompromised rat model of retinitis pigmentosa that was generated by crossing the RNU rat with the retinal degenerative rhodopsin s334ter rat 67 allowing the transplantation of human embryonic stem cell–derived neural progenitor cells that survived in its subretinal space for more that 180 days following transplant 67 . In 2018, Thomas and colleagues reported production of a similar model by crossing the RNU rat with the Royal College of Surgeons (RCS) Mertk mutant rat 68 , which also accepted human subretinal xenografts without immune rejection 68 . However, the lack of commercial availability of these animals precluded their use for this large toxicity study.
Among the remaining translational challenges for the development of photoreceptor cell replacement therapy is the study of factors affecting cell survival and synaptic integration. Because these factors involve delicate interactions between the transplanted cells and host retina, these experiments are best carried out in vivo using animal models of retinal degeneration. Although immune suppressed backgrounds that support xenografts may be suitable for this purpose, an alternative approach would be to develop strain specific rat iPSC lines69–72 that could be transplanted as autologous grafts in the absence of immunosuppression. However, there is no guarantee that results derived from such animal models would directly translate to adult humans. Thus, the field will need to weigh the potential risk of initiating human clinical trials with imperfect efficacy data against the potential benefits that would be afforded should blind patients regain some useful vision. We believe that the favorable safety profile presented here provides compelling support for initiating a human clinical safety trial in patients with advanced disease and very poor vision. Such safety trials could be performed in parallel with continued in vitro and animal research focused on enhancing graft axon extension and promoting host-donor synaptic integration.
Footnotes
Ethical Approval
Approval to obtain dermal fibroblast cells and generate iPSCs from patients with inherited retinal degeneration and normal controls was obtained under informed consent. This study was reviewed and approved by the University of Iowa Institutional Review Board (IRB# 200202022).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the University of Iowa’s Institutional Review Board (IRB# 200202022), the University of Iowa’s Animal Care and Use Committee (animal welfare assurance #8051317) and the ARVO statement for the use of animals in ophthalmic and vision research.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article (IRB# 200202022).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Iowa Institute for Vision Research, University of Iowa, Iowa City, IA, USA; the Elmer and Sylvia Sramek Charitable Trust, Research to Prevent Blindness; and National Institutes of Health Grant (NIH; Bethesda, MD, USA): R01 EY 024605 and P30 EY025580.
