Abstract
Glucocorticoid (GC)-induced avascular osteonecrosis of femoral head (AOFH) is a devastating complication, and no cures are currently available for it. Previous studies have demonstrated that implantation of bone marrow mesenchymal stem cells (BMMSCs) may prevent the progression of pre-collapse AOFH. Based on previous observations, we hypothesized that GCs induce AOFH via the COX-2 (cyclooxygenase-2)–PGE-2 (prostaglandin E2)–HIF-1α (hypoxia-inducible factor-1α) axis, and that modification of BMMSCs may improve the efficacy of their implantation. BMMSCs isolated from wild-type (WT) mice were treated with dexamethasone (Dex) and the results showed that Dex repressed the expression of COX-2. Femoral head samples harvested from both WT and COX-2 knock-out (COX-2−/−) mice were subjected to micro–computed tomography and histological examinations. Compared with their WT littermates, COX-2−/− mice had larger trabecular separations, diminished microvasculature, and reduced HIF-1α expression in their femoral heads. In vitro angiogenesis assays with tube formation and fetal metatarsal sprouting demonstrated that Dex repressed angiogenesis and PGE-2 antagonized its effects. An AOFH model was successfully established in C57BL/6J mice. In vitro experiment showed that BMMSCs infected with Lentivirus encoding HIF-1α (Lenti-HIF-1α) resulted in a robust increase in the production of HIF-1α protein. Implantation of BMMSCs overexpressing HIF-1α into femoral heads of AOFH mice significantly reduced osteonecrotic areas and enhanced bone repair, thus largely preserving the structural integrity of femoral heads. Our studies provide strong rationales for early intervention with core decompression and implantation of modified BMMSCs for GC-induced AOFH, which may spare patients from expensive and difficult surgical procedures.
Introduction
Glucocorticoids (GCs) are the most commonly prescribed drugs for autoimmune rheumatic diseases (ARDs). However, some devastating complications, such as avascular osteonecrosis of femoral head (AOFH), may occur. Initial and accumulated doses are important factors for AOFH formation 1 , and primary types of ARDs are also associated with its pathogenesis. For example, the incidence of AOFH is higher in patients with systemic lupus erythematosus (SLE), possibly due to vasculopathy 2 . It is estimated that newly diagnosed AOFH cases are over 8 million annually in China, and most of these patients had a history of GC usage and a female predominance has been observed 3 . While the mechanisms underlying the pathogenesis of GC-induced AOFH are not completely understood, several factors may hold responsible, including impaired angiogenesis, osteogenesis, adipogenesis, and so on4,5. Femoral heads gradually collapse, leading to the destruction of hip joints, at which time the treatment of choice is total hip arthroplasty (THA). However, major concerns remain because a large majority of patients are relatively young and active, and the quality of bone is poor, which often lead to unacceptably high rate of aseptic loosening of prostheses, and revision arthroplasty is needed6,7. Common interventions for pre-collapse AOFH include core decompression, osteotomy, bone grafting, and so on. Nevertheless, the treatment outcomes are not satisfactory8–10. Nonoperative treatments cannot prevent AOFH progression 11 . Therefore, it is imperative to develop effective treatments to halt AOFH progression in its early stage to avoid costly and technically challenging surgical procedures.
Emerging data suggest a promising role of bone marrow mesenchymal stem cells (BMMSCs) in the treatment of GC-induced AOFH progression12,13. BMMSCs transplanted in osteonecrotic femoral heads under hypoxic conditions can survive, proliferate, and differentiate into osteoblast to promote osteogenesis 14 . In vitro exposure of BMMSCs to hypoxia results in an upregulation of vascular endothelial growth factor (VEGF), which is vital for neoangiogenesis of osteonecrotic bone15,16. Enhanced osteogenesis and angiogenesis render BMMSCs therapeutic potentials for early-stage AOFH. A meta-analysis indicates that autologous BMMSC implantation to core decompression areas may relieve pain and prevent femoral head collapse of AOFH patients 17 . Despite these promising results, BMMSC implantation therapy has its drawbacks, notably, the limited supply of BMMSCs18,19. Increasing the osteogenic and angiogenic potentials of BMMSCs through rapidly evolving techniques of gene therapy is expected to improve the treatment outcome of AOFH.
GCs such as dexamethasone (Dex) have long been used as anti-inflammation drugs, partially via their inhibition of cyclooxygenase-2 (COX-2) expression and activity. The major product of COX-2, namely, prostaglandin E2 (PGE-2), is closely involved in skeletal repair 20 . GC treatment significantly inhibits COX-2 expression 21 . GC-induced leucine zipper is a transcription factor that can repress COX-2 expression in mouse BMMSCs 22 . In addition, COX-2 and PGE-2 are linked to angiogenesis23–25. Angiogenesis is critical for bone development and regeneration through increasing bone remodeling units and providing a conduit for vessel-derived factors required for bone formation 26 .
PGE-2 increases the expression of hypoxia-inducible factor-1α (HIF-1α) under normoxic conditions. Nuclear translocation of HIF-1α is necessary for the expression of many hypoxia-regulated genes, including VEGFs 27 . Importantly, HIF-1α can coordinate osteogenesis and angiogenesis 28 . VEGFs play a central role in neoangiogenesis. HIF-1α enhances the expression of VEGF in BMMSCs and chondrocytes29,30. RUNX-2 is a pivotal transcription factor for osteoblast formation and differentiation, and it can be regulated by HIF-1α31,32. All pathophysiological processes, namely, hypoxia, angiogenesis, and osteogenesis, exist in the pathogenesis of AOFH, which leads to our central hypothesis that GCs induce AOFH through the COX-2–PGE-2–HIF-1α axis in BMMSCs, and that modification of certain molecules in this axis may result in enhanced efficacy of BMMSC implantation.
Our current study aimed to clarify the role of COX-2–PGE-2–HIF-1α axis in BMMSCs during the process of Dex-induced AOFH in mice. Our results showed that Dex repressed the expression of COX-2, leading to reduced production of PGE-2. Subsequently, PGE-2-mediated HIF-1α expression was downregulated, which impaired both osteogenesis and angiogenesis. We successfully established an AOFH model in C57BL/6J mice by adding Dex to the drinking water. Implantation of BMMSCs overexpressing HIF-1α resulted in significant shrinkage of osteonecrotic areas. Our study paves a new way for AOFH treatment although different modifications of BMMSCs may be used to maximize the treatment efficacy and preserve the integrity of femoral heads.
Materials and Methods
Mice
The following mice were used in this study: wild-type (WT) mice with C57BL/6J background. Global gene knock-out mice included COX-2−/−33, EP1 receptor knock-out (EP1−/−),34,35 EP2 receptor knock-out (EP2−/−)34,35, and EP4 receptor floxed (EP4flox/flox) mice 36 . Green fluorescent protein (GFP) transgenic mice were purchased from Jackson Laboratories (Farmington, CT, USA). All of the experimental procedures involving mice were approved by the Ethics Committee of Zhengzhou University First Affiliated Hospital, Zhengzhou, China (approval number: 2021050604).
Viruses
Adenoviruses and lentiviruses encoding different genes were purchased from Vector Biolabs (Palo Alto, CA, USA) and Origene Technologies, Inc. (Rockville, MD, USA).
Isolation and Culture of BMMSCs From Mice
Bone marrow cells were isolated from WT C57BL/6J, COX-2−/− mice, EP1−/−, EP2−/−, EP4flox/flox, and GFP transgenic mice. Flow cytometry was conducted to purify BMMSCs using surface markers such as CD105, CD166, and CD29. The purified BMMSCs were cultured in α-MEM containing 15% fetal bovine serum (FBS).
Western Blotting, and Immunohistochemical and Immunofluorescent Staining
Different cells were lysed with Gold lysis buffer, and Western blotting was performed with different antibodies: COX-2 (Cayman Chemicals, Ann Arbor, MI, USA), HIF-1α, and CD31 (Abcam, Cambridge, MA, USA). Immunofluorescent and immunohistochemical staining were done with a monoclonal rabbit anti-mouse antibody to HIF-1α using previously described methods37,38.
Mouse Model of AOFH
WT C57BL/6J mice (12 weeks old) were used for this experiment. Dex was added to the drinking water (5 mg/l). Trace amount of dimethyl sulfoxide (DMSO) was added to the drinking water of control group mice. The continuous treatment lasted for 16 weeks. Mice were anesthetized with an intraperitoneal injection of ketamine (60 mg/kg) and xylazine (4 mg/kg) and examined in a Faxitron Cabinet X-ray system (Faxitron X-ray Corporation, Lincolnshire, IL, USA). The results showed that approximately 60% of mice treated with Dex developed osteonecrotic lesion in femoral heads. BMMSCs (10 μl, 5 × 105 cells) infected with either Lenti-HIF-1α or Lenti-GFP were injected into the femoral head. Pilot study showed that the efficacy peaked 2 weeks after local injection of BMMSCs. Thus, we collected femoral head samples at this time point.
In Vitro Angiogenesis Assay
Human umbilical vein endothelial cells (HUVECs) were seeded at 2 × 105 viable cells per 75 cm2 tissue culture flask using Medium 200PRF. Single-cell suspensions were harvested after the following treatments: vehicle control, Dex (10−6 μM), PGE-2 (10−6 μM), and Dex plus PGE-2. These cells were placed onto the Geltrex-coated six-well plates. After culturing at 37°C for 72 h, Calcein was added and tube formation was assessed under fluorescent microscope.
Fetal Metatarsal Sprouting
Fetal metatarsals isolated from E17.5 mice were cultured in 24-well plates in α-MEM containing 15% FBS for 3 days. Then, the medium was replaced by fresh medium containing Dex, PGE-2, or both. The medium was changed every 3 days and the treatment lasted for 14 days. Immunohistochemical staining was then performed with an antibody against PECAM (CD31), and in vitro neoangiogenesis was evaluated under microscope by measuring metatarsal sprouting.
Micro–Computed Tomography (μCT) Scanning and Three-Dimensional Reconstruction
Mouse femoral head samples were harvested at indicated times and scanned using a Viva micro-CT system (Scanco Medical, Bassersdorf, Switzerland) at a voxel size of 10.5 μm to image bone or vasculature. For all scans, a global threshold was applied to separate mineralized and soft tissues and kept consistent within each analysis region. For morphometric analyses of trabecular bone, a direct three-dimensional (3D) model was used to determine trabecular separations.
Results
Dex Inhibited COX-2–PGE-2–HIF-1α Axis in Mouse BMMSCs
BMMSCs isolated from WT mice were treated with Dex of different doses for 24 h. Cell lysates were subjected to Western blotting analysis with a rabbit anti-mouse COX-2 antibody. The results demonstrated that Dex inhibited COX-2 protein expression in a dose-dependent manner (Fig. 1A). As PGE-2 is a major effector of COX-2 enzymatic activity, we then examined the effect of PGE-2 on HIF-1α expression. BMMSCs were treated with PGE-2 of different concentrations for 24 h. Cell lysates were subjected to Western blotting analysis with a rabbit anti-mouse HIF-1α antibody. The results showed that PGE-2 dose-dependently upregulated the expression of HIF-1α protein (Fig. 1B). HIF-1α is a transcription factor and may regulate certain gene expression in the nucleus. Thus, we treated BMMSCs with PGE-2 and monitored nuclear translocation of HIF-1α. The results demonstrated that PGE-2 induced its nuclear transcription (Fig. 1C). Primary BMMSCs isolated from either WT or COX-2−/− mice were cultured under normoxic (20% O2) or hypoxic (5% O2) conditions for 24 h. While hypoxia significantly increased the expression of HIF-1α protein in WT BMMSCs, no significant change was detected in COX-2−/− BMMSCs, suggesting that COX-2 deficiency impaired BMMSC response to hypoxia regarding HIF-1α expression (Fig. 1D).
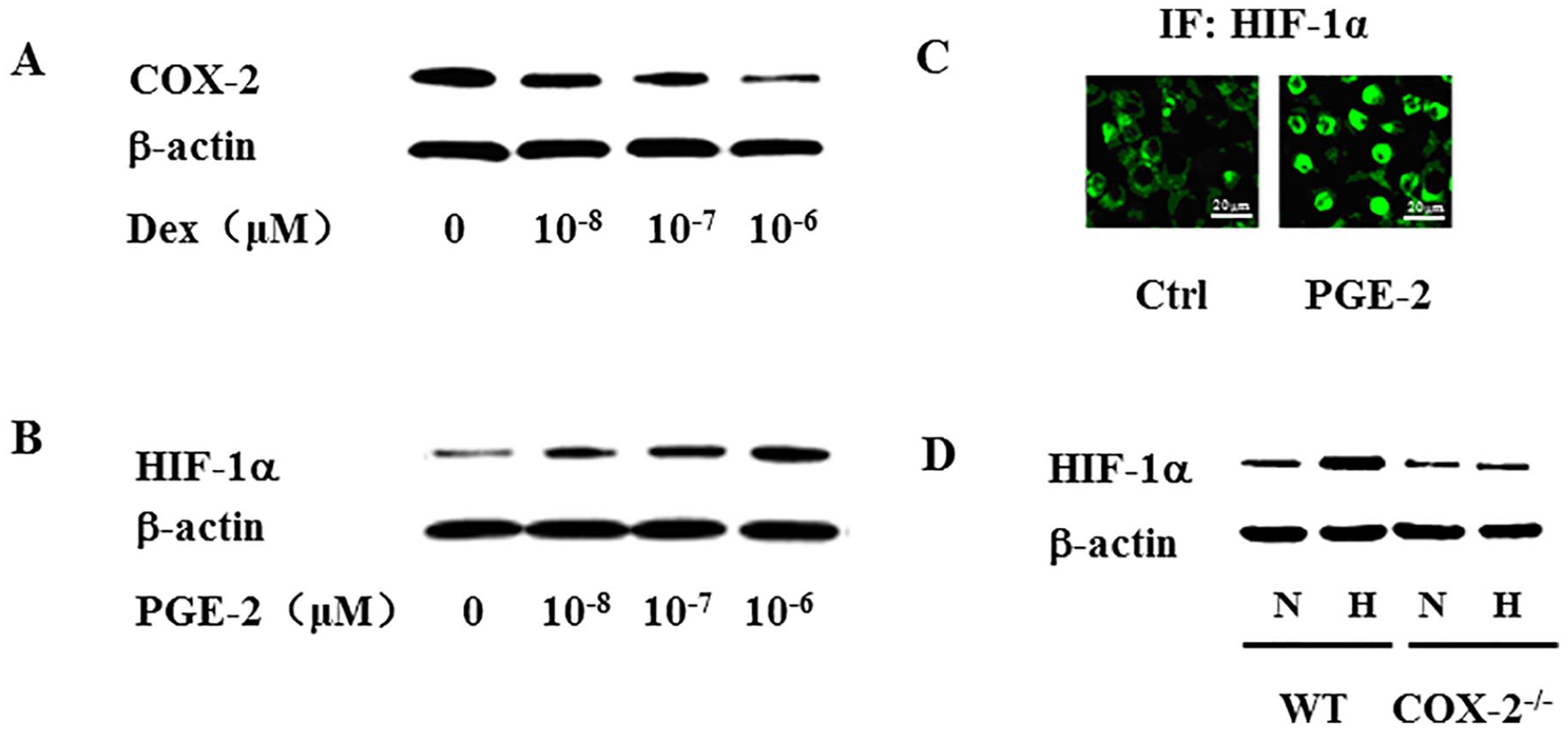
Mouse BMMSCs were cultured and treated with Dex of different doses for 24 h, and cell lysates were subjected to Western blotting analysis. The results showed that Dex dose-dependently repressed COX-2 expression (A). Mouse BMMSCs were treated with PGE-2, and Western blotting analysis showed that PGE-2 upregulated HIF-1α expression in a dose-dependent manner (B). Immunofluorescence staining showed that PGE-2 induced nuclear translocation of HIF-1α (scale bar = 20 μm) (C). BMMSCs isolated from either WT or COX-2 knock-out (COX-2−/−) mice were cultured in normoxic or hypoxic conditions for 24 h. Western blotting analysis showed that COX-2 deficiency resulted in decreased responsiveness to hypoxia (D). BMMSCs: bone marrow mesenchymal stem cells; COX-2: cyclooxygenase-2; PGE-2: prostaglandin E2; HIF-1α: hypoxia-inducible factor-1α; WT: wild type; Dex: Dexamethasone; μM: micromole; Ctrl: vehicle control; N: normoxic; H: hypoxic; IF: immunofluorescence.
PGE-2 Increased HIF-1α Expression Mainly Through Its EP4 Receptor
PGE-2 takes its effect by binding to its different receptors including EP1, EP2, EP3, and EP4. As EP3 mediates a negative effect of PGE-2, our study focused on the other three receptors. BMMSCs were isolated from EP1−/−, EP2−/−, and EP4flox/flox mice. While BMMSCs isolated from EP1−/− and EP2−/− mice were treated with PGE-2 directly for 24 h, BMMSCs from the EP4flox/flox mice were first subjected to in vitro gene deletion of EP4 receptor through infection of adenovirus encoding Cre (Ad-Cre), with Ad-GFP as the infection control. While PGE-2 largely preserved its ability to upregulate HIF-1α expression in the BMMSCs from EP1−/− and EP2−/− mice (Fig. 2A, B), deletion of EP4 significantly abrogated PGE-2-mediated HIF-1α expression (Fig. 2C).

Mice with different genetic backgrounds were divided into three groups (n = 8 per group): EP1 receptor knock-out (EP1−/−) mice, EP2 receptor knock-out (EP2−/−) mice, and EP4 receptor floxed (EP4flox/flox) mice. BMMSCs were isolated from these mice and cultured cells were treated with PGE-2 (10−6 M) for 24 h. Cell lysates were subjected to Western blotting analysis with an antibody against HIF-1α. After treatment of BMMSCs from EP1−/− mice or EP2−/− mice with PGE-2, Western blotting analysis showed that the deficiency of these two receptors did not affect PGE-2-induced HIF-1α expression (A and B). BMMSCs isolated from EP4flox/flox mice were infected with Adeno-CRE, with Adeno-GFP as the infection control. The results showed that deletion of EP4 receptor in vitro resulted in the loss of responsiveness to PGE-2 stimulation regarding HIF-1α expression (C). BMMSCs: bone marrow mesenchymal stem cells; PGE-2: prostaglandin E2; HIF-1α: hypoxia-inducible factor-1α; BMSC: bone marrow stem cell; GFP: green fluorescent protein.
Pathological Changes in the Femoral Head of COX-2−/− Mice
Chinese ink was injected into the femoral heads of WT and COX-2−/− mice via greater trochanter. The femoral head samples were collected 6 h later for cryostat sections. Microscopic examination revealed that the sinusoid sizes were smaller in COX-2−/− mice compared with their WT littermates (Fig. 3A). Femoral head samples from WT and COX-2−/− mice were subjected to μCT scanning, and the results after 3D reconstruction showed that trabecular separations were increased in COX-2−/− mice compared with their WT littermates (Fig. 3B). Immunohistochemical staining was performed with a rabbit anti-mouse HIF-1α antibody, and the results demonstrated that the number of HIF-1α-positive cells was decreased in COX-2−/− femoral heads compared with WT ones (Fig. 3C).

WT and COX-2−/− mice were sacrificed and Chinese ink was injected into the femoral heads via greater trochanter. Femoral head samples were cut into 10 μm pieces in a cryostat machine, and the results showed that the sinusoid sizes were smaller in COX-2−/− mice than in their WT littermates (n = 8 per group) (scale bar = 400 μm) (A). Femoral head samples harvested from either WT or COX-2−/− mice were subjected to μCT scanning and IHC staining with an HIF-1α antibody. The results from μCT scanning and reconstruction showed that trabecular separations were larger in COX-2−/− mice than in their WT littermates (n = 8 per group) (scale bar = 1mm) (B). IHC staining showed that the number of HIF-1α-positive bone marrow cells was smaller in COX-2−/− mice than in their WT littermates (n = 8 per group) (scale bar = 200 μm) (C). WT: wild type; COX-2−/−: cyclooxygenase-2 mutant; μCT: micro–computed tomography; IHC: immunohistochemistry; HIF-1α: hypoxia-inducible factor-1α.
PGE-2 Partially Reversed Dex-Mediated Inhibition of Angiogenesis
HUVECs were used for in vitro angiogenesis assay. These cells were cultured in gel-coated dishes and treated with PGE-2, Dex, or both. Calcein was added to dishes and tube formation was evaluated under fluorescent microscope. The results demonstrated that Dex inhibited tube formation, while PGE-2 antagonized the Dex effect (Fig. 4A). Fetal metatarsals isolated from E17.5 mice were used for endothelial sprouting. Our results showed a robust inhibition of endothelial sprouting by Dex, while PGE-2 partially antagonized such effect (Fig. 4B).

HUVECs were used for in vitro angiogenesis (tube formation): After culturing on the gel in a cell culture incubator, these cells were treated with Dex, PGE-2, or both. Calcein was added to the cultured HUVECs after different treatments. The results showed Dex inhibited in vitro angiogenesis, while PGE-2 enhanced tube formation. In addition, PGE-2 antagonized the inhibitory effect of Dex on tube formation (scale bar = 300 μm) (A). Fetal metatarsal assay: Fetal metatarsals dissected from E17.5 mice were cultured in 24-well plates and treated with Dex, PGE-2, or both. At day 14, immunohistochemical staining was performed with an antibody against PECAM (CD31). The results showed Dex inhibited endothelial sprouting, while PGE-2 enhanced this process. In addition, PGE-2 antagonized the inhibitory effect of Dex on endothelial sprouting. D+P: dexamethasone plus PGE-2 (scale bar = 200 μm) (B). PGE-2: prostaglandin E2; PECAM: platelet endothelial cell adhesion molecule; HUVECs: human umbilical vein endothelial cells.
Establishing a Mouse AOFH Model
WT C57BL/6J mice were assigned to two groups: control and treatment groups. In the treatment group, Dex was added to the drinking water. The mice were subjected to radiographic examinations at weeks 4, 8, 12, and 16. Based on the incidence of osteonecrotic changes, the duration of treatment with Dex was decided to be 16 weeks. In the control group mice, trace amount of DMSO was added to the drinking water in micro-isolators as Dex was dissolved in DMSO. Femoral head samples were collected and subjected to μCT and histological evaluations. The results from 3D reconstruction after μCT scanning revealed the crescent signs (red arrow head) in femoral heads of mice from the treatment group (Fig. 5A). Histological examination demonstrated that Dex induced osteonecrosis of femoral heads as evidenced by red acellular areas (blue arrow head) and appearance of empty osteocyte lacuna (blue arrow) (Fig. 5B).
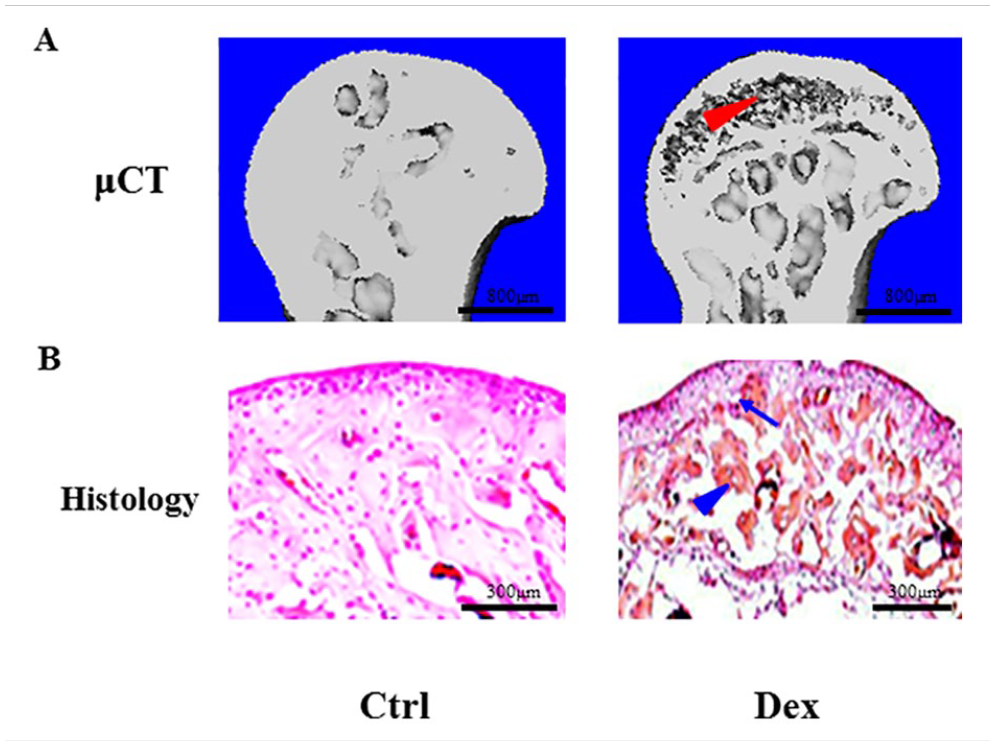
WT C57BL/6J mice (12 weeks, average 12 g) were assigned to two groups: control and treatment groups (n = 10 per group). In the treatment group, Dex (5 mg/l) was added to the drinking water, and the water containing Dex was supplied to this group of mice for 16 weeks. Then, femoral head samples were collected and subjected to μCT and histological evaluation. μCT scanning revealed the crescent signs in femoral heads from the treatment group mice (red arrow head) (scale bar = 800 μm) (A). Histological examination revealed that Dex caused osteonecrosis of femoral heads, which appeared as red-stained acellular areas (blue arrow head) and empty osteocyte lacuna (blue arrow) (scale bar = 300 μm) (B). WT: wild type; μCT: micro–computed tomography.
Implantation of BMMSCs Overexpressing HIF-1α Prevented the Progression of Dex-Induced AOFH
As it was technically challenging, we tried to insert the needles at different sites and angles, and we were successful when a 30-gauge needle was inserted via greater trochanter. Radiographic examination confirmed that the needle was placed at the center of the mouse femoral head (Fig. 6A). Next, we assessed the viability of the implanted cells in mouse femoral heads. BMMSCs isolated from GFP transgenic mice were injected to the femoral heads of WT C57BL/6J mice. Femoral head samples were harvested and the tissue sections from these samples were examined under fluorescent microscope. Our results showed that BMMSCs may survive and proliferate at least 7 days after implantation (Fig. 6B). The hematoxylin and eosin (H&E) staining picture corresponding to the fluorescent figure showed that the structural integrity of femoral head was largely preserved (Fig. 6C).

Radiographic examination confirmed that the 30-gauge needle can be inserted at the center of the mouse femoral head (scale bar = 10 mm) (A). BMMSCs isolated from GFP transgenic mice were injected into the femoral heads of WT C57BL/6J mice. Femoral head samples were harvested 5 days after injection, cut in cryostat, and examined under fluorescent microscope. The result showed that BMMSCs may survive in hypoxic condition (scale bar = 200 μm) (B). H&E staining showed that the structural integrity of femoral head was largely preserved after pin insertion (scale bar = 400 μm) (C). Mouse BMMSCs infected with Lenti-HIF-1α significantly increased the production of HIF-1α protein by BMMSCs (D). AOFH mice confirmed by in vivo μCT were injected with BMMSCs infected with Lenti-HIF-1α to femoral heads of these mice, and BMMSCs infected with Lenti-LacZ were used as control (n = 10 per group). The results from μCT examination showed that BMMSCs overexpressing HIF-1α reversed the osteonecrotic process of AOFH (scale bar = 800 μm) (E). The results were confirmed by histology (scale bar = 300 μm) (F). BMMSCs: bone marrow mesenchymal stem cells; WT: wild type; HIF-1α: hypoxia-inducible factor-1α; AOFH: avascular osteonecrosis of femoral head; μCT: micro–computed tomography; GFP: green fluorescent protein; H&E: hematoxylin and eosin.
As implantation of cells may increase the pressure inside femoral heads, only small number of BMMSCs may be injected. Thus, we tried to enhance the regenerative capability of BMMSCs by overexpressing HIF-1α. The reason we chose this molecule was that it may enhance osteogenesis and angiogenesis simultaneously. Western blotting analysis confirmed that Lenti-HIF-1α had a satisfactory efficacy of infection as evidenced by a robust increase in the expression of HIF-1α protein in BMMSCs (Fig. 6D). AOFH mice confirmed by in vivo μCT scanning were injected with BMMSCs infected with Lenti-HIF-1α at the center of femoral heads, and BMMSCs infected with Lenti-LacZ were used as control. The results from μCT examination showed that implantation of BMMSCs overexpressing HIF-1α reversed the osteonecrotic process of AOFH (Fig. 6E). The results were confirmed by histological examination (Fig. 6F).
Discussion
GCs are the most commonly prescribed drugs, particularly by rheumatologists. The incidence of GC-induced AOFH is relatively high in diffuse connective tissue diseases such as systemic lupus erythematosus (SLE) 2 . Currently, nonsurgical treatments are not so effective as to prevent or reverse the progression of osteonecrotic process. Thus, a large majority of patients will end up with complete joint destruction, and at that time, the only treatment of choice is THA. Core depression is a routine procedure for early AOFH patients. However, the outcome of such procedure is often suboptimal. Inspiring results were achieved by Gangji and colleagues when they treated AOFH patients with core depression and simultaneous implantation of autologous BMMSCs 39 . Additional studies have demonstrated that BMMSC implantation is a safe, effective, and feasible treatment for early-stage AOFH12,14,17.
The pathogenesis of GC-induced AOFH is multifaceted. Impaired osteogenesis due to inhibition of osteoblastic functions and reduced angiogenesis are critical factors for AOFH formation and progression. GCs exert anti-inflammatory effects partially via their inhibition of COX-2 expression and activity. COX-2 and its major product PGE-2 play an essential role in angiogenesis and skeletal repair20,23,25.
Because of ischemia and hypoxia in AOFH, HIF-1α may be activated 40 . Previous studies from Dr. Clemens group and other groups have demonstrated that HIF-1α is a pivotal molecule capable of coupling osteogenesis and angiogenesis during skeletal repair28,41. Based on these observations, we posited that GC-induced AOFH may be reversed by core depression combined with local implantation of BMMSCs overexpressing HIF-1α and confirm such a hypothesis may pave a new and effective way for clinical treatment of AOFH.
We first demonstrated that COX-2-deficient BMMSCs lost their response to hypoxic stimulation, suggesting a protective role of COX-2/PGE-2 for stem cells in a detrimental environment. Consistently, the number of HIF-1α-positive cells was significantly reduced in COX-2−/− mice, and PGE-2 upregulated HIF-1α expression and enhanced its nuclear translocation. Our results confirmed that PGE-2 took its effect on HIF-1α expression via the EP4 receptor in BMMSCs, which was different from EP1 receptor–mediated upregulation of HIF-1α in human embryonic kidney cells 42 . VEGF-A is a major downstream molecule regulated by HIF-1α 29 . In addition, a previous study demonstrates that HIF-1α upregulates the expression of RUNX-2, a pivotal transcription factor for osteoblast differentiation and osteogenesis 43 . Thus, we postulated that local implantation of BMMSCs overexpressing HIF-1α may create a microenvironment favorable for both osteogenesis and angiogenesis.
Although higher dose of Dex and longer period of treatment are needed compared to the report from Yang and colleagues 44 , we successfully established an AOFH model in C57BL/6J mice. As most genetically engineered mice are in C57BL/6J background, our model may be widely used in future study, including those using conditional knock-out of HIF-1α. It is convenient to add GCs into the drinking water compared with subcutaneous or intraperitoneal injection. μCT and histological evaluation demonstrated a satisfactory rate of Dex-induced AOFH and the number of AOFH mice was sufficient for the proposed studies. In our pilot experiments, we tried to treat AOFH with systemic application of BMMSCs by injecting them via tail vein. The results were disappointing as very few GFP-BMMSCs could enter the femoral heads of AOFH mice. While direct cell injection is technically challenging, we managed to inject Chinese ink into the center of femoral heads by inserting the needle at the greater trochanters. Then, we successfully implanted BMMSCs from GFP transgenic mice to femoral heads and confirmed that these cells could survive in hypoxic condition for a couple of weeks.
Our final set of experiments aimed to test the therapeutic effects of BMMSCs overexpressing HIF-1α based on its potential to couple osteogenesis and angiogenesis. Lentivirus was chosen to overexpress HIF-1α because such virus can infect both proliferating and nonproliferating cells. Our results suggested that implantation of mouse BMMSCs infected with Lenti-HIF-1α to femoral heads may simultaneously reduce pressure in osteonecrotic femoral heads and enhance osteogenesis and angiogenesis. The efficacy was satisfactory in that all mice that received cell injection demonstrated a significant reduction in osteonecrotic areas and the shape of femoral heads was largely preserved, which is consistent with a study by Laboubier and colleagues 45 . In addition, HIF-1α-modified BMMSCs and exosomes can halt the progression of AOFH in rabbit model46,47. However, other intervention approaches such as intravascular and intramuscular injections are not as effective as direct bone marrow implantation48,49.
However, the concerns remain for stem cell–related therapies because of the difficulty in maintaining the quality consistency of BMMSCs obtained from different donors. Recent studies have demonstrated that MSC derived from the same parental pluripotent stem cell (PSC) may overcome many disadvantages of adult MSC, such as batch-to-batch variation in MSC quality, and limited capacity for proliferation and differentiation50,51. In addition, a phase I clinical trial of MSCs derived from induced pluripotent stem cells (iPSCs) for the treatment of acute steroid-resistant graft versus host disease has shown satisfactory efficacy, safety, and tolerability 52 . The BMMSCs in our current study were obtained from mice with different genetic backgrounds, and BMMSCs used for implantation to femoral heads were isolated and pooled from WT C57BL/6J mice. In our follow-up studies, we will take use of PSC and iPSCs to treat AOFH in mice and compare the efficacy of different stem cells in reversing disease progression.
Collectively, our study laid a solid foundation for potential clinical application of modified BMMSC implantation to prevent AOFH progression. More comprehensive studies are warranted to reduce the variation in stem cell populations and unveil the most appropriate molecule(s)/pathway(s) for possible modification of BMMSCs to achieve better outcomes. In addition, there is a much room for the improvement of delivery systems for BMMSCs.
Conclusion
We demonstrated that Dex hindered angiogenesis and osteogenesis by inhibiting the COX-2–PGE-2–HIF-1α axis in BMMSCs. We first confirmed that implantation of BMMSCs overexpressing HIF-1α prevented osteonecrotic changes in Dex-induced AOFH mice, which may, to some extent, contribute to the development of effective treatment for human AOFH.
Footnotes
Author Contributions
X-XZ and XL participated in study design, and molecular and animal studies, S-RL and K-JG took charge of mouse surgery, micro-CT scanning, and reconstruction. D-FL and T-FL were in charge of experimental designs, trouble-shooting, and result interpretation and manuscript polishing.
Ethical Approval
This study was approved by the Ethics Committee of Zhengzhou University First Affiliated Hospital, Zhengzhou, China (approval number: 2021050604).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Zhengzhou University, China, and approved by the Ethics Committee of Zhengzhou University First Affiliated Hospital, Zhengzhou, China (approval number: 2021050604).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grants from the National Natural Science Foundation of China (U1704177 and 81871811) and China Postdoctoral Science Foundation (2020TQ0281).
