Abstract
Traumatic spinal cord injury (SCI) results in direct and indirect damage to neural tissues, which results in motor and sensory dysfunction, dystonia, and pathological reflex that ultimately lead to paraplegia or tetraplegia. A loss of cells, axon regeneration failure, and time-sensitive pathophysiology make tissue repair difficult. Despite various medical developments, there are currently no effective regenerative treatments. Stem cell therapy is a promising treatment for SCI due to its multiple targets and reactivity benefits. The present review focuses on SCI stem cell therapy, including bone marrow mesenchymal stem cells, umbilical mesenchymal stem cells, adipose-derived mesenchymal stem cells, neural stem cells, neural progenitor cells, embryonic stem cells, induced pluripotent stem cells, and extracellular vesicles. Each cell type targets certain features of SCI pathology and shows therapeutic effects via cell replacement, nutritional support, scaffolds, and immunomodulation mechanisms. However, many preclinical studies and a growing number of clinical trials found that single-cell treatments had only limited benefits for SCI. SCI damage is multifaceted, and there is a growing consensus that a combined treatment is needed.
Introduction
Spinal cord injury (SCI) is a devastating injury that is a source of extensive psychological and economic burden for patients and healthcare systems 1,2 . It is estimated that SCI affects more than 1 million people in the United States alone, with approximately 17,000 new cases each year 3 . Current treatments include spinal decompression surgery, treatment for spasticity, and rehabilitation therapy. Despite some advances in clinical management that improve patient’s quality of life 4,5 , SCI recovery is very limited, and finding alternative treatments for paralysis remains a top priority.
The time-sensitive and complex pathophysiology make it particularly difficult to investigate therapeutic targets for SCI 6 . After the initial mechanical injury, there are a series of secondary events that worsen the condition of patients 7 . The inflammatory response, gliosis hyperplasia, formation of an inhibitory environment 8 , and scar formation impede axonal regeneration and limit the potential for many therapeutic interventions (Fig. 1).
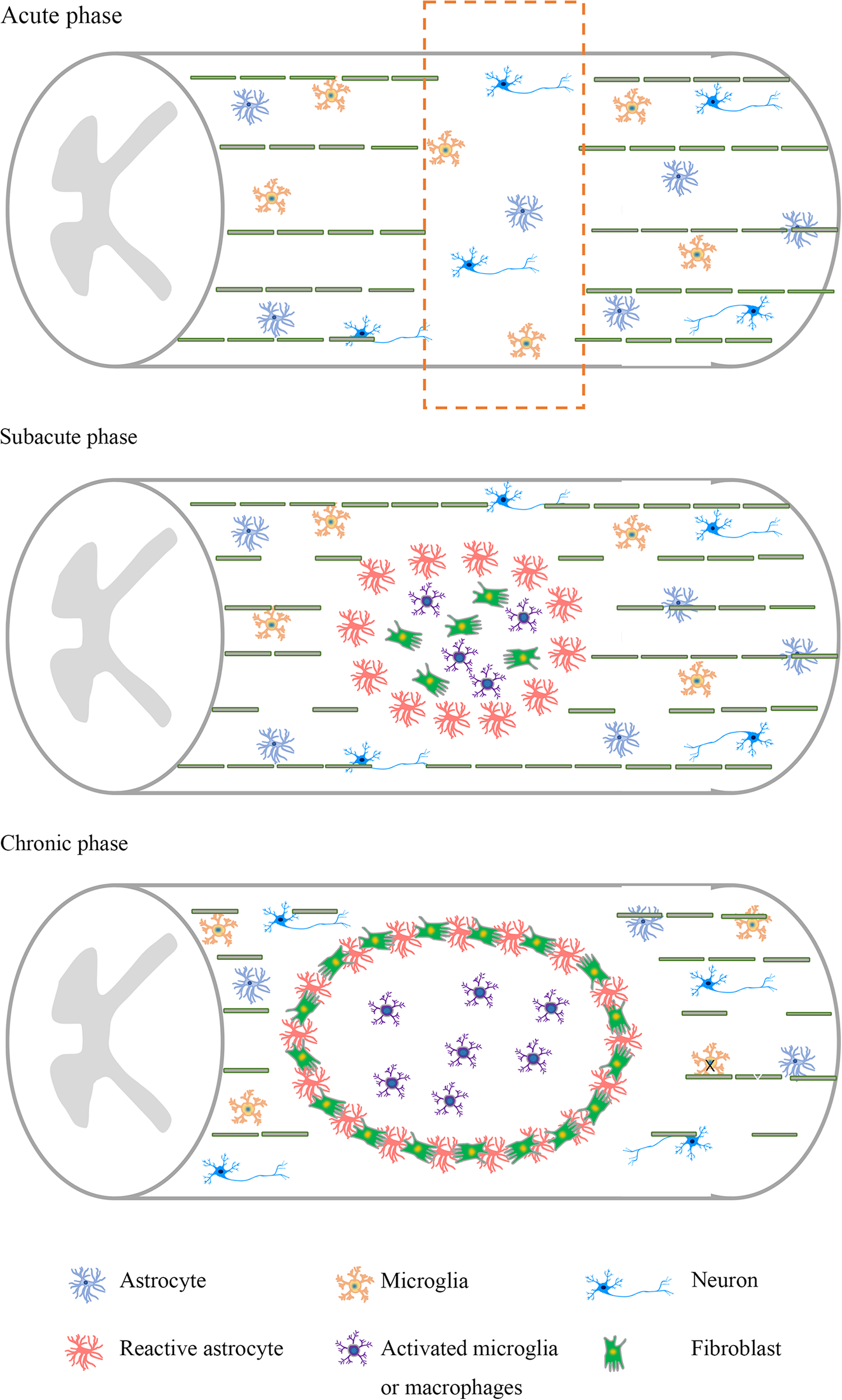
Pathological characteristics of spinal cord injury at different stages. Neuronal apoptosis and axonal damage are abundant in the acute stage. At the subacute stage, there is a large loss of neurons, axons, and myelin. Activated astrocytes, activated microglia, and macrophages accumulate in the injury site. At the chronic stage, a glial scar and an injury cavity further develop, and the inhibitory microenvironment is formed.
Cell therapies exhibit neuroprotective and nerve regeneration potential in SCI with different targets and responses to stimuli, such as regulating inflammatory responses, providing nutritional support, and improving plasticity. With these excessive potential mechanisms, various cells from different tissue sources, including bone marrow mesenchymal stem cells (BM-MSCs), umbilical mesenchymal stem cells (U-MSCs), adipose-derived mesenchymal stem cells (AD-MSCs), neural stem cells (NSCs), neural progenitor cells (NPCs), embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and extracellular vesicles (EVs), were studied. Previous reviews discussed cell therapy for SCI, but there is a lack of systematic elucidation, such as the original function of these cells, the function of modified cells, and the effect of combined therapy. This review performed an up-to-date summary of the current research status, challenges, and prospects for stem cell therapy in SCI to provide an overview of this field 9 –13 (Table 1).
The Effects of Different Stem Cells on Spinal Cord Injury.

AD-MSC: adipose-derived mesenchymal stem cell; BM-MSC: bone marrow mesenchymal stem cell; BSCB: blood-spinal cord barrier; CSPG: chondroitin sulfate proteoglycan; ESC: embryonic stem cell; EV: extracellular vesicle; iPSC: induced pluripotent stem cell; NPC: neural progenitor cell; NSC: neural stem cell; SSEP: somatosensory-evoked potential; U-MSC: umbilical mesenchymal stem cells.
Stem Cell Transplantation Strategy
Bone Marrow Mesenchymal Stem Cells
BM-MSCs are partially differentiated progenitor cells that are present in adult bone marrow and support sustained hematopoiesis and bone regeneration 60 . These cells were originally considered pluripotent, with the ability to differentiate into neurons and glial cells. However, additional studies showed that BM-MSC therapy primarily involved in cell fusion and transdifferentiation instead of cell differentiation. Early in vivo studies demonstrated that BM-MSC introduction into the lesion site of spinal cord contusion rats resulted in the formation of tissue bundles of astrocytes and neuronal predecessors 15 . The introduction of BM-MSCs to the injury site reduced inflammatory reactions 17 , astroglial scarring density 16 , and blood-spinal cord barrier (BSCB) leakage 18 ; modulated astrogliosis; alleviated neuropathic pain; and improved the functional recovery of hindlimb movement, which may involve the matrix metalloproteinase (MMP) 2/STAT3 pathway 61 . Conditioned medium from MSCs exhibited a therapeutic effect on SCI and may regulate the autophagy- and survival-related proteins Olig 2 and HSP70 19 .
Further investigation of the BM-MSC intravenous graft model indicated that functional recovery was achieved via the expansion of neurotrophic factors, including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and vascular endothelial growth factor (VEGF) 14 . NGF and BDNF are key regulators of neuronal differentiation, and VEGF is a key factor in the initiation and maintenance of angiogenesis and vasculogenesis induction 62,63 . Besides, BM-MSCs may be used as carriers due to their tropism to the injury sites and of interleukin-13 (IL-13), which is an inducer of the anti-inflammatory microglia/macrophage phenotype that significantly improved motor function recovery and decreased demyelination 64 .
Genetic engineering of BM-MSCs is an encouraging method to enhance their therapeutic effect, such as the regulation of specific factors or proteins. Insulin-like growth factor 1 (IGF-1) is an important factor for maintaining the characteristics of NPCs. IGF-1 overexpression of BM-MSCs strengthens antioxidant reactions and improves basso mouse scale (BMS) scores 65 . Other approaches, such as modification of the microRNA-124 gene 66 , silencing the Nogo-66 receptor gene 67 , inhibition of tumor necrosis factor α (TNF-α) 68 , and overexpression of neurotrophin-3 (NT-3) 69 , the chemokine stromal-derived factor-1 70 , and neurotrophic factor-derived glial cell (GDNF) genes 71 , exhibited better efficacy than original BM-MSCs in motor function and surrounding axon densities. The effects of individual cell transplantation are enhanced by cotransplantation with cells from other sources. These coupling strategies are primarily focused on MSCs and Schwann cells (SCs) because these cells regulate the microenvironment and improve the survival, differentiation, and proliferation of cotransplanted cells. Various studies reported that MSCs enhanced the effects of SCs 72 and olfactory ensheathing cells (OECs) by decreasing cell apoptosis 73 .
A longitudinal study of BM-MSC-based treatment of cervical SCI patients expanded autologous BM-MSCs and introduced these cells via intradural injection. Improved upper limb motor function and magnetic resonance imaging (MRI) images were observed in 6 of 10 candidates 6 months after transplantation 21 . Six patients with complete SCI received autologous MSC and SC therapy, and the results showed improvements in american spinal cord injury association (ASIA) grade, bladder compliance, and axonal regeneration. Similarly, a patient with chronic SCI received MSC therapy, and neurological function and the ability to walk were improved 20 . However, a phase III clinical trial demonstrated that single MSC application was safe but had little therapeutic effect. This result may be related to the timing of MSC transplantation because the homing capacity of stem cells is not substantial in chronic SCI 74 . Because of the controversial reports on the extent of patient responses to BM-MSC therapies, the efficacy of BM-MSCs must be further confirmed 75,76 . Several trials are ongoing, and completion of these studies will provide needed information to initiate a larger investigation of the efficacy of BM-MSC therapies. Overall, BM-MSC therapy is beneficial for SCI recovery by improving the microenvironment of the injury site, enhancing nutritional support, modulating the inflammatory response, and alleviating BSCB leakage. Patients avoid immunoreaction by receiving autologous cell transplantation. Therefore, BM-MSCs have huge potential for SCI treatment due to their reduced immunogenicity and improved availability. However, the therapeutic effects, homing ability, survival, and proliferation of single-cell types are limited. Further studies should focus on these aspects and combinational therapy to improve the efficacy of BM-MSCs.
Umbilical MSCs
Recent studies investigated MSCs separated from umbilical cords and adipose tissue 77,78 . U-MSCs possess the ability to develop into a homogeneous population that expresses neural markers and develops neural phenotypic features 79 . An early study found that U-MSCs migrated into the injury site but not noninjured areas after transplantation 80 , which lays the foundation for their therapeutic effects. Previous studies demonstrated that U-MSCs protected neurons from apoptosis 22 , inhibited the formation of glial scars via regulation of MMP2 23 , attenuated ischemic compromise of the spinal cord 25 , decreased reactive astrocytes 24 , improved motor function, and alleviated allodynia and hyperalgesia after SCI in animal experiments 26 –28 . U-MSCs demonstrated a better effect for a wide dynamic range of neurons than BM-MSCs 28 . Park and colleagues found that transplanted U-MSCs exhibited a better effect 1 week after SCI than at 12 h and 2 weeks, which indicates a potential time point for the treatment of SCI 81 .
Wnt proteins are involved in neural precursor (NP) differentiation and axon development, and Wnt-3a plays important roles in spinal cord dorsal interneuron differentiation. To enhance the efficacy of U-MSCs, researchers established Wnt3a-secreting U-MSCs by gene modification, which showed a better therapeutic effect than primary U-MSCs in SCI rats. Rats that received Wnt3a-MSCs had increased motor function scores and elevated expression of axonal regeneration-related proteins, including choline acetyltransferase, growth-associated protein 43, and microtubule-associated protein 2 82 . Cotransplantation may complement and synergize to improve single-cell therapies 83 . The cotransplantation of human U-MSCs and human NSCs exhibited the best efficacy compared to that of transplantation of hU-MSCs or hNSCs alone 84 .
U-MSCs improved motor function in the lower limb and expanded the atrophied spinal cord after injection into the subarachnoid, intradural, or extradural space of the spinal cord in patients with compressed fractures 85 . After U-MSC transplantation, 7 of 10 patients with thoracolumbar SCI had obvious improvements in movement, muscle tension, bladder function, and urine control compared to those of patients who received rehabilitation therapy alone 29 . The somatosensory-evoked potential (SSEP) and clinical manifestations of neuropathic pain of a patient with 2-year complete cervical SCI were significantly improved and alleviated 1 year after U-MSC transplantation, and the physiological function of myelinated large fibers was reflected by the SSEP 30 . U-MSCs are conveniently obtained because the umbilical cord is generally discarded. U-MSCs are obtained from umbilical blood, perivascular regions, and the umbilical vein subendothelium without ethical issues, and these cells are beneficial in the recovery of SCI via different mechanisms 24 . Further efforts are needed to fully assess the effectiveness of UC-MSC transplantation.
Adipose-derived MSCs
AD-MSCs and BM-MSCS share some similarities, such as morphology and cell surface antigen expression, but they differ in proliferation rates and multilineage capabilities 86 . Adipose tissue contains more somatic stem cells than bone marrow, which makes AD-MSCs a good candidate for MSCs, especially with adipose tissue availability 87,88 .
AD-MSC transplantation demonstrated satisfactory effects in chronic and acute SCI. Intravenous administration of AD-MSCs activates angiogenesis and upregulates ERK and Akt, which improves hindlimb motor function 37 . AD-MSCs also promote cell survival and tissue repair by increasing the expression of beta3-tubulin, BDNF, and ciliary neurotrophic factor (CNTF) 35 . AD-MSCs may protect neurons and ameliorate erectile dysfunction in rats with SCI 31 –34 .
In addition to the direct effects, human adipose-derived stem cells transdifferentiate into neuron/motoneuron-like cells, which reduce the formation of cavities and suppress immune activity via the inhibition of astrocyte reactivation and secretion of anti-inflammatory factors 36 . Hypoxic preconditioning-treated AD-MSCs promoted cell survival and increased the expression of marker genes in DsRed-engineered neural stem cells, which enhanced the effect of the combined treatment of stem cells and gene therapy for SCI 89 .
Although AD-MSCs transplantation has been investigated in animal SCI models, large longitudinal clinical trials using stem cells derived from adipose tissue are lacking. Early studies investigating the safety of intravenous AD-MSCs showed no tumorigenicity or other adverse side effects. One study investigated the effects of autologous transplantation of AD-MSCs in 14 patients with SCI who underwent intrathecal transplantation. ASIA sensory and motor scores and electrophysiological evaluations, including MRI and electromyography, were used to determine the effect. After the intervention, 10 patients showed sensory improvement, but the size of the lesion visualized using MRI remained stable. None of the patients treated with AD-MSCs had serious adverse events 90 . Some barriers should be elucidated before clinical translation, such as standard protocols of cell generation, cell characteristics, and clear disclosure of the underlying mechanism, and larger experimental animals that are closer to humans should be used.
NSCs and NPCs
NSCs and NPCs are pluripotent cells that are isolated from the subventricular region of the ventricles and hippocampus of the brain and the ependymal region of the central canal of the spinal cord 1,91 –96 . These cells are capable of differentiating into specific neuronal or glial cells, enhancing remyelination and providing nutritional support, which makes them suitable for cell transplantation therapy in SCI 38 .
NPCs primarily differentiate into oligodendrocytes 97,98 , increase myelination 40 , and improve hindlimb function. One study also demonstrated that transplantation of NPCs obtained from the subventricular zone promoted respiratory recovery after SCI, which did not work by differentiation 42 . NPC transplantation increased the expression of NGF, CNTF, BDNF, IGF-1, and GDNF, which are beneficial for SCI recovery 39 . NPCs also modulate the inflammatory response 41 via inhibition of the secretion of reactive macrophages and T cells and neuroprotective cytokines 99 . Previous studies revealed that the transplantation of NPCs during the acute stage demonstrated better efficacy than during the subacute and chronic stages 100 , and transplantation in intact soft tissue may produce better efficacy than transplantation in the injury site during the subacute period 101 .
Modified NSCs may exhibit better therapeutic efficacy than naïve cells. Inhibition of leucine-rich repeat and immunoglobulin domain-containing protein (LINGO)-1 in NSCs facilitated neuronal differentiation and recovery in SCI rats 102 . Transplantation of recombinant NSCs with VEGF reduced transient receptor potential vanilloid (TRPV1), increased the release of neurotrophic factors, and promoted neuronal recovery 103 . NSCs with high expression of E-cadherin, a transmembrane adhesion protein, increased the survival of NSCs, decreased the release of inflammatory factors, and promoted functional recovery 104 . Overexpression of the antiapoptotic gene Bcl-XL 105 , upregulation of miR-124 106 , upregulation of NT-3 107 , or polarization toward a more oligodendrogenic fate 108 also achieved better recovery. Mild hypothermia 109 or hypoxia pretreated 110 of NSCs showed a more favorable effect on SCI than untreated NSCs by improving cell proliferation and upregulating neurotrophic and growth factors. Combined with MSCs 111 , SCs 112 and OECs 113 also enhanced neuronal differentiation and cell survival, which further improved motor recovery.
A 2018 study demonstrated that perilesional intramedullary injections of NSCs were safe, but the dose should be verified 114 . Twelve amyotrophic lateral sclerosis patients received transplantation of human spinal cord–derived NPCs, and the results showed that NPC transplantation was safe, which initiated further clinical trials 115,116 . NPCs showed great potential for SCI treatment, but the functional recovery was limited. Quintessential combinational methods have raised much hope to enhance the efficacy of NPCs. However, rodents were generally used as subjects in previous studies, and some specific larger animals that are closer to humans should be used as experimental subjects to address the problems and move toward clinical translation 117 .
Embryonic Stem Cells
ESCs are multipotent stem cells that are capable of differentiating into new cell types in the body. ESCs differentiate into neurons and glial cells to replace nonfunctional cells or tissues in SCI 118,119 . However, their undifferentiated form is rarely used due to the risk of tumorigenicity. Previous studies demonstrated that ESC transplantation was effective for SCI recovery 120 –122 . ESCs transfected with cell adhesion molecule L1, which promotes neuronal survival and neurite sprouting, had promising potential for SCI treatment 122 .
ESC-derived definitive neural stem cells express myelin basic protein 46,123 , support nodal architecture, and display multilayer myelination in SCI animal models 46,47 . Human embryonic stem cell–derived oligodendrocytes 43,124 or oligodendrocyte progenitor cells 43,44 and motoneuron progenitors promote astrogliosis and enhance motor recovery. ESC-derived neural lineage cells enable axons to pass through chondroitin sulfate proteoglycan (CSPG), which is a tremendous barrier to axonal regeneration, and exhibit therapeutic potential for SCI treatment. The expression of nerve glial antigen 2 and MMP9 45 is involved in this process. Transplantation of GABAergic neurons derived from mouse ESCs attenuated neuropathic pain and increased the paw withdrawal threshold and vocalization threshold 48 .
A clinical study in 2014 showed that human ESC-derived oligodendrocyte progenitor cell transplantation was safe for SCI patients 125,126 . Another two studies in 2016 demonstrated that SCI patients had restored body functions after intervention with human ESCs 127 , and there were no serious complications. However, the pluripotency of ESCs may result in tumor formation due to their considerable proliferative ability. There may be genetic changes during the cell culture process 128 . Therefore, it is critical to optimize the differentiation protocol to decrease tumor occurrence and control cell populations to match the different recovery requirements in SCI patients 129 .
Induced Pluripotent Stem Cells
There is significant controversy about ESCs due to their origin. iPSCs, which share the same pluripotent characteristics as ESCs, may neutralize this problem. iPSCs are generated from reprogrammed somatic cells 12,130 –132 , which are separated from accessible tissue, such as autologous skin, which avoids ethical issues, allows autologous cell transplantation, and prevents rejection.
NPs derived from a clone of human iPSCs led to restoration of the injury site 133 . IPSCs-derived neural stem/progenitor cells (iPSC-NS/PCs) inhibited demyelination 51,52 and promoted synapse formation 53 and neurotrophic factor secretion, which improved functional recovery in common marmosets after SCI without tumor formation 49 . Researchers found that only spinal cord-type NPCs from human iPSCs exhibited efficacy, compared to that with forebrain-type NPCs from human iPSCs, which indicates the importance of the regional identity 134 . A comparative study demonstrated that iPSC-NPs exhibited the best effect due to their strong graft survival, glial scar inhibition, and axonal sprouting enhancement compared to those of BM-MSCs and NPs derived from an immortalized spinal fetal cell line (SPC-01) 50 . Different transplantation regions may lead to different effects, and researchers found that intraspinal implantation (cells present in the tissue) may produce better long-term efficacy than intrathecal implantation (paracrine only mechanism) 135 .
Modified human iPSC-derived astrocytes reduced lesion size and morphological denervation of respiratory phrenic motor neurons and improved respiratory function 54 . Similarly, γ-secretase inhibitors promoted iPSC-derived NPCs maturation and increased neuronal commitment via regulation of the NOTCH signaling pathway 136 .
A case report demonstrated that NSCs derived from iPSCs obtained from a healthy 86-year-old male differentiated into neurons and glia, and axons extended long distances and formed synapses after cell transplantation 137 . Another study suggested that the iCaspase9 gene alleviated adverse events after iPSC-derivative transplantation 138 . Another study demonstrated that hydrogels modified with an RGD peptide and platelet-derived growth factor (PDGF-A) promoted cell survival and differentiation and reduced teratoma formation 139 . However, there are opposite results that human iPSC-derived NPCs do not provide beneficial results for SCI therapy. Some of these studies had limitations with graft survival or time to transplant 140,141 . The tumorigenesis of iPSCs and the prohibitively high cost–benefit for developing treatments 142 hinder the clinical translation 143 . It is crucial to develop optimized solutions, including standard protocols for collecting cells, the ideal time for cell delivery, and the safe and effective routes of administration in clinical treatment.
EVs Derived From Stem Cells
EVs have come into the spotlight in recent years because of their satisfying therapeutic potential. They are small vesicles (100–1,000 nm) secreted from a variety of cells and have a lipid bilayer membrane. EVs work as cell communication messengers by carrying nucleic acids, proteins, and lipids 144,145 . EVs are not a single type of vesicle but consist of ectosomes, microvesicles, and exosomes. Exosomes, with diameters of 50–150 nm, are remarkable carriers with low immunogenicity and high biocompatibility 146 , which protect their cargo from degradation and maintain their biological activity 147 .
EVs exhibit robust chemotaxis to the injury site and cooperate with neurons. Recent studies reported that MSC- 57 and NSC-derived 55 EVs inhibited inflammation, protected cells from apoptosis and reduced injury size, and the mechanism may involve autophagy 55 and the microRNA-21-5p/FasL gene axis 58 . Lankford et al. found that exosomes accumulated in the injury sites of the spinal cord and spleen after IV injection 148,149 . Other studies demonstrated that exosomes derived from BM-MSCs were primarily incorporated in microglial cells, downregulated nuclear factor kappa-B 150 , protected the integrity of the BSCB 49 , inhibited the activation of A1 astrocytes 56 , and played a protective role in rats after SCI.
Exosomes derived from gene-modified stem cells showed more therapeutic potential than exosomes derived from native stem cells. For example, exosomes derived from miR-133b-modified adipose-derived stem cells regulated axon regeneration and improved neurological function after SCI 55 . Phosphatase and tensin homolog (PTEN) exists in neurons and axons, and it plays an inhibitory role in the growth of axons. Therefore, suppression of PTEN in MSC-derived exosomes showed desirable therapeutic effects on SCI 151,152 . Similarly, the downregulated expression of phosphatase and tensin homolog pseudogene 1 (PTENP1) in exosomes derived from differentiated P12 cells and MSCs promoted neuronal survival and functional recovery by regulating the expression of miR-19b and miR-21 153 . There was an obvious decrease in miR-544 expression after SCI, and exosomes derived from miR-445-modified MSCs improved functional recovery in rats after SCI 154 . MiR-126 loaded in MSC-derived exosomes enhanced angiogenesis, inhibited inflammation, and had an encouraging effect on SCI 155 . Similarly, miR-21 deficiency in exosomes derived from MSCs also displayed desirable effects 156 . Iron oxide nanoparticles (IONPs) carried by exosome-mimetic nanovesicles (NVs), which were derived from IONP-treated MSCs, enhanced NV homing capacity and further promoted the therapeutic potential of NVs in SCI 157 . Since few studies demonstrated the pathophysiology of EVs in SCI, further studies are needed to identify the molecular mechanism and related signaling pathways of the therapeutic effects of EVs. Some nontargeted EVs have also been reported 158 , and normalizing the isolation and acquisition of EVs is paramount before translating this therapeutic method to SCI patients clinically 159 .
Other Combinatorial Methods
Neuroprotection
Neuroprotective drugs aim to minimize pathological damage and preserve neural tissue. Only methylprednisolone has been clinically proven to provide benefits post-SCI, but it also brings some risks, including gastrointestinal bleeding, wound infection, and thromboembolism 160 . However, increasingly promising neuroprotective interventions are under investigation (e.g., chondroitinase 161,162 , alginate scaffolds 163 , TNF-α antagonists, anti-Nogo antibodies, minocycline, and Lavandula angustifolia extract 164 ). These interventions may be used before or during cell transplantation to create a microenvironment that improves stem cell efficacy. Therapeutic hypothermia in combination with cell therapy has been successfully used for neuroprotection. Hypothermia lowers the basal metabolic rate and reduces inflammation to provide synergistic action in SCI 165 . Minocycline synergistically improved the anti-inflammatory effects of MSCs 166 . Although the initial results are encouraging, additional work is needed to optimize the efficacy of combination treatment. The combination of BM-MSC transplantation and propofol injection effectively improved neuroprotection 167 , increased horseradish peroxidase-positive nerve fibers, and shortened the latencies of SSEPs and motor-evoked potentials in the hindlimb 168 . Zhang et al. injected NSCs into the tibial nerve and investigated the effect of lithium chloride (LiCl) on the survival of neurons and axons. They found that LiCl promoted NSC differentiation, and this combinational therapy increased the regeneration of axons in the tibial nerve and decreased the formation of glial scars 169 . Electroacupuncture 170 , folic acid, substance P 171 , and granulocyte-macrophage colony-stimulating factor 172 also exhibited synergetic effects by improving NSC proliferation and neuron survival in SCI rats.
Biomaterials
Although stem cell therapy has gained momentum in the field of SCI therapy, it has room for improvement. Biological material use is an encouraging approach for cell therapy by bridging the lesion cavity, replacing damaged extracellular matrices, and integrating the host tissue and transplanted cells. Matrigel is primarily composed of laminin, collagen IV, heparan sulfate proteoglycans, and growth factors that support cell survival and differentiation 173 ; increase neuronal markers; decrease fibrosis, astrogliosis markers, and inflammatory factors 174 ; and enhance behavioral recovery in SCI animals. Hydrogels possess a three-dimensional (3D) network structure that provides the benefits regarding electrostatic forces, steric hindrance, and entanglement. These gels are injected or implanted directly because of their soft texture. Laminin-coated hydrogel enhanced the viability of IPSC-NPs and promoted host axon and astrocyte growth in the lesion site 175 . Ischemia and hypoxia following the primary injury may exacerbate the pathological process of SCI and extremely impede functional recovery after SCI. To address the ischemia and hypoxia in SCI, prevascularized nerve conduits based on the stem cell sheet were designed and implanted in the injury spinal cord, which exhibited satisfactory potential 176 . Self-assembling peptides form 3D nanofibers via self-assembly after direct injection to the injury site, act as structural framework, and regulate the microenvironment. The use of NPCs with the self-assembling peptide QL6 reduced cystic cavity formation and inflammation and enhanced synaptic connections by reducing astrogliosis and CSPG, which improved forelimb function in a cervical injury SCI model 177 . A previous study reported that chondroitinase ABC (ChABC) enhanced the therapeutic effect of NPCs in SCI, but the ChABC delivery efficiency was unsatisfactory. Nori et al. manufactured NPCs biased toward an oligodendrogenic fate and upgraded the ChABC delivery system via a crosslinked methylcellulose biomaterial, and this combinatorial therapy promoted oligodendrocyte differentiation, remyelination, and synaptic connectivity 178 . An N-cadherin-modified linearly ordered collagen scaffold promoted the migration and differentiation of endogenous neural/progenitor stem cells and produced a desirable therapeutic effect in rats after SCI 179 . The collagen microchannel scaffold and paclitaxel-liposome combination induced neuronal differentiation of NSCs and growth of neurons and axons, which exhibited great potential for SCI treatment 180 . Other scaffolds, such as silk fibroin combined with neurotrophic factors 181,182 , fibrin scaffolds containing growth factors 183 , polycistronic delivery of IL-10 and NT-3 184 , also promoted the differentiation, proliferation, and viability of transplanted cells, which has desirable therapeutic potential for SCI treatment.
Many kinds of biomaterial scaffolds have been used to deliver MSCs to damaged spinal cords. Unlike NPCs, MSCs likely provide nutritional support, promote axonal regeneration and angiogenesis, and reduce inflammation. Modified biodegradable chitin conduits in combination with BM-MSC transplantation improved the microenvironment for MSCs, prevented scar formation, and promoted recovery after right spinal cord hemisection injury 185 . Superparamagnetic iron oxide labeling of BM-MSCs coupled with magnetic guidance offers a promising avenue for the clinical treatment of SCI by enhancing the homing efficiency of cells 186 . AD-MSCs encapsulated in a fibrin matrix, which is a biopolymer that simulates the natural microenvironment, inhibited injury cavity expansion, increased tissue retention, and promoted recovery of function and structure 187 . However, some previous studies demonstrated that some biomaterials stimulated a disadvantageous microenvironment in the lesion site, such as a proinflammatory milieu 188 . Other tissue engineering scaffolds, such as acellular spinal cord scaffolds 189 , polycaprolactone 190 , 3D gelatin methacrylate hydrogels 191 , and 3D fibrin-based scaffolds 192 , enhanced axonal regeneration and tissue remodeling and improved the therapeutic effect of stem cells. In general, the use of biological materials is a promising combination approach for SCI cell therapy by improving cell implantation, delivering certain factors, promoting neural marker expression and axonal regeneration, inhibiting the inflammatory response, and contacting the injured central nervous system (CNS) tissue.
Challenges and Prospects
Stem cells have neuroregenerative and neuroprotective effects in SCI cell therapy. Cell-based therapies in SCI have different mechanisms in functional recovery, such as immunomodulation, cell replacement nutrition, and scaffold support. However, stem cell therapies present particular safety concerns. First, cell therapy–related immunotoxicity, immunogenicity, and tumorigenicity are often discussed in preclinical studies. Second, limited cell survival and limited integration were common obstacles in previous studies with different experimental designs, including cell number, timing of treatment 193 , and strategies of transplantation 194 . Third, it is important to ensure the genetic stability, generation consistency, and storage safety 162 of stem cells 195 . The quality and repeatability of stem cell transplantation are critical to clinical translation. Small differences in cell origin and growth conditions may have a significant impact on the outcomes 196,197 . Fourth, the mechanism of the effects and biological properties should be further investigated to guide the clinical application 187 . Finally, small sample size, limited supervision, and poor quality are the common problems of most registered clinical trials that hinder the development of stem cell therapy 198 . Standard protocols are difficult to confirm due to the heterogeneity of the injury type and level, the particular time of treatment, and the different number of transplanted cells.
Encouraging preclinical studies, coupled with publicity, led to early clinical deployment, but the results were mixed. One specific type of stem cell achieves only a limited therapeutic effect. Therefore, many researchers are committed to enhancing the efficacy of stem cells. The use of genetic engineering technology, cell coupling, combinational therapy with neuroprotective agents, trophic factors, biomaterials, and rehabilitation may help improve the therapeutic effectiveness of stem cells in heterogeneous patient populations. Research is needed to optimize their use.
Conclusion
Although cell therapy offers important promise for SCI treatment, there are many obstacles to clinical translation. These obstacles include suitable cell types and sources, cell survival, quality and repeatability of stem cells and optimal transplantation dosage and timing. There are endogenous differences between experimental animals and humans, and much work should be completed before clinical transformation. Each type of stem cell has unique benefits. Previous studies already focused on how to enhance the efficacy of stem cells and made positive achievements. Future treatments may use a variety of novel strategies to address the problems of SCI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
