Abstract
Stem cell therapy has been explored for the treatment of cerebral stroke. Several types of stem cells have been investigated to ensure the safety and efficacy in clinical trials.
Cryopreserved umbilical cord blood (UCB) mononuclear cells (MNCs) obtained from healthy donors have a more stabilized quality, thereby ensuring a successful therapy. A phase I study was conducted on patients aged 45–80 years who sustained acute ischemic stroke. An UCB unit was obtained from a public cord blood bank based on ABO/Rh blood type, HLA matching score (6/6), and cell dose (total MNC count of 0.5–5 × 107 cells/kg). In addition, to facilitate blood brain barrier penetration of UCB, 4 doses of 100 mL mannitol was administered intravenously after 30 min after UCB transplantation and every 4 h thereafter. The primary outcomes were the number of disease (GVHD) within 100 days after transfusion. The secondary outcomes were changes in the National Institutes of Health Stroke Scale (NIHSS), Barthel index, and Berg Balance Scale scores. A 46-year-old male patient with identical ABO/Rh blood type, HLA matching score of 6/6, and MNC count of 2.63 × 108 cells/kg was enrolled. The patient did not present with serious AEs or GVHD during the 12-month study period. The patient’s NIHSS score decreased from 9 to 1. Moreover, the Berg Balance Scale score increased from 0 to 48 and the Barthel index score from 0 to 90. This preliminary study showed that an adult patient with hemiplegia due to ischemic stroke completely recovered within 12 months after receiving allogeneic UCB therapy.
Introduction
Currently, acute stroke is the second and third leading cause of death and disability, respectively, worldwide. More than 15 million people present with stroke annually. Approximately 30%–35% die and nearly 75% of survivors sustain permanent disability 1 . The current treatments in the acute phase include the use of thrombolytic, anticoagulant, and antiplatelet agents. Thrombolytic agents must be administered within 4.5 h of stroke onset. However, the use of this agent increases the incidence of hemorrhage by 15%–20%. After ischemic stroke, about 120 million neurons die every hour, which is equal to 3.6 years of aging brain function 2 . Furthermore, the dead neurons cannot regenerate. Hence, treatment with new cells to facilitate regeneration is essential. The developing cell therapy is a breakthrough in the treatment of stroke. Our research team used granulocyte colony stimulating factor (G-CSF) injection combined with autologous hematopoietic stem cells (CD34+) brain transplantation for the treatment of patients with chronic ischemic stroke (n=15) 3 . Based on the current study, this treatment was found to be feasible and safe. However, results also showed that the patient’s cell could significantly affect the degree of improvement. That is, autologous stem cell proliferation and differentiation in an elderly patient (aged over 60 years) is not as good as that of younger patients.
Compared with human adult peripheral blood, human umbilical cord blood contains richer hematopoietic primitive cells and numerous endothelial primitive cells, which have strong replication capacity in vitro and in vivo. Samples of cryopreserved plasma depletion (PD) cord blood products were assayed to determine cytokine profiles using a R&D Human XL Cytokine Discovery 14 Plex panel. The in-vitro analytic results listed in Table 1, which showed the level of anti-inflammatory cytokine, interleukin (IL)-10, is significantly higher than those of pro-inflammatory cytokine, IL-1-beta, IL-2, IL-6, interferon gamma (IFN)-gamma and tumor necrosis factor(TNF)-alpha. The relatively higher levels of growth factors (GFs), epidermal growth factor (EGF), FGF-basic, vascular endothelial growth factor (VEGF), G-CSF, and granulocyte-macrophage colony-stimulating factor (GM-CSF) were observed in comparison with those of cytokines, IL-1-beta, IL-2, IL-4, IL-5, IL-6, IFN-gamma and TNF-alpha. The high amounts of EGF, VEGF, G-CSF, and IL-10 in PD cord blood (CB) products allows us to hypothesize that the infusion of PD cytokine products may not only restore immune homeostasis but also enhance the repairing of the damaged brain nervous system in cerebral stroke patients 4 .
Cytokine and Growth Factor Profiles in Cryopreserved PD Cord Blood Products vs. Adult Plasma/Serum.
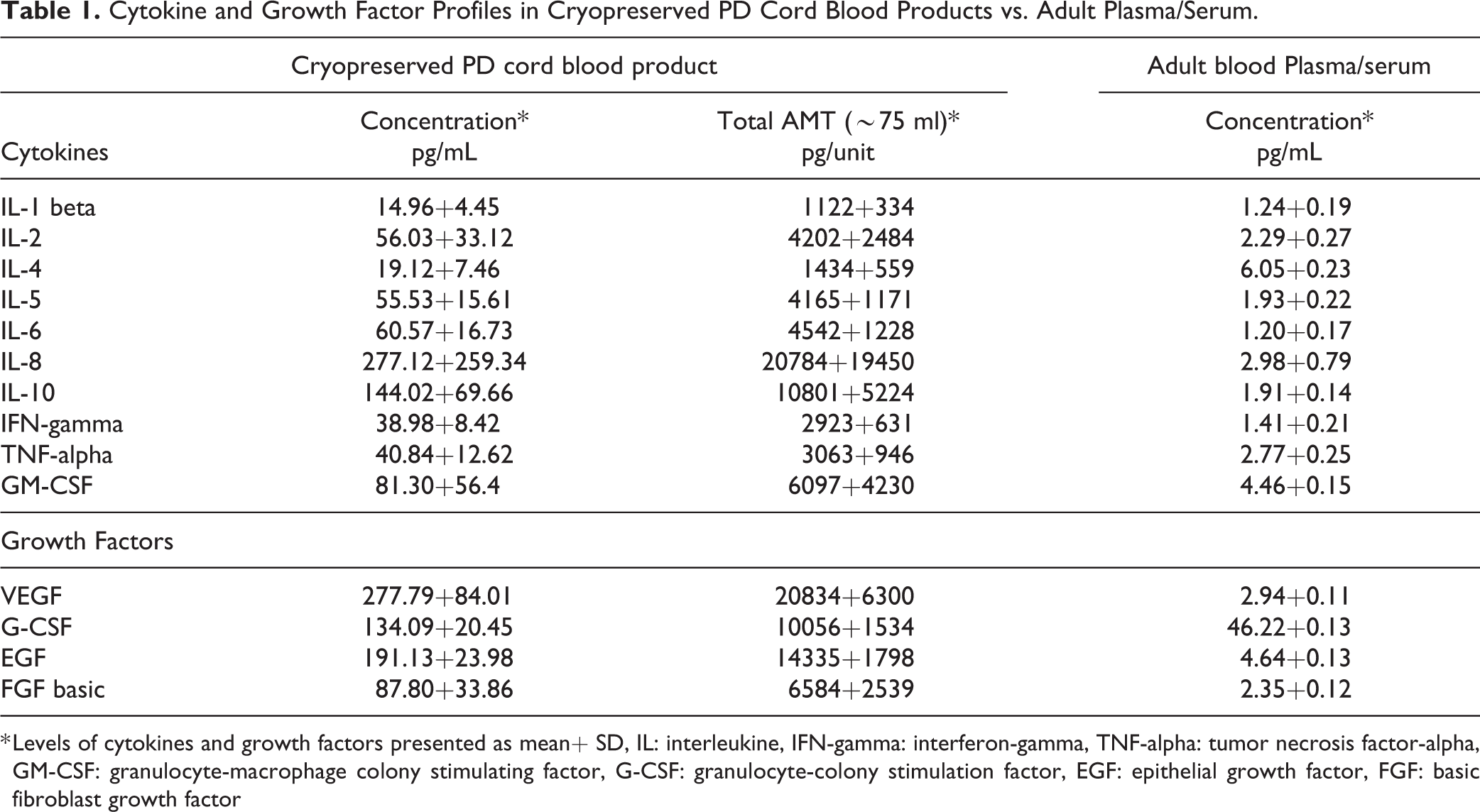
* Levels of cytokines and growth factors presented as mean
Moreover, cord blood stem cells proliferate into neural cells, and they were found to be effective in the treatment of several neurodegenerative diseases 5,6 . In cerebral stroke, the intravenous injection of umbilical cord blood (UCB) mononuclear cells (MNCs) could restore exercise abilities, and it has neuroprotective effects 7 . After transplantation of human UCB, the expression of inflammatory markers such TNF-alpha, IL-1β, and IL-2 decreased. Meanwhile, that of inflammatory markers including IL-6, IL-10, and transforming growth factor beta 1 increased. Consequently, after transplantation of human UCB, the anti-inflammatory processes can achieve the protective effect of neural cells 7,8 . In addition to the ant-inflammatory effect, UCB MNCs can spontaneously transfer to the damaged central nervous system and the spleen 7,9 . Therefore, UCB MNCs can participate in the biosynthesis of lymphocytes. Some studies have confirmed that lymphocytes are associated with neuroprotection in rats with acute stroke 10 –12 .
A phase I clinical trial on the intravenous administration of human UCB in acute ischemic stroke was conducted. To the best of knowledge, this study presented the first patient who completely recovered from right hemiplegia 12 months after receiving UCB therapy.
Materials and Methods
Study Design
This is a multicenter phase I safety study on patients aged 45–80 years who sustained acute stroke but did not receive t-PA therapy. In total, six participants will be enrolled. The inclusion and exclusion criteria are presented in Tables 2 and 3, respectively. The participants will undergo a series of baseline neurological assessments, blood tests, and MRI. UCB units will be selected from a public CB bank based on the ABO/Rh blood type, HLA matching score (6/6), and cell dose (total nucleated cell count of 0.5–5 × 107 cells/kg). The UCB will be administered intravenously as a single infusion between 3 and 10 days after stroke onset. The participants will be monitored for 6 h after infusion, and follow-up will perform after 24 h. Subsequent follow-up phone calls will be conducted at 1, 6, and 12 months. The calls will include telephone surveys on post-stroke rehabilitation and functioning. The follow-up assessment after 90 days will include neurological examination, MRI, and blood tests. The Research and Ethical Review Committees and Internal Review Board of Tzu Chi Hospital approved the protocol and procedures for the project (IRB105-71-A).
Inclusion criteria.

Exclusion Criteria.

Selection of Umbilical Cord Blood Unit
An assessment was performed upon receipt of the participant’s information, including HLA typing, from the transplant center. The report was used to create a Summary Search Report for Acute Stroke Clinical Trial, which included information on candidate CB units meeting the following criteria: at least a matching score of 4/6 based on low-resolution HLA typing, identical ABO/Rh blood type between the subject and donor, and total mononucleated cell (MNC) count of 2–5 × 108 cells/kg.
Case Presentation
A 40-year-old male patient, with a history of hypertension and hemodialysis for end-stage renal disease, presented with acute left hemiplegia at an emergency department. Emergent assessment conducted by a neurologist and MRI revealed infarction in the right ICA territory. Recombinant tissue plasminogen activator was not recommended owing to a time delay >3 h. Hence, the patient was enrolled in this study and the informed consent was written by patient himself. The baseline NIHSS score was 8, and it increased by 1 point after 24 h. A Summary Search Report for Acute Stroke Clinical Trial was created after assessing information from the CB bank. The report showed an identical ABO/Rh blood type, HLA matching score of 6/6, and total MNC count of 2.63 × 108 cells/kg. UCB transfusion was performed on the 8th day after stroke onset. Then, 4 doses of 100 mL mannitol were administered intravenously after 30 min after UCB transplantation and every 4 h thereafter. The patient was discharged 8 days after UCB transfusion, and he was followed-up at 1, 3, 6, and 12 months for the assessment of NIHSS score, neurological function, and MRI results.
Results
The patient’s neurological function improved gradually after UBC transfusion. Moreover, the NIHSS score decreased from 9 at baseline to 1 at 12 months after UCB transfusion (Fig. 1A). Moreover, the Berg Balance Scale score increased from 0 to 48 (Fig. 1B), and the Barthel index score from 0 to 90 (Fig. 1C). The patient did not present with splenomegaly on abdominal ultrasonography during the 12-month observation period. However, he presented with insomnia and upper respiratory tract infection and was treated with medications, which led to satisfactory outcomes. No serious adverse event was observed. Diffusion-weighted imaging (DWI) performed 2 h and 8 days after stroke revealed infarction (increased white intensity) in the right corona radiata, which scattered after 3 months and disappeared after 6 months (Fig. 2A–D). T2-weighted images obtained 2 h, 8 days, 3 months, and 6 months after the onset of infarction revealed increased white intensity in the right corona radiata (Fig. 3A–D).
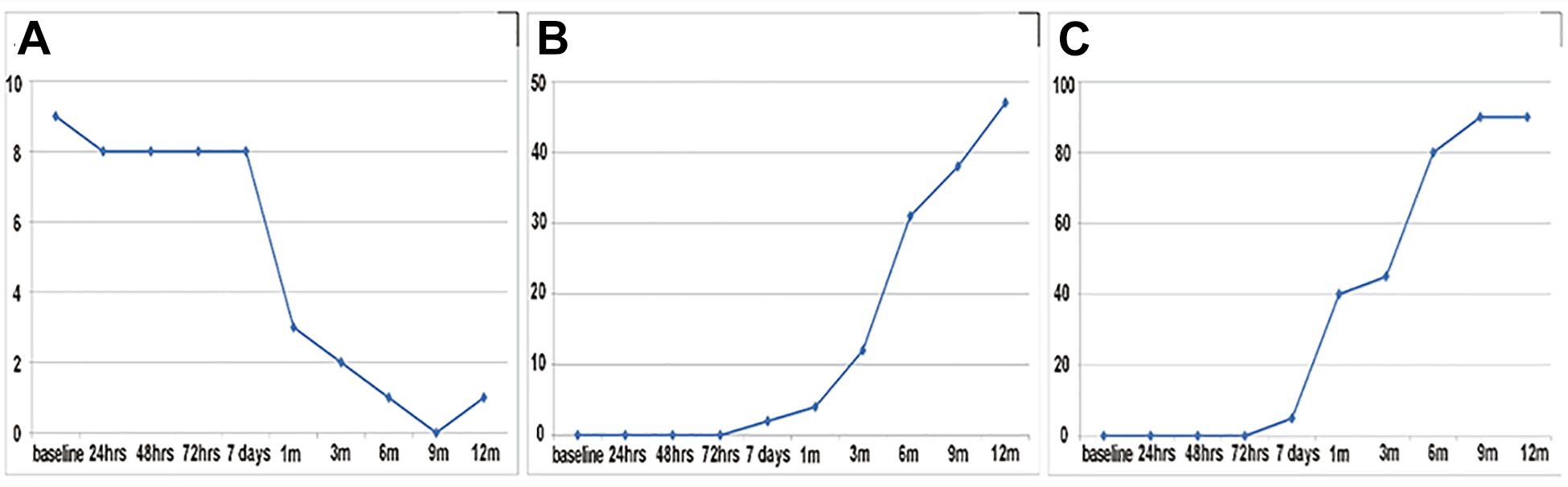
Neurological function from day 1 prior to UCB transfusion (baseline) to 12 months after UCB transfusion. (A) National Institutes of Health Stroke Scale score, (B) Berg Balance Scale score, and (C) Barthel index score
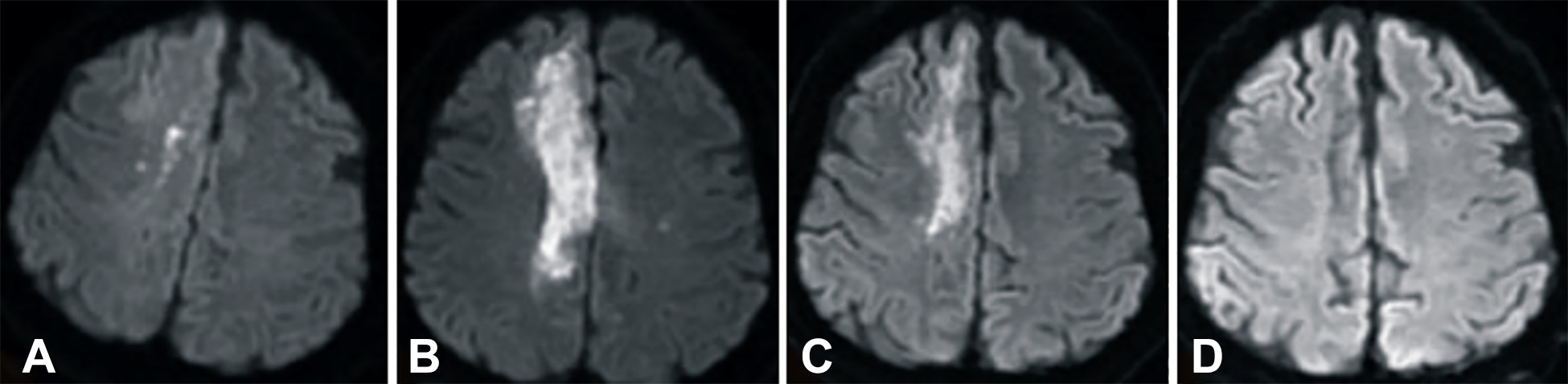
Diffusion-weighted images obtained 2 h after stroke onset and 6 months after mononucleated cell UCB transfusion. (A) 2 h after stroke, (B) 1 day after umbilical cord blood transfusion, (C) 3 months after UCB transfusion, and (D) 6 months after MNC transfusion
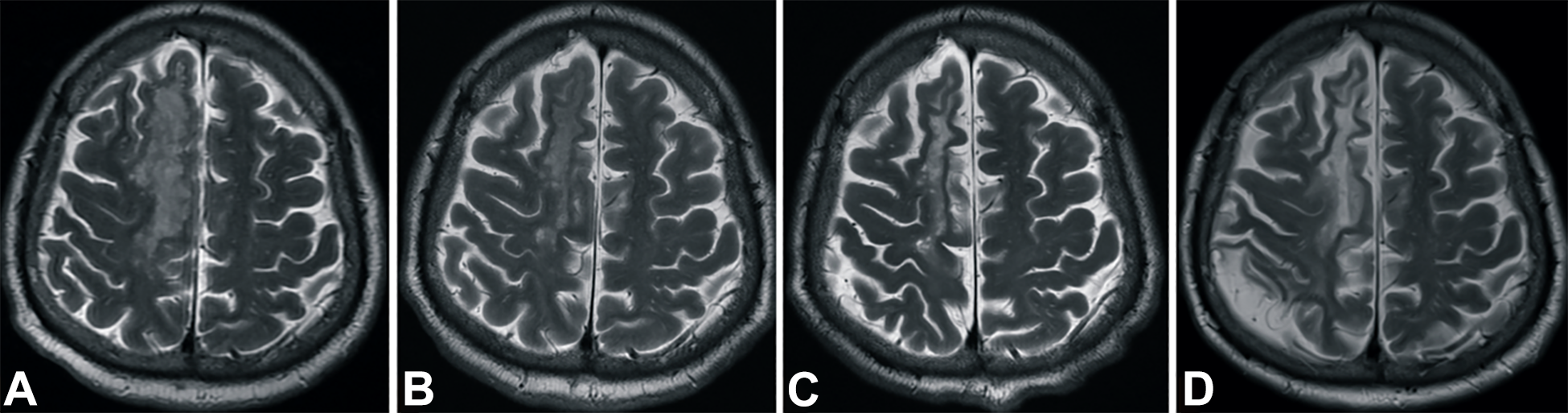
T2-weighted images obtained 2 h after stroke and 6 months after UCB transfusion. (A) 2 h after stroke, (B) 1 day after UCB transfusion, (C) 3 months after UCB transfusion, and (D) 6 months after UCB transfusion
Discussion
The efficacy of stem cell therapy for cerebral stroke in terms of cell type, cell origin, cell number, timing of cell treatment, and route of cell delivery has been subjected to optimization evaluations. The optimal cell transplant strategy has been examined in several preclinical and clinical studies. Although a conclusion has not been established, translational guidance on the safe and effective transplant regimen has been recognized. The time window for neural cell repair or renewal after the initiation of stroke is within 72 h based on an animal experiment 13 . Early cell therapy in the acute or subacute stage of stroke might result in better outcomes. This patient presented with subclinical second stroke on MRI 3 months after stroke. The T2-weighted images revealed loss of brain parenchyma and CSF acumination only in the second stroke region, but not in the first stroke region, which has been treated with cell therapy in the acute stage (Fig. 3c, 3d).
Human UCB MNCs have been shown to exert robust therapeutic effects in experimental models of acute and subacute stage of stroke. These cells have immunomodulatory and anti-inflammatory effects in addition to regenerative effects. Hence, they can protect the penumbral tissue from further injury caused by inflammation after stroke 14 . MNCs have immunomodulatory effects by changing the phenotype of splenocytes 9 . In the acute or subacute stages, intravenous MNC therapy may achieve neural protection via the modification of systemic immunomodulation.
MNCs, and other adult tissue-derived stem cells can be easily isolated and expanded in the laboratory. The safety profile of MNCs has been consistently demonstrated clinical trials. However, their efficacy is unclear 15,16 . The quality, activity, and stemness of donor stem cells are the fundamental requirement for a successful stem cell therapy. The cell conditions are significantly correlated to donor origin. In autologous transplantation, the donor cells are collected from sick patients thus the stem cells are possibly unhealthy. Cryopreserved MNCs in allogenic transplantation, unlike autologous transplantation, are collected from a healthy donor, and they have a stabilizing quality.
The appropriate stem cell number for stroke therapy is not fully elucidated. However, a minimum single dose of 840 million is required for the intravenous injection of mesenchymal stem cells 17 . A benefit-to-risk compromise between cell number and toxicity should be based on the maximal benefit and the lowest cell dosage 18 . Enhancing the permeability of stem cells when crossing the blood–brain barrier (BBB) may reduce the required amount of stem cells. Mannitol can break the BBB to facilitate the peripheral delivery of stem cells 19 . Although the cell count in this case was 263 million, mannitol was used to open the BBB 30 min after MNC infusion. In addition to increasing the permeability of stem cells, the expression of neurotrophic and neural growth factors across the BBB can be enhanced by this drug 20,21 . Therefore, aside from MNC cell regeneration, the effects of by-products derived from transplanted MNCs can contribute to synaptogenesis, immature neuron proliferation, and neuronal cell migration.
Conclusion
This preliminary result showed that an adult patient with ischemic stroke significantly recovered within a short period after receiving allogeneic umbilical cord blood MNCs with an HLA matching score of 6/6.
Footnotes
Abbreviations
Author Contributions
T.K.L, C.Y.L. S.T.T. and P.H.T. designed the research, performed the experiments, and wrote the manuscript; Y.C.L., and S.Z.L analyzed the data and interpreted data; J.C.W., and C.Y.H. contributed analytical tool. All authors were involved in editing the manuscript and gave final approval of the submitted final published format.
Tian-Kuo Lee and Sheng-Tzung Tsai shares equal contribution.
Declaration of Conflicting Interest
PRS and CVB hold patents with the University of South Florida for the use of human umbilical cord blood-derived cells, including MNCs, as a cell therapy for stroke and several disorders. PRS and CVB provided the intellectual and technological expertise in the use of mannitol and human umbilical cord blood-derived cells. The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethical approval to report this case series was obtained from *The Research and Ethical Review Committees and Internal Review Board of Tzu Chi Hospital (IRB105-71-A)*.
Statement of Human and animal Rights
All procedures in this study were conducted in accordance with the *The Research and Ethical Review Committees and Internal Review Board of Tzu Chi Hospital (IRB105-71-A)* approved protocols.
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge financial supported by a grant from StemCyte International Ltd. USA.
