Abstract
Cerebral palsy (CP) is a common disability which results in permanent chronic motor disability appearing in early childhood. Recently human umbilical cord blood mesenchymal stem cell (hUCB-MSC) infusion has emerged as a promising therapeutic strategy for CP, and the treatment efficacy remains to be confirmed by clinical trials. All 54 patients received basic rehabilitation as a background treatment. The infusion group comprising 27 patients received 4 infusions of hUCB-MSCs (intravenous infusions at a fixed dose of 5 × 107) and basic rehabilitation treatment, whereas 27 patients in the control group received 0.9% normal saline and basic rehabilitation treatment. Several indices were tested from baseline up to 24 months posttreatment regarding efficacy and safety evaluations, including the gross motor function measurement 88 (GMFM-88) scores, the comprehensive function assessment (CFA), lab tests, electroencephalogram (EEG), routine magnetic resonance imaging (MRI), and adverse events. The changes in the total proportion of GMFM-88 and total scores of CFA in the hUCB-MSC infusion group were significantly higher than that in control group at 3, 6, 12, 24 months posttreatment. Less diffuse slow waves were noticed after hUCB-MSC infusion in patients with slowing of EEG background rhythms at baseline. Based on the routine MRI exams, improvements in cerebral structures were rare after treatment. Serious adverse events were not observed during the whole study period. The results of the study indicated that hUCB-MSC infusion with basic rehabilitation was safe and effective in improving gross motor and comprehensive functions in children with CP.
Introduction
Cerebral palsy (CP) is one of the leading causes of childhood disability and affects the individual's development and ability to function 1 . CP encompasses a group of movement disorders and affects posture. Motor limitations are attributed to a static disturbance in the developing brain, often accompanied by associated impairments and secondary health conditions 2 . The pathogenesis of CP is still unclear, and several factors are considered to contribute to CP, such as prenatal factors (hypoxia and infections) 3 , birth weight 4 as well as genetic factors 5 . Despite new developments in the field of obstetrics and perinatology, incidence of CP is reported as 2 out of 1,000 live births in premature deliveries and 1.1 out of 1,000 live births in mature babies (40+ weeks’ gestation) 6 . The weighted prevalence of CP-related motor impairment is 1.25/1,000 children in China. For mild, moderate, severe, and extremely severe groups of motor impairment, weighted proportions of CP are 14.12%, 20.35%, 27.44%, and 38.09%, respectively. Weighted proportions of concurrent visual, hearing, and cognitive impairment are 5.00%, 6.98%, and 71.06%, respectively 7 .
Of all available treatments for CP, mesenchymal stem cell (MSC) infusion was considered as a promising therapeutic alternative in clinical practices and numerous trials in recent decades 8,9 . Additionally, developments in rehabilitation have also improved performance in several aspects, including intensive upper extremity training to improve bimanual performance, strength training to improve muscle strength, hippotherapy to improve muscle symmetry, and activities and balance training to improve reactive balance 10 . It was more convenient to obtain human umbilical cord blood-derived MSCs (hUCB-MSCs), as their use has fewer associated ethical issues, and the cells have low immunogenicity when compared to other types of MSCs 11 . Compared to bone marrow-derived MSCs, hUCB-MSCs are more appealing due to their higher proliferative capacity, lower immunogenicity, and stronger immunosupressive potential 12 . Based on the therapeutic potential and convenient procurability, clinical research has been performed to study the safety and efficacy of hUCB-MSC infusion in children with CP, and the results were promising in some reported cases 13 . For further confirmation of the safety and effectiveness of cord blood-derived stem cell infusion, several clinical studies have been conducted 11,14,15 . Only 1 randomized double-blind, placebo-controlled trial was registered in the clinical trial database to date (clinicaltrials.gov identifier NCT01988584) 16 , which is still ongoing. Nowadays, combined therapies have become a common strategy for CP treatment, and rehabilitation training was usually applied as a basic adjunctive therapy. Based on the data from our previous study, a significant improvement in gross motor function was observed when hUCB-MSC infusion and basic rehabilitation were combined 17 . The efficacy and safety in combining hUCB-MSC infusion and rehabilitation would be further clarified with a randomized controlled clinical trial.
Materials and Methods
Ethics
This trial was approved by the institutional review board (IRB) of the Taihe Hospital affiliated to Hubei Medical College (Hubei, China, Ethical approval No. 2010-06). Informed consent forms were approved by the IRB and were signed by the patients or guardians before the commencement of the study.
Target Population
Patients fulfilling the inclusion criteria as stated below were eligible for participating in the study. must be diagnosed with CP according to the national standard criteria
18
including clinical history and physical examination, aged between 3 and 12 years, no prior history of epilepsia gravior within 15 d of infusion and no seizure attack within 24 h of treatment, must be able to comply with scheduled visits, treatment plans, physical examinations, laboratory tests, performance scales, and other study procedures, and showing willingness by signing the informed consent form, which was approved by the IRB for patients or parents.
Patients having liver or renal dysfunction at enrollment, any known genetic or immunological disorder, coagulation disorder, malignancy history, known allergy to more than 2 kinds of food or medications, current severe infection, or any other features that hampered the compliance with the requirement of the protocol according to the clinical judgment of the investigator were excluded from study. All the patients were recruited from China.
Study Design
This trial was designed as a placebo-controlled, single-blind study. A total of 56 children with CP were enrolled during the recruitment phase which began on September 20, 2010, and 2 patients dropped out before the second course and were lost to follow-up. The last follow-up visit was in September 2015. All of the patients were randomly assigned to 2 groups on a 1:1 allocation. All of the patients and their families were blinded to the group assignment, and the patients received hUCB-MSC infusion with basic rehabilitation in the infusion group, whereas patients in the placebo-controlled group received basic rehabilitation and normal saline (0.9% NS). The investigators and charge nurses were made aware of the treatment information to handle emergencies, if any. The placebo and hUCB-MSC injectates could not be distinguished from one another by their appearances since hUCB-MSC turbid liquid was diluted with 0.9% NS before infusion.
Assessment Parameters and Schedule
The study schedule is shown in Table 1 with detail information as below.
Study Schedule Flow Diagram.

Abbreviations: ECG, electrocardiogram; EEG, electroencephalogram; MRI, magnetic resonance imaging; GMFM, gross motor function measurement; hUCB-MSCs, human umbilical cord blood mesenchymal stem cells; NS, normal saline.
aAllowed a time window of 7 d ahead or after the scheduled day.
bThe study process items with an X mark should be completed as scheduled.
cThe study process items with this mark was optional to perform according to patients’ status judging by investigators.
Baseline assessments
The baseline assessments of the patients were done 7 d prior to first dose. They consisted of physical exams, various laboratory functional tests, electrocardiogram (ECG), electroencephalogram (EEG), magnetic resonance imaging (MRI), gross motor function measure 88 (GMFM-88) scale, and comprehensive functional assessment (CFA) scale. Among laboratory functional tests, hematology test, biochemistry test, blood coagulation, serum pathogen test, liver and renal function test, immunologic tests, and urinalysis were performed.
Safety measures
The safety assessments were performed during baseline and follow-up phases. Follow-up visits were conducted as they were scheduled, at the 3rd month (± 7 d), 6th month (± 7 d), 12th month (± 15 d), and 24th month (± 15 d) posttreatment, respectively. The physical exam, lab tests, and ECG were performed at follow-up visits. Regarding lab tests, only hematology, biochemistry, liver and renal function tests, immunologic tests, and urinalysis were required in follow-up phases. MRI was performed at baseline and posttreatment at the 6th month (± 7 d), 12th month (± 15 d), and 24th month (± 15 d).
The adverse events were also considered as safety outcomes, and relative information was collected including the event title, severity grade, and relation to the study process.
Efficacy measures
The efficacy outcomes were assessed as follows. The GMFM-88 scale was usually employed to evaluate the recovery of gross motor ability of children with CP. Eighty-eight questions were addressed in the GMFM-88 scale for 5 function areas including “lying and rolling,” “sitting,” “crawling and kneeling” “standing,” and “walking, running, and jumping.” Raw scores were collected. The proportion of raw scores in total scores for each function area was calculated since the full score in each area was different. Then the sum of raw scores in each function area was shown as a total score, and the proportion of total raw score in full mark at each study phase was evaluated. The changes in total score proportion and each functional score proportion between different phases were processed for analysis. The CFA scale was used for functional assessment in 5 functional areas including cognizance, language competence, self-care, motor function, and social adaptability. Raw scores were collected in each functional area, and the total scores were calculated as their sum. Then the changes in total scores and each functional score were processed for analysis.
Scales were finished at baseline and post-treatment during the 3rd month (± 7 d), 6th month (± 7 d), 12th month (± 15 d), and 24th month (± 15 d). The outcome results of scales were expressed as the main efficacy indices to show the overall functional improvement in the treatment. The difference between baseline and post-treatment scores was calculated. The changes in total score proportion in the GMFM-88 and the total score changes in the CFA were evaluated as clinical effective power. The clinical efficacy evaluation results could be divided into 3 levels according to the effective power: significantly effective when effective power reached 20%; effective when effective power reached 10% but less than 20%; and ineffective when effective power was less than 10%.
Exploratory measures
EEG was believed to be an exploratory measure to demonstrate the accompanied changes in cerebral electrophysiology, if any. It was performed at baseline and posttreatment during the 6th month (± 7 d), 12th month (± 15 d), and 24th month (± 15 d).
Besides, changes in cerebral structure were checked by routine MRI to explore the correlation between cerebral electrophysiology and structure, if any. All the patients were scanned with a GE HDX 3.0 T system (GE Healthcare, Pittsburgh, PA, USA) for MRI exams with sequence of 3 dimensional longitudinal relaxation time weighted imaging(T1WI ), transverse relaxation time weighted imaging (T2WI), and transverse relaxation time-Flair (T2-Flair).
Study treatment
hUCB-MSC source
Allogeneic hUCB-MSCs were acquired from the UCB bank of Beike Biotechnology Company (Shenzhen, China). All manufacturing processes and laboratories met the standards for good manufacturing practices and good tissue practices. The resources were from the umbilical cord blood and tissues of healthy puerperal women with a negative result for all the following tests: syphilis, HIV, hepatitis B virus (HBV), and toxplasma, nubellavirus, cytomegalo virus, herpes simplex virus, and other virus (TORCH 5). Several tests were performed on the collected samples following donor’s requirement from the United States Food and Drug Administration (USFDA) and American Association of Blood Banks. Then the hUCB-MSCs were separated by centrifugation and cultivated in Dulbecco’s modified Eagle’s medium (Thermo Fisher Scientific, Waltham, MA, USA) enhanced with basic fibroblast growth factor (bFGF) and epidermal growth factor (EGF, Thermo Fisher Scientific) instead of any animal products. The culture medium was washed away when the cells were harvested. The hUCB-MSCs were passaged and collected at the fourth generation, before storing them at −196°C in cryopreservation storage equipment. A series of testing was carried out to ensure the quantity and quality of hUCB-MSCs before transporting for clinical usage, including tests for cytomegalovirus, human T-cell leukemia virus, and microorganisms, such as aerobic bacteria, anaerobic bacteria, and fungi. All these tests were negative. Additionally, the parameters of hUCB-MSC vitality, and biological characteristics were tested to meet with International Society for Cellular Therapy standards 19 and documented in test reports for each batch of cell product, including morphology, cell count (5× 10 7 cell/mL, range 90% to 120%, and 1 mL/vial), viability rate (≥90%), cell surface markers, colony-forming ability, differential capacity, and so on. Regarding the cell surface markers, it was found that CD90, CD73, CD105, and CD29 were higher than 95%, and CD45, CD34, CD79a/ CD19, CD14, and HLA-DR were less than 2%.
Study treatment in hUCB-MSC infusion group
All patients in the hUCB-MSC infusion group received intravenous infusions and basic rehabilitation therapies as scheduled. The patients were infused with hUCB-MSCs at day 1 after randomization and then given 3 infusions in each course with an interval of 7 d between infusions. The infusion procedure was carried out twice with a 3-month interval between courses. At each treatment course, the hUCB-MSCs were infused at a fixed quantity as 5 × 107 cells after they were dispersed and mixed in 30 mL 0.9% NS. All the treatments were performed in a specific room for hUCB-MSC infusion. Immunosuppressant was not administered in our study, considering the immunosuppressive properties of MSCs12. Allogeneic hUCB-MSCs maintained low immunogenicity in vitro and in vivo, suggesting it was immunologically safe for use in allogeneic clinical applications 20 . Moreover, it was suggested that hUCB-MSCs could be successfully transplanted even when they were major histocompatibility complex (MHC) mismatched 21 . Therefore, the compatibility test between donor and recipient was not considered before hUCB-MSC infusion. Additionally, the safety of allogeneic hUCB-MSC infusion without concomitance of immunosuppressant was shown in a case report 17 . In order to monitor the rejection reaction, if any, immunologic tests were performed before and after infusion, including immounoglobin (Ig)A, IgM, IgG, C3, C4, rheumatoid factor (RF), hypersensitive C reactive protein (hsCRP), and anti-streptolysin O (ASO). All the test results were within normal ranges.
Basic rehabilitation treatments were performed by rehabilitation physiatrists for CP, including Bobath therapy and conductive education. They were carried out as scheduled. Each session lasted 40 min, twice a day, and 6 d per week.
Study treatment in control group
All the patients in the control group were given 30 mL 0.9% NS and basic rehabilitation with the same procedure as that in the hUCB-MSC infusion group. Bobath therapy and conductive education were performed by rehabilitation physiatrists for CP, as scheduled. Each session lasted 40 min, twice a day, and 6 d per week.
Statistical analysis
Statistical analysis was carried out using SPSS version 19.0 (IBM, Armonk, NY, USA), and efficacy data were treated as continuous variables for analysis and presented as mean ± standard error of mean. Differences in GMFM-88 and CFA scores between groups were evaluated by analysis of variance (ANOVA), followed by least-significant difference (LSD) as post hoc test. Statistical significance was considered if
Results
Patient Demographics
Except for 2 patients who dropped out and were lost to follow-up without efficacy assessments, 54 patients in total completed all the required study evaluations at scheduled time points and were included in the statistical analyses. No significant difference in demography was noticed between 2 groups (Table 2). There were more males in the patient pool seeking treatment. In both groups, immature labor was noticed as a significant factor for CP. Preterm incidence reached 40.7% in the hUCB-MSC infusion group and 37% in the control group. The top 3 pathogenesis risk factors were hypoxia (29.0%), low birth weight (25.8%), and infection (22.6%) among the patients, and the other factors included neonatal jaundice, trauma, hydrocephalus, and genetic disease. Several patients in both groups with previous rehabilitation treatment showed limited benefit.
Patient Demographic Characteristics at Baseline.

Abbreviations: hUCB-MSCs, human umbilical cord blood mesenchymal stem cells; SD, standard deviation.
Safety Assessments
Safety assessments were performed by adverse event (AE) documentation and analyses according to study schedule. Any complaints, symptoms, or abnormal results with clinical significance in physical exams, lab tests, ECG, and other examinations were considered as AEs. No serious adverse events (SAEs) were observed during the whole study period. Among all observed non-SAEs, upper respiratory tract infection and diarrhea were the most frequently reported. None of the observed AEs influenced the study significantly. Furthermore, no immunologic rejection event, such as fever or leucopenia, was detected. MRI results demonstrated that there was no sign of cerebral tumor over the complete study course. Furthermore, no other AEs were observed in general laboratory results and physical examinations. The results of AEs were shown in Table 3.
Adverse Event (AE) Case Number and Severity Grades.

Abbreviation: hUCB-MSCs, human umbilical cord blood mesenchymal stem cells.
aGrade referred to the severity of adverse events based on the following general guideline of International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use- Good Clinical Practice (ICH-GCP): grade 1: mild AE. Clinical intervention was not involved usually; grade 2: moderate AE. Clinical intervention depended on the case; grade 3: severe AE. Clinical treatment was needed usually; grade 4: life-threatening or disabling AE; grade 5: death related to AE.
Efficacy Assessments
No considerable difference was observed in baseline functional assessments between the 2 groups, including GMFM-88 scale scores and the CFA scores. The improvement versus baseline status after hUCB-MSC infusion was significantly higher in the infusion group than that in the control group.
Functional improvement—GMFM-88
The GMFM-88 total score proportion at the baseline level was evaluated as 84.99 ± 0.85 in the hUCB-MSC infusion group and 85.03 ± 0.76 in the control group. No significant difference was observed between groups at baseline. The change in total score proportion for the GMFM-88 assessment was evaluated (Table 4), and the clinical efficacy evaluation result was effective in the hUCB-MSC infusion group at 12 and 24 months posttreatments. The clinical effective power did not reach effective level in the control group though the change in the total score proportion increased slightly with treatment. Superior efficacy was shown significantly in hUCB-MSC infusion group, based on the change in total score proportion for GMFM-88 assessment over 3, 6, 12, and 24 months postinfusion (
The Change in Total Score Proportion in GMFM-88 Evaluation.a
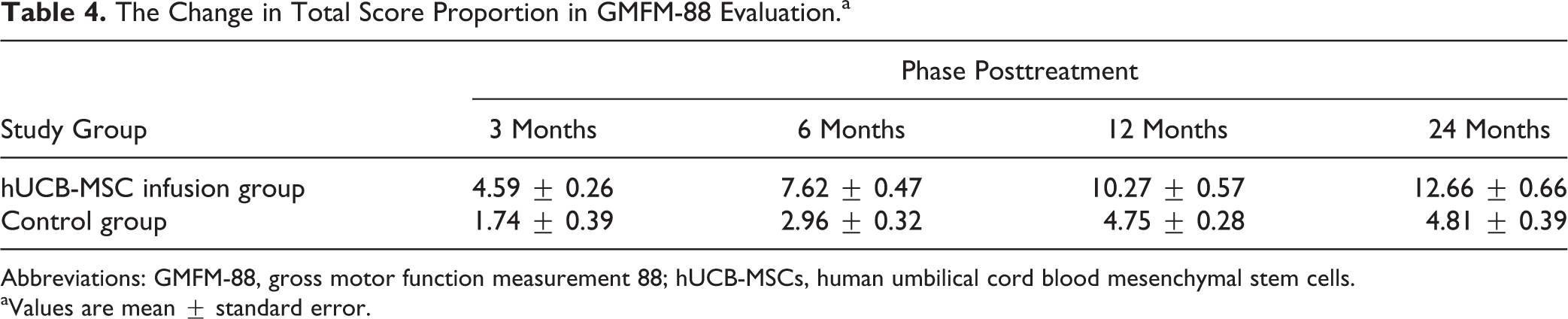
Abbreviations: GMFM-88, gross motor function measurement 88; hUCB-MSCs, human umbilical cord blood mesenchymal stem cells.
aValues are mean ± standard error.

The change in gross motor function measurement 88 (GMFM-88) total score proportion in 3, 6, 12, and 24 months posttreatment.

The change in gross motor function measurement 88 (GMFM-88) proportion in each function area at 3, 6, 12, and 24 months posttreatment.
Functional improvement—CFA
CFA total scores at baseline level were 66.04 ± 1.48 in the hUCB-MSC infusion group and 65.70 ± 1.37 in the control group. No significant difference was observed between groups. The total score change in CFA was evaluated (Table 5), and the clinical efficacy evaluation result was effective at 6 months and 12 months posttreatment, while significant efficacy was observed at 24 months posttreatment in the hUCB-MSC infusion group. The clinical effective power did not reach the lower limitation of effective level until 24 months posttreatment in the control group. The differences between total score changes in 2 groups were also significant over 3, 6, 12, and 24 months posttreatment (
The Change in Total Score in CFA.a

Abbreviations: CFA, the comprehensive functional assessment; hUCB-MSCs, human umbilical cord blood mesenchymal stem cells.
aValues are mean ± standard error.

The change in comprehensive functional assessment (CFA) total score in 3, 6, 12, and 24 months posttreatment.

The change in comprehensive functional assessment (CFA) score in each function area in 3, 6, 12, and 24 months posttreatment.
Exploratory measure— EEG and MRI
All 54 patients underwent EEG exams, while abnormal results were shown in 20 patients at baseline. Among the 7 patients with slowing of EEG background rhythms in EEG reports, less diffuse slow waves were noticed in all 4 patients in the infusion group after hUCB-MSC treatments. It appeared that the cerebral electrophysiology might be improved after hUCB-MSC infusions. However, we could not come to a definite conclusion due to the limitation of data. Furthermore, EEG status did not improve with regard to increasing θ waves, δ waves, sporadic cusp, or sharp waves in other patients.
According to the MRI results, pathological changes were noticed in 41 patients at baseline, including periventricular leukomalacia (41.5%), ventriculomegaly of lateral ventriclesp (19.5%), hydrocephalus (17.1%), subcortical atrophy (12.2%), callosum hypoplasia (7.3%), and other cases of cerebral congenital hypoplasia (2.4%). Based on the routine MRI exams, the improvements in cerebral structures were rare after treatments.
Discussion
The therapeutic benefits of hUCB MSCs 22,23 , mononuclear cells (MNCs) 24 , and plasma 25 were investigated for various neurological diseases. Although the development of neuroregeneration provided some new options for CP therapy, alternative therapies were still limited. Based on the current data in clinical trials and case reports, hUCB-MSC infusion was a promising therapy compared to other types of MSCs. They were immunologically safe and easily accessible 26 . The clinical application of MSC infusion was limited by several technical hurdles, such as source, transplant viability, therapy timing, dose, and immunogenic toxicity 11 . Different studies had various therapy timelines, frequencies, and doses. It was believed that the minimum necessary cell dosage for cell engraftment was 1 × 10 7 cells/kg 27 . Regarding the risk of overdose infusion, it was also reported that the umbilical cord blood (UCB) single-dose infusion units with total nucleated cell dose of more than 21×107 cells/kg was one of the factors leading to higher mortality 28 . Single-dose infusion of UCB was reported in some studies with cell dose as 2 × 107 cells or 3 × 107 cells/kg 15,29 . Furthermore, it was suggested that the neuroprotective effect of hUCB MNCs could be enhanced by repeated cell administrations in a mouse model 30 . Multiple-dosing infusion was also used in some case reports 13 and was shown effective at a fixed quantity of 5 × 107 cells/dose, in our study. No difference was observed in AE incidence between groups. According to our results, it could be safely concluded that the hUCB-MSC infusion with basic rehabilitation was safe and effective to improve the gross motor function in children with CP.
In particular, significant improvement in gross movement was shown after hUCB-MSC infusion. The beneficial effects observed in the hUCB-MSC infusion group was superior to the control group with basic rehabilitation therapy only. The improvements in GMFM-88 total score proportion reached effective level in clinical efficacy evaluation 12 months posttreatment, while it failed to reach the effective level in the control group during the whole study phase (Table 4). It was coincident with clinical observation. The therapeutic efficacy of basic rehabilitation was influenced by several factors, such as treatment duration, patient compliance, efficacy evaluation system, and so on. Regarding the benefits of basic rehabilitation to gross motor function, the improvement was limited, based on the clinical effective power assessment within the 24-month follow-up phase, though slight improvement was observed with the treatments. The clinical effective power evaluation in CFA total scores was effective at 6 months and significantly effective at 24 months posttreatment in the hUCB-MSC infusion group. It was noticed that the clinical effective status in CFA was observed 6 months ahead of that in GMFM-88. It indicated that improvements in comprehensive function could occur even before significant development of gross motor function in clinical practice. Besides, in the control group, comprehensive function was improved at the effective level at 24 months posttreatment (Table 5). Taking the evaluation of GMFM-88 and CFA in the control group together, the efficacy of basic rehabilitation was limited, though it seemed inclined to improve slightly. The efficacy of basic rehabilitation was still ambiguous. The persistent efficacy of basic rehabilitation to CFA in the control group could not be evaluated after 24 months posttreatment due to the limitation of the study follow-up phase. Further study was needed since the lower limitation of the clinical effective level was just reached in the control group at 24 months posttreatment.
The safety of hUCB-MSC infusion was ensured at the 24-month follow-up phase after the last dose was administered. The patients did not show any signs of SAE nor immunologic rejection.
The cerebral function might be improved since better cerebral electrophysiologic trace was observed in the EEG of some patients. However, cerebral structure improvements were not observed in routine MRI results. This might be due to insufficient sensitivity of routine MRI to measure the changes at the cellular level. Diffusion tensor imaging (DTI) was superior to routine MRI on corticospinal tract (CST) imaging, and the correlation between DTI parameters of the injured CST and the severity of motor dysfunction had been confirmed in previous studies 31 .
Moreover, after rehabilitation treatment, the DTI parameters could also reflect the motor function outcomes of pediatric patients with hemiplegic CP 32 .
Furthermore, increased metabolic activity in various areas of the brain could be shown in 18F-fluorodeoxyglucose positron emission tomography (18F-FDG-PET/CT) scan images post autologous bone marrow mononuclear cell therapy in CP patients 33,34 . It was indicated cerebral functional improvement could be induced by MSC therapy. The functional improvement was detectable given a proper technical approach. Further functional neuroimaging would be needed to explore the effects of hUCB-MSC therapy on cerebral functional areas, such as DTI and 18F-FDG-PET/CT.
Various preclinical studies on CP models showed that infusion of MSCs could lead to homing, viability, and differentiation of these cells into neurons, oligodendrocytes, astrocytes, and so on 27,35 . hUCB-MSCs were studied extensively in rat models with neonatal hypoxia/ ischemia and significant improvements were observed in behavioral function. The possible mechanisms of action include reducing proinflammatory cytokine levels 36,37 , and paracrine effects 27 to stimulate recovery in the injured brain. However, it might not be realistic to improve neurological function through neuronal replacement by hUCB-MSC infusion intravenously, considering the limited quantity of MSCs through the hematoencephalic barrier 27 . Although the safety of hUCB-MSC infusion was ensured up to 24 months in our long-term follow-up phase, further double-blind, randomized, placebo-controlled trials are necessary to obtain more evidence of efficacy. Further neuroimaging monitoring would be of help to identify the influence of hUCB-MSC therapy on cerebral function in our future studies.
Footnotes
Acknowledgments
We acknowledge the assistance of all the patients and their families in this study.
Authors’ Contribution
Li Huang and Che Zhang contributed equally to this work.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This study was approved by the Institutional Review Board (IRB) of the Taihe Hospital Affiliated to Hubei Medical College (Hubei, China, Ethical approval No. 2010-06).
Statement of Informed Consent
Informed consent forms approved by IRB were signed by the patients or legally authorized representative before commencement of study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The present work was supported by Hubei Province Technology Fund No. 2013BCB002 and was conducted at the Children’s Medical Centre of Taihe Hospital (Hubei, China).
