Abstract
Prior to transplantation into individuals with type 1 diabetes, in vitro assays are used to evaluate the quality, function and survival of isolated human islets. In addition to the assessments of these parameters in islet, they can be evaluated by multiparametric morphological scoring (0–10 points) and grading (A, B, C, D, and F) based on islet characteristics (shape, border, integrity, single cells, and diameter). However, correlation between the multiparametric assessment and transplantation outcome has not been fully elucidated. In this study, 55 human islet isolations were scored using this multiparametric assessment. The results were correlated with outcomes after transplantation into immunodeficient diabetic mice. In addition, the multiparametric assessment was compared with oxygen consumption rate of isolated islets as a potential prediction factor for successful transplantations. All islet batches were assessed and found to score: 9 points (n = 18, Grade A), 8 points (n = 19, Grade B), and 7 points (n = 18, Grade B). Islets that scored 9 (Grade A), scored 8 (Grade B) and scored 7 (Grade B) were transplanted into NOD/SCID mice and reversed diabetes in 81.2%, 59.4%, and 33.3% of animals, respectively (P < 0.0001). Islet scoring and grading correlated well with glycemic control post-transplantation (P < 0.0001) and reversal rate of diabetes (P < 0.05). Notably, islet scoring and grading showed stronger correlation with transplantation outcome compared to oxygen consumption rate. Taken together, a multiparametric assessment of isolated human islets was highly predictive of transplantation outcome in diabetic mice.
Keywords
Introduction
Type 1 diabetes (T1D) is an autoimmune disease that results from the destruction of insulin-producing pancreatic islet β-cells. The majority of T1D individuals depend on insulin replacement to provide glucose homeostasis. However, in T1D, blood glucose control within a physiological range is not easily achieved, and uncontrolled hyperglycemia degrades the function of multiple organs including the heart, vasculature, kidneys and eyes 1,2 . Further, despite glycemic control with insulin, some T1D individuals experience life-threatening hypoglycemia and hypoglycemia unawareness. Islet transplantation (IT) provides insulin independence and mitigates life-threatening hypoglycemia unawareness and organ damage in T1D 3 –5 . Several factors influence the quality of isolated islets and subsequent islet transplantation outcomes. Donor characteristics including sex, body mass index, and age, as well as technical details of the isolation process, impact islet quality and outcomes following IT 6 –9 .
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)-funded Clinical Islet Transplantation Consortium released detailed standards for determining isolated islet quality that take into account islet equivalent (IEQ), purity, viability, and glucose-stimulated insulin secretion (GSIS) 10 –12 . While providing release criteria metrics, these assays do not predict IT outcomes well, except IEQs transplanted 13 . Recently, analyses of islet oxygen consumption rate (OCR) were reported to predict IT outcomes. OCR normalized to DNA content (OCR/DNA) and increment of OCR stimulated by the glucose predict IT outcomes in rodents 14 –16 , and OCR dose, the product of islet dose and OCR/DNA, predicts clinical allo- and auto-IT outcomes 17,18 . We developed a computer-based semi-automated viability assessment tool as an alternative to conventional manual viability assessment 19 . Other methods such as islet cellular composition assessment using laser scanning cytometry and the ADP/ATP ratio of isolated islets can assist in grading the quality of isolated islets 20,21 . Regardless of the assay employed, timely evaluation of islet quality is paramount. Unfortunately, current in vitro assays of isolated islet quality and function are time consuming. Thus, there remains a need for islet assessment tools that require little time and yet accurately predict IT outcomes. Herein, we tested a multiparametric assessment including islet morphological scoring (0–10 points) and grading (A, B, C, D, and F) based on the islet characteristics (shape, border, integrity, single cells, and diameter) to predict islet function after transplantation in immunodeficient diabetic mice.
Materials and Methods
Study Design
This study retrospectively determined the correlation between a multiparametric assessment of isolated islets prior to transplantation (n = 55, period of islet isolation: March 2015 – June 2018) and outcome of islet graft function following transplantation into immunodeficient diabetic mice. The parameters were also compared with OCR (n = 49).
Human Donor Pancreata for Islet Isolation
Human pancreata from deceased donors were obtained from a local organ procurement organization (OneLegacy, Los Angeles, CA, USA) for islet isolation. The pancreata were stored on ice in cold preservation solution (University of Wisconsin solution or histidine-tryptophan-ketoglutarate solution) and transported to the current Good Manufacturing Practices facility at City of Hope. All pancreata processed in this study were approved for research by the Institutional Research Board of City of Hope (IRB # 01046) and informed consent was obtained from family or relatives of the donors.
Isolation of Human Islets
Islet isolations were conducted as described 22 . Briefly, the pancreas was trimmed of fat and connective tissue before perfusion with a collagenase/protease solution through the main pancreatic duct using an automatic perfusion apparatus (Biorep Technologies, Miami Lakes, FL, USA). The pancreas was then cut into 7–12 pieces and further digested in a Ricordi digestion chamber at 37°C 23 . Isolated islets were purified with continuous density gradient centrifugation using a COBE 2991 cell processor system (Terumo BCT, Lakewood, CO, USA) 24 . Islet fractions were pooled according to purity as determined by Dithizone (DTZ) staining (iDTZ, Gemini Bio-products, CA, USA) 25 . Islet particulate number (IPN) was determined, and total islet counts were then converted into islet equivalent (IEQ) which represent islets of 150 µm diameter IEQ 26 . Islet morphological scoring was performed followed by culture at 37°C with 5% CO2 and subsequent analyses.
Islet Multiparametric Assessment
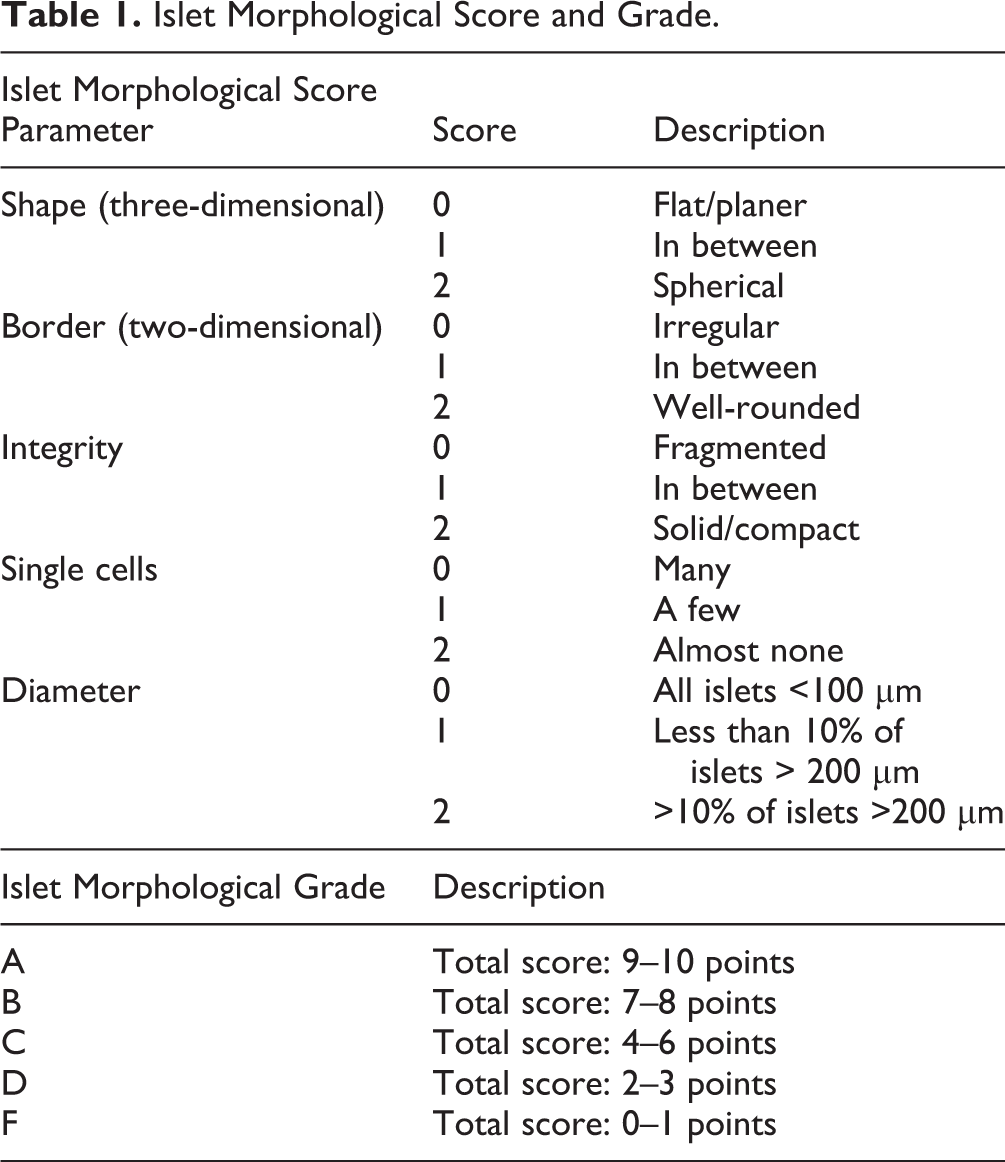
Scoring of DTZ-stained islets was completed on the day of isolation using a bright-field microscope (CKX31, Olympus, Tokyo, Japan) and a 4× objective lens. Details of the five scored parameters (shape, border, integrity, single cells, and diameter) are found in Table 1 12 . Each individual parameter was scored from 0–2 points, and the total score was calculated. The total scores were then categorized as A (best), B, C, D, and F (worst). Islet assessment was performed for the available islet fractions, as defined by the purity of each batch (fraction 1, ≥70% pure; fraction 2, 40–70% pure, and fraction 3, ≤40% pure).
Islet Morphological Score and Grade.
Metabolic Assessment of Isolated Islets
The oxygen consumption rate (OCR) of isolated islets was measured using a Seahorse XFe24 analyzer (Agilent, Santa Clara, CA, USA) 27,28 . Briefly, islets (70–100 IEQ) were plated into 4 or more separate wells of a Seahorse XFe islet capture plate. The islet OCR was measured at base line (3 mM glucose for 45 minutes), after glucose stimulation (20 mM glucose for 53 minutes) and during inhibition of mitochondrial respiration (5 nM oligomycin for 105 minutes). Measurements were repeated every 7.5 minutes until the end of the experiment. The basal OCR (OCR basal) was calculated as the average OCR obtained on incubation in 3 mM glucose solution. The maximum OCR was obtained at least 1 hour after the initiation of glucose stimulation. The OCR fold increase was defined as the maximum OCR/basal OCR (OCR_SI).
Other Post-Isolation Assessments
In addition to the islet multiparametric assessment and metabolic assessment of isolated islets, several post-isolation assessments were routinely performed in vitro, including purity, viability, and GSIS (static incubation), as described previously 19,29 . The viability of isolated islets were assessed with both conventional and semi-automated methods as previously reported 10,19 .
Human Islet Transplantation Into Diabetic Immunodeficient Mice
Animal studies were approved by the City of Hope Beckman Research Institute Institutional Animal Care and Use Committee. Male 8–12 week-old non-obese diabetic, severe combined immunodeficiency (NOD/SCID) mice (Charles River Laboratories, Wilmington, MA, USA) were used as recipients for islet transplantations as described 19,28 . Mice were rendered diabetic by intraperitoneal injections of 50 mg/kg of streptozotocin (Sigma-Aldrich, St. Louis, MO, USA) for three consecutive days. Mice with a blood glucose level >350 mg/dL underwent IT and blood glucose levels were monitored twice a week. Diabetes reversal was defined as a blood glucose <200 mg/dL on at least three consecutive measurements. A total of 202 mice were transplanted with isolated human islets from 55 separate isolations (donors). For each islet isolation, 1–8 mice were transplanted with 1200 IEQ under the left kidney capsule 28,30 . For quantitative evaluation of post-transplant blood glucose control in individual mice, the area under the curve (AUC) of blood glucose from days 0–28 (AUC_0-28) was determined 19 . These quantitative parameters correlated well with diabetes reversal (Supplemental Fig. S1A). AUC also correlated with the number of days to reverse diabetes in the mice that achieved a blood glucose <200 mg/dL (Supplemental Fig. S1B). As well, the diabetes reversal rate for each islet batch, defined as the number of mice with diabetes reversal/number of mice transplanted, was determined. Five islet isolation batches were excluded from the analysis since in these instances only 1 or 2 mice were transplanted.
Statistical Analysis
Comparisons between two factors were analyzed using the Student’s t-test. Multivariate analysis was performed to identify the statistical correlations between several variables (Pearson’s correlation). Receiver operating characteristic (ROC) curves were used to calculate the optimal cutoff value to distinguish two groups among the data set. The comparisons between two groups in the cumulative diabetes reversal assessment were analyzed using a log-rank test. All statistical analyses were performed using JMP 9.0.0 software (SAS Institute, Cary, NC, USA). A P < 0.05 was considered significant.
Results
Islet Characteristics and Morphological Scoring
Donor information and islet characteristics are shown in Table 2. All 55 islet preparations scored either 1 or 2 points for all parameters (Fig. 1). The distribution of the scores for each parameter is shown in Fig. 2A. Most islet isolation batches scored 2 points for all parameters except for islet integrity. The cumulative islet morphological score ranged between 7 and 9 points (full score = 10 points), with an average of 8.0 ± 0.1 points (Fig 2B). Islet isolation batches evenly distributed into each total score: 18 batches for 7 points, 19 batches for 8 points, and 18 batches for 9 points. Based on the grading shown in Table 1, 18 batches fell into Grade A (9–10 points) and 37 batches Grade B (7–8 points). These data confirmed that our standard operating procedure for pancreatic islet isolation delivered morphologically high-quality islets.
Donor and Islet Characteristics.
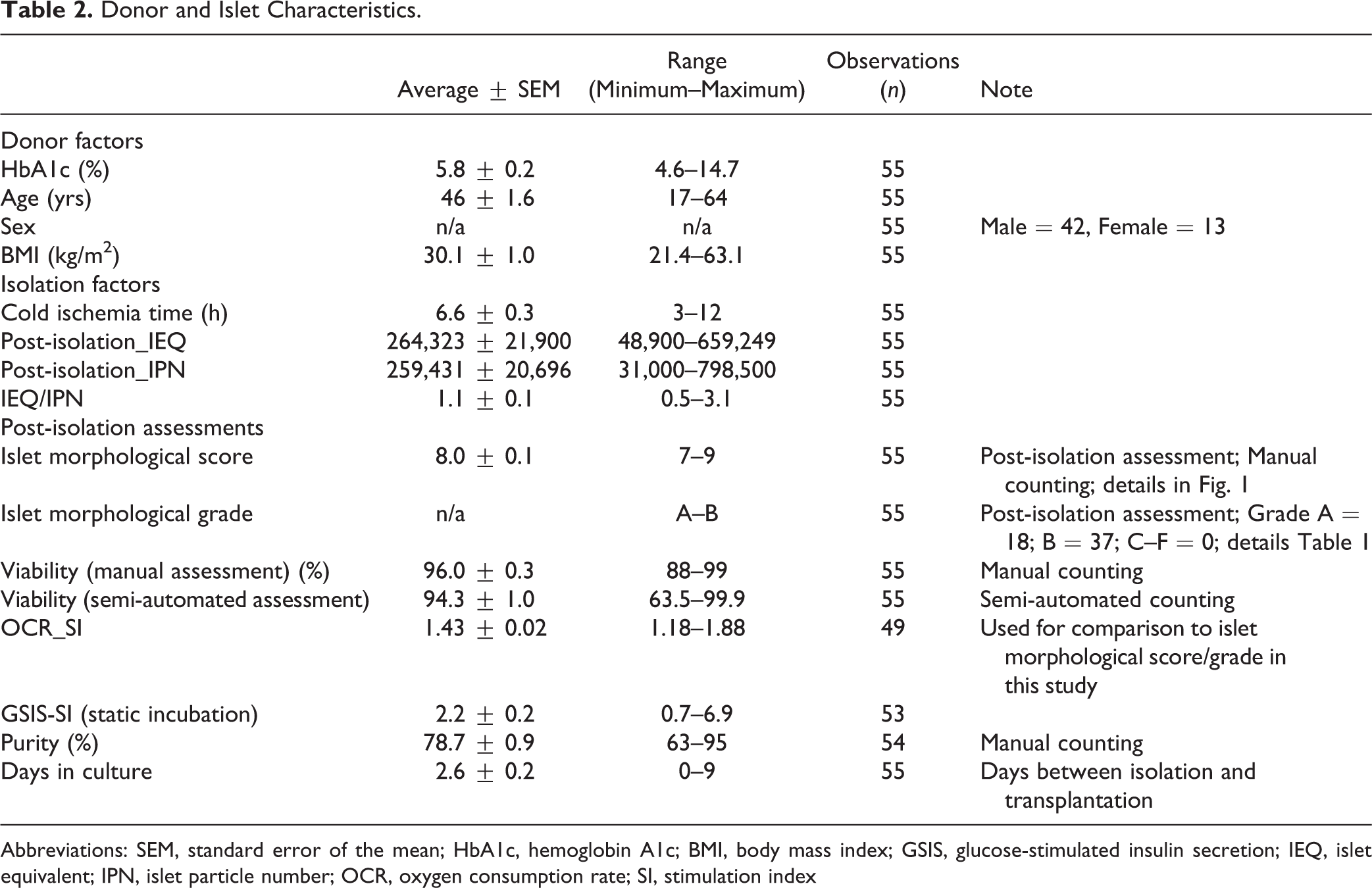
Abbreviations: SEM, standard error of the mean; HbA1c, hemoglobin A1c; BMI, body mass index; GSIS, glucose-stimulated insulin secretion; IEQ, islet equivalent; IPN, islet particle number; OCR, oxygen consumption rate; SI, stimulation index
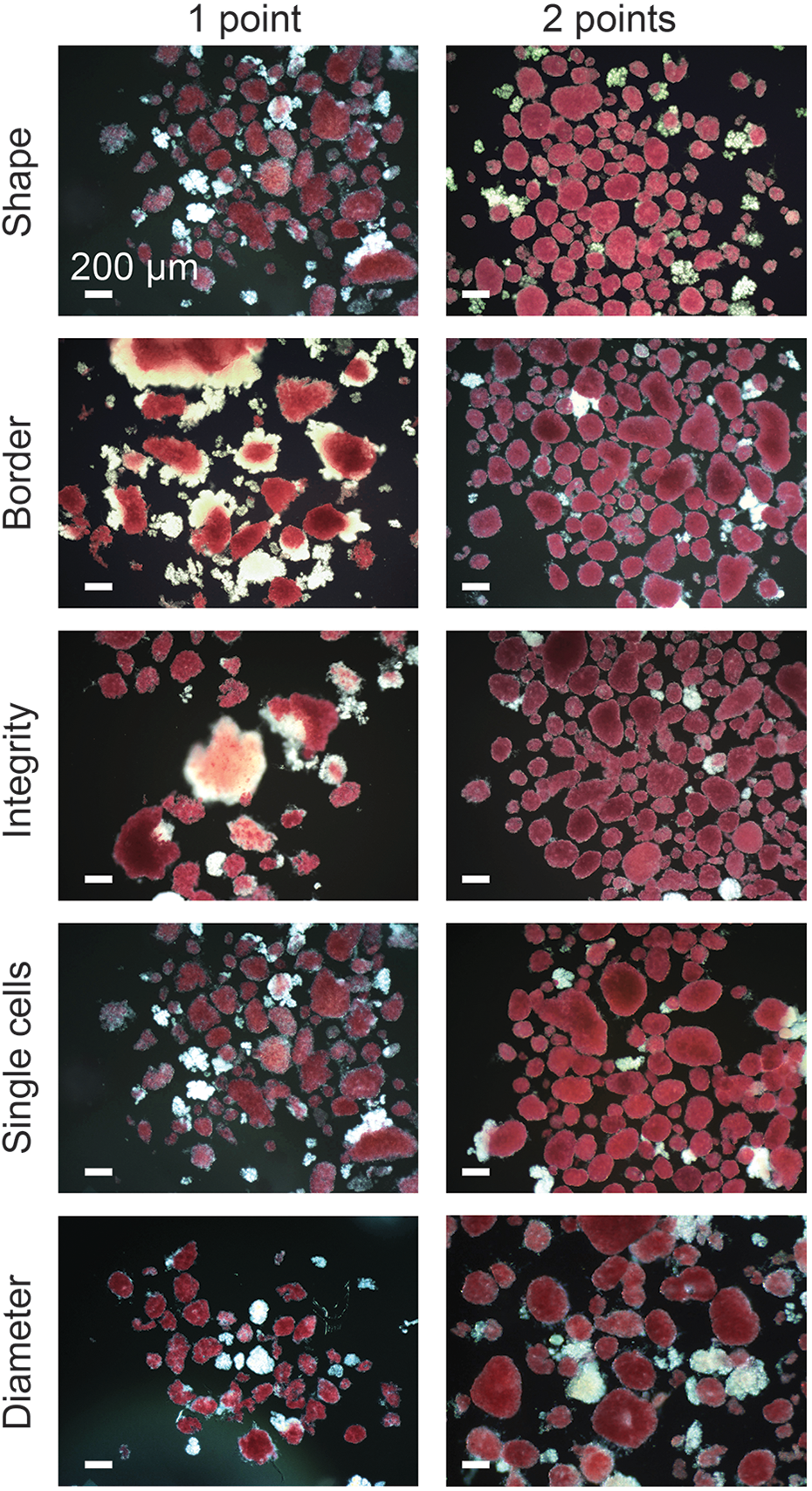
Representative images of isolated human islets displaying typical morphologic features. Images are from the dataset of the current study. Islets were scored for shape (three-dimensional), border (two-dimensional), integrity, single cells, and diameter. Each individual parameter was scored from 0 to 2 point(s). No islets scored 0 in any parameter. Islets were stained with iDTZ solution. Scale bar, 100 μm. See Table 1 for additional details.
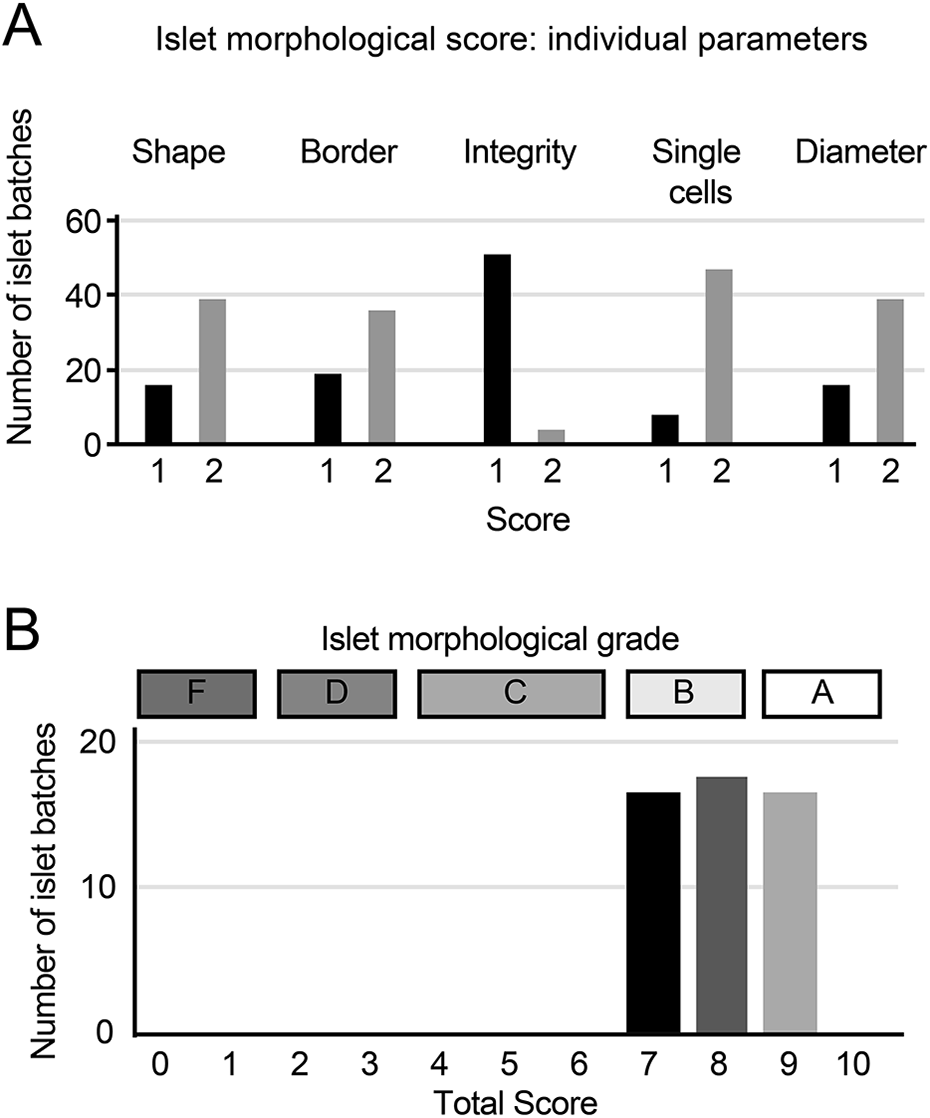
Distribution of islet morphological scores and grades. (A) Distribution of the scores of individual parameters (n = 55 islet batches in each parameter). (B) Distribution of the total scores and grades (n = 55 islet batches).
Islet Morphological Score Strongly Correlates with in vivo Islet Transplantation Outcomes
The results of human islet transplantation in mice are summarized in Table 3. We analyzed the correlations between islet assessment data and transplantation outcomes. The AUC_0-28 was used to quantify transplantation outcome in individual mice while the diabetes reversal rate of each islet batch was taken as an indicators of post-transplant glycemic control. Interestingly, islet morphological score was highly and inversely correlated to transplantation outcomes (r = −0.4048, P < 0.0001) (Table 4). Notably, it showed stronger correlation to the AUC_0-28 as compared to OCR_SI (r = −0.282, p = 0.0001) and semi-automated viability (r = −0.204, p = 0.0039). As well, the islet morphological score was the only variable that significantly and positively correlated with the diabetes reversal rate (r = 0.2876, p = 0.0428).
Characteristics of Mouse Transplantation Data.
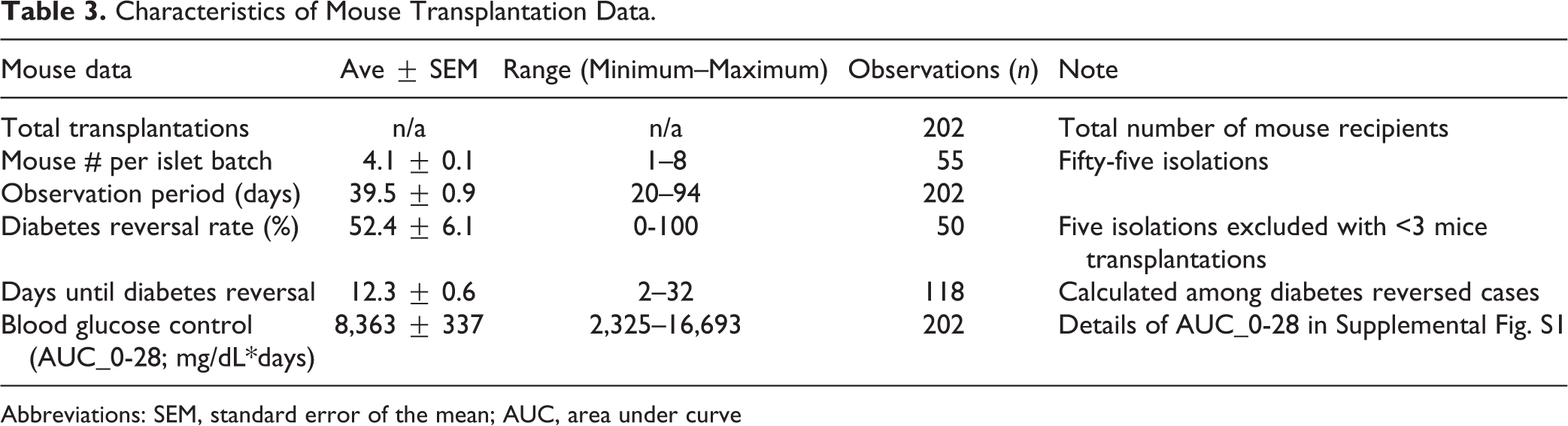
Abbreviations: SEM, standard error of the mean; AUC, area under curve
Post-Isolation Assessments with Significant Correlations to Transplantation Outcomes.
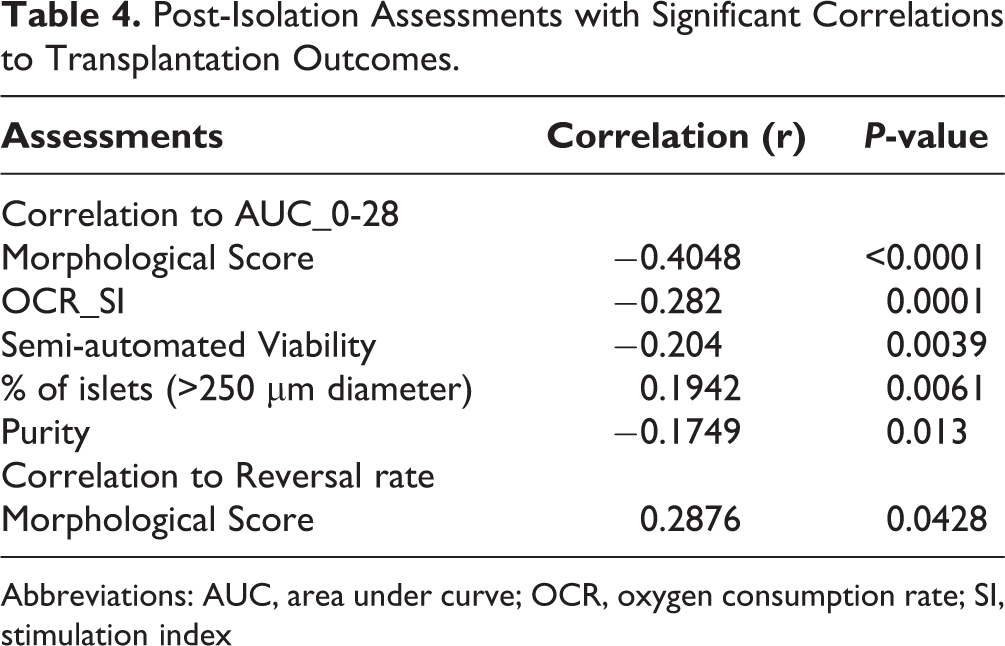
Abbreviations: AUC, area under curve; OCR, oxygen consumption rate; SI, stimulation index
We further investigated if transplantation outcomes varied in relation to islet morphologic score. We found that islet morphological scores significantly impacted the post-transplant glycemic control as measured by AUC_0-28 (Fig. 3A). Indeed, diabetes reversal rates tracked quite closely with islet morphological scores (Fig. 3B, P < 0.0001). Mice that received islets that were scored 9, 8, and 7, showed reversal of diabetes in 81.2%, 59.4%, and 33.3% of animals, respectively.
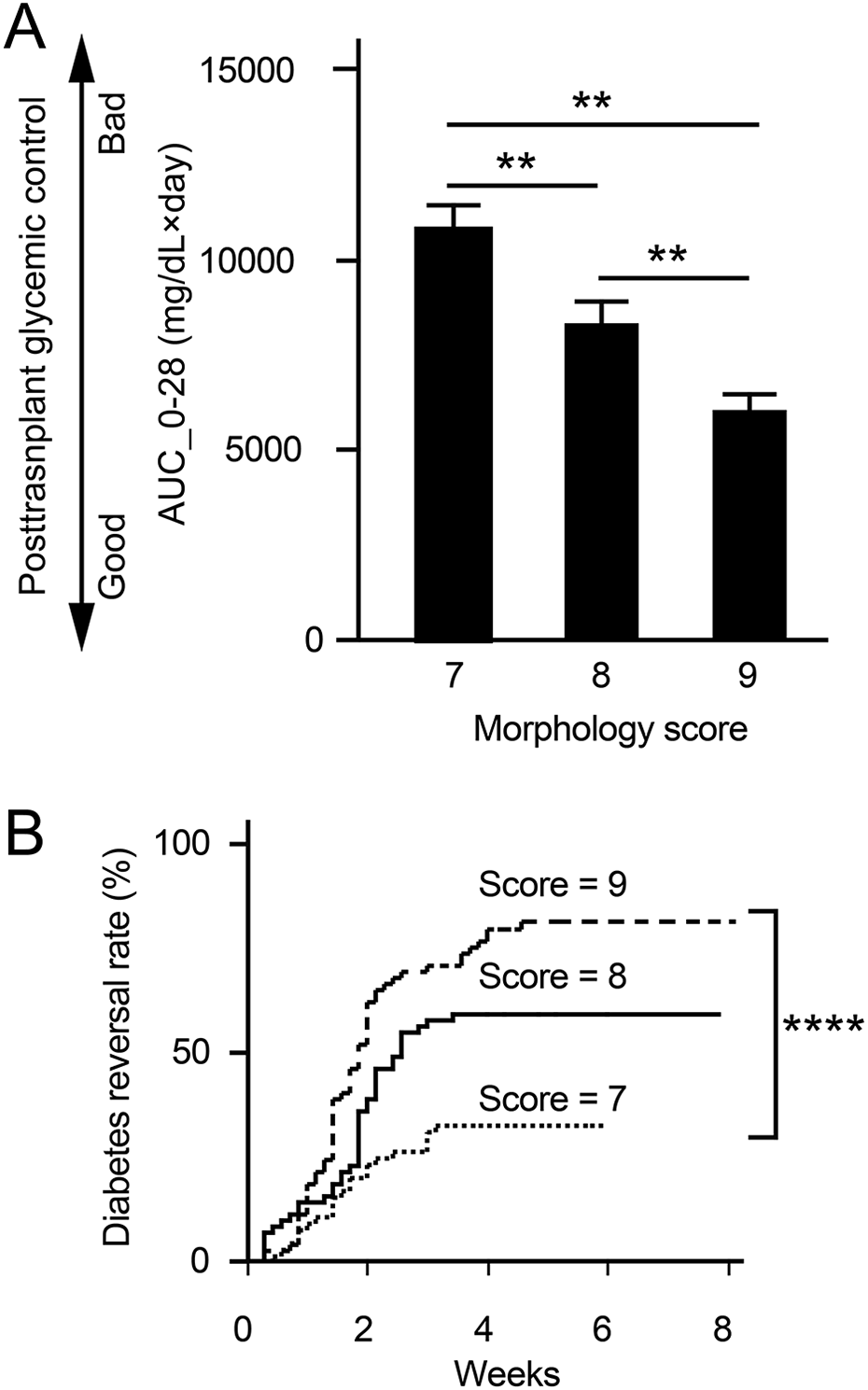
Islet morphological scores positively correlate with islet transplantation outcomes. (A) Correlation of islet morphological scores (7–9 points) to AUC_0-28; n = 62 mice transplanted with islets graded 7 points, n = 70 mice transplanted with islets graded 8 points, and n = 70 mice transplanted with islets graded 9 points. (B) Cumulative curves of diabetes reversal derived from blood glucose levels from diabetic animals transplanted with islets from the indicated scored islet groups. ** P < 0.01, **** P < 0.0001. Student’s t-test and Log-Rank test (Prob>ChiSq) were used to determine significance in Fig. 3A, B, respectively.
Islet Morphological Grade is Comparable to Metabolic Assessment (OCR) as a Predictor of Transplantation Outcome
We compared the potential of islet morphological grade and metabolic assessment (OCR) to predict transplantation outcome. Data from each assay were categorized into two groups: Grade A (score 9–10) or B (score 7–8) for islet morphological grade; <1.24 or >1.24 for OCR-SI. Using a ROC curve, 1.24 was selected as an optimal cutoff for OCR-SI (Supplemental Fig. S2). Mice that received islets graded A, and >1.24 in OCR-SI demonstrated significantly better glycemic control, as defined by lower AUC_0-28 values, when compared to other groups (P < 0.0001 in morphological grade B, Fig. 4A; P < 0.0001 in OCR-SI<1.24, Fig. 4B). Mice that received Grade A islets showed reversal of diabetes in 81.2% of animals, whereas those received Grade B islets showed reversal in 46.6% of animals (Fig. 4C, P < 0.0001). Mice that received islets of OCR_SI >1.24 showed diabetes reversal in 66.0% of animals, whereas those received islets of OCR_SI <1.24 showed reversal in 22.3% (Fig. 4D, p = 0.0001). Both islet assessment methods predicted islet transplantation outcome but vary in their sensitivity and specificity (Table 5).
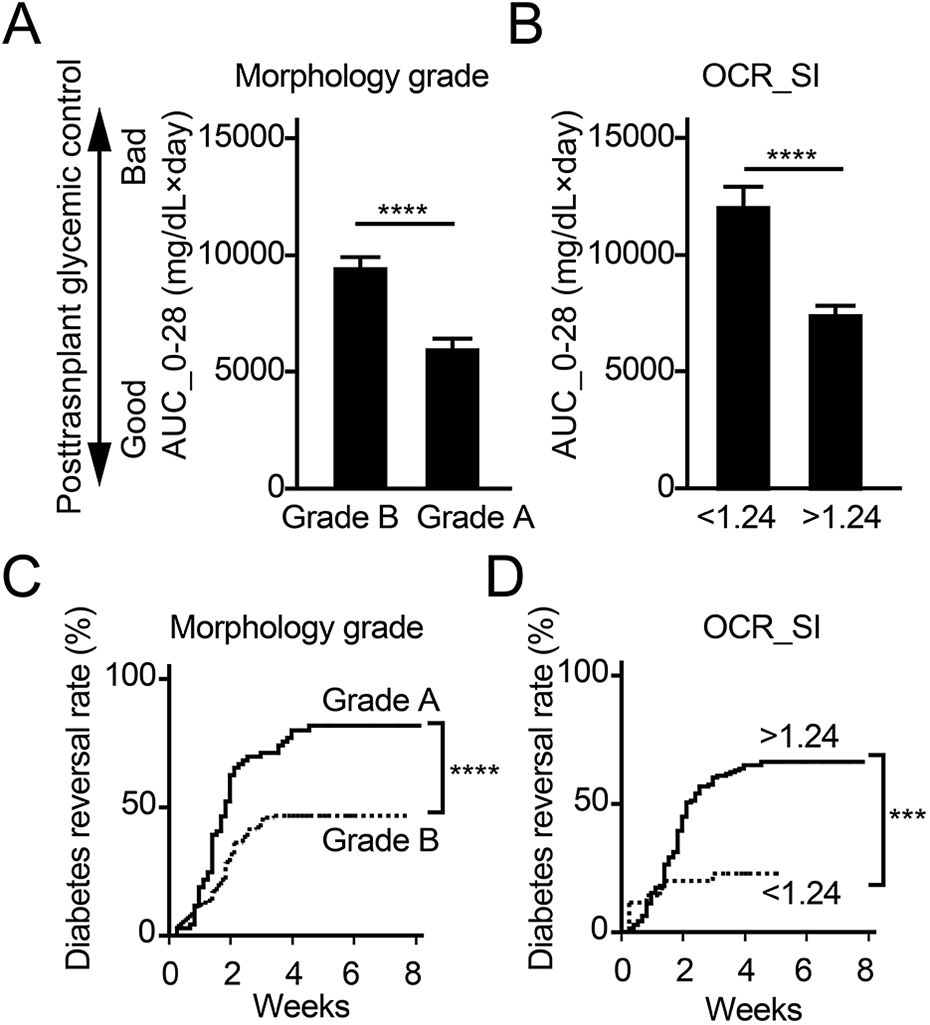
Islet morphological grade is effective at predicating islet transplantation outcome compared to OCR. (A) Correlation of islet morphological grade (Grade A vs. Grade B) to AUC_0-28; n = 70 mice transplanted with Grade A and n = 132 mice transplanted in Grade B islets. (B) Correlation of OCR-SI (<1.24 vs. >1.24) to AUC_0-28; n = 36 mice transplanted with islets having an OCR <1.24 and n = 145 mice transplanted with islets having an OCR >1.24. (C) Cumulative curves of diabetes reversal according to the morphological grade. (D) Cumulative curves of diabetes reversal according to the OCR-SI. *** P < 0.001, **** P < 0.0001. Student-t test and Log-Rank test (Prob>ChiSq) were used to calculate significance in Figures 4A, B, and Figures 4C, D, respectively.
Sensitivity and Specificity of Pre-Transplant Assessments to Predict Islet Transplantation Outcome.

Abbreviation: OCR, oxygen consumption rate.
Individual Parameters in Islet Evaluation Correlate with Transplantation Outcome
Total islet score and grade predicted islet transplant outcomes. However, it was not clear if one or more parameters more closely tracked with outcome. We analyzed transplantation outcome (AUC_0-28) in relation to the score of each parameter (Fig 5). A high score (2 points) in three parameters (border, integrity, and single cells) individually correlated with better post-transplant glycemic control when compared to a low score (1 point) (p = 0.0213, p = 0.0225, and p = 0.0001, respectively). The interaction analysis of the five parameters revealed that each parameter was relatively independent (Supplemental Fig. S3).
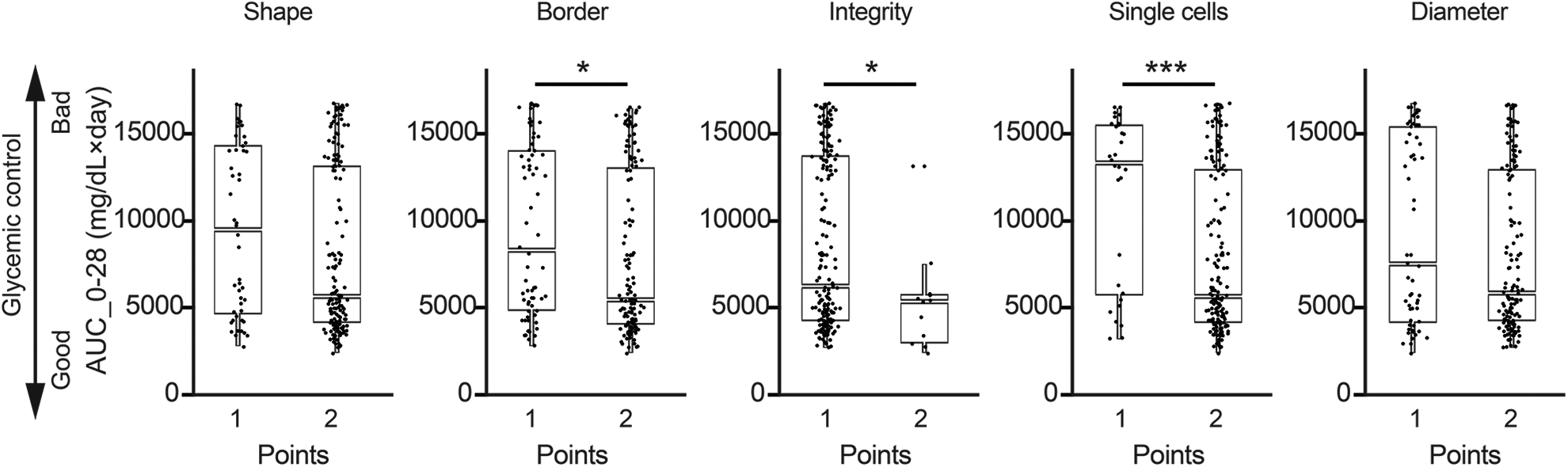
Individual parameters in the islet morphological score positively correlate with transplantation outcome. Post-transplant glycemic control values of mice (AUC_0-28) were plotted according to the individual parameters (shape, border, integrity, single cells, and diameter). Boxplots demonstrate data distribution, interquartile range, and median. The Student’s t-test was used to determine significance. *P < 0.05, ***P < 0.001.
Discussion
In this study, a multiparameter islet assessment based on shape, border, integrity, single cells, and diameter was found to significantly correlate with islet transplant outcomes in immunodeficient diabetic mice. Accordingly, the multiparametric islet assessment showed advantage over other assays including stimulation index in OCR assay and semi-automated viability for assessing islet transplantation outcomes. The multiparametric assessment requires only DTZ staining of islets 31 and should be performed immediately after islet isolation using light microscopy 25 . As the technical requirements for morphologic assessment are modest, this method can be applied widely. Five parameters were used to score the islets (shape, border, integrity, single cells and diameter) with points assigned from 0 to 2. The narrow range of scoring may limit the consideration of subtle differences in islet morphology but has the advantage of being easier to master and implement. It should be pointed out that scoring reflects certain unique aspects derived from our experience. This is exemplified in the score we assigned to islet size. Indeed, other data suggested that islet size might not reflect transplant outcome 32 . Islets were then further categorized into several grades (A, B, C, D, and F) based on the total score for overall islet quality and integrity. Counting of DTZ-stained islets is a gold standard for calculating islet particle number (IPN) and to determine the final islet equivalent (IEQ) 33 . The islet scoring system employed in the present study uses the well-established methods of DTZ staining and observation under light microscopy. And, while being somewhat operator-dependent, morphologic scoring was found superior in certain aspects compared to the stimulation index in OCR assay and semi-automated viability. There are several potential reasons for this. First, all scored parameters showed the same trend in relation to islet transplant outcome, that is the higher score in any parameter positively correlated with better post-transplantation glucose control. Second, as demonstrated in the interaction analysis, each of the five scored parameters were relatively independent of the other. Therefore, adding together the score of each individual parameter acted to enhance the predictive value of the combined score.
Islet transplantation is largely restricted to centers with substantial experience in isolating human islets for clinical and research applications. Characterization of islets is essential and performed immediately after isolation. Consistent with this, in the present analysis, among the 55 islet batches scored, the average scores assigned by the two study investigators were 8.03 ± 0.16 (range: 7–9) versus 7.96 ± 0.16 (range: 7–9). The distribution of the scores by each investigator were also similar. These results indicate that islet assessments are reproducible between individuals. Expanding upon this, the data generated from this multiparametric assessment could aid in developing computer-based automated platforms for morphometric assessment 34 –37 . For instance, shape and border may be digitally evaluated using the shape factor, a numerical factor describing the shape of a particle of two-dimensional images 38,39 . Single cells and islet diameter could be detected by computer-based image analysis to further improve objectivity 19,29 .
Uniformity in islet characterization for clinical transplantation is needed for rationale interpretation of outcomes that then inform therapeutic decisions. Wider utilization of this approach and sharing of data may permit refinements such as development of automated non-biased assessment software tools. In regard to the clinical application, Grade A and B islets are designated as transplant-quality islets especially if the islet yield/volume of fraction 1 are high, islet recovery is more than 30% post-culture, and donor age is less than 50 years old. It is worth noting that Grade B islets have also been used in clinical transplantation and are administered particularly for 2nd or 3rd infusion 4,40,41 . In this study, fraction 1 was used to assess islet quality. However, the multiparameter assay performed for islets from fraction 2 or 3 often found identical to those of fraction 1. It is important to point out that establishing a multiparametric assessment and biomimetic potency tests would assist in the effort to obtain a biological license for IT from the US Food and Drug Administration 41 –44 .
In conclusion, multiparametric assessment of isolated human islets highly predicted transplantation outcomes in diabetic mice. In this regard, multiparametric assessment compared favorably to other more costly and time-consuming techniques of islet evaluation. Application of multiparametric assessment of islets to transplant quality and research grade islets could improve clinical islet transplant outcomes and decrease the heterogeneity of research results.
Supplemental Material
Supplemental Material, sj-jpg-1-cll-10.1177_09636897211052291 - A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice
Supplemental Material, sj-jpg-1-cll-10.1177_09636897211052291 for A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice by Hirotake Komatsu, Meirigeng Qi, Nelson Gonzalez, Mayra Salgado, Leonard Medrano, Jeffrey Rawson, Chris Orr, Keiko Omori, Jeffrey S. Isenberg, Fouad Kandeel, Yoko Mullen and Ismail H. Al-Abdullah in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-2-cll-10.1177_09636897211052291 - A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice
Supplemental Material, sj-jpg-2-cll-10.1177_09636897211052291 for A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice by Hirotake Komatsu, Meirigeng Qi, Nelson Gonzalez, Mayra Salgado, Leonard Medrano, Jeffrey Rawson, Chris Orr, Keiko Omori, Jeffrey S. Isenberg, Fouad Kandeel, Yoko Mullen and Ismail H. Al-Abdullah in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-3-cll-10.1177_09636897211052291 - A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice
Supplemental Material, sj-jpg-3-cll-10.1177_09636897211052291 for A Multiparametric Assessment of Human Islets Predicts Transplant Outcomes in Diabetic Mice by Hirotake Komatsu, Meirigeng Qi, Nelson Gonzalez, Mayra Salgado, Leonard Medrano, Jeffrey Rawson, Chris Orr, Keiko Omori, Jeffrey S. Isenberg, Fouad Kandeel, Yoko Mullen and Ismail H. Al-Abdullah in Cell Transplantation
Footnotes
Acknowledgments
We thank Sung Hee Kil, Ph.D., for critical reading and editing of the manuscript; and Taro Yoshida for figure production.
Authorship
H.K. designed the study. H.K., M.Q., and I.H.A. wrote the manuscript. H.K., M.Q., I.H.A., N.G., M.S., L.M, J.R., C.O., K.O. collected and analyzed data. J.S.I, F.K. and Y.M. provided scientific feedback, and reviewed and edited the manuscript.
Disclosure
All authors certify that there are no conflicts of interest with any financial organization regarding the material discussed in the manuscript.
Ethical Approval
This study was approved by the institutional review board of Beckman Research Institute, City of Hope.
Statement of Human and Animal rights
All of the experimental animal procedures in this study were conducted in accordance with the Institutional Animal Care and Use Committee of Beckman Research Institute, City of Hope.
Statement of Informed Consent
Human islets were isolated from the human pancreata of deceased donors with informed research consent in place.
Data availability statement
The data that support the findings of this study are available to researchers upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) diclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support was provided by the Nora Eccles Treadwell Foundation (Grant Period: July 1, 2012–June 30, 2020, P.I.: Yoko Mullen, M.D., Ph.D.; July 1, 2020–June 30, 2024, P.I.: Hirotake Komatsu, M.D., Ph.D.).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
