Abstract
Hearing is one of our most important means of communication. Disabling hearing loss (DHL) is a long-standing, unmet problem in medicine, and in many elderly people, it leads to social isolation, depression, and even dementia. Traditionally, major efforts to cure DHL have focused on hair cells (HCs). However, the auditory nerve is also important because it transmits electrical signals generated by HCs to the brainstem. Its function is critical for the success of cochlear implants as well as for future therapies for HC regeneration. Over the past two decades, cell transplantation has emerged as a promising therapeutic option for restoring lost auditory nerve function, and two independent studies on animal models show that cell transplantation can lead to functional recovery. In this article, we consider the approaches most likely to achieve success in the clinic. We conclude that the structure and biochemical integrity of the auditory nerve is critical and that it is important to preserve the remaining neural scaffold, and in particular the glial scar, for the functional integration of donor cells. To exploit the natural, autologous cell scaffold and to minimize the deleterious effects of surgery, donor cells can be placed relatively easily on the surface of the nerve endoscopically. In this context, the selection of donor cells is a critical issue. Nevertheless, there is now a very realistic possibility for clinical application of cell transplantation for several different types of hearing loss.
Introduction
Over 450 million people suffer disabling hearing loss (DHL), equivalent to 6.1% of the world’s population (https://www.who.int/deafness/estimates/en/). Hearing loss affects our most important means of communication, and it may lead to social isolation, depression, and even dementia in the elderly 1 .
Traditionally, significant efforts to cure DHL have focused on hair cells (HCs). No less important, however, is the auditory nerve, which contains the sensory neurons that transmit electrical signals generated by HCs to the brainstem 2,3 .
Auditory nerve damage may occur as a result of various types of insult. These include internal causes, such as neuropathies and intracranial mass lesion, and head trauma, which is a representative external cause 3 . Over several decades, cell transplantation has emerged as a promising therapeutic option to rebuild lost auditory nerve function. Numerous studies in vitro and in vivo have explored different combinations of cells and delivery methods, and two successful studies have provided proof of principle that cell transplantation can lead to functional recovery. The challenge now is to focus on how the human auditory system can be approached in the clinic, including the selection of donor cells and how auditory nerve function can be restored with surgically acceptable techniques involving minimal intervention.
Degeneration Pattern of Auditory Neurons Following Insult
Insight into why cell transplantation works comes from the nature of tissue degeneration (Fig. 1). When auditory nerve axons are compromised, for example, in neuropathies, closed head injury, microsurgery (MiS), or radiation exposure in radiotherapy (RT) (see the following section), degeneration proceeds centripetally away from the soma, as in Wallerian or anterograde degeneration (Fig. 1B, ). Cochlear nucleus cells and upper neurons up to the cerebrum subsequently degenerate transneuronally 4 (Fig. 1B, ). At the same time, retrograde axon degeneration proceeds toward the soma of spiral ganglion cells (SGCs) within the cochlea, leading eventually to the death of the neurons and loss of the peripheral processes (Fig. 1B, ). If auditory neurons degenerate, the HCs that they innervate can still survive. Auditory neurons express the tyrosine receptor kinase B (TrkB) and tyrosine kinase receptor C (TrkC) (Fig. 1A) for brain-derived neurotrophic factor (BDNF) and neurotrophin 3 (NT-3), which are produced mainly by HCs 3,5 . Thus, damage to HCs can lead to degeneration of auditory neurons and transneuronal degeneration of the cochlear nucleus cells and upper relay neurons (Fig. 1, , ) 3 . It is well known clinically that degeneration of the HCs is triggered by systemic use of pharmacological agents such as aminoglycoside antibiotics and platinum-based drugs 6 and also exposure to intense noise 7 .
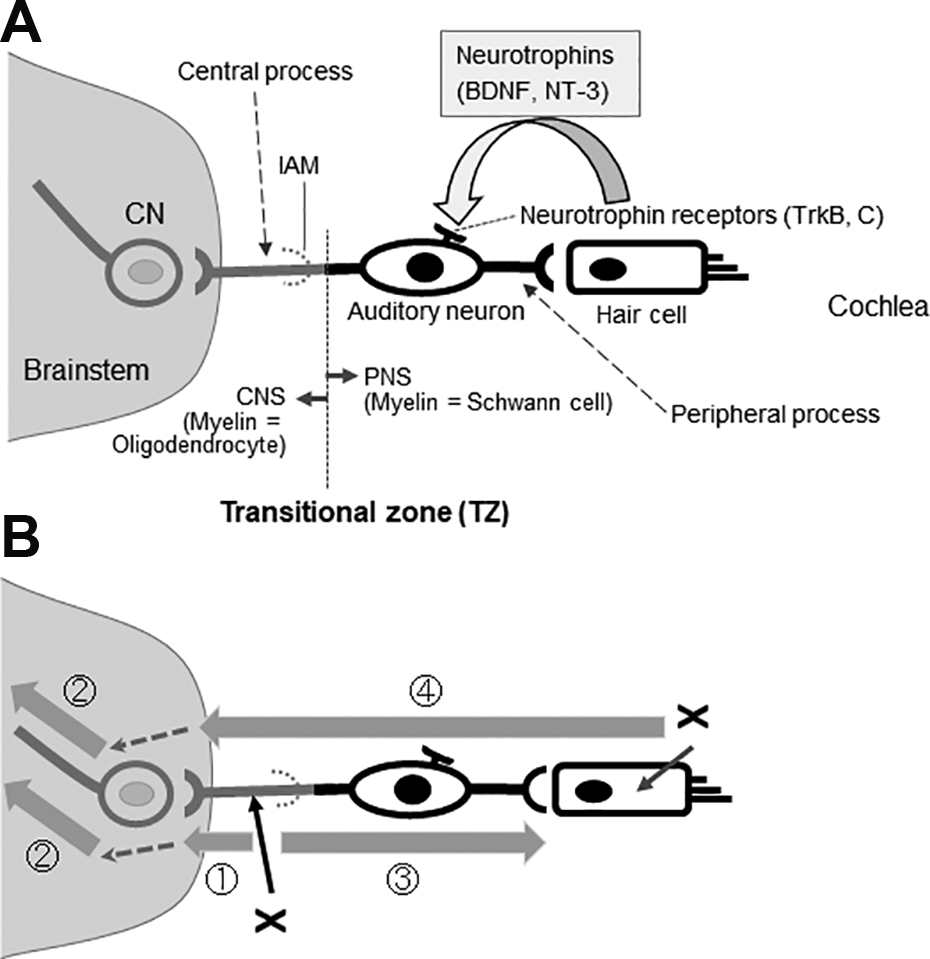
Auditory neurons and their degeneration patterns. (A) The auditory nerve is a bundle of bipolar auditory neurons. The peripheral processes of auditory neurons form synapses with HCs and the central processes with CNs in the brainstem. HCs provide much of the trophic support required for the maintenance and survival of auditory neurons, including BDNF and NT-3. Auditory neurons synthesize the high-affinity tyrosine receptor kinases, TrkB and TrkC. The interface between the PNS and CNS is called the TZ, which is distal to the IAM. Myelin sheaths are formed by oligodendrocytes centrally from the TZ, and the surrounding milieu is astrocytic. Peripheral to the TZ, the myelin sheaths are formed by Schwann cells that are enveloped in endoneurium. The interface is penetrated only by axons. (B) The onset of anterograde (Wallerian) (), trans-neuronal (), and retrograde degeneration () of the auditory nerve depends on the initial site of injury (x). In HC damage, neurodegeneration involves the auditory neuron entirely () and neurodegeneration proceeds to higher-level neurons including the CNs (). Shaded arrows indicate the progression of degeneration, and dotted arrows indicate transneuronal degeneration. BDNF, brain-derived neurotrophic factor; CNS, central nervous system; CNs, cochlear nucleus cells; HC, hair cells; IAM, internal auditory meatus; NT-3, neurotrophin 3; PNS, peripheral nervous system; TZ, transitional zone.
Hearing levels can deteriorate progressively in closed head injury patients 8 . Similarly, in MiS and RT for vestibular schwannoma (VS), the hearing preservation rates measured within a few years of treatment can be misleading because hearing loss that is unrelated to tumor recurrence continues to progress even after 7 to 8 years 9 –14 . Various mechanisms are responsible for such delayed hearing loss, but one contributing factor is likely to be the unusually slow speed of auditory nerve degeneration, which can be protracted for years 15 . There are several reasons for the slow degeneration of the auditory nerve. First, the soma of human SGCs contact each other and can provide mutual trophic support 16 . Second, non-myelinated Schwann cells (SCs) and satellite glial cells surrounding the soma prevent the SGCs from dying even after HCs are damaged 17 . Third, SGCs depend on neurotrophins provided mainly by HCs but supporting cells are also a source of neurotrophins 18 even after the HCs degenerate. Cochlear implants (CIs) stimulate auditory neurons directly and they exploit the protracted course of auditory nerve degeneration 15 . Cell transplantation is more likely to succeed for the same reason because degenerated auditory neurons can be replenished progressively by donor cell-derived neurites that seem to regenerate over several months 2 .
Causes of Auditory Nerve Degeneration and Related Clinical Issues
Neuropathies
The classical description of auditory neuropathy (AN) is that auditory nerve function is impaired but outer HCs in the cochlea are functional 19 . In AN, speech comprehension is compromised although pure tone audiograms are disproportionately well maintained so patients can hear but cannot understand 19 . This type of hearing loss is observed in various diseases including a subset of neuropathic and presbycusis patients 20,21 . Nowadays, the causative sites for AN include not only the auditory nerve and outer HCs but also the inner HCs and inner HCs ribbon synapses (auditory synaptopathy) 19 . Nevertheless, AN due to auditory nerve dysfunction and auditory neuropathic hearing loss is a potential candidate for cell transplantation 2,3,22 . Some patients with genetic disorders have polyneuropathic disorders, such as auditory neuropathic hearing loss and optic neuropathy with bilateral blindness 20 , and their anguish would be alleviated remarkably even if only their hearing was restored. For patients with pathologies in both the auditory nerve and HCs, auditory nerve regeneration would most effectively be coupled with HC regeneration, should that eventually prove to be successful in mammals.
Tumors
VS develops from the vestibular nerve, but the surgical removal of VS inevitably imposes direct mechanical stress to the auditory nerve, potentially leading to the severance of continuity of auditory neurons or to the initiation of auditory nerve degeneration. VS surgery can also have far-reaching effects on the cochlea through the vasculature (the internal auditory artery or labyrinthine artery), leading to cochlear ischemia and reflow phenomena that are inevitably repeated during surgery, eventually leading to HC death. The latter presumption is supported by recordings of distortion product otoacoustic emissions (DPOAEs), which are sounds generated within the cochlea recorded by a microphone fitted into the ear canal 23 . The amplitude of DPOAEs reflects the blood flow to the cochlea 24 , and an intraoperative decrease in DPOAEs indicates cochlear ischemia due to mechanical pressure upon the vasculature 25,26 . Several minutes of cochlear ischemia are sufficient to cause morphological changes of the distal ends of the auditory neurons, and longer periods can cause cessation of internal auditory artery blood flow leading to HC death 27,28 . Postmortem histological examinations of VS patients without surgery reveal structural changes within the cochlea, including degeneration of HCs and the stria vascularis in the outer wall of the scala media 29 .
Radiation
Radiotherapy (RT) for the central nervous system (CNS) and peripheral nervous system (PNS) lesions incur multiple pathological processes, including vascular endothelial damage, neuroinflammation, genetic/epigenetic alterations, apoptosis/necrosis of neurons and glial cells, reactive gliosis, and demyelination and deterioration of stem cell and progenitor cell proliferation 30 . It is extremely difficult to avoid radiation injury to the auditory nerve in RT for VS 31 . To make matters worse, not only the cochleovestibular nerve but also the facial nerve and other vital structures, such as HCs and the stria vascularis 32 , are packed in a confined space of the cochlea (Fig. 2). The horizontal diameter of the internal auditory canal is only about 4.5 mm 33 . Within this narrow canal, the cochleovestibular and facial nerves are compressed by the tumor and take a tortuous course. Reports revealed that radiation doses in the cochlea and cochlear nucleus during RT are correlated with patients’ hearing outcome 31,34 , implying radiation injury to auditory neurons is responsible for hearing deterioration after RT in addition to that to HCs and the stria vascularis, both vital to hearing 32 . Patients with small VS in which auditory neurons degenerate but HCs are still functional 35 are an ideal candidate for auditory nerve replacement 3 , and this is the case in a subset of presbycusis or auditory neuropathic patients as mentioned above 20,21 .
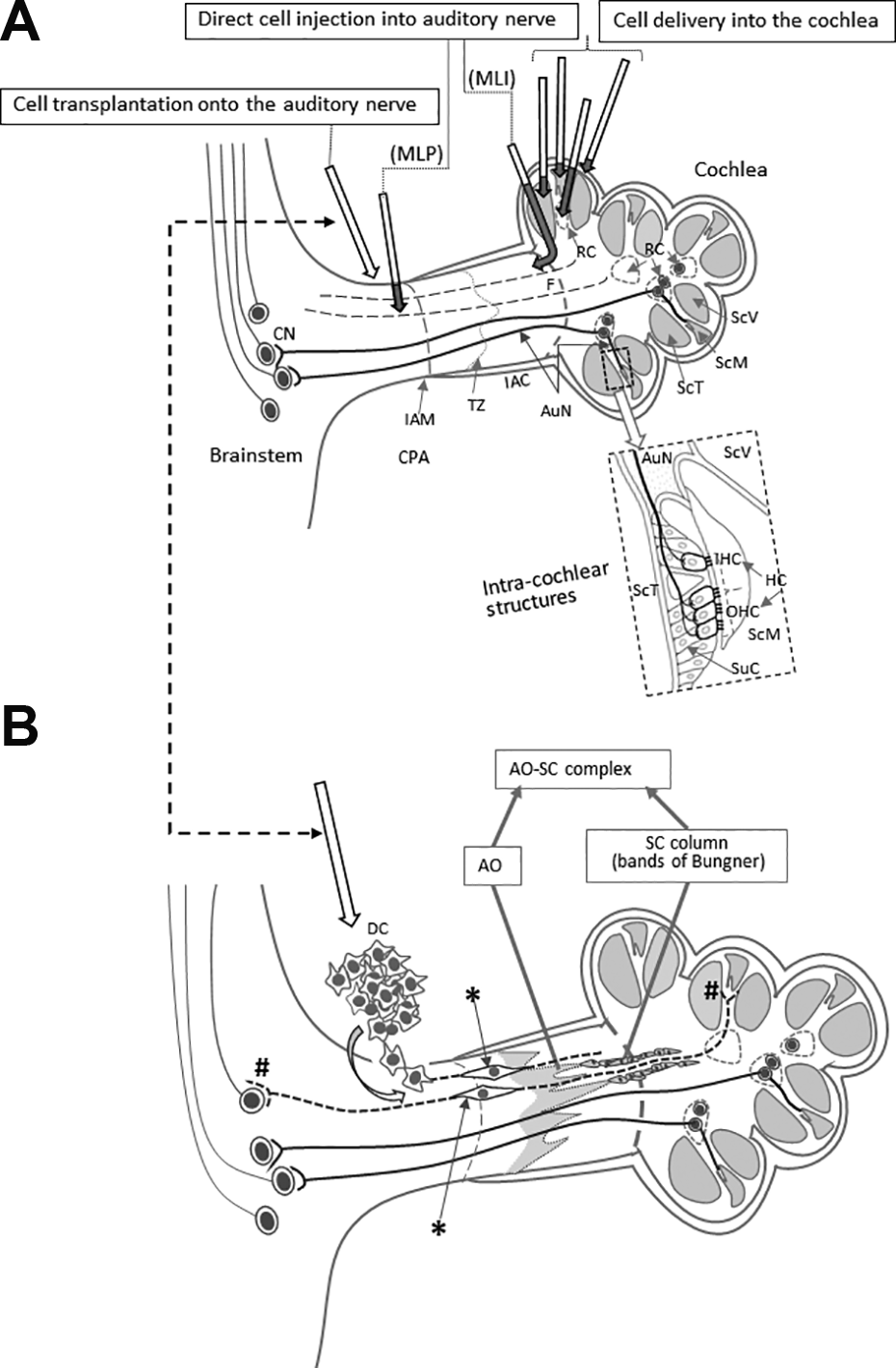
Reported cell delivery methods to restore AuN function. (A) Reported cell delivery methods in Table 1 are shown with arrows in the upper panel. Dark shaded parts of each arrow indicate intracochlear or intraneural portions of each route. Arrows show each route conceptually and do not trace each anatomical route precisely. The dotted rectangle is enlarged to illustrate intracochlear structures in detail. (B) Surface transplantation of DCs on degenerated AuN. DCs transplanted onto the surface of degenerated AuN autonomously enter the nerve, differentiate (*) and form functional synapses with HCs and CNs (#). In degenerated AuN, the AO and SC columns form a continuous, “naturally occurring autologous cell bridge”, the AO–SC complex (a part is shown here), which acts as an anatomical scaffold for DC migration to connect between the PNS and the CNS (see the text). Note regenerating axons run parallel with the AO–SC complex. Studies using systemic delivery of donor cells are not shown here. AO, astrocyte outgrowth; AuN, auditory nerve; CNS, central nervous system; CN, cochlear nucleus cell; CPA, cerebellopontine angle; DC, donor cells; HC, hair cell; IAC, internal auditory canal; IAM, internal auditory meatus; IHC, inner hair cell; OHC, outer hair cell; MLI, membranous labyrinth injured; MLP, membranous labyrinth preserved; PNS, peripheral nervous system; RC, Rosenthal’s canal; ScM, the scala media; SC, Schwann cell; ScT, the scala tympani; ScV, the scala vestibuli; SuC, supporting cell; TZ, the transitional zone.
Head Injury
In patients with a closed head injury even without temporal bone fractures, damage to auditory neurons is observed primarily and/or secondarily following HC damage 36 –42 . Clinically, cases in which the auditory nerve is damaged without damage to HCs are most suitable for cell transplantation because the HCs can provide trophic support, as mentioned above (Fig. 1). The auditory nerve is particularly vulnerable to external force in the regions of the fundus of the internal auditory canal and the transitional zone (TZ) 36 –41 . When the medial displacement of the brainstem is greater than that of the cochlea in the temporal bone, the resultant force on the auditory nerve may avulse the auditory neurons from the fundus of the internal auditory canal 36 –41 . The TZ is the interface between the CNS and PNS 43 , and it can be highlighted by immunostaining for glial fibrillary acidic protein (GFAP), which is expressed by astrocytes only in the CNS 2 and it lies within the internal auditory canal 44 (Fig. 3). The TZ is vulnerable to external stretch and shear forces probably because there is an abrupt anatomical change at this point; central to the TZ, myelin sheaths are formed by oligodendrocytes, and the supporting tissue is astrocytic, whilst peripheral to the TZ, the sheaths are SCs enveloped in endoneurium, although the axons are continuous 3,39 . HC damage in closed head injury may occur due to a breach of the sealing of the membranous labyrinth and/or impairment of blood supply to the cochlea 42 .
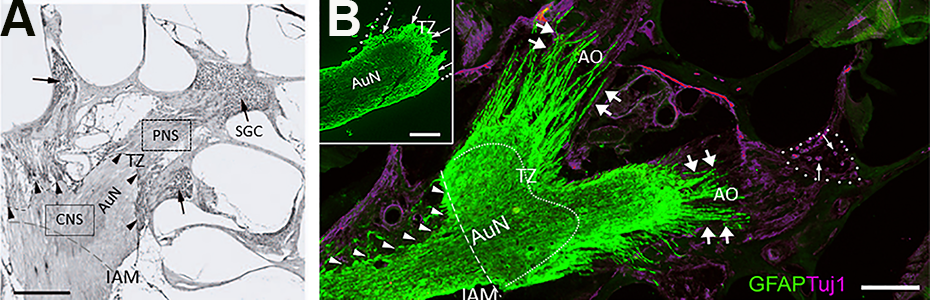
Transitional zone and the astrocyte outgrowth following auditory nerve mechanical compression. (A) Normal TZ. The CNS portion of the AuN extends peripherally, with a dome-like shape (arrowheads with dotted line). Rosenthal’s canals are densely packed with auditory SGC (arrows). Rat, Hematoxylin and Eosin stain, Scale bar, 200 µm. Cited from Sekiya et al. (2007) with publisher’s permission. (B) Gliotic AuN after compression. A glial scar is induced following mechanical compression applied to the CNS portion of the auditory nerve in the cerebellopontine angle (arrowheads). Marked AO is indicated by double arrows. Most auditory SGCs degenerate following sustained compression (single arrows in dotted circle, Rosenthal’s canal). With GFAP antibody, an astrocyte marker, the glial scar is also stained because it contains many reactive astrocytes. An antibody Tuj1 against beta-tubulin stains neurons and neurites, including SGCs. The curved dotted line indicates the default position of the TZ. (Inset) Normal rat AuN. The TZ (arrows) is clearly observed as a peripherally convex, dome-like shape. The PNS portion of the nerve (outlined by dotted line) is GFAP-negative because astrocytes exist only in the CNS. Scale bars, 200 μm. AO, astrocyte outgrowth; AuN, auditory nerve; CNS, central nervous system; GFAP, glial fibrillary acidic protein; IAM, internal auditory meatus; PNS, peripheral nervous system; SGCs, spiral ganglion cells; TZ, transitional zone.
Cochlear Implants (CIs)
Cell transplantation could potentially enhance the performance and candidature for CI in patients, who generally have too few remaining functional auditory neurons 45,46 . In fact, the minimum number of functional auditory neurons needed for the successful performance of a CI is astonishingly few and estimated to be 5% to 10% of the normal number 15 . CIs are beneficial to neurofibromatosis type 2 (NF2) patients with bilateral VS 47 , but a significant number of patients experience a decline in performance as the VS grows 48 . Hence, replenishing auditory neurons would potentially benefit NF2 patients.
Surgical Options for Cell Delivery
There is extensive literature on cell transplantation to the auditory system, but in this context, the relevant studies are those done in vivo on deafened animals and on deaf humans (Table 1). To establish proof of principle, two main conditions must be met in the analysis of cell integration and functional recovery 49 . First, there must be an electrophysiological analysis of the restoration of nerve function with an objective method such as the auditory brainstem response (ABR). Second, to link recovery to the transplanted cells, it is important to provide morphological evidence for synaptic connections, not only with HCs in the cochlea but also with neurons of the cochlear nuclei within the brainstem. In other parts of the nervous system, functional improvements have been recorded without morphological integration of the donor cells 50 –52 by indirect mechanisms, including trophic effects, immunomodulation, and other bystander effects 53 –55 .
In vivo studies to restore auditory nerve function.
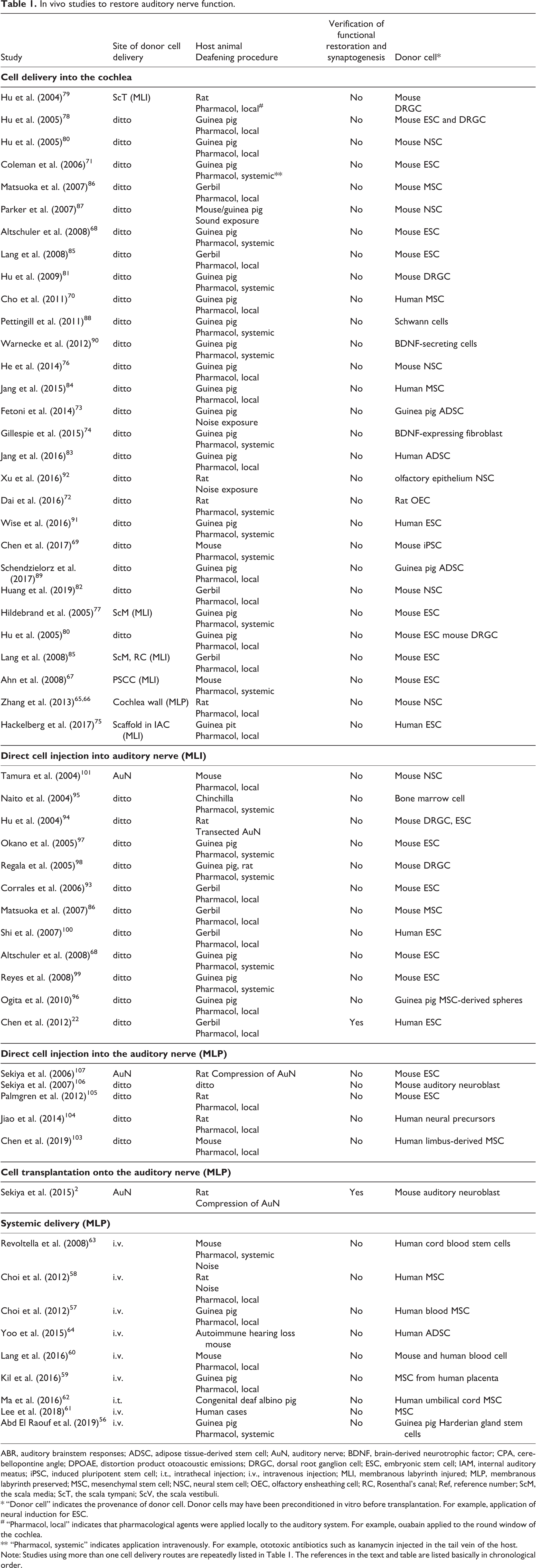
ABR, auditory brainstem responses; ADSC, adipose tissue-derived stem cell; AuN, auditory nerve; BDNF, brain-derived neurotrophic factor; CPA, cerebellopontine angle; DPOAE, distortion product otoacoustic emissions; DRGC, dorsal root ganglion cell; ESC, embryonic stem cell; IAM, internal auditory meatus; iPSC, induced pluripotent stem cell; i.t., intrathecal injection; i.v., intravenous injection; MLI, membranous labyrinth injured; MLP, membranous labyrinth preserved; MSC, mesenchymal stem cell; NSC, neural stem cell; OEC, olfactory ensheathing cell; RC, Rosenthal’s canal; Ref, reference number; ScM, the scala media; ScT, the scala tympani; ScV, the scala vestibuli.
* “Donor cell” indicates the provenance of donor cell. Donor cells may have been preconditioned in vitro before transplantation. For example, application of neural induction for ESC.
# “Pharmacol, local” indicates that pharmacological agents were applied locally to the auditory system. For example, ouabain applied to the round window of the cochlea.
** “Pharmacol, systemic” indicates application intravenously. For example, ototoxic antibiotics such as kanamycin injected in the tail vein of the host.
Note: Studies using more than one cell delivery routes are repeatedly listed in Table 1. The references in the text and table are listed basically in chronological order.
Equally important from the experimental aspect are clinical relevance, which is reflected in the animal model used to replicate human clinical pathology, and clinical feasibility, which relates to whether or not the surgical techniques can be used in the clinic. In the following sections, we consider a number of in vivo studies in these terms. We focus on local delivery of cells to parts of the inner ear because trials with systemic cell delivery have not led to successful migration of donor cells to the auditory system 56 –64 (Table 1).
Cell Delivery into the Cochlea with Injury to the Membranous Labyrinth
The soma of spiral ganglion neurons (SGNs) are located within the cochlea, and it is worth knowing whether or not cells delivered into the cochlear fluid spaces or cochlear wall are able to find their way into Rosenthal’s canal in which the SGN soma are housed (Fig. 2A). This does not seem to be the case, and none of the relevant studies have led to functional recovery (Table 1). With the exception of two studies 65,66 , the membrane that seals intracochlear fluid-containing spaces (the membranous labyrinth = the scala tympani, scala vestibuli, the scala media, and posterior semicircular canal) was breached and/or trespassed (membranous labyrinth injured [MLI]) 67 –92 (Table 1). Importantly, invasion into the membranous labyrinth is clinically unacceptable because it leads to hearing loss 3 . Furthermore, the cochlea in small experimental animals is easily accessible as it is conspicuous within the hollow dome-like bulla, but it is not as accessible in humans as it is deeply buried in the temporal bone. Thus, this method is not suitable for human patients.
Direct Cell Injection Into Auditory Nerve With Injury to the Membranous Labyrinth
For targeting donor cells to the auditory nerve, this method seems to be more dependable 22,68,86,93 –101 . Unlike injection into the cochlear fluids, the cells are located into the appropriate neural tract with morphological continuity with the relevant target cells. Nevertheless, the membranous labyrinth is injured as with direct injection into the cochlea. Moreover, intraneural injection with a syringe needle can damage the morbid, fragile auditory nerve and trigger an inflammatory reaction along with reactive gliosis around the needle and transplant 102 (see the following sections for further discussions). This method has proved successful in one study 22 and offers important proof of principle for clinical translation, especially from the viewpoint of donor cell selection (Table 1; Fig. 2A). However, it remains possible that leakage of the donor cells outside the cochlea might have played a predominant role (see below for the details) in addition to indirect bystander effects that could account for the observed improvement of the ABRs.
Direct Cell Injection Into or Onto Auditory Nerve With Preservation of the Membranous Labyrinth
Even without damaging cochlear structures, cell injection into the cerebellopontine angle portion of the auditory nerve trunk through a hole posterior to the mastoid process (the retromastoid region) has not restored auditory nerve function 103 –107 .
Surprisingly, functional restoration was observed if donor cells were simply placed onto the surface of the auditory nerve via the retromastoid route, thus preserving the integrity of both the nerve and the membranous labyrinth (membranous labyrinth preserved [MLP]) 2 (Fig. 2A, B) (Table 1). This “surface transplantation” method can be regarded as a more promising option for cell transplantation, and it is, thus, considered in more detail in the following sections.
What is the Nature of Nerve injury and Degeneration and How Could Cell Transplantation Work for Specific Clinical Conditions?
The success of in vivo experiments with animal models is encouraging, but it is important to understand the biology that underlies the pathology of nerve degeneration and the subsequent structural and biochemical environment that underlies the successful integration of transplanted cells. This not only informs the optimal technique for cell delivery but also the selection and possibly the conditioning of donor cells.
Structural and Biochemical Cues for Cell Transplantation
Protective Addition
In principle, regenerative medicine should add new functional elements without causing further damage, following the principle of “protective addition”. This principle is most effectively met for the auditory nerve by placing donor cells on the tissue surface 2 . As discussed above, all other delivery techniques involve significant tissue damage.
The Scaffold
The scaffold is an indispensable element for the formation of the nervous system. For example, radial glia plays a crucial role as the scaffold for cell migration from the ventricular zone toward the brain surface 108,109 . Hence, various artificial scaffolds such as collagen-rich acellular matrices and matrices such as hydrogel with in vitro expanded donor cells attached have been intensively investigated in many neurodegenerative disorders, including spinal cord injury (SCI) with efforts to overcome various obstacles including provocation of host immune responses 110 –114 . Currently, another practical issue to be solved aiming at clinical translation is the surgical maneuverability of artificial materials in the delicate and confined space of the CNS.
The Scaffold Within: A Natural Autologous cell Scaffold
One of two successful studies was serendipitous but demonstrated that an autologous cell scaffold had been spontaneously formed in collaboration with SCs during the progression of auditory nerve degeneration. This naturally occurring autologous cell scaffold plays key roles in cell integration as described below. A number of donor cells incidentally spilled onto the nerve surface from a hole through which a syringe needle had been inserted for traditional intra-neural injection of donor cells. These “leaked” donor cells autonomously entered the nerve, gradually transformed into the bipolar shape characteristic of auditory SGCs in the nerve, and finally formed synapses with target HCs and cochlear nucleus cells 2 (Fig. 2B and 4). Intriguingly, donor cell migration and axon elongation apparently recapitulated processes observed during development. These processes include glia-guided migration 115 and migration within GFAP-positive astrocytic, tube-like structures in the rostral migratory stream 116,117 . Even residual neurons appeared to be used as a migration guide 118 (Fig. 4D).
Natural Autologous Cell Scaffold—the Astrocyte Outgrowth and SCs Form a Bridge Between the CNS and PNS
When neurons in the CNS die, astrocytes react to form the glial scar (the astrocyte scar), irrespective of the cause, which may be ischemia, mechanical trauma, irradiation, or genetic disorder 119 –122 . In mouse SCI, reactive astrocytes of the glial scar form characteristic elongated and overlapping processes at the periphery of the lesion core about a week after the insult 123 . In a rat stroke model, reactive astrocyte processes were apparently longer than in sham rats in the penumbra even 30 days after ischemic and hemorrhagic stroke 124 . After a stab lesion in the cerebral cortex of mice, one subset of astrocytes directed their processes toward the lesion 125 . After injection of iron into mice, reactive astrocytes around the lesion core extended long and overlapped processes 126 .
Similarly, at the cranial and peripheral nerve roots, such elongated processes of reactive astrocytes are observed as a conspicuous tongue-like protrusion toward the periphery, the astrocyte outgrowth (the AO) (Fig. 3). One clinical study demonstrated that auditory nerve specimens taken during VS surgery were gliotic, indicating that reactive astrocytes had invaded the auditory nerve 127 . Other than damaged auditory nerve, the AO has been reported in a plethora of diseases in which motor and sensory neurons die, including amyotrophic lateral sclerosis 128 –133 . Electron microscopy shows that the AO comprises processes of reactive astrocytes of the glial scar, which have been known as “glial bundles”, extending from the spinal cord/brainstem 128,130,133 . It should be noted that the polarity of the AO plays pivotal roles in cell migration and axon elongation 134 .
Normally, astrocytes in the CNS and SCs in the PNS are apart and mutually exclusive but their mutual repulsion decreases following motor and sensory neuron death in the brainstem/spinal cord 135 . As a result, the distal tip of the AO extensively apposes with SCs or is directly wrapped by SC cytoplasm within a common basal lamina 133,135 . Distally, SCs form structures called SC columns or bands of Bungner that can guide regenerating axons back to their targets 136 . Thus, a continuous structure, the AO–SC complex, forms autonomously and can act as an anatomical bridging scaffold connecting the CNS and the PNS 137 (Fig. 2B). In fact, in one study on the auditory nerve, the AO–SC complex appeared to be the only continuous scaffold between the PNS and CNS 2 .
Furthermore, upon injury, both astrocytes and SCs become rich sources of pro-regenerative molecules, including laminin, N-cadherin neural cell adhesion molecule, nerve growth factor, BDNF, NT-3, and fibroblast growth factor, glial cell line-derived neurotrophic factor, artemin, and vascular endothelial growth factor 136,138,139 .
Intraneural Injection and Surface Transplantation
It is difficult to compare the different cell transplantation experiments in the auditory system because the donor cells, surgical techniques, and animal models are so varied. However, when intraneural injection and surface transplantation were compared under the same parameters 2 , surface transplantation was clearly more successful. There was no ABR improvement with intraneural transplantation, and the transmission of electrical activity failed to pass the transplantation site 140 (Fig. 4A). Morphological examination revealed a failure of cell migration with cell debris mainly at the site of cell transplantation with a few cells stuck in the midst of the gliotic auditory nerve (Fig. 4C,1). Another finding was cavity formation in the nerve, apparently due to infusion pressure during injection and the large volume of the infused cell mass that might also have damaged residual host neurons and vascular networks (Fig. 4C, 2). In contrast, the animals in which cells were delivered by surface transplantation demonstrated statistically significant improvement of the ABRs measured 3 months after cell transplantation (Fig. 4B). Morphologically, various modes of cell migration were observed as mentioned above (Fig. 4D, 1–4), and synaptic connections with HCs and the cochlear nucleus cells were morphologically confirmed (refer to ref. 2 for the original images).
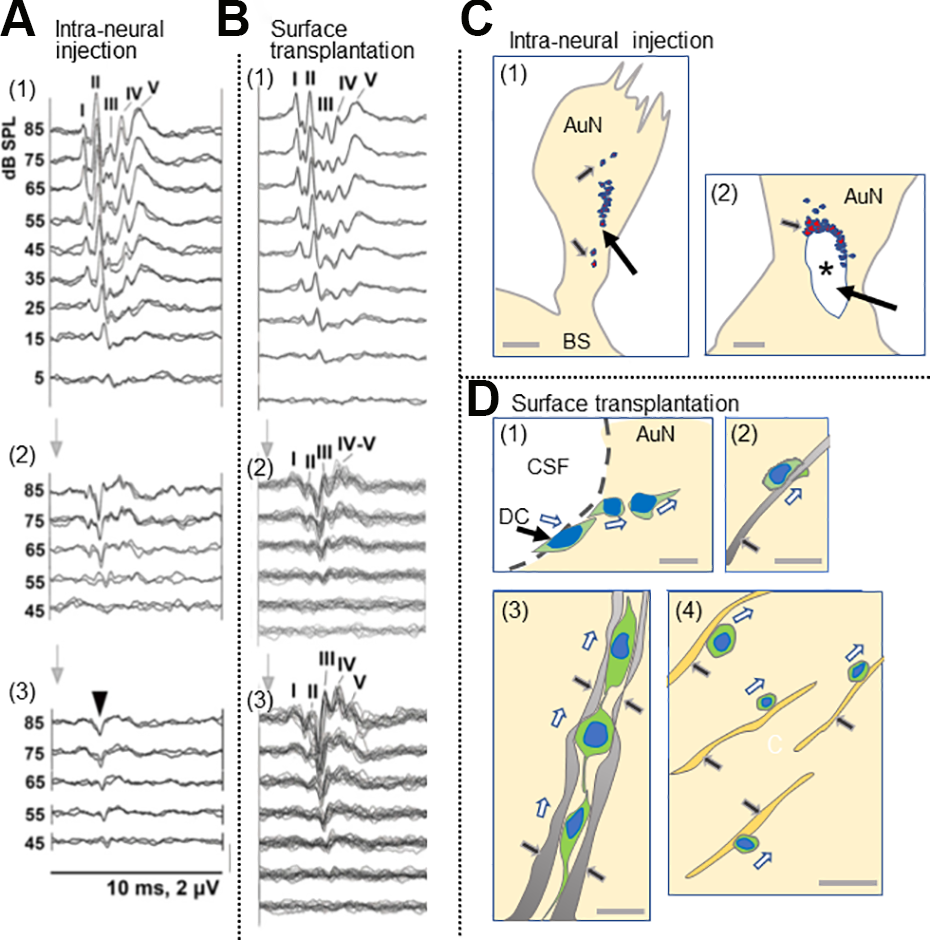
Comparison between intraneural and surface transplantation of cells. (A) Intraneural transplantation of DCs. ABRs before compression (1), 5 weeks after compression before cell transplantation (2), and 3 months after (3). Arrowhead in panel 3, monophasic positive potential indicating electrical failure of nerve impulse transmission. I–V, ABR wave I–V. (B) Surface transplantation of donor cells. ABRs before compression (1), 5 weeks after compression (2), and 3 months after surface transplantation (3). Note a significant improvement of ABRs 3 months after surface transplantation (see Sekiya et al., 2015 for more details). (C) Schematic drawing of fate of intraneurally injected cells. (1) Cell debris mainly in the site of cell transplantation (large arrow), and a few cells are seemingly stuck in the gliotic auditory nerve tissue (small arrows). (2) Large arrow indicates cavity formation (asterisk) in the nerve due to infusion pressure during injection and the infused cell mass. Small arrow indicates cell debris around the cavity. (see Sekiya et al., 2015 for original images). Scale bars: (1), 200 μm; (2) 50 μm. (D) Schematic drawing of various modes of cell migration of donor cells transplanted on the surface of the auditory nerve (see Sekiya et al., 2015 for original images). (1) The DCs autonomously enter the AuN in a chain formation (hollow arrows). CSF, cerebrospinal fluid in the cerebellopontine angle subarachnoid space. (2) Within a gliotic auditory nerve, a transplanted cell is intimately associated with a GFAP+ process (black arrow) derived from the glial scar and migrated (hollow arrow). (3) three migrating donor cells (hollow arrows) form chains within GFAP+ sheaths (2 pairs of black arrows). (4) Migrating transplanted cells (hollow arrows) associated with neurons (black arrow), possibly for guidance. Scale bars: (1, 2, 4), 20 μm; (3) 10 μm. Cited from Sekiya et al. (2015) with publisher’s permission. ABR, auditory evoked brainstem responses; AuN, auditory nerve; BS, brainstem; CSF, cerebrospinal fluid; DCs, donor cells; GFAP, glial fibrillary acidic protein.
The Glial Scar, is it Friend or foe?
Emerging evidence challenges the traditional belief that the glial scar is a physical and molecular barrier to neural regeneration 141 –143 . An in vivo experimental study on SCI showed that regenerating axons skirted around the surface of the glial scar 144 , indicating that they can negotiate its surface and benefit from the structural and chemical cues that it contains. In unilateral cerebral stroke of the motor cortex in mouse, axons of the contralesional corticospinal tract normally sprout into the denervated spinal cord and contribute to motor functional recovery. In a double knockout of GFAP and vimentin (the principal genes responsible for glial scar formation), corticospinal axons only rarely crossed the midline and the reduced astrocytic reactivity led to impaired neurological recovery 142 . Another study showed that scar-forming reactive astrocytes do not only have a protective function but also promote axonal regrowth after SCI. In two different transgenic mouse models to either prevent or inhibit glial scar formation, the study showed that there is a failure in axonal regrowth following removal of reactive astrocytes in both acute and chronic glial scars 141 . Reactive astrocytes in cerebral infarct play a crucial source of a pro-regenerative molecule, the stromal cell-derived factor 1 (SDF-1) 145 . Blocking SDF-1 action with a neutralizing antibody against a receptor for SDF-1, CXC chemokine receptor 4 (CXCR4), strongly attenuated progenitor migration 146 , indicating that SDF-1/CXCR4 promotes migration of stem/progenitor cells toward the lesion 147 .
Pro- and Anti-Regenerative Astrocytes
Astrocytes are not homogenous and are composed of at least five distinct subpopulations, although it is not clear how each subpopulation responds to different insults in different locations 148 –150 . Astrocytes not only conform to different environmental niches but also show different transcriptional changes induced by different types of injuries 149,151 . In a non-penetrating lateral fluid percussion brain injury model in adult rats, the morphology of reactive astrocytes is regionally distinct; those in the injured cortex, subcortical white matter tracts, and CA3 region of the hippocampus show a distinct morphology with an enlarged cell body and long intertwined processes, but those in the thalamic nuclei have thicker shorter processes 152 . Following experimental occlusion of the middle cerebral artery, reactive “A2” astrocytes are likely to be protective as they lead to increased expression of neurotrophic factors and cytokines, transferring mitochondria to injured neurons 143,148,153 . In contrast, neuroinflammation with systemic endotoxin lipopolysaccharide injection induces neurotoxic “A1” astrocytes 143,148,153 . A recent study reported that such molecular and functional diversity of astrocytes in the healthy adult brain depends on cues from neurons through neuron-derived sonic hedgehog (Shh) 154 . This is also another example of glia–neuron interaction (see above).
Thus, it is more likely that there are pro- and anti-regenerative reactive astrocytes, and further research is required to identify those subsets of reactive astrocytes that can aid and contribute to axon elongation efficiently for auditory nerve regeneration.
What is the Ideal Animal Experimental Model?
Studies of the auditory nerve require animal models in which the auditory nerve is selectively, quantifiably, and reproducibly damaged without confounding factors such as concomitant HC damage.
In pharmacological models, to induce auditory nerve degeneration, ouabain is most commonly used 22,57,59,60,65,66,70,75,76,82,85,86,93,96,100,103 . However, with this approach, SGNs are hard to damage reproducibly to avoiding “sudden and all-or-none type cell death”. It is technically difficult to titrate the dose, so ouabain treatments destroy nearly all SGNs in most of the studies 60,85,86,93,96,155 –158 . This makes it hard to assess any further damage that may occur through surgical intervention.
Instead, clinically relevant animal experimental models of neurodegenerative disorders, including hearing loss, should ideally involve a reproducible, “intermediate degree” of stable injury to reflect the gradual progression of tissue degeneration and a suitable opportunity to systematically test potential therapies. In fact, this critical issue has long been discussed when creating animal models of SCI 159 .
Ouabain is usually applied to the round window in the middle ear. It enters the cochlea across the round window membrane and is diluted in the perilymph of the scala tympani before reaching the SGN through Schuknecht’s canaliculae perforantes 3,160 . Pharmacological agents, including ouabain, that are applied even locally to the cochlea generally diffuse throughout the cochlear fluid space in an uncontrolled manner and tend to affect not only auditory neurons but also HCs 158,161 . Moreover, the effect of ouabain is different between species; ouabain selectively destroys SGNs in gerbils and mice, whereas in guinea pigs, it preferentially damages HCs 158 . In rats, if high doses applied to the round window are not sufficiently diluted, then both HCs and SGN can be damaged 158 .
Ouabain is a potent inhibitor of the ubiquitous Na+-K+ pump 162 , which maintains a low Na+ and high K+ concentration within most cells to ensure their excitability and to provide the driving force for the transport of glucose, amino acids, and other nutrients into the cell 162,163 . Thus, a caveat with ouabain is that it may affect not only neurons but also cells in the surrounding epithelial, connective, and muscle tissues. This also applies in systemic administration of pharmacological agents 164 . Thus, the majority of animal models are not ideal for clinical translation.
In contrast, if mechanical compression is applied to the CNS portion of the auditory nerve, it can produce selective, “intermediate” degree of degeneration of auditory neurons with HCs preserved 2,165,166 (Fig. 1B, , 4A, B). This leads to transneuronal death of CNS cells (cochlear nucleus cells) and formation of a protruded bundle of reactive astrocytes of the glial scar (the AO), which plays a crucial role with distal Schwann cell columns for auditory nerve regeneration as elucidated above (Figs. 2B, 3B, 4). Unique to this model, the transneuronal degeneration of cochlear nucleus cells (Fig. 1B, ) can be quantitatively analyzed 165,167,168 . Mechanical compression is thus likely to be the most realistic model for the clinical conditions that lead to auditory nerve degeneration.
Donor Cells for Auditory Nerve Regeneration
Cell Source
Selection and preparation of donor cells are not the focus of this review, but they are critical issues because the cells must be competent to respond to regenerative cues within the damaged tissue.
As depicted in Table 1, embryonic stem cells (ESC) and neural stem cells (NSC) were most frequently used as xenografts or allografts after preconditioning in vitro with diverse factors such as bFGF, BMP4, and the bHLH transcription factor neurogenin 2 68,80,93,100,169 . These approaches carry a greater risk of immune rejection compared with autologous transplantation 170 . Even using autologous-induced pluripotent stem cells (iPSCs) as donor cells, immune rejection can be an issue 170 –172 , despite major histocompatibility complex matching 173 . Furthermore, the phenotypes of individual iPSCs are not entirely predictable, and preconditioning can be complex, time-consuming, and expensive 174 –177 .
Human cells, particularly autologous human cells, are the most likely candidates for clinical translation and those derived from mesenchymal stem cells and adipose tissue-derived stem cells are being studied intensively as donor cells in human disease 178 –184 . Tissue-specific autologous stem cells are naturally strong candidates because they are more closely adapted to the host environment 185,186 . The human inner ear contains endogenous adult stem cells, as has been shown in other organs 187,188,189 , although their potential at the clinical level is not yet clear.
Bipolarity, a Key Requisite as Donor Cells for Auditory Nerve Regeneration
Auditory neurons are bipolar, and donor cells must connect both peripherally with HCs and centrally with cochlear nucleus cells. Table 1 shows that functional recovery of the auditory nerve has been achieved only in two studies, one with human ESCs 22 and the other with a mouse cell line 2 . In both cases, the donor cells adopted a bipolar phenotype 2,22 . The ESCs were conditioned as otic progenitors by simulating the initial, developmental specification of the otic placode with sequential application of selected factors, including NT-3, BDNF, bFGF, and Shh 22 . The mouse cells were from a conditionally immortal mouse otic neuroblast cell line, US/VOT-N33, derived from a mouse otocyst (inner ear anlage) 190 . They show that ontogenetic-stage/region-restricted precursors can be successfully integrated into the host tissue, which has also been shown in a study of retinal regeneration 191 .
Ultimately, the selection of appropriate donor cells must be made in the context of the animal model most closely allied to the clinical application. It cannot be assumed that a given cell type would be equally effective with both intraneural injection and cell surface delivery because the biochemical cues encountered from the damaged tissue may be different. Thus, there is a need for more systematic research with a number of potential donor cell types in carefully controlled animal models. This is recognized more generally in cell transplantation to address a number of technical hurdles, not least those of phenotype instability, cost versus benefit, and ethical issues 170,183 .
A Minimally Invasive Technique for Cell Transplantation
Endoscopic surface transplantation
Minimal invasiveness of cell delivery is an indispensable requisite for clinical translation 192 . Endoscopy may fulfill this requirement most efficiently (Fig. 5). It has long been used in clinical otorhinolaryngology, has been reported in neurosurgical procedures since the 1970s 193,194 , and its safe maneuverability in the CPA has been established 195,196 .
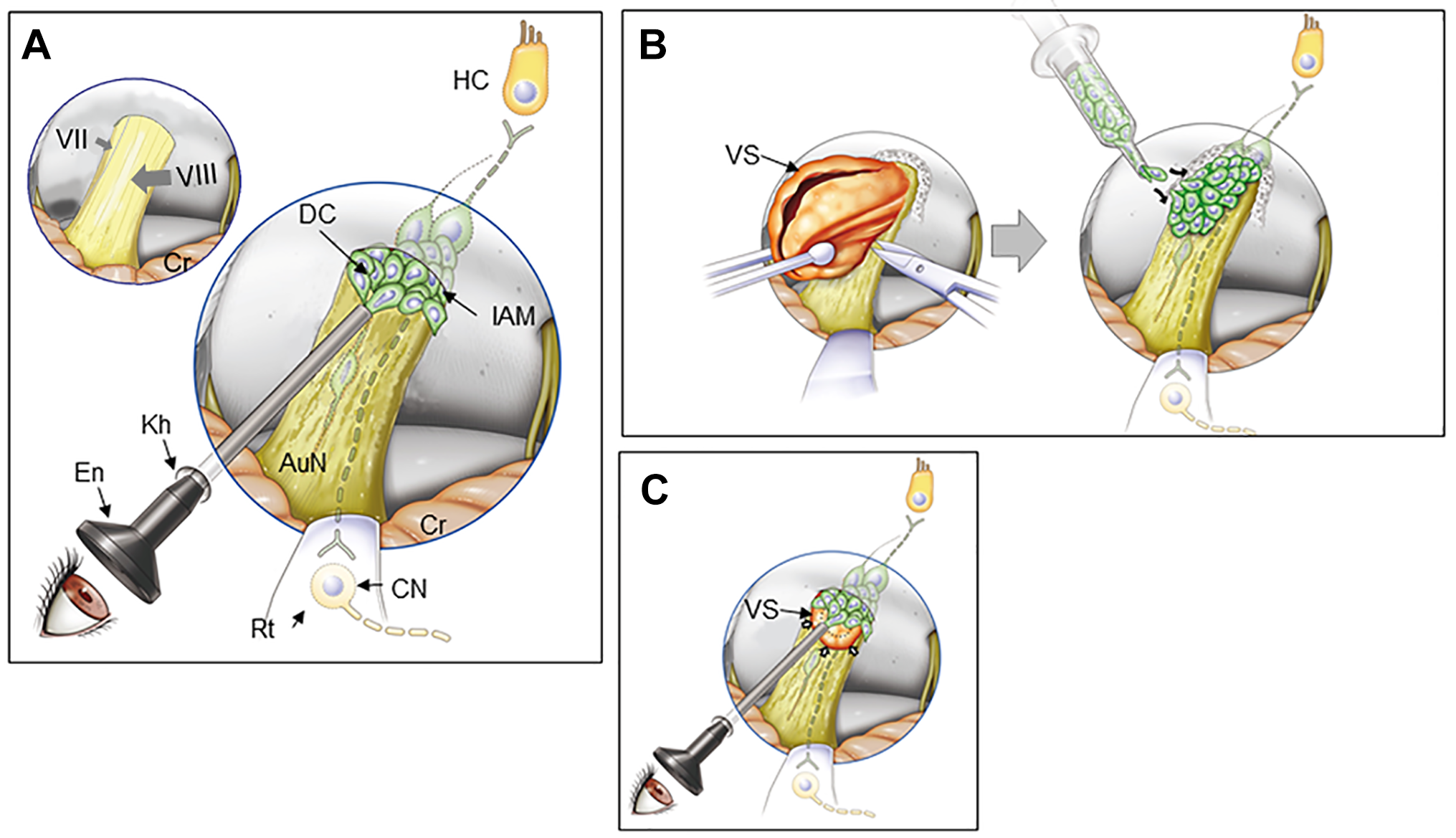
Endoscopic surface transplantation of donor cells. (A) In patients with non-tumorous auditory neuropathic hearing loss, an endoscope can be introduced in the cerebellopontine angle cistern and DCs placed onto the AuN. Normal facial nerve (VII) and vestibulocochlear nerve (VIII) are shown in the left upper corner of the panel. (B) In open surgery for larger VS, following tumor removal (left), donor cells can be placed on the surface of the AuN. The posterior wall of the internal auditory meatus is drilled to expose the tumor entirely. (C) In radiotherapy for small to medium-sized VS (arrows), a similar approach shown in A can be undertaken immediately after treatment. DCs are placed both on the distal side of the tumor through the internal auditory meatus (shown in this figure) and on its medial side if possible (not shown here). The dotted line indicates tumor shrinkage after radiotherapy. Regenerated bipolar neurons (dotted line) are shown in the nerve in each panel. AuN, auditory nerve; CN, cochlear nucleus cells; Cr, cerebellum; DC, donor cell; En, endoscope; HC, hair cell; IAM, internal auditory meatus; kh, keyhole; Rt, retractor; VS, vestibular schwannoma.
Surface transplantation of donor cells to the auditory nerve can be done with an endoscope introduced intracranially through a single keyhole in the retromastoid area. It could be applied to diseases such as auditory neuropathic hearing loss in neuropathies and head trauma (Fig. 5A), VS immediately after tumor removal (Fig. 5B), and VS following RT (Fig. 5C). This simple procedure requires only local anesthesia under sedation, and it is, thus, applicable to physically more sensitive patients, including the elderly.
Surface transplantation has several other advantages. Excessive numbers of cells are not required to compensate for the very high rates of donor cell death as observed in intraparenchymal injection 197 because donor cells apparently autonomously enter the host tissue in proportion to the demand and capacity of the host environment 2 . Moreover, in contrast to intraneural injection, donor cells transplanted onto the nerve are immediately nourished by cerebrospinal fluid, which is a very rich source of nutrients including proteins, ions, lipids, hormones, cholesterol, glucose and metabolites, and pro-regenerative molecules such as BDNF and IGF-2 198,199 , before they establish a link to the blood supply. In transplantation experiments of Parkinson’s disease, most dopamine neurons injected into the brain died due to apoptosis within the first 24 h of transplantation 200 , and subsequently, more than 90% of transplanted neurons died by the end of a typical several week transplantation study 201,202 . In rats, more than 1 week is required after transplantation before sufficient neovascularization is established between the host and transplants 203,204 . Until then, the intraparenchymally transplanted cells suffer insufficient nutrients diffusing from host vessels located outside the graft perimeter, resulting in apoptotic cell death (see the sections above).
It is worth noting that “surface transplantation” of donor cells is distinct from “stem cell sheet technology” such as that explored in heart, kidney, and liver disorders 205,206 . In surface transplantation for auditory nerve regeneration, a uniformly molded cell sheet manufactured before cell transplantation cannot be applied to the target area. On the contrary, to facilitate the integration of donor cells into the host, it is important to drop them freely into the narrow spaces within the irregular and complex contours of the tissue surface.
Conclusion
We conclude that there is great potential for clinical translation of cell transplantation in the auditory nerve. Proof of principle has been established; appropriate clinical techniques are available, and there is considerable theoretical support from wide-ranging studies on neurodegeneration and tissue repair. Auditory nerve damage may occur as a result of neuropathies, intracranial mass lesions, head trauma, and even therapeutic intervention. The finding that donor cells placed on the surface of the gliotic auditory nerve autonomously entered the nerve tissue, migrated, and functionally integrated into the host neural circuit makes clinical surgery much more realistic 2 . For clinical translation, endoscopy provides the best way to deliver viable cells to the tissue surface with minimal damage to residual functional elements in the nerve.
Whilst proof of principle is an important step, there is clearly a need for focused animal experiments that recreate the combination of approaches necessary for clinical application. “From the bench to the clinic” is a slogan that has been repeated also in regenerative medicine. Now it can be accomplished if an appropriate cell transplantation method is applied to humans, choosing potent human stem cells or human cochlear precursors 207,208 that may or may not be conditioned to achieve integration.
Notably, the auditory nerve holds an advantageous and suitable position for cell transplantation therapy because HCs, auditory neurons, and cochlear nucleus cells are aligned over a relatively short distance 209,210 . This contrasts with the recovery of the injured pyramidal tract in SCI, which involves not only axon sprouting but also recruiting endogenous relay neurons 211,212 .
Finally, transdisciplinary combinations with regenerative studies for both auditory nerve and HCs would pave a new path for more widespread treatment of DHL and even for a number of other neurodegenerative conditions.
Footnotes
Acknowledgment
We appreciate Emeritus Professor Juichi Ito and Professor Koichi Omori, Department of Otolaryngology, Head and Neck Surgery, Kyoto University Graduate School of Medicine for their support.
Author Contributions
TS and MCH conceived the original idea. TS curated and analyzed the data, obtained the funding and wrote the original draft of the article. MCH also contributed to the writing and editing of the article.
Ethical Approval
Our university and hospital do not require ethical approval for any papers based on article review.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Japan Society for the Promotion of Science (MEXT) (#26931051, 24931046, 13557112, 1559150, 12470281, 09557113-3, 10877209, 08457356-3, 09557113, 08457356, 07457303, 06454407, 03454341, 58770916), Univers Foundation, the General Insurance Association of Japan, the Japan Health Foundation, Osaka Gas Group Welfare Foundation, Zenkyoren and Mitsui Sumitomo Insurance Welfare Foundation.
