Abstract
Recently, mesenchymal stem cells have demonstrated a potential for neurotrophy and neurodifferentiation. We have recently isolated mobilized dental pulp stem cells (MDPSCs) using granulocyte-colony stimulating factor (G-CSF) gradient, which has high neurotrophic/angiogenic potential. The aim of this study is to investigate the effects of MDPSC transplantation on peripheral nerve regeneration. Effects of MDPSC transplantation were examined in a rat sciatic nerve defect model and compared with autografts and control conduits containing collagen scaffold. Effects of conditioned medium of MDPSCs were also evaluated in vitro. Transplantation of MDPSCs in the defect demonstrated regeneration of myelinated fibers, whose axons were significantly higher in density compared with those in autografts and control conduits only. Enhanced revascularization was also observed in the MDPSC transplants. The MDPSCs did not directly differentiate into Schwann cell phenotype; localization of these cells near Schwann cells induced several neurotrophic factors. Immunofluorescence labeling demonstrated reduced apoptosis and increased proliferation in resident Schwann cells in the MDPSC transplant compared with control conduits. These trophic effects of MDPSCs on proliferation, migration, and antiapoptosis in Schwann cells were further elucidated in vitro. The results demonstrate that MDPSCs promote axon regeneration through trophic functions, acting on Schwann cells, and promoting angiogenesis.
Keywords
Introduction
Peripheral nerve injuries are often caused by trauma or surgical complications and result in considerable disabilities (25). Autologous nerve graft is the gold standard for nerve repair/regeneration. Requirement of a second surgery with sacrifice of a functional nerve, however, results in loss of sensation, potential formation of neuroma, and neuropathic pain (23,34). Recently, much of the research effort has focused on the use of mesenchymal stem cells (MSCs), such as bone marrow mesenchymal stem cells (BM-MSCs), adipose-derived stem cells (ADSCs), and amniotic fluid stem cells (AFSs), to develop a novel treatment for peripheral nerve injuries (24). Nevertheless, autologous nerve graft remains the most successful treatment in peripheral nerve injury (14,16). MSCs ameliorate peripheral nerve regeneration through multiple mechanisms: direct differentiation into Schwann cell phenotype, secretion of trophic factors, and promoting remyelination (24). Differentiation of BM-MSCs into Schwann cell lineage has been reported in a rat sciatic nerve gap after implantation in vivo (2,33). Various neurotrophic factors, including glial cell line-derived neurotrophic factors (GDNF), nerve growth factor (NGF), ciliary-derived neurotrophic factor (CDNF), neu-rotrophin-3 (NT-3), and brain-derived neurotrophic factor (BDNF) from MSCs have been demonstrated to promote endogenous repair of neurologically damaged areas (6,29). Those neurotrophic factors are suggested to provide a favorable microenvironment for neural cell survival and neurite extension, and stimulate the growth, maturation, and differentiation of Schwann cells (32). In addition, another possible effect of the transplanted MSCs may be enhanced homing and migration of endogenous cells to the injured site for remyelination signaled by the stromal cell-derived factor (SDF-1)/CXCR4 axis such as reported in injured central nervous system (22). In the peripheral nervous system, a report demonstrated that transplanted ADSCs evoke greater Schwann cell proliferation/intrusion in the distal stump (3).
Transplanted dental pulp stem cells (DPSCs) combined with polylactic glycolic acid (PLGA) tubes promoted 7-mm-long facial nerve gap repair (27,28). In our previous study, central nerve regeneration and angiogenesis/ vasculogenesis have been demonstrated after transplantation of DPSCs in ischemic cerebrum models and in ischemic hindlimb models, respectively, by the trophic effects including migration, proliferation, and antiapoptosis (12,13,30). Direct differentiation of the transplanted DPSCs into neurons, endothelial cells, and glial cells are not demonstrated in these models. The mechanisms by which DPSCs enhanced peripheral nerve regeneration, however, should be further investigated. Mobilized dental pulp stem cells (MDPSCs) using granulocyte-colony stimulating factor (G-CSF) gradient showed higher trophic effects including migration and angiogenic/neurogenic induction compared to mobilized bone marrow mesenchymal stem cells (MBM-MSCs), mobilized adipose-derived stem cells (MADSCs), and colony-derived nonisolated DPSCs isolated from the same individuals (11,17). The transplantation of MDPSCs into pulpectomized teeth regenerated pulp tissue in higher volume with higher vascularization and innervation compared with the transplantations of MBM-MSCs, MADSCs (17), and DPSCs (11).
In the present investigation, we examined the potential utility of MDPSCs for peripheral nerve regeneration compared with autografts and control conduits without MDPSCs in an in vivo system of rat sciatic nerve defect. To further elucidate the possible mechanisms of MDPSCs, trophic effects on Schwann cells were evaluated in vivo by histochemical and in situ hybridization analyses and in vitro using the conditioned medium of MDPSCs.
Materials and Methods
Isolation of Human DPSC Subsets, MDPSCs
Normal human third molars were collected from adults (18-29 years of age) at the Aichi-Gakuin University Dental Hospital under approved guidelines set by the School of Dentistry, Aichi-Gakuin University, and the National Center for Geriatrics and Gerontology, Research Institute. DPSCs were isolated from human dental pulp tissue with a slight modification of a previously described method (12). Briefly, dental pulp tissue was enzymatically digested, and the isolated cells were plated at 3 × 104 cells on 35-mm dishes (BD Biosciences, San Jose, CA, USA) after filtration using a 40-μm Cell Strainer (BD Biosciences) and lysis of red blood cells with IOTest3 lysing solution (Beckman Coulter, Fullerton, CA, USA). Once the cells reached 60-70% confluency, they were detached and subcultured.
Human DPSC subsets were further isolated by G-CSF (Neutrogin® Chugai Pharmaceutical Co., Ltd., Tokyo, Japan)-induced stem cell mobilization (MDPSCs) as described previously (18). Briefly, chemically treated Costar Transwell® to prevent cell attachment was used as the upper chamber, and 24-well tissue culture plates (BD Biosciences) were inserted as the lower chamber. At the third cell culture passage, human DPSCs (2 × 104 cells) were added to the upper chambers, and DMEM (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% human serum (Lonza, Basel, Switzerland), and G-CSF at the final concentration of 100 ng/ml was added to the lower chambers. After 48-h incubation, the medium was changed to DMEM supplemented with 10% human serum without G-CSF. Once the cells reached 60-70% confluency, they were detached with 0.05% trypsin-EDTA (Gibco, Carlsbad, CA, USA) and subcultured.
Surgical Procedures
All animal protocols were approved by the Ethical Committee of Aichi-Gakuin University School of Dentistry. Adult male Fischer 344 rats (Japan SLC, Inc., Shizuoka, Japan) weighing 190-210 g were used. Before transplantation, all collagen conduits (provided by Prof. Hirata) were immersed in Hank's balanced salt solution (Gibco) containing 20 μg/ml fibronectin (Nitta Gelatin, Osaka, Japan) and 10 μg/ml laminin (Invitrogen, Carlsbad, CA, USA) for 3 h. Animals were initially anesthetized with 3.0% isoflurane (Intervet, Summit, NJ, USA) and maintained under anesthesia with 1.5% isoflurane. A 5-mm segment of the left sciatic nerve was excised. Immediately after transection, a 5-mm gap was bridged by a 9-mm collagen conduit with a 1.3-mm lumen, with each nerve end being sutured 2 mm into the conduit using 10-0 nylon (Bear Medic Corporation, Ibaraki, Japan) resulting in a 5-mm nerve gap (Fig. 1). A 20% dilution of the original collagen scaffold (1% Atelocollagen Implant; Koken, Tokyo, Japan) in Hank's balanced salt solution containing 20 μg/ml fibronectin and 10 μg/ml laminin was prepared for cell suspensions. A 5-mm gap was filled with 3.0 × 105 MDPSCs in the collagen scaffold (n=4) and the collagen scaffold only (n=4). For autograft, a 5-mm segment of the excised nerve was reversed and resutured with 10-0 nylon (n=4). DiI-labeled cells were used in some of MDPSC transplants. The wound was closed with 4-0 silk sutures (Natsume Seisakusho, Tokyo, Japan). To avoid immunorejection, intraperitoneal administration of tacrolimus (Prograf® Astellas Pharmaceutical Inc., Tokyo, Japan) was performed at 0.05 mg/kg every day starting at the preoperative day for 2 weeks after transplantation, and every other day for the following 3 weeks. All surgical procedures were performed aseptically. All animals were allowed to access water and foods ad libitum.

Sciatic nerve defect model with collagen conduits. A 5-mm defect in the left sciatic nerve was bridged with a 9-mm collagen conduit.
Histological Preparation
Histological evaluation of sciatic nerve regeneration was evaluated by hematoxylin and eosin (H&E; Muto Pure Chemicals, Tokyo, Japan) and toluidine blue staining on days 21 and 35. The left sciatic nerve was dissected out, placed in 4% paraformaldehyde (PFA) (Nacalai Tesque, Kyoto, Japan) and processed for paraffin embedding (Sigma-Aldrich). Cross sections, 2.5 μm in thickness, were cut for H&E staining and vascular staining. Staining of vascular endothelium was performed with Fluorescein Griffonia (Bandeiraea) Simplicifolia Lectin 1/fluorescein-Galanthus nivalis (snowdrop) lectin (20 mg/ml; Vector Laboratories, Burlingame, CA, USA). A fluorescence microscope BIOREVO, BZ-9000 (Keyence, Osaka, Japan) was used for calculating the vessel area. Semithin sections prepared from samples embedded in epoxy resin (Epon812; Taab Laboratories Equipment Ltd., Berks, UK) as mentioned below were stained with 0.1% toluidine blue (Amresco, Solon, OH, USA). Sections were observed under a light microscope (6000B-4; Leica, Wetzlar, Germany). At 400x magnification, three fields per nerve were measured to determine the density (axons/mm2), the diameter of regenerated axons, and the total number of myelinated axons (axons/nerve) using ImageJ (National Institutes of Health, Bethesda, MD, USA).
Transmission Electron Microscopic Analysis
Electron microscopy was used to evaluate remyelination and growth of fibrous tissue. Samples were fixed by a solution of 2.5% glutaraldehyde (Taab Laboratories Equipment Ltd.) and 2% paraformaldehyde in 0.1 M sodium cacodylate (pH 7.4) (Karnovsky's fixative) (Wako, Osaka, Japan) for 6 h at 4°C and postfixed in 1% osmium tetroxide (Taab Laboratories Equipment Ltd.), according to conventional procedures. After dehydration through a graded ethanol series, the specimens were embedded in Epon 812 (Taab Laboratories Equipment Ltd.). Ultrathin sections were cut with a diamond knife and were stained with uranyl acetate (Wako) and lead citrate (Wako), then examined with an electron microscope (Type 1010; JEOL, Tokyo, Japan). Six fields per nerve were measured to calculate the g-ratio value (the ratio of the inner axonal diameter to the outer diameter).
Immunohistochemical Analysis
The animals were sacrificed by pentobarbital (Kyoritsu Seiyaku, Tokyo, Japan) overdose at day 14 after transplantation. Left sciatic nerve was dissected out, fixed in 4% PFA overnight, and immersed into 15% sucrose solution and further into 30% sucrose solution and were embedded in OCT compound (Sakura Finetek Japan, Tokyo, Japan). Twelve-micrometer-thick cross sections were cut on a cryostat.
To evaluate the fate of the transplanted cells, immunostaining with S100 antibody was performed using DiI-labeled sections. The sections were preincubated in 2% goat serum (Sigma-Aldrich) at RT for 30 min and incubated at 4°C overnight with mouse anti-human S100 antibody (1:200, ab4066; Abcam, Cambridge, UK) and secondary Alexa 488-conjugated anti-mouse IgG antibody (1:50; Life Technologies, Carlsbad, CA, USA) for 30 min at room temperature (RT).
To evaluate the antiapoptotic effect of MDPSCs, immunostaining with caspase 3 antibody was performed using nonlabeled sections. The sections were preincubated in 2% goat serum at RT for 30 min, and incubated at 4°C overnight with rabbit anti-human caspase 3 (1:50, #9661; CST, Danvers, MA, USA) and secondary Alexa 594 conjugated anti-rabbit IgG antibody (1:50; Life Technologies) at RT for 30 min. Nuclei were counterstained with Hoechst 33342 (Sigma-Aldrich). Antiapoptotic effect was analyzed on a BZ-9000 BIOREVO fluorescence microscope (Keyence).
Double immunostaining with Ki-67 and S100 antibody was performed for the evaluation of Schwann cell proliferation activity using nonlabeled sections. The sections were preincubated in 2% goat serum at RT for 30 min, and incubated at 4°C overnight with rabbit anti-Ki-67 antibody (1:100, ab15580; Abcam) and secondary Alexa 594-conjugated anti-rabbit IgG antibody (1:50; Life Technologies) at RT for 30 min. Further, the sections were incubated at 4°C overnight with mouse-antihuman S100 antibody (1:200, ab4066; Abcam) and secondary Alexa 488-conjugated anti-mouse IgG antibody (1:50; Life Technologies) for 30 min at RT. To evaluate the proliferation of transplanted cells, single immunostaining with Ki-67 antibody was also performed using DiI-labeled sections. The sections were preincubated in 2% goat serum at RT for 30 min and incubated at 4°C overnight with rabbit anti-human Ki-67 antibody (1:100, ab15580; Abcam) and secondary Alexa 488-conjugated anti-rabbit IgG antibody (1:50; Life Technologies) for 30 min at RT. Slides were examined by confocal laser microscopy (TCS SP5 conventional inverted microscope; Leica). Three-dimensional structures were reconstructed by Leica Application Suite Advanced Fluorescence (LAS AF; Leica) software.
In Situ Hybridization Analysis
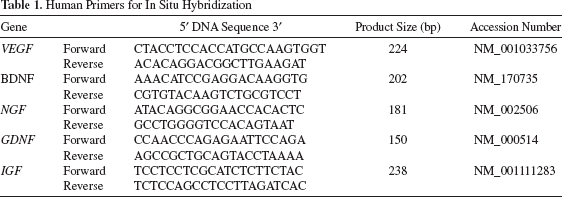
Neurotrophic/angiogenic factors expressed in the transplanted cells (nonlabeled) were examined by in situ hybridization in the cryosections on day 14. The left sciatic nerve was dissected out, fixed in 4% paraformaldehyde solution (Nacalai Tesque) overnight and immersed into 15% sucrose solution and further into 30% sucrose solution, and was embedded in OCT compound (Sakura Finetek Japan). Cross sections were obtained. Human cDNA of brain-derived neurotrophic factor (BDNF) (202 bp), glial cell-derived neurotrophic factor (GDNF) (150 bp), insulin-like growth factor (IGF) (238 bp), nerve growth factor (NGF) (181 bp), and vascular endothelial growth factor (VEGF)-A (224 bp) linearized with NcoI, NcoI, SpeI, SpeI, and NcoI, respectively, were used as antisense probes, and those linearized with SpeI, SpeI, NcoI, NcoI, and SpeI, respectively, were used as sense probes. The probes were constructed out of the plasmids after subcloning of the PCR products using the primers as Table 1. In situ hybridization analysis was performed as previously described (19). In brief, the cryosections were treated with 20 μg/ml proteinase K for 6 min and hybridized with BDNF, GDNF, IGF, NGF, and VEGF antisense and sense probes in hybridization solution at 57°C for 16 h. Then, after treatment with RNase buffer at a concentration of 20 μg/ml, the sections were washed in high stringency buffer, 2x SSC and 0.1x SSC, each for 20 min. They were incubated with anti-DIG-POD (Roche, Indianapolis, IN, USA) after equilibration in TNB buffer (pH 7.5) and further developed in fluorescein tyramide reagent (PerkinElmer, Boston, MA, USA) for 10 min. They were examined by confocal laser microscopy (TCS SP5 conventional inverted microscope; Leica). Three-dimensional structures were reconstructed by Leica Application Suite Advanced Fluorescence (LAS AF; Leica) software.
Human Primers for In Situ Hybridization
Migration, Proliferation, and Antiapoptotic Assays
For collecting the conditioned medium (CM), the medium was changed into DMEM without fetal bovine serum (FBS) at 70% confluence, and the CM from MDPSCs were collected 24 h later and concentrated by Amicon® Ultra-15 Centrifugal Filter (Millipore, Billerica, MA, USA).
To assess the proliferative effect of the CM, RT4-D6P2T cells were seeded at 1 × 103 cells per well in a 96-well plate (BD Biosciences) in DMEM supplemented with 0.2% BSA (Sigma-Aldrich) and 10 μg/ml of CM. Ten microliters of Tetra-color one® (Seikagaku Corporation, Tokyo, Japan) was added to the 96-well plate, and the cell numbers were measured using a spectrophotometer at 450 nm absorbance at 2, 12, 28, 36, 51, and 60 h of culture. Wells without cells served as negative controls.
To examine migration activity of RT4-D6P2T (rat Schwann cells, EC93011415-F0; DS Pharma Biomedical, Osaka, Japan) in CM from MDPSCs, a horizontal chemotaxis assay was performed as described previously (10). In brief, each cell fraction (1 μl of 105 cells/ml) was placed into the single hole with which the device was held together with a stainless steel holder, and 10 μg/ml of CM was placed into the contra-hole. Video images of cell migration were taken every 3 h for 18 h.
To assess the antiapoptotic effect of the CM, RT4-D6P2T cells were incubated with 500 nM staurosporine (Sigma-Aldrich) in DMEM supplemented with 8 μg/ml of CM from MDPSCs. After 5 h, the RT4-D6P2T cells were harvested, and the cell suspensions were treated with annexin V-FITC and propidium iodide (Roche) for 15 min and then analyzed by flow cytometry.
Statistical Analysis and Significance
Data are reported as means ± SD. Values of p were calculated using Mann-Whitney U test and Bonferroni correction in SPSS 21.0 (IBM, Armonk, NY, USA).
Results
Histomorphometric Analysis
Five weeks after cell transplantation, the animals were sacrificed to study the morphology of response to the transplants. Gross observation of the transplants revealed that the transplanted collagen conduit was covered with a thin capsule of fibrous tissue and that it remained intact and was not degraded. No apparent immunoreaction was observed in the surrounding tissue, which proved that collagen conduits had good histocompatibility (Fig. 2A-C). Light microscopy of sections with H&E staining demonstrated that regenerating nerves were enclosed in collagen conduits. In the MDPSCs transplants, excellent angiogenesis was observed. A few inflammatory cells were also observed (Fig. 2D). In the collagen control group, angiogenesis was observed, but the response and diameter was smaller than cell transplanted groups. In addition, relatively high degree of inflammation was noticed (Fig. 2E). Five weeks after transplantation, the regenerated axons, which were surrounded by the thin myelin sheath, were observed in all groups (Fig. 2G-I). The axon diameter and density were calculated in the toluidine blue-stained sections (Fig. 2J, K). The autograft group (Fig. 2I) showed a significantly superior axon diameter (2.15 ± 0.22 μm) when comparing to other groups (vs. MDPSCs p<0.05; vs. collagen p<0.01). No statistical difference was observed between the MDPSC transplants (1.53 ± 0.43 μm) and the collagen group (1.39 ± 0.23 μm) (Fig. 2J). On the other hand, the highest axon density was found in the MDPSC transplants (23,678 ± 5,306 fibers/mm2), which was superior when compared to the collagen group (13,280 ± 1,757 fibers/mm2; p<0.01) and the autograft group (16,981 ± 2,137 fibers/mm2; p<0.05). The lowest axon density was found in the collagen group, which showed a significant statistical difference compared with other groups (vs. MDPSCs p<0.01; vs. autograft p<0.05) (Fig. 2K). The total number of regenerated myelinated axons was largest in the autograft group (14,345.83 ± 3,496.44 axons/nerve), followed in MDPSC transplants (11,492.54 ± 1,534.93 axons/nerve), and the collagen group (5,417.72 ± 2,062.39 axons/nerve). There was no statistical difference between MDPSC transplants and the autograft group (Fig. 2L). The g-ratio values were 0.73 ± 0.059 (MDPSC transplants), 0.75 ± 0.040 (collagen group), and 0.73 ± 0.016 (autograft group) (Fig. 2M). Immunofluorescent staining analysis with BS-1 lectin demonstrated that there was a trend toward a larger amount of neovascularization in the MDPSC transplants (Fig. 2N). The electron micrograph confirmed the massive fibrous tissue in the collagen group (Fig. 3A), which was less in the MDPSC transplants (Fig. 3B) and the autograft group (Fig. 3C). In the MDPSC transplants, the amount of the inflammatory cells was comparatively less than that of the collagen group.

Macroscopic and microscopic findings of regenerated nerve. (A-C) Macroscopic findings 35 days after transplantation. (D-F) H&E staining 21 days after transplantation (cross section). v, vessel. (G-I) Toluidine blue staining 35 days after transplantation (cross section). Arrows indicate myelinated axon (A, D, G: MDPSC transplants; B, E, H: collagen group; C, F, I: autograft group). Scale bars: 25 μm. (J) Myelinated axon diameter. (K) Density of myelinated axons per square millimeter. (L) Total number of myelinated axon. (M) Calculated g-ratio value. (N) Vascularization area. **p<0.01, *p<0.05.

Transmission electron microscopic analyses in cross-sections 35 days after transplantation. (A) Collagen group. (B) MDPSC transplants. (C) Autograft group. (D) Normal sciatic nerve. Ax, axon; Sc, Schwann cell; Ly, lymphocyte; Fb, fibroblast. Scale bars: 2 μm. *unmyelinated nerve.
Pulp Stem Cell Outcome
To investigate the outcome of the transplanted cells, the rats transplanted with DiI-labeled MDPSCs were sacrificed on day 14 and immunostained with S100 antibody (Fig. 4A-C). DiI-labeled transplanted MDPSCs, which were alive on day 14, were characterized by round-to-oval nuclei with minimal variable cytoplasm. The transplanted cells localized in the close proximity of the S100-positive cells and did not directly differentiate into Schwann cell phenotype. Instead, the expression of several neurotrophic factors, BDNF, GDNF, IGF, NGF, and VEGF, was detected in the DiI-labeled MDPSCs on day 14 by in situ hybridization analysis (Fig. 4D-R), demonstrating trophic factor release in the vicinity of the transplanted cells.

Immunohistochemical and in situ hybridization analyses in cross sections of regenerated nerve 14 days after transplantation. (A-C) Immunofluorescent staining with S100 antibody. (green: S100, red: MDPSCs) (A: S100; B: transplanted cells; C: merge) (D-R) Expression of neurotrophic factors in the transplanted cells. In situ hybridization with BDNF (D-F), GDNF (G-I), IGF (J-L), NGF (M-O), and VEGF (P-R) [D, G, J, M, P: each factor (green); E, H, K, N, Q: transplanted cell (red); F, I, L, O, R: merge] Scale bars: 10 μm.
Proliferation and Antiapoptosis
In order to further characterize the response to dental pulp stem cells, proliferation and antiapoptotic effect was evaluated (Fig. 5A-F). In the MDPSC transplants, the number of caspase 3-positive cells was markedly decreased compared with the collagen group on day 14 (Fig. 5A-C). As for cell proliferation, Ki-67-positive cells were observed more in the MDPSC transplants compared with collagen group on day 14 (Fig. 5D, E). There were no transplanted cells that were positive for Ki-67 in the MDPSC transplants (Fig. 5F).

Evaluation of antiapoptotic and proliferation activities in vivo 14 days after transplantation. (A-B) Immunofluorescent staining with caspase 3 antibody (red: caspase 3; blue: Hoechst 33342) (A: MDPSC transplants; B: collagen group). (C) Percentage of caspase 3-positive cells. (D, E) Double immunofluorescent staining with S100 and Ki-67 antibodies. Arrows indicate Ki-67-positive nucleus (green: S100, red: Ki-67) (D: MDPSC transplants; E: collagen group). (F) Immunofluorescent staining with Ki-67 antibody (green: Ki-67, red: transplanted cells). Scale bar: 10 μm.
Response to Conditioned Medium
To clarify the effect of MDPSC CM on Schwann cells, migration, proliferation, and antiapoptotic assays were performed in vitro. Although the proliferation activity of RT4-D6P2T was not enhanced by supplementation of 5% FBS, the MDPSC CM significantly enhanced proliferation of RT4-D6P2T cells by 28 h (p<0.01) (Fig. 6A). The MDPSC-conditioned medium also significantly enhanced the migratory activity of RT-D6P2T cells compared with FBS (p<0.01) (Fig. 6B). In addition, the MDPSC-conditioned medium significantly decreased apoptosis of RT-D6P2T cells (p<0.05) (Fig. 6C).

The effect of CM on RT4-D6P2T (rat Schwann cells). (A) Proliferative activity (**p<0.01 vs. 0.2% BSA). (B) Antiapoptotic activity (**p<0.01 vs. FBS). (C) The migration activity (**p<0.01, *p<0.05).
Discussion
There are many stem cell sources for the repair and regeneration of injured peripheral nerves. Dental pulp is derived from neural crest, and DPSCs are easily isolated from discarded teeth following extraction with very low morbidity and no ethical issues. The promoting effects of DPSCs on nerve regeneration such as replacement of lost cells by differentiating into mature oligodendrocytes, antiapoptotic effect on neurons, astrocytes and glial cells, and acceleration of migration and differentiation of neuronal progenitor cells have been demonstrated in the central nerve lesion (spinal cord injury model and focal cerebral ischemia model) (26,30). Although transplantation of DPSCs has also been reported to promote facial nerve regeneration, comparison with autologous nerve graft has not been reported (27,28). In this investigation, the MDPSC group exceeded the autograft group in axon density. The total number of myelinated axons was largest in the autograft group, followed in MDPSC transplants, and collagen group, and there was no statistical difference between the autograft group and MDPSC transplants. That means bridging nerve defects with a collagen conduit may restrict the regenerating area and caused relative increase in axon density in MDPSCs transplants. In this way, the difference in axon diameter in both groups may be due to the difference of the regenerating area. The g-ratio value is about 0.6-0.7 in normal peripheral nerves. All groups showed g-ratio value around 0.7-0.8, suggesting a good degree of remyelination.
MDPSC groups showed large blood vessel formation, consistent with the previous reports on mesenchymal stem cells to increase the neovascularization (15,21). During axonal growth, neovascularization is an important prerequisite step. Schwann cells and axons extend together with angiogenesis, which precedes axonal regeneration. Blood vessel regeneration increases the axonal regrowth and Schwann cell proliferation in the presence of VEGF (8,9). In this study, MPSCs expressed VEGF both in vivo and in vitro. MDPSCs may support angiogenesis by secreting VEGF, which result in good nerve regeneration.
In the peripheral nerve regeneration, Schwann cells are important, as they support and promote axonal regrowth and target tissue innervation (7). For example, ADSCs express Schwann cell proteins in a rat sciatic nerve gap after intravenous injection, suggesting differentiation potential of ADSCs into Schwann cells (20). BM-MSCs also express Schwann cell proteins in a rat sciatic nerve gap after transplantation, which implies differentiation into Schwann cells (2,31,33). Neurotrophic factors also play an indispensable role for neuronal survival and axonal regeneration, which is also necessary for effective functional reinnervation of injured peripheral nerves. For instance, ADSCs are capable of secreting neurotrophic factors such as insulin-like growth factor-1 (IGF-1) and BDNF, which are involved in axonal growth and sprouting required for peripheral nerve regeneration in vivo (5,15). BM-MSCs express neurotrophic factors, such as BDNF, basic fibroblast growth factor (bFGF), and ciliary neurotrophic factor (CNTF), which promote neurite extension and Schwann cell proliferation and maintain differentiated neurons in vivo (1,4,33). In the present study, direct differentiation of the transplanted MDPSCs into Schwann cell phenotype was not detected, but the expression of several neurotrophic factors, BDNF, GDNF, IGF, NGF, and VEGF, was detected in the DiI-labeled MDPSCs, which localized near the Schwann cells, by in situ hybridization analysis. Based on experimental evidence, MDPSCs may have stimulatory influence on residual Schwann cells by secreting neurotrophic factors. In vitro experiments showed antiapoptotic, proliferative, and migratory effects on Schwann cells. In vivo, less apoptotic cells and more proliferation were observed in the MDPSC-transplanted groups compared with the control collagen group. In conclusion, MDPSCs contribute to peripheral nerve regeneration by secretion of neurogenic/angiogenic factors in the close proximity of newly migrated Schwann cells and also regulating their apoptosis and proliferation. We predict, in the near future, dental pulp stem cell transplantation may become a possible candidate to take the place of autologous nerve grafts in peripheral nerve repair and regeneration.
Footnotes
Acknowledgments
This work was supported by the Research Grant for Longevity Sciences (23-10) from the Ministry of Health, Labour and Welfare (M.N.) and Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture, Japan, No. 23593007 to K.K. The authors declare no conflicts of interest.
