Abstract
This study tested the impact of single dose and two doses of endothelial progenitor cells (EPCs) and EPCs-derived condition medium (CM) on protecting the left-ventricular myocardium (LVM) from acute ischemia-reperfusion (IR) injury. In vitro study showed EPCs and CM had comparably higher capacity for enhancement of angiogenesis as compared with the controls (all
Introduction
Despite state-of-the-art treatments, including modern pharmacomodulation 1,2 , procedural advancements such as percutaneous coronary intervention (PCI) and coronary bypass surgery (CABG) 3 –7 , progressive guideline renewals 8,9 and continuous education regarding secondary prevention of coronary artery disease (CAD) 10 , CAD remains the leading cause of death worldwide, suggesting the need for further development of management options.
Endothelial progenitor cells (EPCs) and mesenchymal stem cells (MSCs) therapies continue to show promise for tissue and organ regeneration and restoration of ischemia-related organ function 11 –13 . Intriguingly, some studies even demonstrated that EPC- or MSC-derived condition medium (CM) therapy effectively improved ischemia-related organ dysfunction 14,15 . Studies by Losordo et al and ourselves 16,17 have shown that bone marrow-derived or circulatory-derived autologous EPC therapy effectively reduced angina and heart failure and improved left ventricular ejection fraction. However, only 5% to 7% increases in left ventricular ejection fraction (LVEF) were achieved after EPC therapy in these studies 16 –18 , suggesting room for further LV improvement. Furthermore, even though autologous cells were available and utilized in these studies 16 –18 , they were administered only once to patients. It is possible that two or more doses of cell therapy could further enhance tissue regeneration and improve ischemia-related organ dysfunction. We designed an animal model study to assess the safety and efficacy of this strategy.
Materials and Methods
Ethics Statement
All animal procedures were approved by the Institute of Animal Care and Use Committee at Kaohsiung Chang Gung Memorial Hospital (Affidavit of Approval of Animal Use Protocol No. 2017102602) and performed in accordance with the Guide for the Care and Use of Laboratory Animals.
Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC; Frederick, MD, USA)-approved animal facility in our hospital with controlled temperature and light cycles (24 oC and 12/12 light cycle).
Animal Groupings and Animal Model of Myocardial Ischemia-Reperfusion (IR) Injury
Pathogen-free, adult male Sprague-Dawley (SD) rats (
The procedure and protocol for myocardial IR injury have previously been reported 20 . All animals were anesthetized with 2.0% inhalational isoflurane on a warming pad at 37°C for the IR procedure and, under sterile conditions, the heart was exposed via a left thoracotomy. IR injury was induced by ligating the left coronary artery for 40 minutes with a 7-0 prolene suture, 3 mm distal to the margin of the left atrium. Regional myocardial ischemia was verified by observing a rapid color change from pink to dull red over the anterior surface of the left ventricle and rapid development of akinesia and dilatation over the affected region. Rats receiving thoracotomy only without ischemia induction served as sham controls. The knot was then released after 40-minutes ischemia to allow 72 h reperfusion. The rats were recovered in a portable animal intensive care unit (ThermoCare®, Paso Robles, CA, USA) for 24 hours, sacrificed at 72 h after the IR procedure, and their hearts harvested for individual study.
Peripheral Blood Was Collected and Cultured for EPCs and Condition Medium
The procedure and protocol have been reported in our previous report 11 . Rats in groups 3 to 6 were anesthetized with inhalational 2.0% isoflurane at day 21 prior to acute IR induction to collect peripheral blood for culturing EPCs and autologous transfusion. Isolated mononuclear cells from peripheral blood were cultured in a 100 mm diameter dish with 10 mL DMEM culture medium containing 10% FBS for 21 days. Flow cytometric analysis was performed to identify cellular characteristics (i.e., EPC surface markers) after cell-labeling with appropriate antibodies on day 21 of cell cultivation prior to implantation. The supernatant was collected during the 3rd to 5th passage of EPC culturing as the EPC-derived “condition medium.” Rats in groups 1 and 2 were anesthetized with inhalational 2.0% isoflurane and only received an identical procedure of peripheral blood sampling without further culture.
Functional Assessment with Echocardiography
The procedure and protocol for echocardiography have recently been reported 21 . Transthoracic echocardiography was performed in each group prior to and on day 3 after myocardial IR induction. The procedure was performed by an animal cardiologist blinded to the experimental design using an ultrasound machine (Vevo 2100, Visualsonics, Toronto, ON, Canada). M-mode standard two-dimensional (2D) left parasternal-long axis echocardiographic examination was conducted. Left ventricular internal dimensions [end-systolic diameter (ESD) and end-diastolic diameter (EDD)] were measured at mitral valve and papillary levels of the left ventricle, according to the American Society of Echocardiography leading-edge method using at least three consecutive cardiac cycles. LVEF was calculated as follows: LVEF (%) = [(LVEDD3-LVEDS3)/LVEDD3] × 100%.
Western Blot Analysis of Heart Specimens
Western blot analysis was performed as described previously 11,19 –21 . In detail, equal amounts (50 μg) of protein extracts were loaded and separated by SDS-PAGE. Separated proteins were transferred to PVDF membranes, and nonspecific sites were blocked by incubation in blocking buffer [5% nonfat dry milk in T-TBS (TBS containing 0.05% Tween 20)] overnight. The membranes were incubated with the indicated primary antibodies [CD31 (1:1000, Abcam, Cambridge, UK), von Willebrand factor (vWF) (1:1000, Abcam, Cambridge, UK), endothelial nitric oxide synthase (eNOS) (1:1000, Abcam, Cambridge, UK), cleaved poly (ADP-ribose) polymerase (PARP) (1:1000, Cell Signaling, Danvers, Ma, USA), cleaved caspase 3 (c-Csp3) (1: 1000, Cell Signaling, Danvers, Ma, USA), Smad3 (1:1000, Cell Signaling, Danvers, Ma, USA), brain natriuretic peptide (BNP) (1:500, Abcam, Cambridge, UK), NADPH oxidase (NOX)-1 (1:1500, Sigma, St. Louis, MO, USA), NOX-2 (1:500, Sigma, St. Louis, MO, USA), cytosolic cytochrome C (cyto-CytC) (1:2000, BD, Franklin Lakes, NJ, USA), mitochondrial cytochrome C (Mito CytC) (1:2000, BD, Franklin Lakes, NJ, USA), p47phox (1:1000, Merck Millipore, Burlington, Ma, USA), transforming growth factor (TGF)-ß (1:5000, Abcam), vascular endothelial growth factor (VEGF) (1:1000, Abcam, Cambridge, UK), CXCR4 (1:1000, Abcam, Cambridge, UK), stromal cell-derived factor (SDF-1α) (1:1000, Cell Signaling, Danvers, Ma, USA) and hypoxia-inducible factor (HIF)-1α (1:1000, Abcam, Cambridge, UK) for 1 hour at room temperature. Horseradish peroxidase-conjugated anti-rabbit IgG (1:2000, Cell Signaling, Danvers, Ma, USA) was used as a secondary antibody. Immuno-reactive bands were visualized by enhanced chemiluminescence (ECL; Amersham Biosciences, Little Chalfont, Buckinghamshire, England, UK) and digitized using Labwork software (UVP, Upland, CA, USA).
Oxidative Stress Reaction in LV Myocardium
Expression of oxidative stress proteins has been described by our previous reports 11,19 –21 . Oxyblot Oxidized Protein Detection Kit was purchased from Chemicon (S7150). DNPH derivatization was carried out using 6 µg of protein for 15 minutes according to the manufacturer’s instructions. One-dimensional electrophoresis was carried out on 12% SDS/polyacrylamide gel after DNPH derivatization. Proteins were transferred to nitrocellulose membranes, which were then incubated in the primary antibody solution (anti-DNP 1: 150) for 2 hours, followed by incubation in secondary antibody solution (1:300) for 1 hour at room temperature. Immuno-reactive bands were visualized by ECL (Amersham Biosciences, Little Chalfont, Buckinghamshire, England, UK) and digitized using Labwork software (UVP, Upland, CA, USA).
Histological Quantification of Myocardial Fibrosis, Infarct and Collagen Deposition Areas
This procedure and protocol were detailed in our previous report 20,21 . Briefly, hematoxylin and eosin (H & E) and Masson’s trichrome staining were used to identify the infarct area and fibrosis of LV myocardium, respectively. Three serial sections of LV myocardium in each animal were prepared at 4 µm thickness by Cryostat (Leica CM3050 S, Wetzlar, Germany). The integrated area (µm2) of infarct area and fibrosis on each section were calculated using the Image Tool 3 (IT3) image analysis software (University of Texas, Health Science Center, San Antonio, UTHSCSA; Image Tool for Windows, Version 3.0, USA). Three randomly selected high-power fields (HPFs) (100×) were analyzed in each section. After determining the number of pixels in each infarct and fibrotic area per HPF, the numbers of pixels obtained from three HPFs were calculated. The procedure was repeated in two other sections for each animal. The mean pixel number per HPF for each animal was then determined by calculating all pixel numbers and dividing by 9. The mean integrated area (µm2) of fibrosis in LV myocardium per HPF was obtained using a conversion factor of 19.24 (since 1 µm2 represented 19.24 pixels).
To analyze the extent of collagen synthesis and deposition, cardiac paraffin sections (6 µm) were stained with picrosirius red (1% Sirius red in saturated picric acid solution) for one hour at room temperature. The sections were then washed twice with 0.5% acetic acid. After dehydration in 100% ethanol, the sections were cleaned with xylene and mounted in a resinous medium. High power fields (100×) of each section were used to identify Sirius red-positive areas in each section. Analysis of the collagen deposition area in LV myocardium was performed analogously.
Immunofluorescent (IF) Staining
The procedure and protocol have been described by our previous reports 11,19 –21 . Re-hydrated paraffin sections were treated with 3% H2O2 for 30 minutes and incubated with Immuno-Block reagent (BioSB, Santa Barbara, CA, USA) for 30 minutes at room temperature. Sections were then incubated with primary antibodies CD31 (1:200, BD Biosciences, East Rutherford, NJ, USA), vWF (1:200, Merck Millipore, Darmstadt, Germany) and CXCR4 (1:200, Thermo, MA, USA). Three sections of LV myocardium from each rat were analyzed. For quantification, three randomly selected HPFs (200× or 400× for IHC and IF studies) were analyzed in each section. The mean number of positively-stained cells per HPF for each animal was determined by summation of all numbers divided by 9.
Model of Left Common Carotid Artery Injury
The procedure and protocol were as described in our previous report 22,23 with minimal modification. Pathogen-free, adult male Sprague-Dawley (SD) rats, weighing around 350 g (Charles River Technology, BioLASCO, Taiwan) were utilized in the present study. All animals were anesthetized by inhalational of 2.0% isoflurane, placed in a supine position on a warming pad at 37°C for isolation of the left external and common carotid arteries in the left neck area. Under flow control with vascular clamps, a small opening was created over the proximal left external carotid artery with a scalpel after adequate exposure in sterile conditions. A coronary angioplasty wire with a diameter of 0.014 inches was used to pass through the small orifice and advance into the left common carotid artery (LCA) and distal to the aorta. A 1.5 × 15 mm angioplasty balloon along the wire was then gently pushed forward and inflated, followed by dilatation within the arteries. This method reliably produced carotid artery endothelial denudation. Each external carotid artery was then ligated at the level of the orifice, followed by closure of the muscle and skin layers.
Carotid-Ring Co-Culture with EPCs and Method for Determining Rat Carotid-Ring Angiogenesis
In detail, by day 5 after the LCA balloon denudation, the harvested carotid rings were utilized for culture with and without EPCs. Carotid-ring angiogenesis assay was conducted in twenty-four-well tissue culture plates embedded with 150 µl of 1 mg/ml type I collagen (BD Biosciences, Franklin Lakes, NJ, USA) and allowed to gel for 60 minutes at 37°C and 5% CO2. The carotid was excised from 6-rats/each group then all extraneous tissue and branching vessels were removed with forceps and a scalpel. The aorta was cut into 1 mm cross-sections, placed on collagen-coated wells then filled with 500 µl of serum free MCDB131 medium. These rings were incubated at 37°C and 5% CO2 for 5 days and photographed with 12.5× magnification. The number and length of sprouting vessels were quantified by OLYMPUS DP72 software (Olympus, Shinjuku, Tokyo, Japan). Experiments were repeated twice.
Assessment of HUVECs Migratory Ability Using Transwell Migratory Assay
The protocol has been described in our previous studies 22 . In brief, to examine the Transwell migratory ability of HUVECs, the Transwell membranes (5 μm; Costar, Germany) were coated on both sides with fibronectin (2.5 μg/mL; Roche, Mannheim, Germany) overnight at 4°C. HUVECs (5.0 × 105 cells/well) were resuspended in M199 medium (Gibco) containing 0.5% FBS (Gibco) and incubated in the upper chamber at 37°C in 5% CO2 whereas the bottom of the chamber contained with or without EPCs (3.0 × 105 cells/well). The HUVECs were allowed to migrate for 4, 8, and 16 hours toward the lower chamber which was filled with M199 containing 20% FBS. Cells remaining on the upper surface of the Transwell membranes were mechanically removed and cells that had migrated to the lower surface were fixed with 4% formaldehyde. For cell quantification, nuclei of the migrated cells were stained with DAPI. Cells migrating into the lower chamber were counted in 5 random microscopic fields using a fluorescence microscope (Olympus, Shinjuku, Tokyo, Japan) with the software Image-Pro Plus (Media Cybernetics, Rockville, MD, USA).
Matrigel Assay for Evaluating Angiogenesis
The procedure and protocol have been described in our previous report 22 . After cell culture in Transwell (i.e., upper chamber with or without EPCs and the bottom of the chamber with HUVECs), the HUVECs were collected for angiogenesis in Matrigel. In details, the HUVECs were then plated in 96-well plates at 1.0 × 104 cells/well in 150 µL serum-free M199 culture medium mixed with 50 µL cold Matrigel (Chemicon international, Inc. Temecula, CA, USA) for 24 hours incubated at 37°C in 5% CO2. Three random microscopic images (200×) were taken from each well to count cluster, tube, and network formations and the mean values were obtained. Both cumulative and mean tube lengths were calculated by Image-Pro Plus software (Media Cybernetics, Bethesda, MD, USA).
Statistical Analyses
Quantitative data are expressed as mean ± SD. Statistical analysis was performed by ANOVA followed by Bonferroni multiple-comparison post hoc test. All analyses were conducted using SAS statistical software for Windows version 8.2 (SAS institute, Cary, NC). A probability value <.05 was considered statistically significant.
Results
In Vitro and Exo Vivo Studies for Determining the Angiogenesis (Fig. 1)
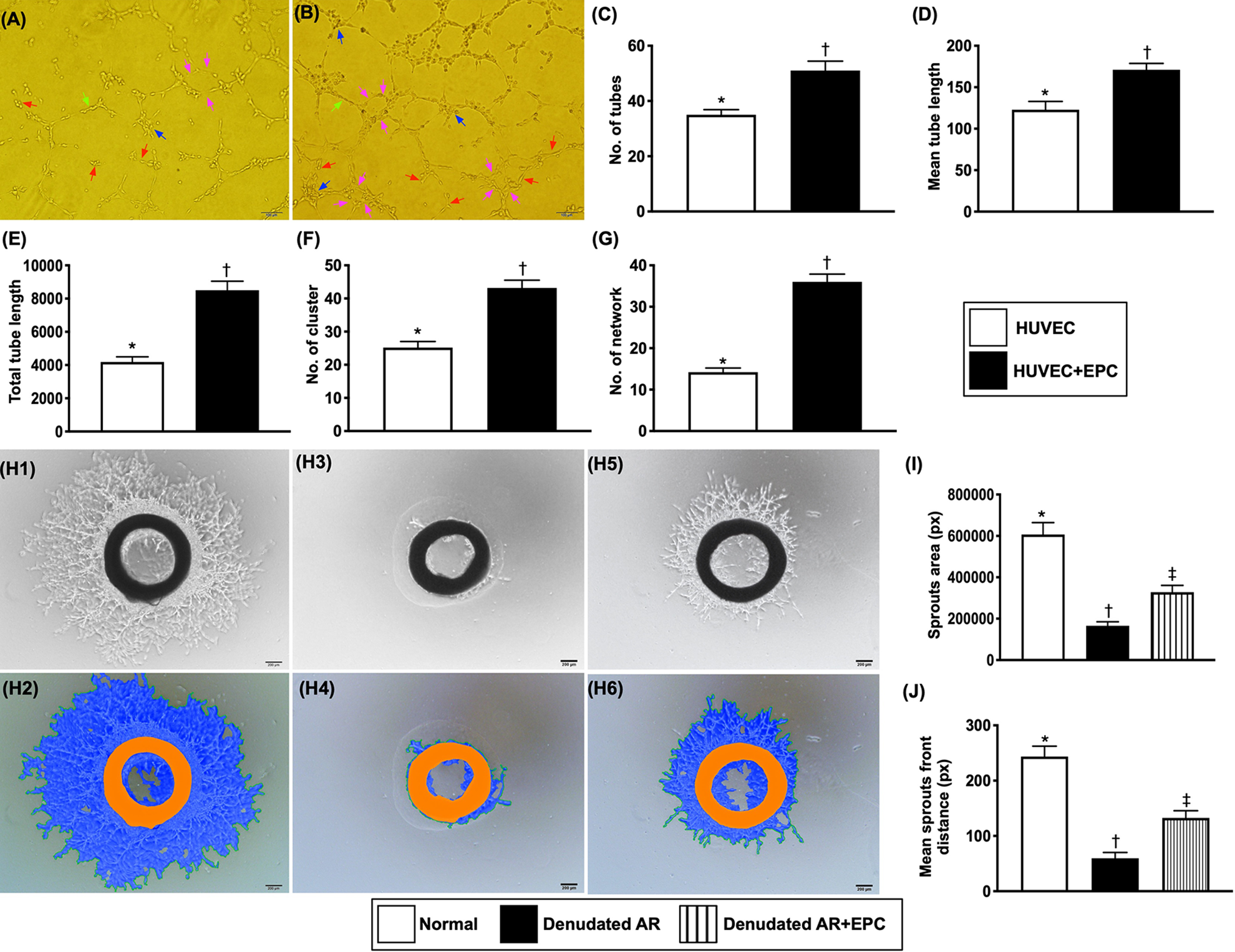
In vitro and exo vivo assessment for determining the angiogenesis (A to B) showing the in vitro study of Matrigel assay for evaluation of angiogenesis of HUVECs without (A) and with (B) EPC-secreted medium. (C) Number of tubules (red arrows), * vs. †,
To elucidate whether EPC’s secretion substance would enhance angiogenesis, the EPCs in upper compartment and HUVECs in bottom of the Transwell were co-cultured fore 24 h, followed by collected the HUVECs for Matrigel assay. The result showed that the angiogenesis capacity was significantly higher in HUVECs with than in without EPC co-culture.
Next, to assess the role of EPCs on the healing of intimal layer and ex vivo angiogenesis capability, the EPCs was co-culture with endothelial denudation of rat carotid artery. The results displayed again the healing of intimal layer and angiogenesis capability of carotid artery was significantly higher in carotid ring with than in without EPC co-culture.
Time Courses of Migratory Ability of HUVECs with and without EPC’s Secretory Medium Treatment (Fig. 2)
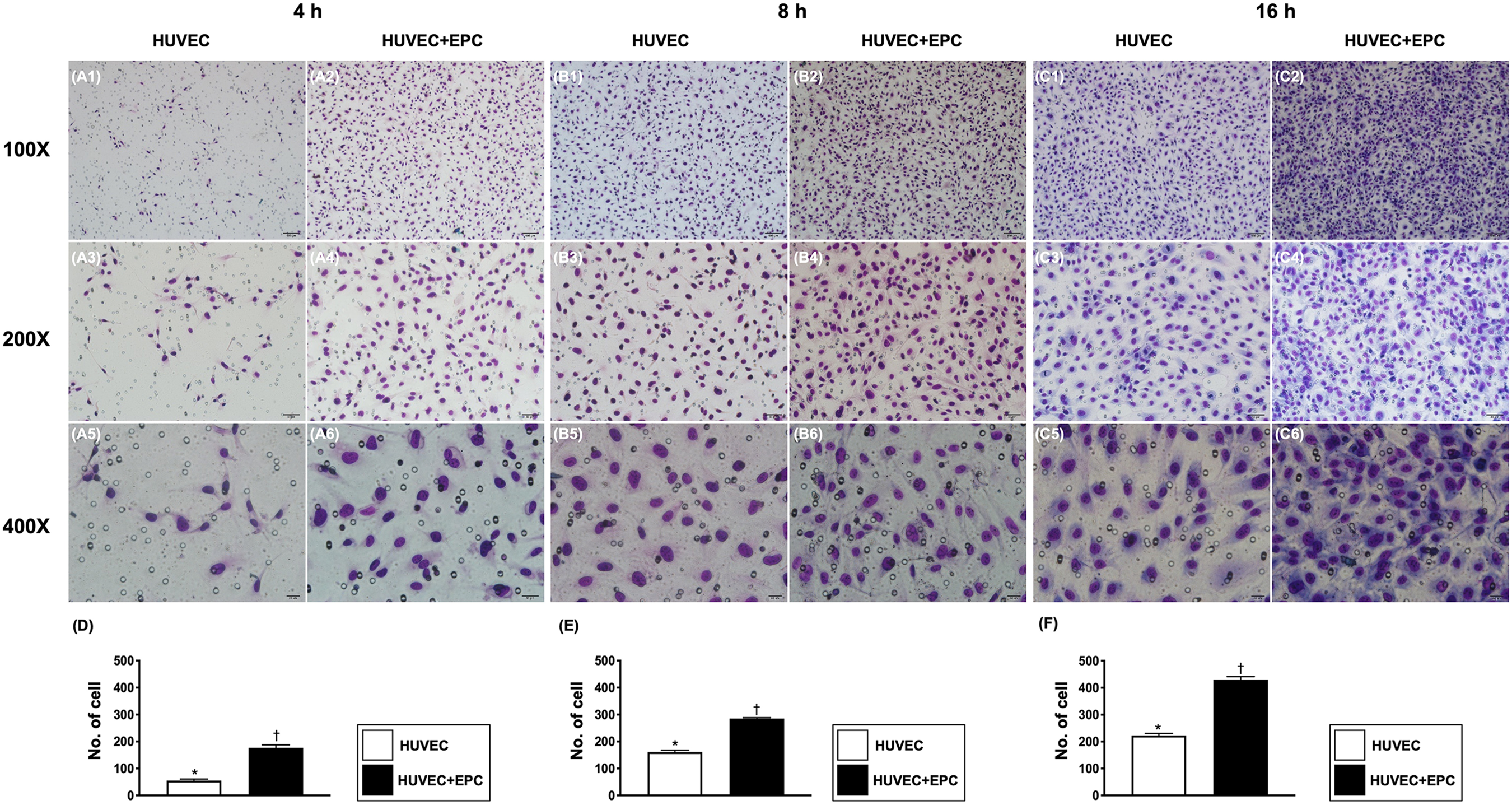
Time courses of migratory activity of HUVECs undergoing with and without EPC’s secretory medium treatment (A1 to C6) illustrating the migratory activity of HUVECs at the time intervals of 4 h (A1 to A6), 8 h (B1 to B6), and 16 h (C1 to C6) hours, respectively. (D to E) Analytical result of migratory activity of HUVECs (i.e., measured by count per field) at time interval of 4 h, 8 h, and 16 h, * vs. †,
On the other hand, for determining the impact of EPCs on HUVEC migratory ability, the culture method now was reversed (i.e., the HUVECs in the upper and the EPCs in the lower compartment of Transwell). The result demonstrated that the migratory ability in the time pints of 4, 8, and 16 hours was significantly higher in HUVECs with than in without EPC co-culture.
Based on the results of Figs. 1 and 2, we encouraged us to perform an animal model of LV myocardial IR injury to test our hypothesis, that is, the therapeutic potential of EPCs/CM therapy on preserving the LVEF and protecting the LV myocardium against IR injury.
Transthoracic echocardiographic findings at baseline and day 3, and Infarct and fibrotic areas of LV myocardium by day 3 after IR procedure (Fig. 3)
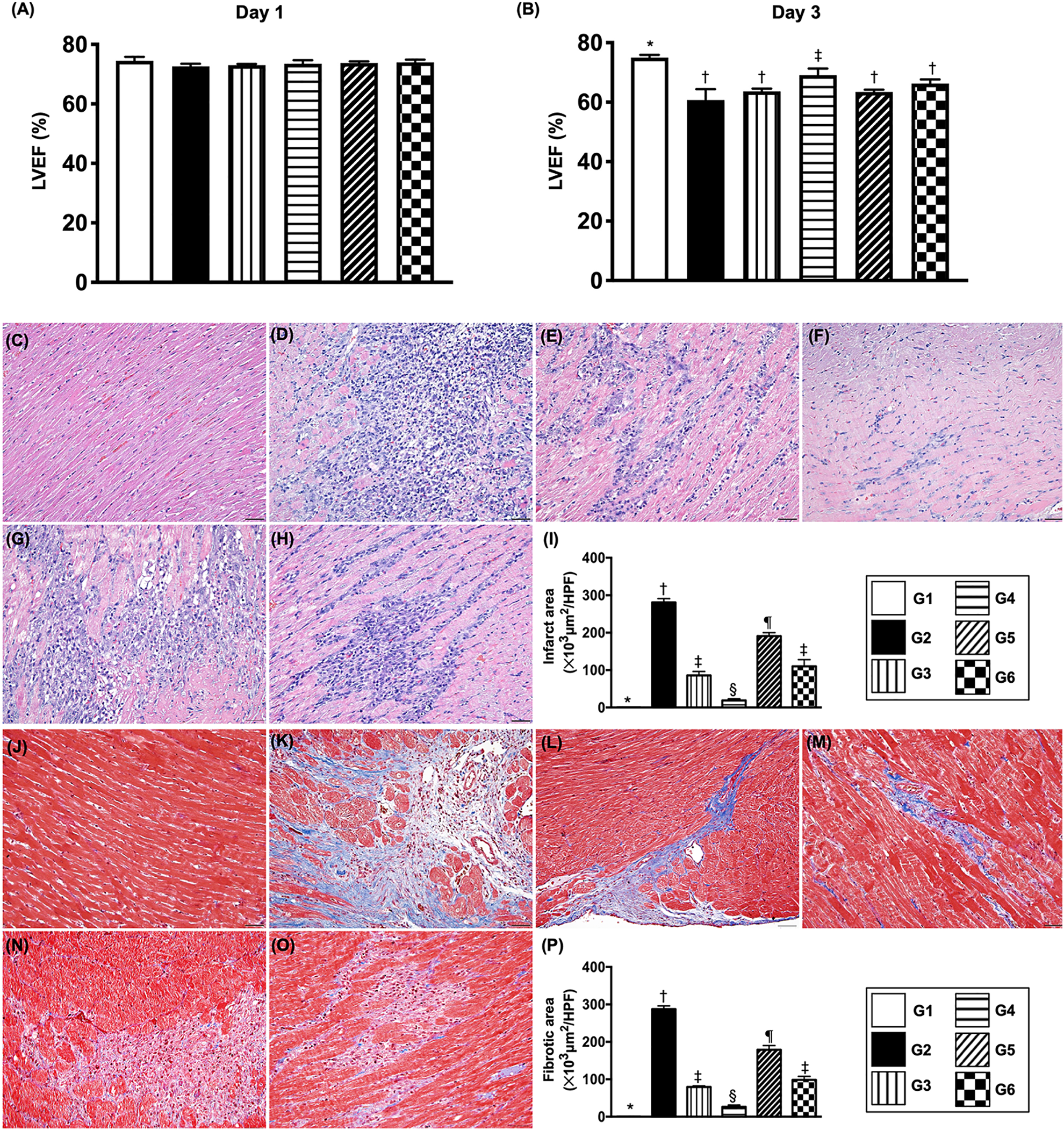
Time points of LVEF and myocardial infarct and fibrotic areas by day 3 after IR induction. (A) Left ventricular ejection fraction (LVEF) at day 0,
By day 0, the L LVEF did not differ among the six groups. However, as our expected, by day 3 after IR procedure, the LVEF was highest in group 1 (i.e., SC), lowest in group 2 [i.e., IR + culture-medium (3.3 cc)/administered at 3 h after IR procedure)], significantly higher in group 4 (i.e., IR + EPCs/administered at 3 and 24 h) than in groups 3 (i.e., IR + EPCs/administered at 3 h), 5 (i.e., IR +CM/administered at 3 h) and 6 (i.e., IR + CM/administered at 3 and 24 h) and significantly higher in group 6 than groups 3 and 5, but it showed no difference between groups 3 and 5.
H&E staining demonstrated that the infarct area of LV myocardium was lowest in group 1, highest in group 2, significantly lower in group 4 than in groups 3, 5 and 6, and significantly lower in group 6 than groups 3 and 5, but this parameter showed no difference between groups 3 and 5. Similarly, Masson’s trichrome staining displayed that the fibrotic area was identical to the infarct area among the six groups.
Collagen-Deposition Area and Number of Small Vessels in Infarct Area of LV Myocardium by Day 3 After IR Procedure (Fig. 4)
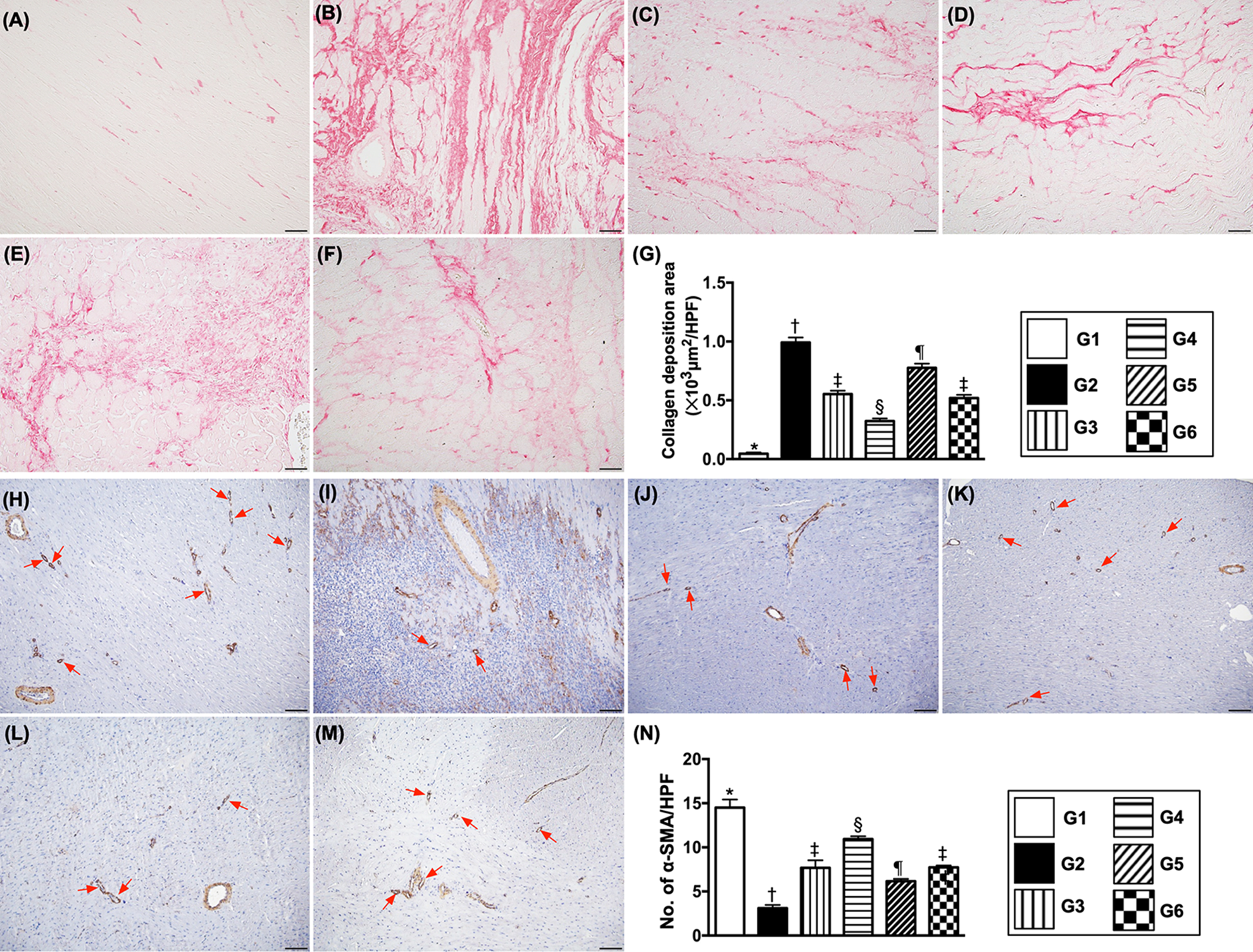
Collagen-deposition area and small vessel density in left ventricular (LV) myocardium by day 3 after IR induction (A to F) illustrating Sirius red stain (200×) for identification of collagen-deposition area (pink color). Scale bars in the right lower corner represent 50 µm. (G) Analytical result, * vs. other groups with different symbols (†, ‡, §, ¶),
Immunofluorescent staining allowed the impact of EPC/MC therapy on reducing the collagen-deposition area and enhancing angiogenesis/vasculogenesis within infarcted LV myocardium to be assessed. Sirius red staining demonstrated that collagen-deposition in LV myocardium was lowest in group 1, highest in group 2, significantly lower in group 4 than in groups 3, 5, and 6, and significantly lower in groups 3 and 5 than in group 6, but this parameter showed no difference between groups 3 and 5. The number of small vessels in LV myocardium exhibited an opposite pattern of collagen-deposition area among the five groups.
Expression of Endothelial Cell Surface Markers in LV Myocardium by Day 3 After IR Procedure (Fig. 5)
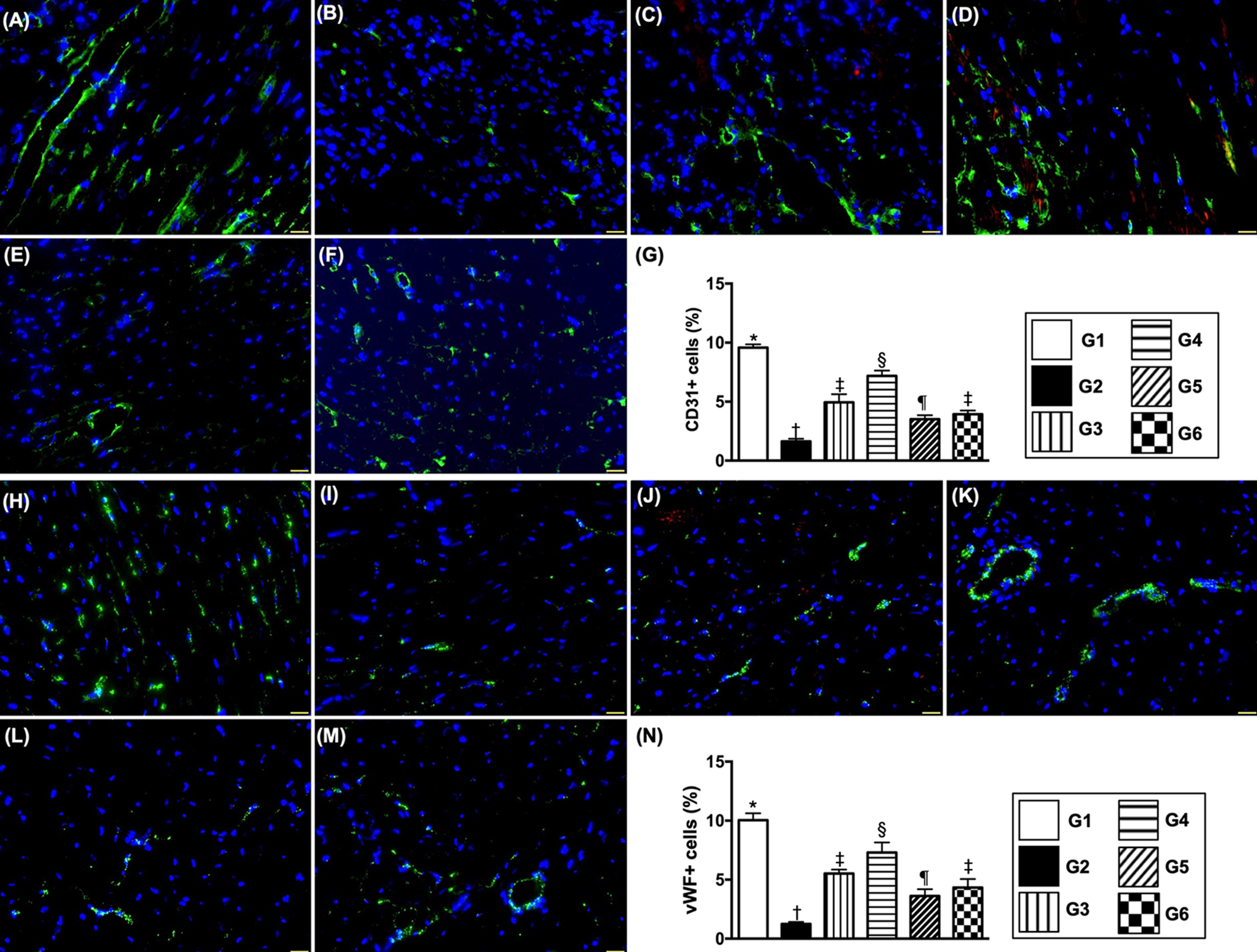
Endothelial cell surface markers in LV myocardium by day 3 after IR induction (A to F) Illustrating immunofluorescent (IF) microscopic finding (400×) for identification of cellular expressions of CD31 (green color). (G) Analytical result of number of CD31+ cells, * vs. other groups with different symbols (†, ‡, §, ¶),
The numbers of CD31+ and vWF+ cells, two indicators of endothelial cell integrity, were highest in group 1, lowest in group 2, significantly higher in group 4 than in groups 3, 5, and 6 and significantly higher in group 3 than in groups 5 and 6, but they showed no differences between groups 5 and 6.
Protein Expressions of Endothelial Cell Biomarkers and Mitochondrial Cytochrome C in LV Myocardium by Day 3 After IR Procedure (Fig. 6)
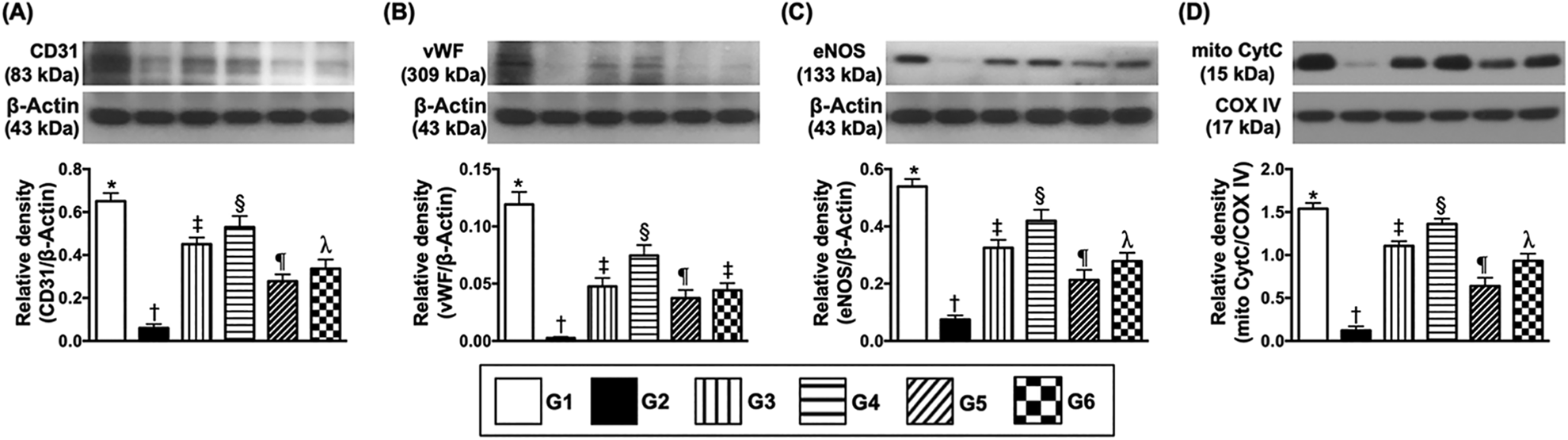
Protein expressions of endothelial cell biomarkers and mitochondrial cytochrome C in LV myocardium by day 3 after IR procedure, (A) Protein expression of CD31, * vs. other groups with different symbols (†, ‡, §, ¶, λ),
The protein expressions of CD31 and vWF, two indices of endothelial cell/angiogenesis biomarkers and mitochondrial cytochrome C, an indicator of mitochondrial integrity, were highest in group 1, lowest in group 2, significantly higher in group 4 than groups 2, 5, and 6, significantly higher in group 3 than in groups 5 and 6 and significantly higher in group 6 than in group 5.
The protein expression of eNOS, another indicator of endothelial cell/angiogenesis biomarker, was highest in group 1, lowest in group 2, significantly higher in group 4 than in groups 3, 5, and 6, significantly higher in groups 3 and 6 than in group 5 and significantly higher in group 6 than in group 3.
Protein expressions of apoptotic, fibrotic mitochondrial-damage and pressure-overload/heart failure biomarkers in LV myocardium by day 3 after IR procedure (Fig. 7)
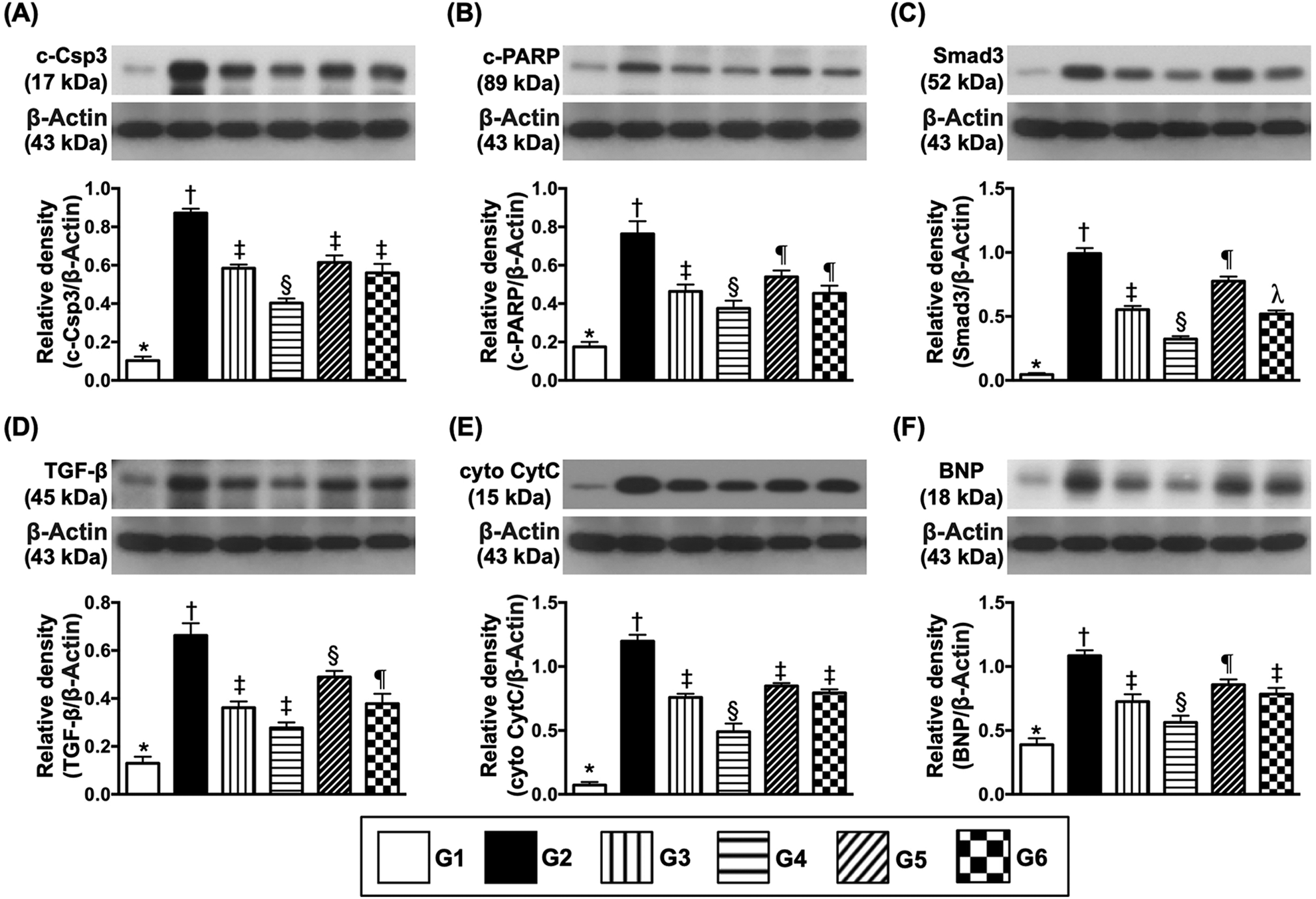
Protein expressions of apoptotic, fibrotic, mitochondrial-damaged, and pressure overload/heart failed biomarkers in LV myocardium by day 3 after IR induction. (A) Protein expressions of cleaved caspase 3 (c-Csp3), * vs. other groups with different symbols (†, ‡, §, ¶),
The protein expressions of cleaved caspase 3 and cleaved PARP, two markers of apoptosis and protein expressions of Smad3 and TGF-ß, two fibrotic markers, were lowest in group 1, highest in group 2, significantly lower in group 4 than in groups 3, 5, and 6, and significantly lower in group 3 and 6 than in group 5, but they showed no differences between groups 3 and 6. Similarly, the protein expression of cytosolic cytochrome C, an indicator of mitochondrial damage and protein expression of BNP, an indicator of pressure overload/heart failure biomarker, exhibited an identical pattern to apoptosis among the six groups.
Protein Expressions of Oxidative Stress and Autophagic Biomarkers in LV Myocardium by Day 3 After IR Procedure (Fig. 8)
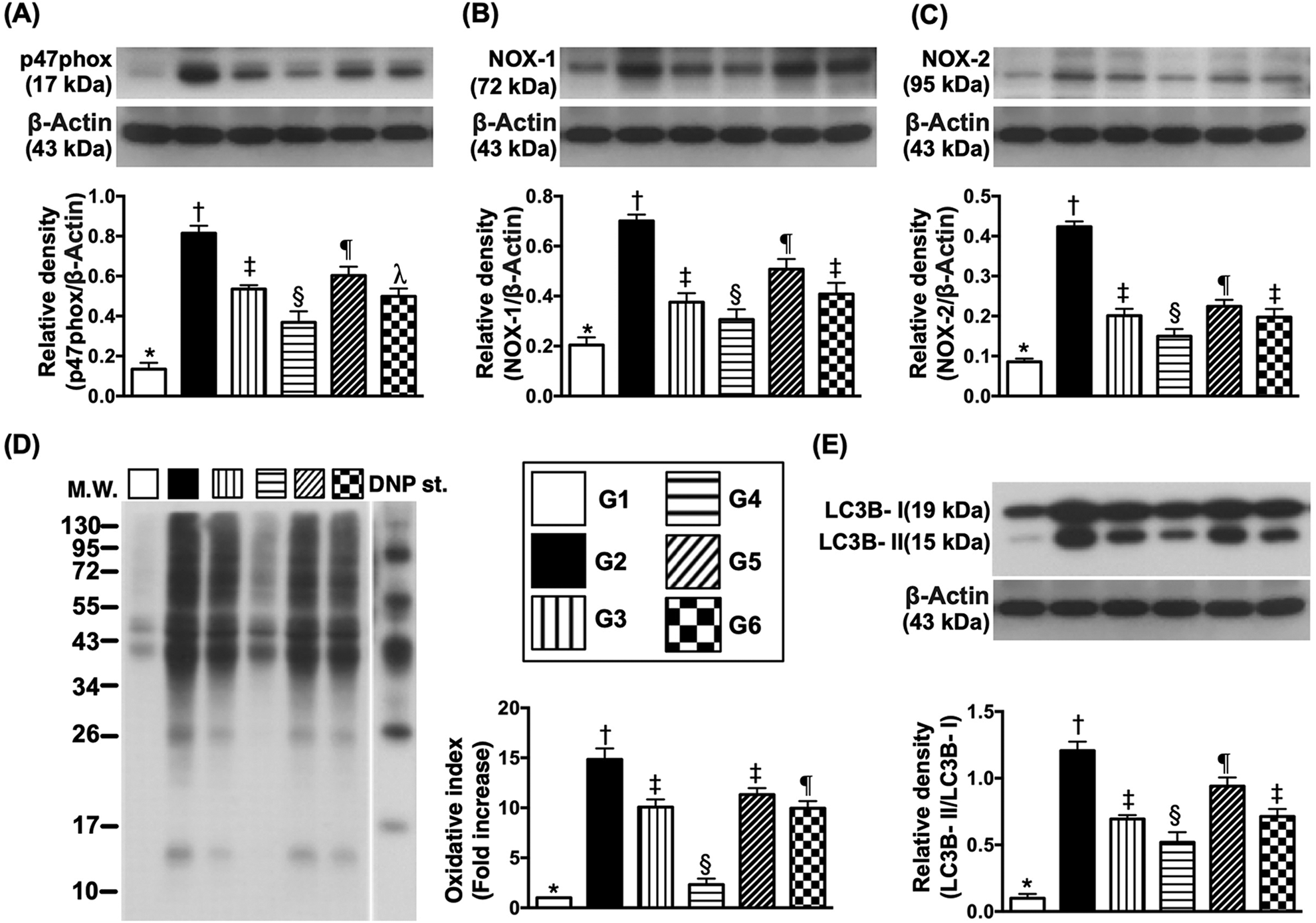
Protein expressions of oxidative-stress and autophagic biomarkers in LV myocardium by day 3 after IR induction (A) Protein expression of p47phox, * vs. other groups with different symbols (†, ‡, §, ¶),
The protein expressions of p47phox, NOX-1, NOX-2, and oxidized protein, four indices of oxidative stress and ratio of LC3B-II/LC3B-I, an indicator of autophagic biomarker, were lowest in group 1, highest in group 2, significantly lower in group 4 than in groups 3, 5, and 6, and significantly lower in group 3 and 6 than in group 5, but not different between groups 3 and 6.
Protein Expressions of Angiogenesis Markers in LV Myocardium by Day 3 After IR Procedure (Fig. 9)
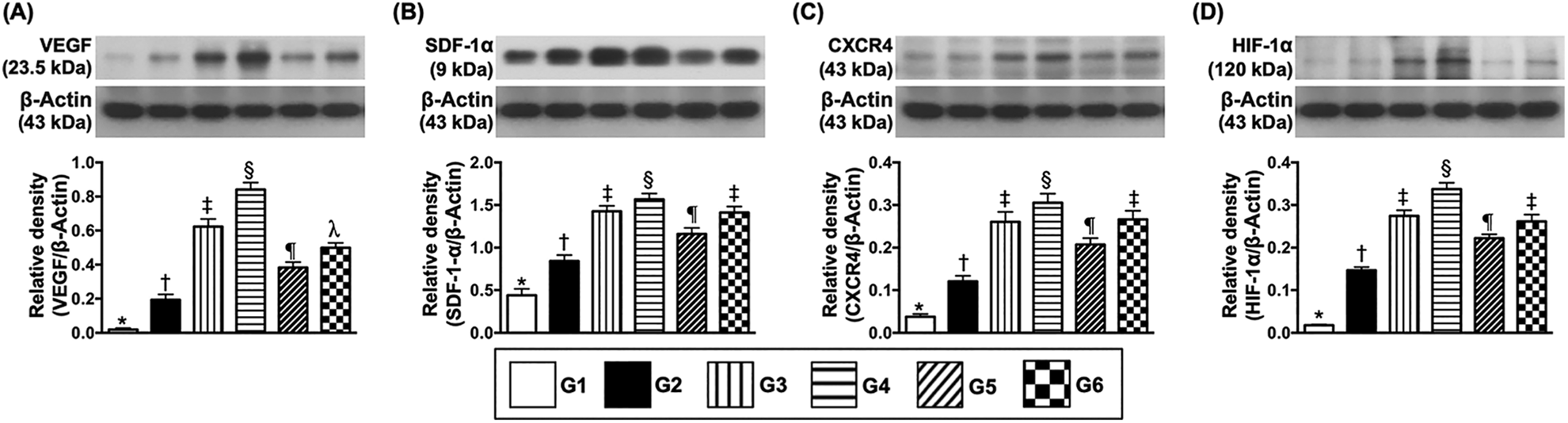
Protein expressions of angiogenesis factors in LV myocardium by day 3 after IR induction. (A) Protein expression of vascular endothelial growth factor (VEGF), * vs. other groups with different symbols (†, ‡, §, ¶, λ),
The protein expressions of VEGF, SDF-1α, CXCR4, and HIF-α, four indices of angiogenesis biomarkers, were lowest in group 1, highest in group 4, significantly higher in groups 3 and 6 than in groups 2 and 5, and significantly higher in group 5 than in group 2 but they showed no differences in groups 3 and 6.
Discussion
This study investigated the therapeutic impact of single and double doses of EPC/EPC-derived CM on heart function in the setting of acute myocardial IR injury in rodents and yielded several clinically relevant implications. First, the therapeutic effect of EPC-derived CM on preservation of heart function was comparable with that of EPCs. Second, providing two consecutive doses of EPCs/EPC-derived CM was superior to the single dose regimen at improving heart function following acute myocardial IR injury in rodents, highlighting that these therapeutic regimens should be seriously considered when cell therapy is considered for tissue/organ regeneration. Third, based on the results of our in vitro and in vivo examinations, we propose that the therapeutic impact of EPCs/EPC-derived CM on protecting LV myocardium against IR injury was mainly mediated by angiogenesis (i.e., from both in vitro and in vivo results) and degradation of oxidative-stress signaling (i.e., from in vivo results).
Recent studies 24,25 have shown that EPC-derived CM which contained plentiful paracrine factors substantially increased viability of brain microvascular cells. An essential finding of our in vitro study revealed that both EPCs and EPC secresome which contained rich soluble angiogenesis factors markedly enhanced angiogenesis endothelial cell migratory ability. Additionally, the cellular and molecular level angiogenesis biomarkers in LV myocardium were substantially higher in IR animals after receiving EPC-derived CM therapy than IR animals without. Furthermore, other previous studies have shown that MSC- and EPC-derived CM therapy effectively improved ischemia-related organ dysfunction 14,15 . Intriguingly, our previous study has further shown that CM usually contained chemokine, paracrine and soluble angiogenesis factors 26 that not only nourished the cells, tissues and organs but also enhanced angiogenesis in ischemic zones, resulting in restored blood flow and improved ischemia-related LV dysfunction. Accordingly, our findings, in addition to being consistent with those of previous studies 14,15,24 –26 , help explain why EPC-derived CM therapy effectively protected LV myocardium against IR injury.
Abundant data have shown that EPC therapy ameliorated angina and heart failure and improved ischemia-related LV dysfunction 16 –18 . A principal finding in the present study was that single dose EPC therapy was better than single dose EPC-derived CM therapy for preserving LV function through better enhancement of angiogenesis at cellular and molecular levels and greater suppression of oxidative stress, autophagy, apoptosis and fibrosis of LV myocardium. Our study further verified the findings of previous studies 16 –18 .
The optimum number of doses of stem cell therapy in this setting remains unclear. Currently, only single doses of EPC or MSC are considered and applied in clinical trials so as to reduce the risk of compromising safety. However, when we assessed the results of these clinical trials 16 –18 , only 5% to 7% improvements in LVEF occurred after EPC therapy, implying that there might be room for further improvement of heart function. The distinctive finding in the present study was that two doses of EPC therapy were safe and also superior to a single dose of EPC therapy for improving heart function in this setting by augmenting angiogenesis and inhibiting oxidative stress and fibrosis. In this way, our finding, in addition to strengthening the findings of previous studies 16 –18 , provides usefully clinically relevant information and encourage to consider two consecutive doses of EPCs for patients with ischemia-related organ dysfunction, especially when they are refractory to conventional therapy.
Study Limitation
This study has limitations. First, although the outcomes were promising, the study period was relatively short; so, the long-term outcomes of two consecutive doses of EPC/EPC-derived CM therapy on improving heart function remains uncertain. Second, although extensive works were completed in the present study, the exact underlying mechanism of EPC/EPC-derived CM therapy on improving heart function might be more complicated than our findings.
In conclusion, the present study demonstrated that EPC and EPC-derived CM therapy effectively protected LV function against IR injury. Additionally, consecutive two doses of EPC/EPC-derived CM therapy were safe and also superior to a single dose for improving heart function in the setting of IR injury.
Footnotes
Authors Contribution
Investigation, Jui-Ning Yeh, Ruan-Ruan Yang, Chi-Ruei Huang, Yi-Ching Chu; Methodology, Chi-Ruei Huang, Yi-Ching Chu, and Hon-Kan Yip; Supervision, Hon-Kan Yip and Jun Guo; Writing—review & editing, Christopher Glenn Wallace and Jun Guo.
Ethical Approval
This study was approved by the Institute of Animal Care and Use Committee at Kaohsiung Chang Gung Memorial Hospital (Affidavit of Approval of Animal Use Protocol No. 2016112402).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Guide for the Care and Use of Laboratory Animals (Affidavit of Approval of Animal Use Protocol No. 2016112402).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants: (1) Chang Gung Memorial Hospital Research Project, Taiwan (NMRPG8H0221). (2) Ministry of Science and Technology, Taiwan (107-2314-B-182A-155).
