Abstract
Fabry disease is caused by a decrease in or loss of the activity of alpha-galactosidase, which causes its substrates globotriaosylceramide (Gb3) and globotriaosylsphingosine (lyso-Gb3) to accumulate in cells throughout the body. This accumulation results in progressive kidney injury due to glomerulosclerosis and in heart failure due to hypertrophy. Enzyme replacement therapy (ERT) has been used as the standard therapy for Fabry disease, but it causes a significant financial burden, and regular administration is inconvenient for patients. Because of the short half-life of alpha-galactosidase in vivo, therapeutic methods that can supplement or replace ERT are expected to involve continuous release of alpha-galactosidase, even at low doses. Cell transplantation therapy is one of these methods; however, its use has been hindered by the short-term survival of transplanted cells. CellSaic technology, which utilizes cell spheroids that form after cells are seeded simultaneously with a recombinant collagen peptide scaffold called a μ-piece, has been used to improve cell survival upon implantation. In this study, syngeneic murine embryonic fibroblasts were used to generate CellSaic that were transplanted into Fabry mice. These spheroids survived for 28 days in the renal subcapsular space with forming blood vessels. These results indicate CellSaic technology could be a platform to promote cellular graft survival and may facilitate the development of cell transplantation methods for lysosomal diseases.
Keywords
Introduction
Fabry disease, considered a representative lysosomal disease, is a genetic disease caused by deficient enzymatic activity of alpha-galactosidase, which is encoded by the GLA gene on the X chromosome. Due to this deficiency, glycosphingolipids such as globotriaosylceramide (Gb3) and globotriaosylsphingosine (lyso-Gb3), which are substrates for alpha-galactosidase, progressively accumulate in intracellular lysosomes and body fluids. As a result, various clinical manifestations such as cardiomyopathy can occur, resulting in heart failure and renal failure, the latter of which requires hemodialysis or kidney transplantation and contributes to the short life expectancy of 58.2 years among males with Fabry disease 1 . At present, a standard treatment for Fabry disease is enzyme replacement therapy (ERT) 2,3 . Although ERT initiated in the early phase of cardiac 4 or renal damage 5 can effectively prevent disease progression, ERT cannot reverse pathophysiology in patients in whom either cardiac hypertrophy or reductions in glomerular filtration rate have already manifested. The economic burden and inconvenience of biweekly outpatient visits are disadvantages of ERT, and a novel therapeutic strategy is needed. Recently, the oral drug migalastat, which functions as a chaperone of specific mutant enzymes that are amenable to therapy, was found to demonstrate effects similar to those of intravenous ERT 6 . Migalastat stabilizes a specific mutant form of an enzyme that is trafficked to lysosomes and hydrolyzes substances 7,8 . It is estimated that 35%–50% of patients with Fabry disease have amenable mutations based on the Sakuraba database of Fabry disease (http://fabry-database.org/).
Physiologically, lysosomal enzymes to be ubiquitously expressed are cotranslationally glycosylated in the rough endoplasmic reticulum, acquire mannose-6-phosphate (M6P) in the cis-Golgi, and are sequentially entrapped by M6P receptors on budding vesicles from the trans-Golgi to the vesicle, resulting in the dissociation of M6P receptors following fusions with endosomes to generate acidic lysosomes where lysosomal enzymes function to hydrolyze their substrates 9 . In the trans-Golgi, in part, oligosaccharides on lysosomal enzymes are remodeled with the removal of mannose and the supplementation of galactose and sialic acids instead of the phosphorylation of mannose, resulting in the secretion of the enzymes without M6P. On the other hand, M6P receptors on plasma membranes facilitate the internalization of extracellular lysosomal enzymes with M6P, which is the underpinning for ERT. Stem-cell-based cell and gene therapies for lysosomal diseases have been thoroughly investigated in animal models, and some clinical trials have been performed based on the assumption that transplanted cells could release lysosomal enzymes into the circulation 10 . Although hematopoietic stem cell transplantation (HSC Tx) offers a successful outcome in terms of life expectancy for Hurler’s syndrome, which is the most severe form of type-I mucopolysaccharidosis (MPS I) 11 , other lysosomal diseases, such as type-II mucopolysaccharidosis (MPS II, Hunter syndrome), type-III mucopolysaccharidosis (MPS III, Sanfilippo syndrome), and Batten disease, do not respond to HSC Tx 12 . Moreover, allogeneic HSC Tx requires myeloablative preconditioning, which is closely associated with perioperative mortality and morbidity. The discovery that these drawbacks can be overcome by ex vivo gene therapy using autologous HSCs with a minimum preconditioning regimen has stimulated interest in the field 13 and leading to the first clinical trial of ex vivo gene therapy for Fabry disease using lentiviruses and autologous CD34+ cells was launched 14 . Preclinical studies of the ex vivo gene therapy indicated that alpha-galactosidase secreted from genetically modified cells into circulation can be an alternative source of recombinant lysosomal enzymes in ERT.
Spheroids, which are cellular aggregates that naturally occur under cell culture conditions, have been intensively investigated in the field of cancer research 15 . Spheroids have allowed the study of developmental mechanisms 16 and have been utilized as materials for tissue engineering 17 . However, because access to nutrition and oxygen for cell survival and growth depend upon diffusion, there is a physical limitation to spheroid size: oxygen cannot reach the center of spheroids greater than 500 µm in diameter, and the absence of oxygen results in central necrosis 18 . This limitation can be overcome by utilizing a scaffolding material such as a recombinant collagen peptide called the µ-piece. This material is manufactured by artificial sequential linkage of a series of three identical fragments containing multiple Arg-Gly-Asp (RGD) motifs. A mixture of cells and a µ-piece easily forms a hollow aggregate in a nonadherent culture dish, called as CellSaic. Large numbers of cells can be integrated into a µ-piece scaffold, and such cells maintain greater viability than 2D-cultured cells 19 . CellSaic has been found to prevent central necrosis in spheroids composed of only cells for a period of time in culture; in addition, upon their transplantation into the subcutaneous tissues of mice, the spheroids survive for long periods and generate internal neovascular networks 20 .
Because CellSaic technology can introduce a massive number of cells into the body and maintain cellular grafts for a long time, we hypothesized that CellSaic technology could be an alternative to HSC Tx or ex vivo gene therapy. To examine the potential that CellSaic technology can provide a platform modality for cell transplantation to treat lysosomal diseases, methods for the mass production of CellSaics were developed, and CellSaics containing murine embryonic fibroblasts (MEFs) without any gene modifications were transplanted into a Fabry disease mouse model. The survival and function of cellular grafts were assessed by pathology and the measurement of the amounts of lyso-Gb3, a biomarker of Fabry disease, in various organs.
Materials and Methods
Animal Experiments
All experiments were performed according to the animal experiment guidelines issued by the Animal Care and Use Committee at the Kyoto Prefectural University of Medicine and approved by the Animal Experiment Ethics Committee of the Kyoto Prefectural University of Medicine (approval number M2019-305).
Cell Culture and CellSaic Stirring Culture
Bone marrow–derived mesenchymal cells in Lewis rats were kindly provided from Jichi university. This cell line was cultured in minimum essential media supplemented with 10% fetal bovine serum (FBS) and 1% penicillin and streptomycin (Thermo Fisher Scientific Inc., Waltham, MA, USA). For MEF isolation, the uteri were removed from C57BL/6 mice that were 13.5 days pregnant and washed using phosphate-buffered saline (PBS) (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). The head and visceral tissues were removed from the isolated embryos. Using two pregnant mice ranging between 18 and 20 days post-coitum, 1 to 5 × 107 cells could be acquired as primary fibroblasts to be used in further experiments. The remaining tissues were washed using fresh PBS, minced with scissors, and transferred into fresh Dulbecco’s modified Eagle medium (DMEM; FUJIFILM Wako Pure Chemical Corporation) supplemented with 10% FBS and 1% penicillin and streptomycin (Thermo Fisher Scientific, Inc.). Three days after incubation, the outgrowth cells were trypsinized, collected by centrifugation (200 × g for 5 min), and resuspended in fresh medium. Following the first passage, 1 × 106 cells were cultured in 150-mm dishes at 37°C in 5% CO2. In this study, we used MEFs within 5–10 passages. The MEFs were cultured in DMEM containing 10% FBS and 1% penicillin and streptomycin at 37°C in 5% CO2.
For CellSaic formation, rat bone marrow–derived mesenchymal cells (2 × 104 per 100 µl of culture medium) were mixed with µ-pieces (FUJIFILM Corporation) and seeded onto a PrimeSurface® 96-well U-bottom plate (MS-9096U, Sumitomo Bakelite Co., Ltd, Tokyo, Japan) to determine the optimization of the contents of µ-pieces. For mass production of CellSaics, 2-ml cell suspensions of MEFs at a density of 1 × 106/ml with 1 mg/ml of µ-pieces were poured into an Elplasia multiple-pore hanging drop microplate, which is a six-well plate with an insert holding 648 pores (MPc500, Kuraray Co., Ltd, Tokyo, Japan). The CellSaic in the microplates was cultured and then transferred into a 30-ml stirred suspension bioreactor (BWV-S03A, Able Co., Tokyo, Japan) on day 2 after seeding, after which it was cultured on a six-channel magnetic stirrer (BWS-S03N0S-6, Able Co.) at 37°C in 5% CO2 for an additional 5–7 days.
Cell Transplantation Under the Renal Capsules
Under general anesthesia using isoflurane (1.5%) (v/v air; Mylan Inc., Tokyo, Japan), C57BL/6 mice were subjected to laparotomy, and the kidneys were exposed. Incisions were made horizontally in the renal capsules at the middle of the ventral aspect. CellSaic was transferred into the renal subcapsular spaces using sterilized tweezers under a stereomicroscope (Leica S6 E, Leica Microsystems GmbH, Wetzlar, Germany). The capsular incisions were closed by a few stiches with 7-0 proline, and the abdominal wounds were closed with two layers.
Tissue Preparation and Alpha-Galactosidase Activity Measurement
Mice were anesthetized and sacrificed 28 days after cell transplantation, and all tissues (liver, kidneys, and heart) were removed. The organs were assessed for alpha-galactosidase activity and Gb3 and lyso-Gb3 levels and were subjected to histological analysis. For determination of alpha-galactosidase activity, samples were homogenized in water and subjected to five freeze–thaw cycles using liquid nitrogen. The supernatant was collected after centrifugation of the homogenate at 10,000 × g for 5 min and was used for the enzyme assays. The protein concentration was measured with a BCA protein assay kit (Thermo Fisher Scientific Inc.) according to the manufacturer’s manual. Ten microliters of cell lysate at a concentration of 50 mg/ml was assayed with 20 µl of 4-methylumbelliferyl-alpha-
Histology and Immunohistochemistry
Tissue samples were fixed with 4% paraformaldehyde (FUJIFILM Wako Pure Chemical Corporation) and embedded in paraffin. Hematoxylin and eosin (HE) staining was performed according to standard procedures. We performed morphological analysis of the grafts and the adjacent blood vessels in the HE-stained sections under light microscopy. Apoptosis detection was performed using an Apoptag apoptosis detection kit (S7100, Merck Millipore, Billerica, MA, USA) for terminal deoxynucleotidyl transferase 2′-deoxyuridine 5′-triphosphate (dUTP) nick end labeling (TUNEL) according to the manufacturer’s instructions.
Statistical Analysis
The results are expressed as the means ± standard errors. The statistical significance of differences among groups was evaluated using t-tests and standard Bonferroni correction (P = 0.01) (Prism 8 software, GraphPad Prism Software Inc., San Diego, CA, USA), and P < 0.05 was considered to indicate significance.
Results
Mass Production of CellSaic
CellSaic technology is a new cell aggregation technology that uses recombinant peptide pieces called µ-pieces for cell transplantation 19 . The mass of µ-piece used to form CellSaic with rat bone marrow–derived mesenchymal cells was optimized by using PrimeSurface® 96-well U-bottom plate (Fig. 1). In view of the uniform distribution of cells from the periphery to the center of CellSaics, a mass of µ-pieces of 20 µg per CellSaic was chosen for further experiments. Large numbers of CellSaics are required for cell transplantation to modulate physiological functioning via the release of lysosomal enzymes from donor cells. Therefore, to facilitate the production of large numbers of CellSaics, we designed a CellSaic mass production system using a sphere-forming vessel and a stirring incubator (Fig. 2). Large numbers of CellSaics were produced by seeding 2 × 106 MEFs plus 2 mg of μ-pieces per well on Elplasia multiple-pore hanging drop microplates. The sphere-forming vessel requires only a one-time pouring of suspension solutions consisting of cells and μ-pieces and drastically reduced the labor required to inject them several hundred times even by using a multipipetter. The culture device limits the time necessary to provide compaction, which is known as the maturation process in spheroid culture, because of the restricted volume of the medium in which the spheroid is soaked. The CellSaics that were formed in a short duration in hanging drop culture were so fragile and did not fulfil the prerequisite that CellSaics have to be physiologically very rigid to tolerate a surgical transplantation procedure. Subsequent stirring culture in the spinner bottle for 5–7 days enabled compaction, which was observed as the downsizing of CellSaics (Fig. 3A). The process was designed as 2 days for hanging drop culture and 5 days for maturation culture; therefore, it took 7 days to complete the process to acquire CellSaic mass production. This production process could be easily expanded several hundred times in the number of CellSaics. From one 6-well plate, 3888 CellSaics containing 3 × 103 cells could be acquired without labor-intensive work. Just after the formation of the spheroids, the histology of CellSaics showed uneven distribution of cells and μ-pieces with sparse density in the center, whereas single-cell aggregates showed compact spheroids with rich density even in the center (Fig. 3B). After spheroids and CellSaic were cultivated for 1 week, they were histologically examined by counting TUNEL-positive cells in each mass had a diameter that ranged from a mean ± 2 standard errors. The TUNEL-positive ratio in spheroids was significantly higher than that in CellSaic (Supplemental Fig. 1). TUNEL staining revealed significant apoptotic cells in single-cell aggregates and almost no apoptotic cells in CellSaics (Fig. 3C). Histological examination following the whole process revealed that following maturation, the CellSaic possessed a uniform cell distribution regardless of the distance from the surface, whereas spheroids without μ-pieces had centers devoid of cells that were surrounded by viable cell rims (Fig. 3C). The cell rims of the spheroids without μ-pieces showed a gradual change from the periphery to the center. The outermost layer was composed of large cells, and the inner portion was formed by smaller cells, with a lower density in the center (Fig. 3C). To evaluate the apoptotic cells inside the cell aggregates with or without µ-pieces, we used a TUNEL assay, a method for detecting apoptotic DNA fragmentation that is widely used to identify and quantify apoptotic cells (Fig. 3C). There were significant numbers of TUNEL-positive cells in the central parts of the spheroids without µ-pieces, whereas far fewer TUNEL-positive cells were found in the centers of the CellSaic structures. The size of the CellSaic structure that could be made by this method was 300–600 µm (78.4%) (Fig. 3D). The activities of alpha-galactosidase in 3D-cultured cells with and without μ-piece were significantly higher than that in 2D-cultured cells (Fig. 3E). The decreased alpha-galactosidase activity in CellSaic might be attributed to the protein content of the µ-piece. Next, alpha-galactosidase activities in 2 ml of the culture medium of CellSaics containing MEFs at a total cell number of 2 × 106 for 48 h were examined. Because the enzyme was contained in FBS, the activity of alpha-galactosidase to be released by MEFs in CellSaics was approximately 2 nmol/h/ml, which was calculated by subtracting the activity in the medium alone (Fig. 3F).

Characteristics of CellSaics HE staining of cell spheres (without µ-pieces) and CellSaics (with µ-pieces) using rat bone marrow–derived mesenchymal cells. These cell structures were cultured in DMEM (10% FBS) for 2 days. The black bars indicate 50 µm. Cells without µ-pieces (A) and with 5 µg/ml (B), 10 µg/ml (C), and 20 µg/ml (D) µ-pieces. DMEM: Dulbecco’s modified Eagle medium; FBS: fetal bovine serum; HE: hematoxylin and eosin.
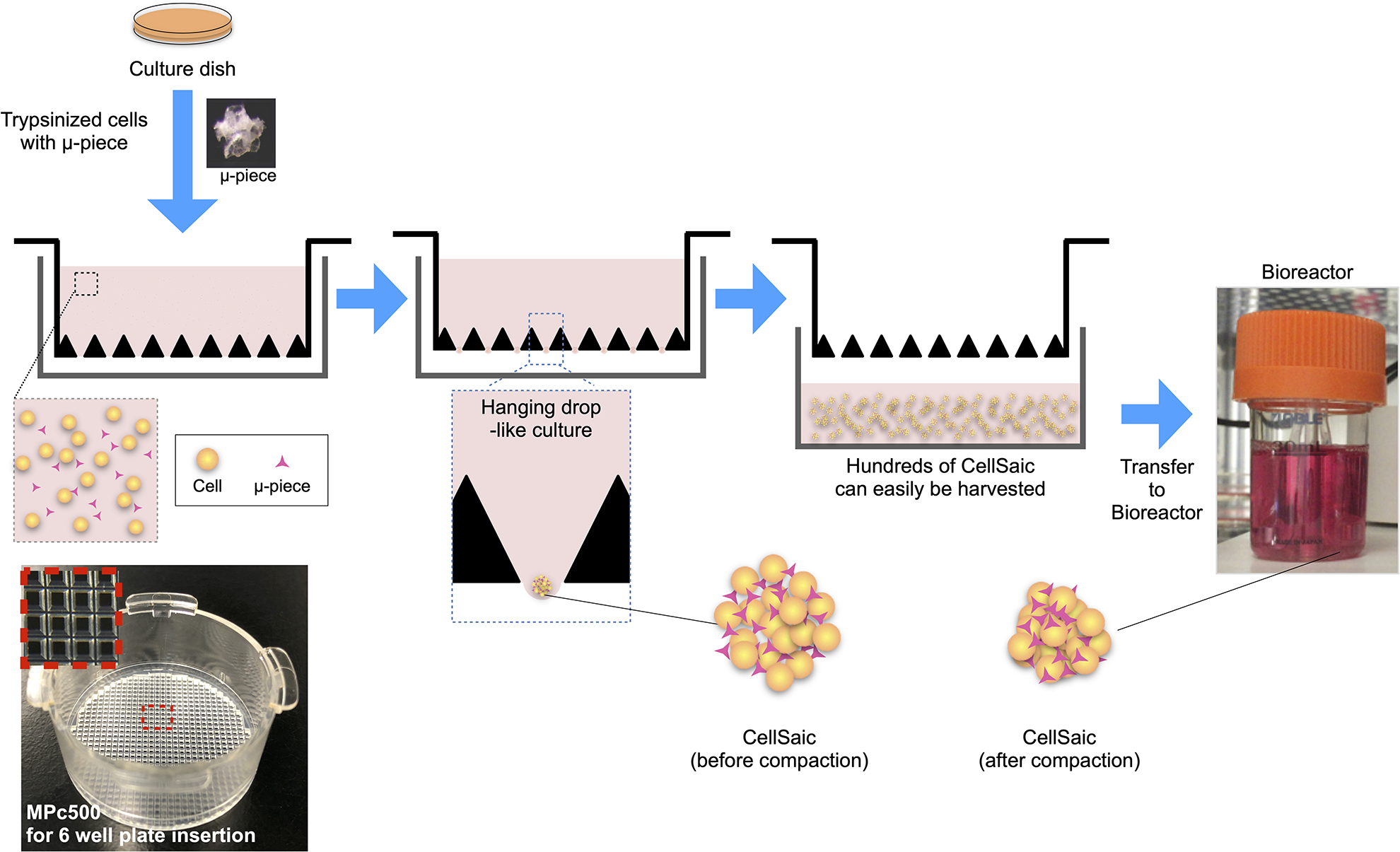
Illustrations and images of the mass production of CellSaics. Cell suspensions were mixed with µ-pieces and seeded into MPc500. Cells and µ-pieces settle by gravity to form CellSaics in a hanging drop-like state. Since the MPc500 has 648 holes per well, it is possible to produce more than approximately 600 CellSaics at a time. Following the generation of CellSaics in hanging drop culture, CellSaics are matured in stirring culture.

MEF-CellSaic transplantation in Fabry disease model mice. (A) Photographs in each step of CellSaic mass production. (B) HE and TUNEL staining of MEF-CellSaic structures before stirring culture. The black bars indicate 100 µm. (C) HE and TUNEL staining of MEF-CellSaic structures after 7 days of stirring culture. The black bars indicate 100 µm. (D) Histogram of the sizes of MEF-CellSaic structures after the maturation process. (E) Alpha-galactosidase activity in MEFs in monolayer culture and MEFs in 3D culture with or without µ-pieces. 2D indicates monolayer culture (2D culture), and 3D indicates 3D culture achieved with spherical structures. (F) Secreted alpha-galactosidase activity in culture medium under each condition. HE: hematoxylin and eosin; MEF: murine embryonic fibroblast; TUNEL: terminal deoxynucleotidyl transferase 2′-deoxyuridine 5′-triphosphate (dUTP) nick end labeling.
Transplantation of CellSaic Containing Fibroblasts into Fabry Disease Model Mice
We examined the effects of CellSaic-containing MEFs on Fabry disease model mice that were generated by using CRISPR technology with a truncated enzyme at exon 3 (Supplemental Fig. 2). We examined the durability of spheroids and CellSaic under the renal capsule prior to the transplantation of CellSaic to Fabry mice on day 28. Spheroids showed central necrosis and an inflammatory reaction in the grafting site, whereas CellSaic maintained macroscopically visible cell clusters (Supplemental Fig. 3). Following the generation of CellSaics, 20 CellSaics were transplanted under the renal capsule in a kidney (Fig. 4A). By day 28 after transplantation into the renal subcapsular spaces, the grafts had generated viable masses, and newly formed blood vessels had entered the grafts. Numerous erythrocytes were observed inside the CellSaic structures (Fig. 4B). On day 28 after transplantation, there were few TUNEL-positive cells in the CellSaic structures. Moreover, on day 28, the amounts of lyso-Gb3 in the heart and the kidney did not significantly change (Supplemental Fig. 4), whereas the liver had significantly decreased in a dose-dependent manner (Fig. 4C). In preclinical studies of ERT for Fabry disease by using Fabry disease model mice, it was proven that recombinant alpha-galactosidase is primarily taken up by the liver. Although the Gb3 substrate was depleted in the liver at a single administration of 0.3 mg/kg, other tissues, including the heart, kidney, and spleen, did not show any significant changes 22 . Following the administration of either agalsidase alfa or beta to Fabry mice, the activities of alpha-galactosidase were one-order greater in the liver than in the heart or the kidney 23 . Our results showed that the supply of alpha-galactosidase released from CellSaic affected only the liver that preferentially ingested the enzyme but was not enough to clear the storage substances in the heart and the kidney.
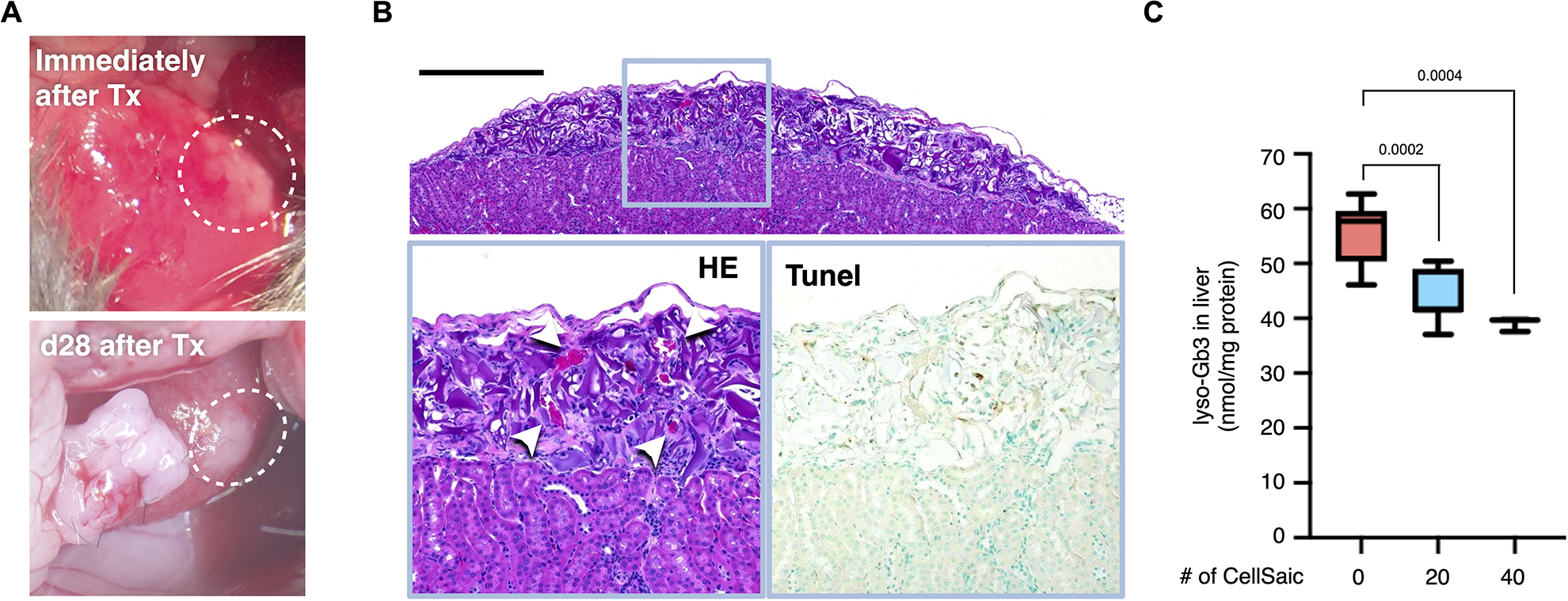
MEF-CellSaic transplantation under the renal capsules of Fabry disease model mice. (A) Macroscopic photographs of MEF-CellSaic transplantation under the renal capsules of Fabry disease model mice. (B) HE staining of Fabry disease model mouse kidneys on day 28. The blue square outlines a magnified view of the same region in the HE and TUNEL images. The white arrow in the HE image indicates red blood cells. The black bar indicates 500 µm. (C) Lyso-Gb3 levels in the livers of Fabry disease model mice transplanted with CellSaics (20 or 40 CellSaics/kidney). HE: hematoxylin and eosin; MEF: murine embryonic fibroblast; TUNEL: terminal deoxynucleotidyl transferase 2′-deoxyuridine 5′-triphosphate (dUTP) nick end labeling.
Discussion
This study demonstrates that CellSaic technology has significant potential to provide a platform for cell transplantation to treat lysosomal diseases. Fabry mice that received CellSaic-containing syngeneic MEFs showed not only long-term survival of grafted cells but also decreased lyso-Gb3 levels in the liver after 1 month. For clinical development using CellSaic technology, the mass production system was successfully developed in this study. In combination with CellSaic technology with gene modifications to release enough lysosomal enzymes enough to be taken up by defective cells and regain their function, CellSaics could offer an essential scaffold.
With respect to pharmacokinetics, steady release of the active enzyme from donor cells might be superior to biweekly administration of the recombinant lysosomal enzyme; this possibility has been supported by observations of improved substrate reduction resulting from HSC Tx relative to that achieved with ERT 24 . The development of a cell transplantation strategy for lysosomal diseases has been hindered by the requirements of large numbers and long survival of cells that release lysosomal enzymes 10 . Although many trials and studies have attempted to verify cell transplantation for the treatment of lysosomal diseases, including Fabry diseases, only bone marrow transplantation (BM Tx) and HSC Tx are recommended for patients suffering from MPS I diagnosed before the age of 2.5 years and clinically presumed Hurler syndrome 13 . HSC Tx and BM Tx could result in relatively measurable comorbidity and perioperative mortality 25 . In the case of alpha-galactosidase, the secreted enzyme does not seem to possess enough M6P moieties to be engulfed by other cells, as the absence of the cross-correction of cells with defective alpha-galactosidase activities was demonstrated by coculturing with wild-type cells 26 and female patients who are heterozygotes for the allele of GLA exist 27 . On the other hand, ex vivo gene therapy for Fabry disease using HSCs 28 and mesenchymal stem cells 29 , in which GLA is overexpressed, reported the correction of the deficiency in a murine model. The overexpression of alpha-galactosidase with M6P driven by an exogenous promoter might saturate the M6P receptors in the trans-Golgi to the fate to lysosomes and leak out of the cells; consequently, alpha-galactosidase might be effectively taken up in functioning lysosomes. Although the risk of provoking malignant transformation of transfected cells with the use of new recombinant lentiviruses is very low 30 , it might not be negligible, especially for benign disorders with established and effective standard therapies such as Fabry disease.
We calculated cell numbers equivalent to ERT by using the measure of the enzyme activities in the culture medium of CellSaics containing MEFs. Agalsidase alfa has 2.1 mmol/h/protein mg of 4-MU-alpha-galactopyranoside-degrading activity 21 and is administered twice weekly at a dose of 0.2 mg/kg to a patient. The pharmacokinetics showed urinary excretion of approximately 80% within the first 24 h, according to the manufacturer’s interview form. Given that the patient’s body weight is 60 kg, it is roughly calculated that 2.4 mg of the drug is taken up into body, resulting in 0.171 mg/day if simply divided by 14. Because the enzyme activity in the CellSaic medium is approximately 1 nmol/h/ml for 24 h (Fig. 3F), 0.171 mg enzyme protein corresponds to approximately 342 l of the culture medium. Because 2 × 106 cells were contained in CellSaics for this 2 ml culture medium, 342 l of the culture medium corresponds to 171 × 109 cells, which requires approximately 86,000 wells holding 648 pores. Although alpha-galactosidase that is released from naïve cells does not possess M6P, it was reported that a lysosomal enzyme could be produced at several thousand–fold increase in transgenic mice compared with naïve mice and then released into the circulation 31 , and 200 times more alpha-galactosidase activity was released from genetically modified fibroblasts in our laboratory, compared with the naïve cells (unpublished data). Two or more digit higher of enzyme release with M6P by gene-modified cells could be feasible, and work to address the number of cells and wells could be applicable to clinical fields.
In addition, the encapsulation of cells via either microencapsulation or macroencapsulation to support the release of biological materials such as insulin has been intensively investigated 32,33 , but the strategy has not reached the clinical arena as a standard therapy mainly due to cellular durability. There have been no reports of the use of these technologies to treat lysosomal diseases. The CellSaic technology used in this study could possess the potential to resolve the main barrier of the cellular macroencapsulation strategy by promoting the survival and viability of grafted cells to be enclosed in the macroencapsulation device. Although this study lasted only 30 days, the results that the histopathological findings showed viable grafts without any apoptotic cells and the release of alpha-galactosidase also suggested that CellSaic technology could be suitable to embed graft cells for cell therapy for lysosomal diseases. We performed this experiment as a first step to a cell transplantation strategy to treat lysosomal diseases. One of the assumed final designs of the treatment could be a combination of CellSaic and macroencapsulation, which could escape from the host immune system, could be metabolically maintained, and could release lysosomal enzymes. As there is substantial evidence that the macroencapsulation of islet cells could rescue diabetes mellitus in animal models, the methodology could work in the setting of lysosomal diseases.
The renal subcapsular space, which is easily vascularized by engrafted foreign tissues and shows advantages to prevent a risk of embolism by remnant of engrafted tissue over intravascular transplantation, was chosen for implantation of CellSaic-containing MEFs. As expected, vascular networks were formed in the CellSaic-implanted sites as early as 1 week following engrafting. Although the use of the renal subcapsular space provided proof of concept that CellSaic-containing fibroblasts can release alpha-galactosidase and decrease lyso-Gb3 levels in the liver, the site and the procedure used for implantation were not clinically relevant because of surgical risks and invasiveness. Although the subcutaneous space has similar characteristics as the renal subcapsular space, it is difficult to identify the implantation site after long periods, which is the reason for the choice of the renal subcapsular space. Conversely, with respect to the clinical situation, the subcutaneous space might be a potential site for CellSaic transplantation, especially due to the low invasiveness.
The results of this study reveal new possibilities in the field of cell therapy research beyond treatment with BM Tx or HSC Tx. Gene modifications could be the next step for clinical therapy along with donor cells to be chosen. Because molecular techniques for cellular genetic modification have been well developed, the use of CellSaic technology could be fruitful for the treatment of lysosomal diseases.
Supplemental Material
Supplemental Material, sj-pdf-1-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-pdf-1-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Supplemental Material
Supplemental Material, sj-pdf-2-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-pdf-2-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Supplemental Material
Supplemental Material, sj-pdf-3-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-pdf-3-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Supplemental Material
Supplemental Material, sj-pdf-4-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-pdf-4-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Supplemental Material
Supplemental Material, sj-xls-1-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-xls-1-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Supplemental Material
Supplemental Material, sj-xls-2-cll-10.1177_0963689720976362 - Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease
Supplemental Material, sj-xls-2-cll-10.1177_0963689720976362 for Cell Transplantation Combined with Recombinant Collagen Peptides for the Treatment of Fabry Disease by Daisuke Kami, Masashi Yamanami, Takahiro Tsukimura, Hideki Maeda, Tadayasu Togawa, Hitoshi Sakuraba and Satoshi Gojo in Cell Transplantation
Footnotes
Acknowledgments
Dr Tsukasa Kitahashi and Dr Kentaro Nakamura, who are employees of FUJIFILM Corporation, provided technical support related to CellSaic.
Author Contributions
DK, HM, and SG designed the research and analyzed the data; DK and SG wrote the paper; MY, TTsu, and TTo performed the animal experiments; and HS reviewed and provided suggestions on the experiments and the manuscript.
Ethical Approval
This study was approved by the Animal Experiment Ethics Committee of the Kyoto Prefectural University of Medicine (Approval Number: M2019-305).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the animal experiment guidelines issued by the Animal Care and Use Committee at the Kyoto Prefectural University of Medicine.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SG and DK received a collaborative research grant from FUJIFILM Corporation. HS received research grants and personal fees from Sumitomo Dainippon Pharma Co., Ltd, and Sanofi Japan Co. outside of the submitted work. TTo received a research grant from Sanofi Japan Co. outside of the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by a collaborative research grant from FUJIFILM Corporation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
