Abstract
Osteosarcoma (OS) is a common primary malignant bone tumor among adolescences, and the emergence of multidrug resistance poses a huge challenge for clinical treatment of OS. LncRNA HOTAIR (HOX antisense intergenic RNA) has been reported to be associated with many malignancies, including OS. However, the underlying mechanisms of HOTAIR involved in drug resistance in OS are obscure. Our study showed that HOTAIR was upregulated in cisplatin (DDP)-resistant OS tissues and cells. HOTAIR knockdown decreased the DDP resistance, drug resistance–related gene expression, cell proliferation, and invasion and promoted apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells. Mechanism researches displayed that miR-106a-5p was downregulated in DDP-resistant OS tissues and cells. MiR-106a-5p directly bound with HOTAIR and was regulated by HOTAIR. Moreover, STAT3 was inhibited by miR-106a-5p at a post-transcriptional level, and the transfection of miR-106a-5p reversed the upregulation of STAT3 caused by HOTAIR overexpression. The increase or decrease of miR-106a-5p suppressed the effect of HOTAIR upregulation or downregulation on DDP resistance, cell proliferation, invasion, and apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells. What’s more, the transfection of STAT3 siRNA reversed the decrease of DDP resistance, cell proliferation, and invasion and rescued the increase of apoptosis induced by miR-106a-5p inhibition. These data suggested that HOTAIR enhanced DDP resistance of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells by affecting cell proliferation, invasion, and apoptosis via miR-106a-5p/STAT3 axis.
Introduction
Osteosarcoma (OS) is a malignant bone tumor among children and adolescents 1 . The usual treatment for OS patients is surgery combined with multidrug chemotherapy, such as cisplatin (DDP), ifosfamide, doxorubicin (Dox), and methotrexate 2 . Although combined chemotherapy has achieved a high survival rate, unfortunately, OS is a relatively drug-resistant disease 3 . Therefore, it is better to decipher and understand the underlying molecular mechanisms of drug resistance and to find novel therapeutic targets.
Long noncoding RNAs (lncRNAs) are a class of noncoding RNAs over 200 nt in length 4 . LncRNAs are involved in many physiological and pathological cellular functions such as cell differentiation, development, apoptosis, proliferation, and metabolism 5 –8 . LncRNA HOTAIR (HOX antisense intergenic RNA) has been found to be overexpressed in various human cancers 9 . HOTAIR alters histone H3K27 methylation and related gene expression profiles through interaction with PRC2, thereby enhancing tumor invasion and metastasis 10 . It has been reported that HOTAIR promotes proliferation and metastasis of OS cells through the AKT/mTOR signaling pathway 11 . Moreover, HOTAIR is a potential target for the treatment of DDP-resistant ovarian cancer 12 . However, the biological function of HOTAIR in DDP resistance in OS is unclear.
MicroRNAs (miRNAs) are a class of endogenous small noncoding RNAs of approximately 22 nucleotides in length that regulate gene expression at the post-transcriptional level 13 . MiRNAs regulate many cellular pathways and functions, including cellular metabolism, differentiation, and apoptosis, so its dysregulation leads to many human diseases 14 . More and more studies have shown that miRNAs play key roles in cancer chemotherapy resistance, and their potential roles in determining drug sensitivity or drug resistance have been confirmed by several researches 15 –17 . MiR-106a-5p is a member of the miR-17 family and is reported to be abnormally expressed in various tumors 18 . It has been proved to exert antitumor effects in astrocytoma, OS, and colorectal cancer 18 .
STAT3 is a member of the STATs (Signal Transduction and Transcriptional Activator) family and acts as a signal messenger and transcription factor 19 . The STATs family plays an important role in regulating cell proliferation and differentiation 20 . The expression and activity of STAT3 are closely related to the occurrence, progression, invasion, and metastasis of various tumors such as breast cancer, colon cancer, and gastric cancer 21 . Besides, the overexpression of STAT3 is strongly associated with the clinical stage of OS and may be a major determinant of OS progression 22,23 .
In the present study, we found that HOTAIR and STAT3 were upregulated, while miR-106a-5p was downregulated in DDP-resistant OS tissues and cells. Subsequent researches demonstrated that HOTAIR enhanced DDP resistance of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells by affecting cell proliferation, invasion, and apoptosis via miR-106a-5p/STAT3 axis. This new regulatory network may provide a promising therapeutic approach for patients with DDP-resistant OS.
Materials and Methods
Tissue Samples
Sixty patients (5–26 years old) including 38 males and 22 females with OS received the same chemotherapy regimen (DDP, methotrexate, ifosfamide, and doxorubicin for two cycles) before surgery, and they all underwent complete resection at Huaihe Hospital of Henan University between 2010 and 2018. The tissues were confirmed by pathological examination and classified according to the Enneking Stage. We collected the tumor tissues surgically removed from all patients and selected 20 cases of DDP-sensitive and 20 cases of DDP-resistant tissues according to the Huvos scoring system. The adjacent normal tissues which were located at least 5 cm away from the tumor tissues were collected as the control. All tissues were frozen in liquid nitrogen and stored at −80°C. The study was approved by the Ethics Committee of Huaihe Hospital of Henan University, and informed consent was provided for all OS patients or their guardians. The clinical parameters of OS patients in this study are shown in Supplemental Table 1.
Cell Culture and Transfection
The Saos2, MG-63, U2OS, and HEK293A cell lines were purchased from the Shanghai Institutes for Biological Sciences (Shanghai, China). DDP-resistant OS cell strains (Saos2/DDP, MG-63/DDP, and U2OS/DDP) were established from the parental cell lines Saos2, MG-63, and U2OS by using an intermittent stepwise selection protocol over 6 months, and finally exposed to 1.2 mg/ml DDP. All cells were cultured using Dulbecco’s modified eagle’s medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco, Rockville, MD, USA) at 37°C in 5% CO2.
The siRNAs against HOTAIR (si-HOTAIR and si-HOTAIR#) and STAT3 (si-STAT3), miR-106a-5p mimic, miR-106a-5p inhibitor, and their respective controls were synthesized by Gene Pharma (Shanghai, China). Full-length cDNA of HOTAIR was inserted into the pcDNA3.1 empty vector. The cells were transfected with si-HOTAIR, si-HOTAIR#, si-STAT3, miR-106a-5p mimic, miR-106a-5p inhibitor, pcDNA3.1-HOTAIR, or their respective controls by using the Lipofectamine® 3000 reagent (Thermo Fisher Scientific).
Western Blotting
Total proteins were extracted by radioimmunoprecipitation assay lysis buffer and 50 µg proteins were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (100 V, 2 h) and electrotransferred onto the polyvinylidene difluoride membranes (Millipore, Boston, MA, USA) (250 mA, 2 h). After blocking with 5% skim milk powder at room temperature for 2 h, the membranes were incubated with primary antibodies overnight at 4°C. The primary antibodies used in this study include anti-MDR1 [#13342; 1:1,000 dilution; Cell Signaling Technology (CST), Boston, MA, USA; Reactivity: Human], anti-ABCB1 (#13342; 1:1,000 dilution; CST; Reactivity: Human), anti-ABCC1 (#14685; 1:1,000 dilution; CST; Reactivity: Human), anti-ABCG2 (#4477; 1:1,000 dilution; CST; Reactivity: Human, Mouse, Rat), anti-MRP5 (sc-376965; 1:1,000 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA; Reactivity: Human, Mouse, Rat), anti-LRP1 (#64099; 1:1,000 dilution; CST; Reactivity: Human, Mouse, Rat), anti-STAT3 (#4904; 1:2,000 dilution; CST; Reactivity: Human, Mouse, Rat, Monkey), and glyceraldehyde 3-phosphate dehydrogenase (#97166, 1:1,000 dilution; CST; Reactivity: Human, Mouse, Rat, Monkey). Horseradish peroxidase–labeled goat anti-rabbit immunoglobulin G (IgG) secondary antibodies (ab6721; 1:3,000 dilution; Abcam, Cambridge, UK) were added and incubated with the membrane at room temperature for 1 h. After elution, the abundances of the proteins were detected in a ChemiDoc XRS Imaging System (Bio-Rad, Hercules, CA, USA). Image J software was used to analyze the gray level of bands.
Apoptosis Assay
Apoptosis analysis was performed by using Annexin V-FITC/propidium iodide (PI) apoptosis detection kit (Multisciences, Shanghai, China). Cells were collected and washed with phosphate buffered saline (PBS). Then they were resuspended, and 5 µl Annexin V-FITC and 5 µl PI were sequentially added. Apoptosis was detected by flow cytometry.
Cell Proliferation Assay
Cell proliferation was measured using Cell Counting Kit-8 (CCK-8, Dojindo, Kumamoto, Japan) according to the manufacturer’s instructions. The treated cells were seeded in a 96-well plate and cultured for 24, 48, and 72 h, respectively. Ten microliters of CCK-8 solution was added at each time point and the cells were incubated at 37°C for 2 h. The absorbance at 450 nm was measured using a microplate reader (Bio-Rad).
Quantitative Polymerase Chain Reaction
Total RNA was extracted using the TRIzol kit (Invitrogen, Carlsbad, CA, USA) to detect the relative expression of genes. Reverse transcription was performed using Transcriptor First Strand cDNA Synthesis Kit (Takara Biotechnology, Dalian, China). SYBR PrimeScript RT-PCR Kit (Takara Biotechnology) was chosen to test the mRNA expression of genes. Quantitative polymerase chain reaction (qPCR) system was designed as follows: cDNA, 2.0 μl; SYBR Fast qPCR Mix, 10.0 μl; reverse primer (10 μM), 0.8 μl; forward primer (10 μM), 0.8 μl; and RNase Free ddH2O, 6.4 μl. The reaction conditions were designed as follows: denaturation at 95°C for 3 min, followed by 35 cycles of 94°C for 15 s, 55°C for 25 s, and 72°C for 30 s. The relative level was calculated by 2−ΔΔCT method.
Transwell Assay
Cells were collected and suspended in serum-free medium and seeded into the upper chamber with Matrigel at a density of 2 × 104 every chamber. The lower chamber was supplemented with RPMI-1640 medium containing 10% fetal bovine serum. After incubated for 48 h, the cells adhering to the lower surface were fixed in methanol for 15 min and stained using 0.05% crystal violet for 10 min. The cells were counted under a light microscope.
Luciferase Activity Assay
StarBase v2.0 (http://starbase.sysu.edu.cn) online database was used to predict the interactions between HOTAIR and miR-106a-5p. The sequence of HOTAIR containing a miR-106a-5p binding site was amplified and cloned to psiCHECK-2 vector (Promega, Madison, WI, USA) to generate HOTAIR-WT (wild-type). The binding site of HOTAIR was mutated to obtain the HOTAIR-MUT (mutant type) using a Site-Directed Mutagenesis Kit (Stratagene, South Carolina, USA). HOTAIR-WT (or HOTAIR-MUT) and miR-106a-5p mimic were cotransfected into HEK293A cells, and relative luciferase activities were measured using the Dual-Luciferase Reporter Assay System (Promega).
TargetScan Human 7.2 (http://www.targetscan.org/vert_72/) online database was used to predict the interactions between miR-106a-5p and STAT3. Wild-type and three mutant-type STAT3 reporters containing miR-106a-5p binding sites were amplified and cloned into psiCHECK-2 vector to obtain STAT3-WT, STAT3-MUT1, STAT3-MUT2, and STAT3-MUT Both. STAT3-WT, STAT3-MUT1, STAT3-MUT2, or STAT3-MUT both were transfected into HEK293A cells together with miR-106a-5p mimic, and relative luciferase activities were measured using the Dual-Luciferase Reporter Assay System (Promega).
RNA Immunoprecipitation
RNA immunoprecipitation (RIP) assay was conducted using Magna RIP RNA-Binding Protein Immunoprecipitation Kit (Millipore, Billerica, MA, USA) according to the manufacturer’s instructions. In brief, cells were washed with ice-cold PBS and then lysed in radioimmunoprecipitation buffer at 4°C for 30 min. The whole cell lysates were incubated with protein A/G sepharose beads conjugated to antibodies against Ago2 (Millipore) or normal human IgG (Millipore). Western blotting and qPCR were used to analyze immunoprecipitated RNA and total RNA from the whole cell lysates (input controls).
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 software (Chicago, IL, USA). The measurement data were expressed as mean ± standard deviation. The comparison between the data of the groups was analyzed by the Student’s t-test and two-way analysis of variance. P < 0.05 was considered statistically significant.
Results
HOTAIR was Upregulated in DDP-Resistant OS Tissues and Cells
To measure the expression of HOTAIR in OS tissues and cells, qPCR was performed. The results displayed that HOTAIR was upregulated in DDP-sensitive and DDP-resistant OS tissues compared to that in paracancerous tissues (Fig. 1A). Patients with increased HOTAIR expression had a reduced overall survival time than those with decreased HOTAIR expression (Supplemental Fig. 5). Also, the data showed higher HOTAIR expression in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells in comparison to that in Saos2, MG-63, and U2OS cells (Fig. 1B and Supplemental Fig. 2A).

HOTAIR was upregulated in DDP-resistant OS tissues and cells, and HOTAIR knockdown decreased the DDP resistance of Saos2/DDP and MG-63/DDP cells. (A, B) The expression of HOTAIR in DDP-sensitive (n = 20) and DDP-resistant (n = 20) OS tissues, Saos2/DDP and MG-63/DDP cells (n = 3), and their matched controls was measured by qPCR. Next, HOTAIR siRNA was transfected into Saos2/DDP and MG-63/DDP cells; following transfection for 48 h, (C) the interference efficiencies were detected with qPCR (n = 3). (D, E) The IC50 values of DDP (n = 3) and (F, G) the protein levels of MDR1, ABCB1, ABCC1, ABCG2, MRP5, and LRP1 (n = 3) were detected by CCK-8 and western blotting. *P < 0.05, **P < 0.01. DDP: cisplatin; OS: osteosarcoma; qPCR: quantitative polymerase chain reaction.
Downregulation of HOTAIR Decreased the Resistance of Saos2/DDP, MG-63/DDP, and U2OS/DDP Cells to DDP
To explore the role of HOTAIR played on OS chemoresistance, the siRNAs specifically against HOTAIR were transfected into Saos2/DDP, MG-63/DDP, and U2OS/DDP cells (Fig. 1C and Supplemental Figs. 1A and 2B). As shown in Fig. 1D, E and Supplemental Figs. 1B, C and 2C, the IC50 values of DDP in Saos2/DDP, MG-63/DDP, or U2OS/DDP cells were observably increased compared with those in Saos2, MG-63, or U2OS cells, but significantly decreased after the interference of HOTAIR. In addition, we confirmed that HOTAIR knockdown in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells effectively decreased the protein levels of MDR1, ABCB1, ABCC1, ABCG2, MRP5, and LRP1, which were multidrug resistance–related genes (Fig. 1F, G and Supplemental Figs. 1D, E and 2D).
Interference with HOTAIR Inhibited Cell Proliferation and Invasion and Promoted Apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP Cells
Based on the above results, the effect of HOTAIR in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells was further investigated. The data showed that the cell proliferative and invasive abilities were prominently suppressed, but the apoptosis was increased in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells by the decrease of HOTAIR (Fig. 2A–F and Supplemental Figs. 1F–K and 2E–G).

Interference with HOTAIR inhibited proliferation and invasion and promoted apoptosis of Saos2/DDP and MG-63/DDP cells. HOTAIR siRNA was transfected into Saos2/DDP and MG-63/DDP cells; following transfection for 48 h, the cell proliferation (A, B), invasion (C, D), and apoptosis (E, F) were detected by CCK-8, transwell, and flow cytometry. n = 3, **P < 0.01.
MiR-106a-5p was Downregulated in DDP-resistant OS Tissues and Cells and Regulated by HOTAIR
We firstly found that miR-106a-5p was dramatically downregulated in DDP-sensitive and DDP-resistant OS tissues and Saos2/DDP, MG-63/DDP, and U2OS/DDP cells in contrast to that in their matched controls (Fig. 3A, B and Supplemental Fig. 3A). Next, StarBase v2.0 online database was used to predict the putative target of miR-106a-5p and HOTAIR, and the data indicated that miR-106a-5p had a binding site with HOTAIR (Fig. 3C). Subsequent luciferase reporter gene assay indicated that the transfection of miR-106a-5p mimic resulted in the decline of luciferase activity of HOTAIR-WT reporter, but the luciferase activity of HOTAIR-MUT reporter had no change (Fig. 3D). RIP assay showed the significant enrichment of miR-106a-5p and HOTAIR using Ago2 antibody compared with IgG antibody (Fig. 3E). Furthermore, as shown in Fig. 3F and Supplemental Fig. 3B, the inhibition of HOTAIR significantly upregulated miR-106a-5p expression in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells.
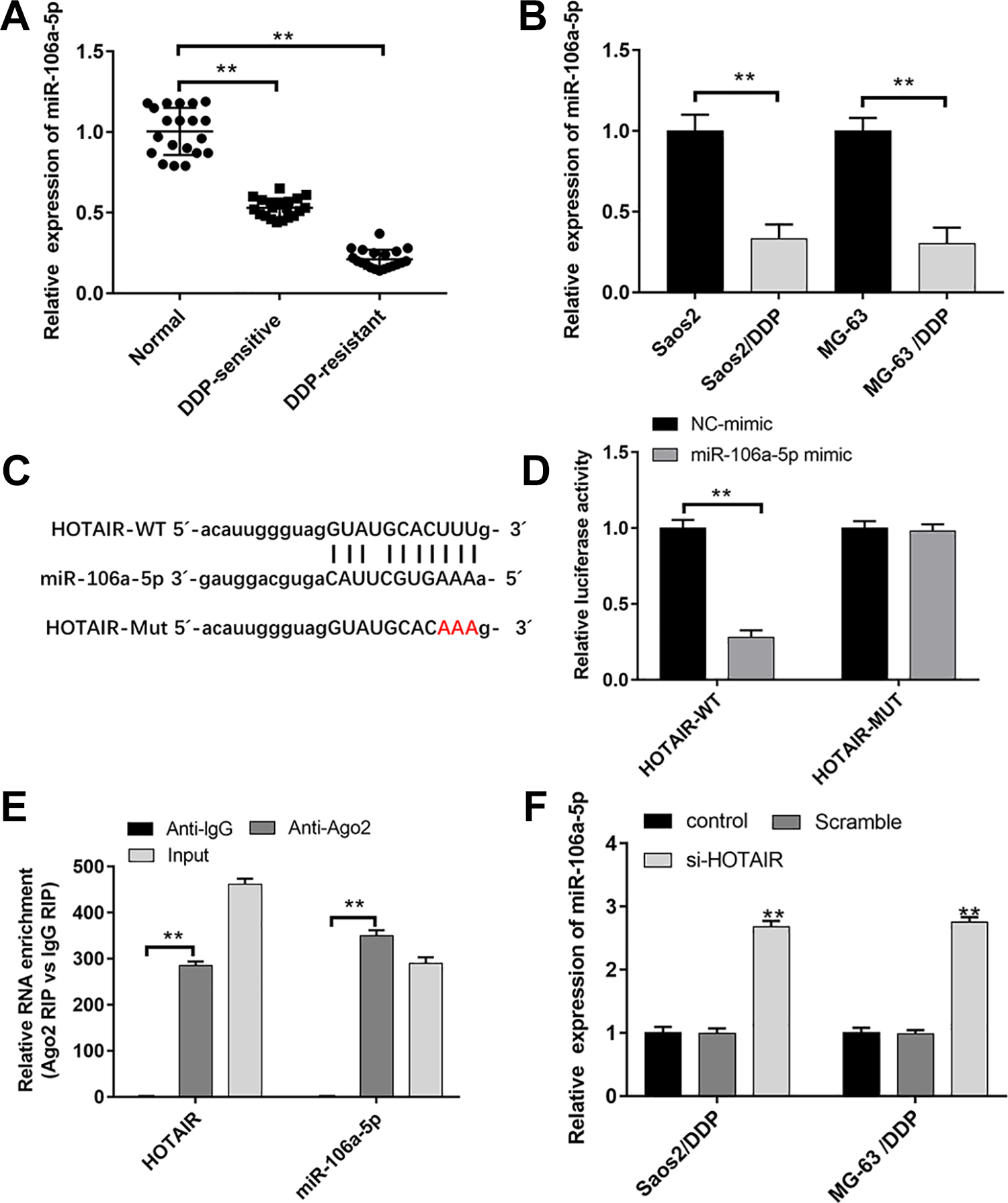
MiR-106a-5p was downregulated in DDP-resistant OS tissues and cells and regulated by HOTAIR. (A, B) The expression of HOTAIR in DDP-sensitive (n = 20) and DDP-resistant (n = 20) OS tissues, Saos2/DDP and MG-63/DDP cells (n = 3), and their matched controls was measured by qPCR. (C) The binding site between HOTAIR and miR-106a-5p was predicted by StarBase v2.0. (D) The luciferase activities of HOTAIR-WT (HOTAIR-MUT) reporters in Saos2/DDP and MG-63/DDP cells cotransfected with miR-106a-5p mimic or negative control (NC) mimic were assessed by Dual-Luciferase Reporter Assay (n = 3). (E) RIP assay was performed to observe the position relation between HOTAIR and miR-106a-5p (n = 3). (F) HOTAIR siRNA was transfected into Saos2/DDP and MG-63/DDP cells; following transfection for 48 h, the expression of miR-106a-5p was detected by qPCR (n = 3). **P < 0.01. DDP: cisplatin; OS: osteosarcoma; qPCR: quantitative polymerase chain reaction; RIP: RNA immunoprecipitation.
HOTAIR Regulated the DDP Resistance, Cell Proliferation, Invasion, and Apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP Cells by Binding miR-106a-5p
The HOTAIR overexpression vector was transfected into Saos2/DDP, MG-63/DDP, and U2OS/DDP cells alone or together with miR-106a-5p mimic. And we demonstrated that the abnormally upregulation of miR-106a-5p reversed the increase of IC50 values, drug resistance–related gene expression, cell proliferation, and invasion induced by HOTAIR overexpression in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells (Fig. 4A–H and Supplemental Fig. 3C–F). Moreover, compared with the HOTAIR-overexpressed group, the transfection of miR-106a-5p mimic promoted apoptosis (Fig. 4I, J and Supplemental Fig. 3G).

HOTAIR regulated the DDP resistance, cell proliferation, invasion, and apoptosis of Saos2/DDP and MG-63/DDP cells by binding miR-106a-5p. HOTAIR overexpression vector was transfected into Saos2/DDP and MG-63/DDP cells alone or together with miR-106a-5p mimic; following transfection for 48 h, (A, B) the IC50 values of DDP, (C, D) and the protein levels of MDR1, ABCB1, ABCC1, ABCG2, MRP5, and LRP1 were detected by CCK-8 and western blotting. Cell proliferation (E, F), invasion (G, H), and apoptosis (I, J) were detected by CCK-8, transwell, and flow cytometry. n = 3, **P < 0.01. DDP: cisplatin.
STAT3 was a Target of miR-106a-5p
We observed that STAT3 was upregulated in DDP-sensitive and DDP-resistant OS tissues and Saos2/DDP, MG-63/DDP, and U2OS/DDP cells compared to that in their respective controls (Fig. 5B, C and Supplemental Fig. 4A). TargetScan Human 7.2 online database was used to predict the interactions between miR-106a-5p and STAT3. We found that STAT3 3′-untranslated region (UTR) had two binding sites with miR-106a-5p sequence (Fig. 5A). Following dual-luciferase reporter gene assay displayed that miR-106a-5p overexpression significantly reduced the luciferase activities of STAT3-WT, STAT3-MUT1, and STAT3-MUT2, whereas the luciferase activity in STAT3-MUT Both group showed no obvious change (Fig. 5D). In addition, overexpression of HOTAIR promoted STAT3 expression, whereas transfection of miR-106a-5p mimic abolished this effect in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells (Fig. 5E, F and Supplemental Fig. 4B).
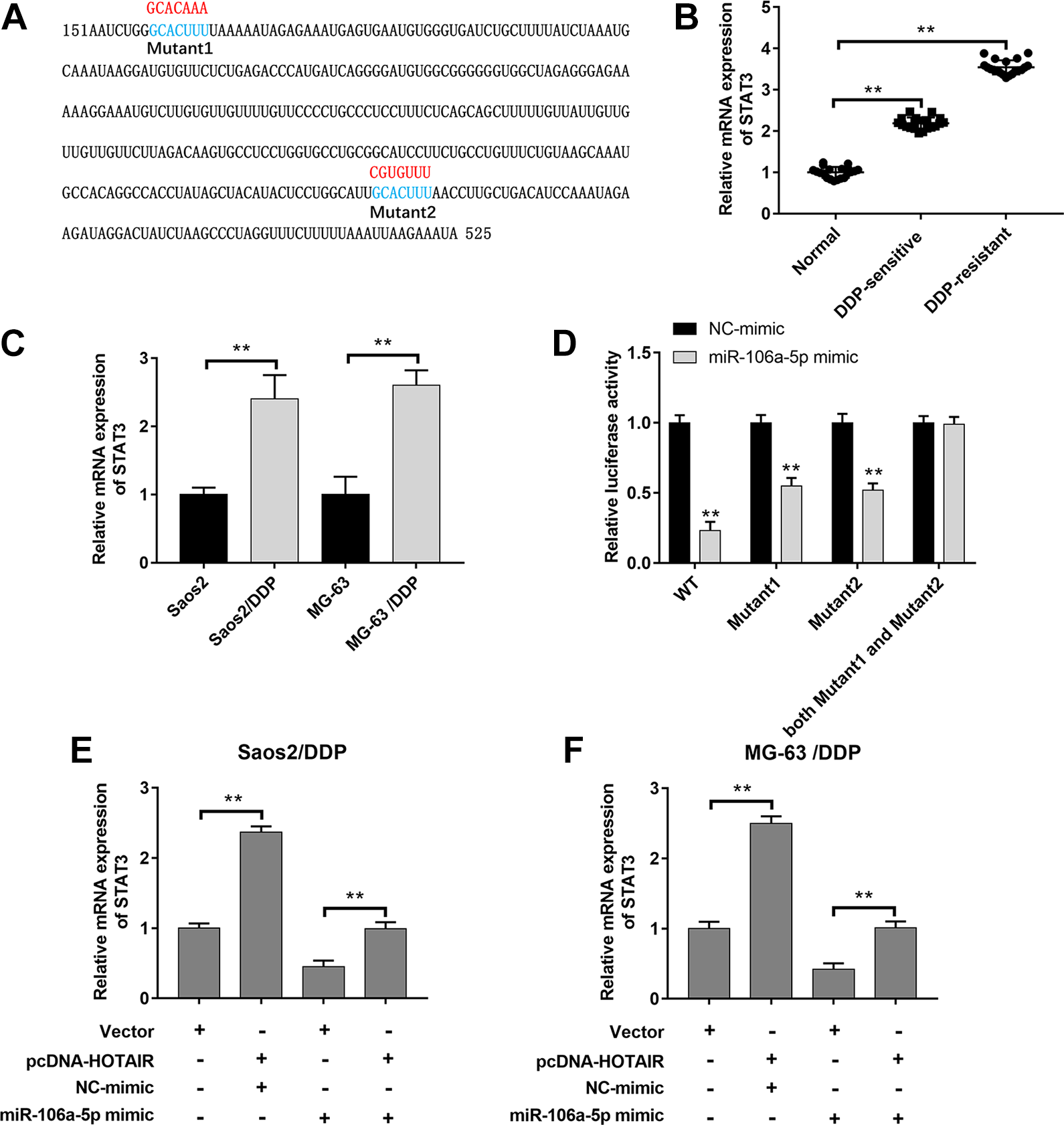
STAT3 was a target of miR-106a-5p. (A) The binding sites between miR-106a-5p and STAT3 were predicted by TargetScan Human 7.2. (B, C) The expression of STAT3 in DDP-sensitive (n = 20) and DDP-resistant (n = 20) OS tissues, Saos2/DDP and MG-63/DDP cells (n = 3), and their matched controls was measured by qPCR. (D) The luciferase activities of STAT3-WT (STAT3-MUT) reporters in Saos2/DDP and MG-63/DDP cells transfected with miR-106a-5p mimic or NC mimic were assessed by Dual-Luciferase Reporter Assay (n = 3). (E, F) HOTAIR overexpression vector and miR-106a-5p mimic were transfected respectively or cotransfected into Saos2/DDP and MG-63/DDP cells, and the expression of STAT3 was detected by qPCR (n = 3). **P < 0.01. DDP: cisplatin; OS: osteosarcoma; qPCR: quantitative polymerase chain reaction.
HOTAIR Regulated the DDP Resistance, Cell Proliferation, Invasion, and Apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP Cells via miR-106a-5p/STAT3 Axis
Next, we investigated whether HOTAIR exerted its effects in Saos2/DDP, MG-63/DDP, and U2OS/DDP cells via regulating miR-106a -5p/STAT3 axis. The results showed that the transfection of miR-106a-5p inhibitor suppressed the decline of IC50 values, the protein levels of drug resistance–related genes and STAT3, cell proliferation and invasion caused by HOTAIR interference, and the transfection of STAT3 siRNA impeded the effect of miR-106a-5p on DDP resistance, STAT3 expression, cell proliferation, and invasion of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells (Fig. 6A–J and Supplemental Fig. 4C–F). In contrast, the inhibition of miR-106a-5p reversed the increase of apoptosis caused by HOTAIR interference, and the inhibition of STAT3 reversed the reduction of apoptosis caused by miR-106a-5p interference (Fig. 6K, L and Supplemental Fig. 4G).
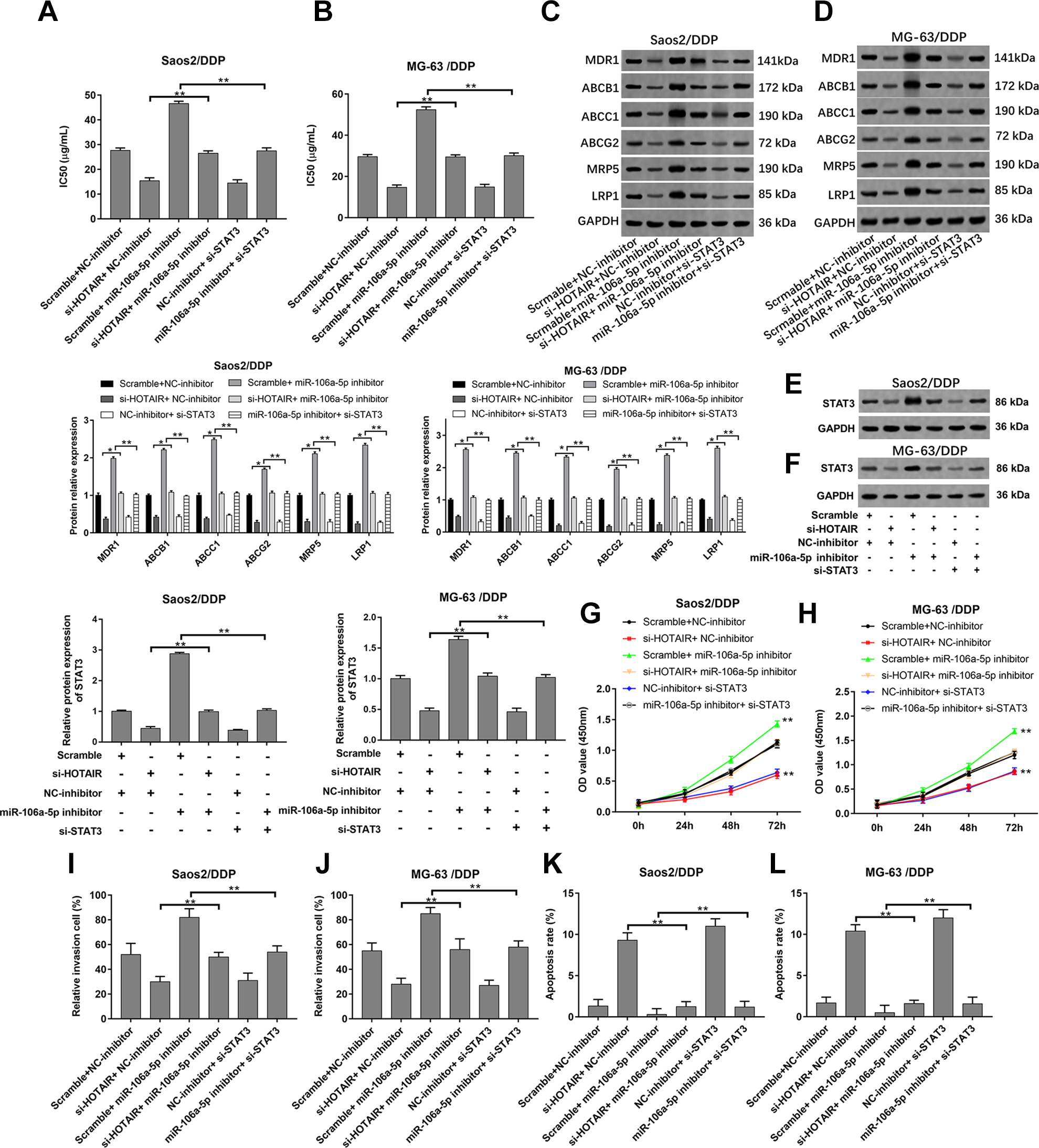
HOTAIR regulated the DDP resistance, cell proliferation, invasion, and apoptosis of Saos2/DDP and MG-63/DDP cells via miR-106a-5p/STAT3 axis. The HOTAIR siRNA, miR-106a-5p inhibitor, HOTAIR siRNA + miR-106a-5p inhibitor, STAT3 siRNA, and miR-106a-5p inhibitor + STAT3 siRNA were transfected into Saos2/DDP and MG-63/DDP cells; following transfection for 48 h, (A, B) the IC50 values of DDP, (C–F) and the protein levels of MDR1, ABCB1, ABCC1, ABCG2, MRP5, LRP1, and STAT3 were detected by CCK-8 and western blotting. Cell proliferation (G, H), invasion (I, J), and apoptosis (K, L) were detected by CCK-8, transwell, and flow cytometry. n = 3, *P < 0.05, **P < 0.01.
Discussion
DDP has been widely used in the treatment of various solid tumors 24 . It is a metal-containing drug, which makes it non-cross-resistance with other drugs in certain key pathways 25 . Therefore, the mere elaboration of the mechanism of OS resistant to DDP alone has important therapeutic value. In this study, we aimed to explore the mechanism and function of HOTAIR in the DDP resistance in OS. Our results suggested that HOTAIR knockdown reduced the DDP resistance of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells.
A number of studies have shown that the expression changes of lncRNAs are closely related to the occurrence and development of OS 26,27 . Studies show that dysregulation of lncRNA HOTAIR is related to tumor chemoresistance. HOTAIR promoted sorafenib resistance by inhibiting miR-217 in hepatic carcinoma 28 . The silence of HOTAIR reverses DDP resistance of ovarian cancer cells by suppressing miR-138-5p-regulated EZH2 and SIRT1 29 . And HOTAIR knockdown promotes the sensitivity of the GC cell lines to DDP, regulates apoptosis-related genes, and inhibits the expression of multidrug resistance genes 11 . Our data showed that HOTAIR interference decreased the DDP resistance and drug resistance–related gene expression of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells, which was consistent with previous studies. HOTAIR is upregulated in clinical OS tissue specimens, suggesting that HOTAIR is a potential OS molecular marker 11,30 . This study revealed that HOTAIR was upregulated both in DDP-resistant OS tissues and cells. It is reported that the silence of HOTAIR inhibits the proliferation, adhesion, migration, and invasion of OS cells, which may play a role in tumor suppression in OS cells 31 . HOTAIR inhibits human chondrosarcoma cell growth by upregulation of miR-454-3p and Stat3 signal inactivation in vivo 31 . In this study, we found that the effect of HOTAIR on OS-resistant cell proliferation and invasion was similar to that reported in the literature. And HOTAIR might act by targeting the miR-106a-5p/STAT3 axis. In brief, lncRNA HOTAIR was involved in the DDP resistance of OS cells.
MiRNAs are reported to be novel DDP sensitivity modulators 32 . Recent studies have shown that miR-22 mediates the DDP resistance of OS cells through suppressing autophagy 33 . MiR-584 hinders cell proliferation and sensitizes OS cells to DDP and taxanes 34 . Besides, miR-504 modulates OS cell chemoresistance to DDP by targeting p53 35 . MiR-106a-5p belongs to miR-17 family, members of which have been reported to promote cancer development by promoting cell proliferation, inhibiting apoptosis, and inducing tumor angiogenesis 18 . As previous research has shown, miR-106a-5p inhibits proliferation, migration, and invasion of OS cells by targeting HMGA2 36 . We found that miR-106a-5p was downregulated in DDP-resistant OS tissues and cells, and its overexpression or inhibition reversed the effect of HOTAIR upregulation or downregulated on DDP resistance, cell proliferation, invasion, and apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells. What’s more, STAT3 was regulated by miR-106a-5p, and miR-106a-5p might play its role by targeting STAT3.
STAT3 is upregulated in multiple cancer tissues 37 . STAT3 has been reported to regulate cell proliferation, migration, and invasion by modulating the expression of Bcl-2 (apoptosis-related gene) and Cyclin D1 (cell cycle-associated genes) 20 . Many studies have revealed that STAT3 expression and dysfunction are associated with OS development, progression, metastasis, and drug resistance 21 . Many miRNAs can target the 3’-UTR of STAT3 to affect the activation of cancer cells 37 . Besides, lncRNA has been found to regulate tumorigenesis and tumor progression by modulating STAT3 20 . We testified in this article that STAT3 was regulated by miR-106a-5p and HOTAIR, and STAT3 knockdown reversed the effect of miR-106a-5p inhibition on DDP sensitivity, proliferation, invasion, and apoptosis of Saos2/DDP, MG-63/DDP, and U2OS/DDP cells, thus revealing that STAT3 was closely related to the resistance of cells to DDP in OS.
In summary, we demonstrated that lncRNA HOTAIR and STAT3 were markedly upregulated, while miR-106a-5p was notably downregulated in DDP-resistant OS tissues and Saos2/DDP, MG-63/DDP, and U2OS/DDP cells. Mechanism studies confirmed that HOTAIR was involved in the formation of DDP resistance in OS via the miR-106a-5p/STAT3 axis. This may offer a promising therapeutic strategy to DDP-resistant OS.
Supplemental Material
Supplemental Material, Supplementary_materials - HOTAIR Promotes Cisplatin Resistance of Osteosarcoma Cells by Regulating Cell Proliferation, Invasion, and Apoptosis via miR-106a-5p/STAT3 Axis
Supplemental Material, Supplementary_materials for HOTAIR Promotes Cisplatin Resistance of Osteosarcoma Cells by Regulating Cell Proliferation, Invasion, and Apoptosis via miR-106a-5p/STAT3 Axis by Jiankuo Guo, Dongmei Dou, Tianlun Zhang and Bo Wang in Cell Transplantation
Footnotes
Ethical Approval
This study was approved by the Ethics Committee at Huaihe Hospital of Henan University.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with Huaihe Hospital of Henan University of Ethics Committee’s or Institutional Review Board’s (Approval Number: HUHH10053) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
