Abstract
The treatment of bone defects has always been a challenge for orthopedic surgeons. The development of tissue engineering technology provides a novel method for repairing bone defects and has been used in animal experiments and clinical trials. However, there are few clinical studies on comparing the long-term outcomes of tissue-engineered bones (TEBs) and other bone grafts in treating bone defects, and the long-term efficiency of TEBs remains controversial. Therefore, a study designed by us was aimed to compare the long-term efficacy and safety of individual tissue-engineered bones (iTEBs) and allogeneic bone granules (ABGs) in treating bone defects caused by curettage of benign bone tumors and tumor-like lesions. From September 2003 to November 2009, 48 patients who received tumor curettage and bone grafting were analyzed with a mean follow-up of 122 mo (range 60 to 173 mo). Based on implant style, patients were divided into groups of iTEBs (
Introduction
Benign bone tumors and tumor-like lesions are common in children and adolescents. As with treating, regular observation is suitable for most cases. However, for the active or aggressive lesions which threaten the structural bone stability, curettage is recommended in consideration of relatively low recurrence rate and favorable limb function 1 . To reduce fracture risk and avoid residual bone defect following curettage, intraoperative bone grafting is required, especially for defects beyond a certain size or located in weight-bearing areas 2 . Based on the excellent osteogenicity, osteoinductivity, and osteoconductivity, autologous bone grafting has been considered the golden standard for repairing bone defects. However, it is associated with complications including donor site morbidity, prolonged operating time, and limited availability 3 . As an alternative, allogeneic bone grafting has been widely employed due to the favorable osteoconductivity and biodegradation property. Nevertheless, the osteoinductive and osteogenic potentials of allogeneic bones are limited by the preparation process of deproteinization, which is obligatory for reducing immunogenicity 4 .
In the past decades, tissue-engineered bones (TEBs), especially patient-specific individual tissue-engineered bones (iTEBs), have been proposed as a promising strategy in repairing bone defects 5 . The general principle of iTEBs involves the incorporation of patients’ own osteoprogenitors (typically mesenchymal stem cells, MSCs) into three-dimensional osteoconductive bio-scaffolds. In this way, all essential elements for an optimal bone graft, including osteoinductivity, osteoconductivity, and bioactive factors, can be integrated into iTEBs. The effectiveness of iTEBs in treating bone defects has been widely documented by large animal experiments and clinical trials 6 . However, there are few clinical studies on comparing the long-term outcomes of TEBs and other bone grafts in treating bone defects, and the long-term efficiency of TEBs remains controversial. In this retrospective study, we tried to compare the long-term results of allogeneic bone granules (ABGs) and iTEBs in treating benign bone tumors and tumor-like lesions.
Materials and Methods
Subjects
This study obtained the approval from the medical ethics committee of Southwest Hospital. Written informed consent was obtained from the patients. From September 2003 to November 2009, 48 patients who suffered from benign bone tumors or tumor-like lesions in long bones and received curettage and bone grafting were included. The series consisted of 32 males and 16 females, with an average age of 15.1 yr (range 5 to 36 yr).The mean follow-up period was 122 mo (range 60 to 173 mo). Based on implant style, the patients were divided into groups of ABGs (
Preparation of Bone Grafts
The ABGs were purchased from BIOGENE, Dasting Bio-Tech Co., Ltd, Beijing, China. The ABGs were processed by decellularization, vacuum freeze-drying, vacuum packaging, irradiation sterilization, low temperature storage, and so on. After the procedures, the antigenic components of allogeneic bone were completely removed. Complying with the standardized protocol previously described by our group
7
, patient-specific iTEBs were constructed using autologous MSCs and allogeneic decalcificated bone matrix (DBM). Briefly, DBM were sectioned into blocks (5 × 5 × 5 mm) and soaked into patient-specific autologous serum for more than 2 d. For cell harvest, 50 ml of StemPro® MSC SFM XenoFree Supplement and 5 ml of 200 mM
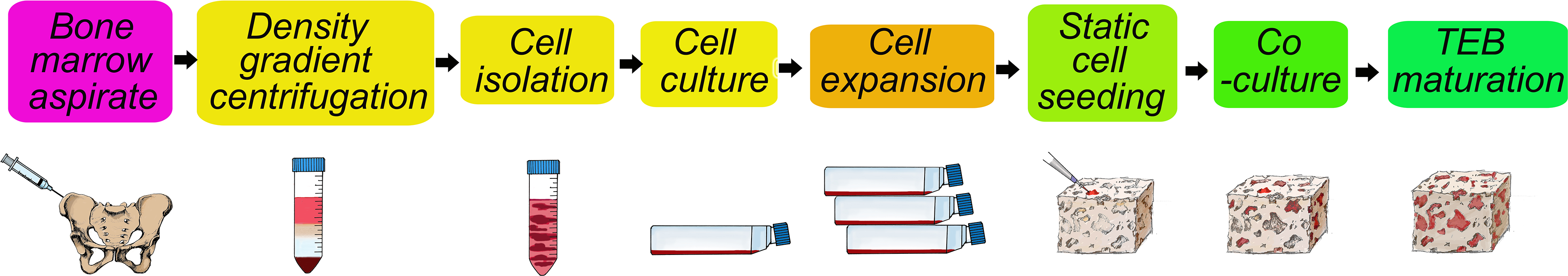
The process of constructing patient-specific individual tissue-engineered bones. TEBs: tissue-engineered bones.
Surgical Procedure
Preoperatively, imaging examinations including anterior–posterior and lateral X-rays, computed tomography (CT) scans, and magnetic resonance imaging were scheduled to determine the lesion location and its relationship with adjacent tissues. The lesion dimension was approximately measured on anteroposterior and lateral X-rays, and the defect size was calculated using the formula of
Postoperative Evaluation and Follow-Up
Postoperatively, X-rays were scheduled once per month until healing and then once per year until the final follow-up. In consideration of cost and radiant exposure, CT scans were performed under certain circumstances, such as persistent pain, suspicion of fixation failure, or tumor recurrence. Bone healing was defined when the cortical bone was thick enough to avoid fracture, as well as disappearance of the lesion and no limit of daily activity. The healing quality was evaluated referring to the modified Neer classification (Table 1) 9 . The function status was assessed at every examination point according to the Musculoskeletal Tumor Society (MSTS) functional evaluation system and the Activities of Daily Living Scale (ADLs) 10,11 .The adverse effect was evaluated according to the common terminology criteria for adverse events (CTCAE).
Modified Neer Classification of Bone Defect Healing.

a With or without small nonprogressive radiolucent area(s) less than 1 cm in size.
Statistical Analysis
Statistical analysis was performed using SPSS, version 20.0, software (IBM Corp., Armonk, NY, USA). Data were presented as mean ± standard deviation. Paired
Results
General Data
No statistical difference was found in age, gender, diagnosis, lesions location, or defect size between the two groups (
General Data of Patients in Two Groups.

ABGs: allogeneic bone granules; iTEBs: individual tissue-engineered bones.
Perioperative Parameters
The average operative time was 113.6 ± 62.9 and 116.7 ± 51.1 min for ABGs and iTEBs, respectively. The average blood loss was 197.2 ± 253.4 and 173.0 ± 230.0 ml for ABGs and iTEBs, respectively. The average hospitalization stay was 11.9 ± 4.1 and 10.5 ± 2.8 d for ABGs and iTEBs, respectively. No significant difference was found in these values (
Comparison of Peri- and Postoperative Data.

ABGs: allogeneic bone granules; CTCAE: common terminology criteria for adverse events; iTEBs: individual tissue-engineered bones.
Radiographic Evaluation
Postoperative X-rays were accessible in all patients with a mean radiological follow-up of 10 yr. The average healing time was notably longer in the group of ABGs (6.2 ± 2.3 mo; range 3 to 12 mo), as compared with iTEBs (4.4 ± 2.0 mo; range 3 to 9 mo). Analysis on healing time is shown in Fig. 2. Moreover, 3 cases (12%) treated with ABGs had tumor recurrence and received autogenous iliac bone transplantation at postoperative 2, 3, and 8 yr, respectively. For iTEBs, only 1 case (4.3%) with tumor recurrence received grafting of iTEBs again at 3 yr postoperatively. In the group of ABGs, 18 cases (72%) achieved complete defect healing (Neer score Ⅰ), 6 cases (24%) healed with defects (Neer score Ⅱ), and 1 case (4%) had persistent defect (Neer score Ⅲ) at the final follow-up. Healing results of iTEBs included 22 cases (95.7%) with complete healing (Neer score Ⅰ) and 1 case (4.3%) healed with defects (Neer score Ⅱ). A statistical difference existed in healing quality between the two groups, as detailed in Table 3. The X-rays of representative cases are displayed in Figs. 3 –5.

Comparison of healing time by Kaplan–Meier survival analysis. The healing time of iTEBs was significantly shorter than that of ABGs (

Images of a 7-yr-old female with bone cyst in the right radius where iTEBs were implanted: (A) the X-ray before surgery; (B) the X-ray at 3 d postoperatively; (C) the X-ray at 3 mo postoperatively; (D) the X-ray at 5 yr postoperatively; (E) the X-ray at 10 yr postoperatively. The bone healing was classified as Neer classification score Ⅰ. iTEBs: individual tissue-engineered bones.

Images of an 11-yr-old male with nonossifying fibroma in the left femur where iTEBs were implanted: (A) the X-ray before surgery; (B) the X-ray at 3 d postoperatively; (C) the X-ray at 6 mo postoperatively; (D) the X-ray at 1 yr postoperatively; (E) the X-ray at 6.5 yr postoperatively. The bone healing was classified as Neer classification score Ⅱ. iTEBs: individual tissue-engineered bones.

Images of a 14-yr-old male with fibrous dysplasia in the left tibia where ABGs were implanted: (A) the X-ray before surgery; (B) the X-ray at 3 d postoperatively; (C) the X-ray at 9 mo postoperatively; (D) the X-ray at 6 yr postoperatively; (E) the X-ray at 10 yr postoperatively. The bone healing was classified as Neer classification score Ⅲ. ABGs: allogeneic bone granules.
Clinical Results
In both groups, the MSTS and ADLs scores were significantly improved at 3 mo postoperatively, as compared with the preoperative records (
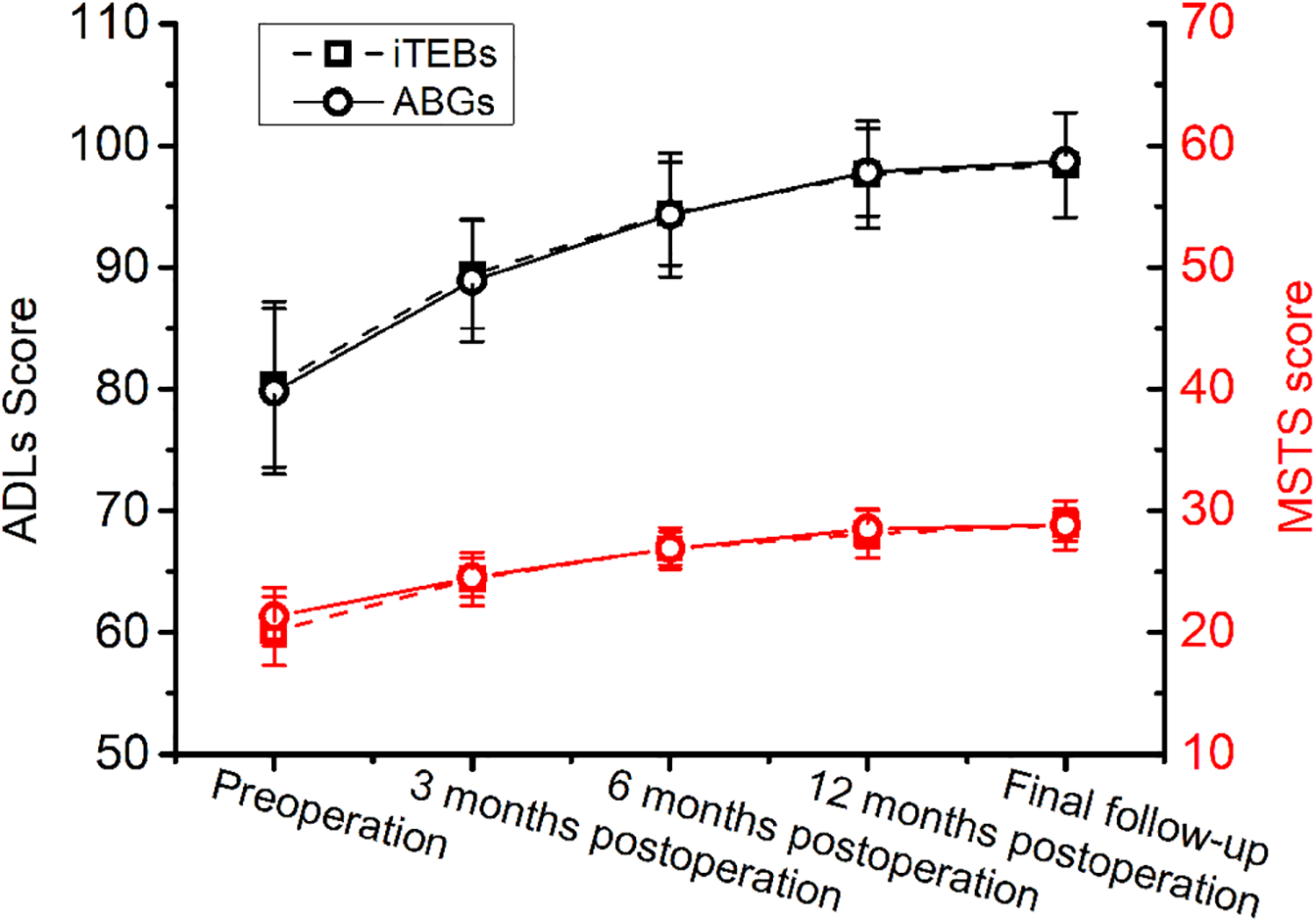
Clinical outcomes at different time points. Compared with the preoperative data, MSTS and ADLs scores were significantly improved at 3 mo postoperatively (
Complications
In the group of ABGs, adverse events were detected in five patients, including lesion recurrence (three), internal fixation failure (one), and pathological fracture (one). According to the CTCAE, three cases were considered as grade 2 and two cases as grade 3. In the group of iTEBs, a 13-yr-old male with bone cyst in the left humerus suffered from lesion recurrence at 3 yr after the implantation of iTEB. Emission computed tomography (ECT) examination was performed and no lesion was found in other sites. After the second extended curettage and iTEB implantation, the bone defect was reconstructed at 6 mo postoperatively. According to the CTCAE, the case was considered as grade 2. The reoperation rate was 20% and 4.3% in the groups of ABGs and iTEBs, respectively. No nerve damage, incision infection, or amputation was recorded in either group. In the long-term follow-up, no disease transmission, tumor formation, or immunological rejection was detected after grafting with iTEBs (Table 3).
Discussion
There has been broad consensus in dealing with benign bone tumors and tumor-like lesion, including correct diagnosis, symptom relief, function retention or recovery, and complication remission. Although various treatment strategies are considerable, curettage with bone defect reconstruction is the most common and reliable method to acquire entire bone remodeling and reduce fracture risk. However, the currently available grafts, including autologous, allogeneic bones, bioactive glass, hydroxyapatite (HA), and tricalcium phosphate, have encountered difficulty in clinical application due to their respective shortcomings.
Since first reported by Quarto et al. 12 , tissue-engineered grafts have been broadly implemented in clinic, covering bone defects caused by trauma, articular cartilage defect 13 , spinal fusion 14 , and particularly oral and maxillofacial surgeries 15 . With regard to their application in bone tumors or tumor-like lesions, Morishita et al. 16 successfully repaired massive bone defects caused by tumor curettage in three patients with grafts fabricated by patients’ MSCs and HA ceramics. In the present study, we retrospectively evaluated the long-term efficacy and safety of iTEBs in repairing bone defects, with ABGs as control. The results suggested that treatment with iTEBs was effective as the clinical symptoms, such as limb pain and activity disorder, were significantly relieved and the limb function was obviously restored.
In this study, iTEBs exhibited evident superiority in healing time, indicating the positive roles of the administrated MSCs. This was consistent with results from our and others’ previous animal experiments, which demonstrated that the introduction of MSCs into scaffolds significantly improved osteogenesis and led to a comparable efficacy to autografts 17 . Moreover, Hernigou et al. 18 found that allografts combined with bone marrow-derived MSCs possessed stronger osteogenic capacity than blank scaffolds and seemed equivalent to autografts in hip revision. However, controversy still existed because some researchers demonstrated that MSCs contributed nothing to bone regeneration induced by DBM 19 . This might be attributed to the impairment of local blood supply during the creation of the bone union model, which influenced the bioactivity of implanted cells or even led to cell death. In the present study, almost 60% patients who received iTEBs transplantation had bone healing after 3 mo, which may be related to the multiple differentiation potential of MSCs. As previous literature reported, MSCs can differentiate into osteoblasts and participate in the bone regeneration 20 . In addition, MSCs can secrete extracellular matrix and osteogenesis-related factors, which induce the recruitment and migration of host cells to aid bone repair 21 . Anyway, this study revealed that the advantage of iTEBs over ABGs in healing time was definite with regard to treating bone defects caused by tumor curettage.
Better bone defect remodeling means lower incidence of complication. Although bone healing was achieved in either group, iTEBs produced better performance in terms of bone healing quality, as revealed by radiological results. It was noteworthy that incomplete bone healing was associated with many factors. Via intramedullary decompression and DBM implantation, Cho et al. 22 treated unicameral bone cyst in 25 cases and reported partial bone healing in 4 cases with no clinical symptom at the final follow-up. As they supposed, partial bone healing was caused by insufficient DBM in quantity. In another study, Horstmann et al. 23 proposed the view point that the deterioration of complete initial grafting resulted from implant resorption and local lesion recurrence. Additionally, in a canine model, Hall et al. 24 reported that cancellous allograft chips without new bone formation were enveloped by fibrous tissue, which could hamper bone reconstruction. In spite of these influencing factors, the advantage of iTEBs in bone healing quality might be partially attributed to the implanted MSCs. Evidence from our studies and current literature suggests that implanted MSCs are beneficial for local angiogenesis and osteogenesis by promoting the migration and proliferation of angiogenic and osteogenic progenitor cells via specific signal pathways 25 –27 .
The incorporation of MSCs brings various influence factors into the properties of TEBs, such as cellular biological characteristics, the in vitro culture, and construction process. Accordingly, controversies occur on the long-term safety of TEBs. Pan et al. 28 reported that the spontaneous tumorigenic transformation could come up after long-term cultures (beyond 5 wk) of human MSCs. Besides, different degrees of foreign body reaction may be caused by internal fixation, prosthesis, or biomaterials 29 . During approximately10-yr follow-up, no immunological rejection or malignant transformation was found in this study. Moreover, the incidence of postoperative complications and reoperation associated with the iTEBs was lower than ABGs. In addition, no blood-borne disease such as viral hepatitis, syphilis, or HIV disease was found in either group. Collectively, these findings indicated the long-term safety of iTEBs. Consistent results were also achieved by other MSCs-based grafts 30 –32 .
Certain limitations exist in the present study. First, bone healing was only assessed through radiographic and clinical observation. Biomechanical and histological evaluation is helpful to increase the confidence level but not available in this study. Second, layer analysis to display and determine other factors associated with bone healing was not performed due to the restriction of sample size. Finally, there were some limitations and difficulties when working with MSCs, such as long construction period, strict quality control and safety criteria, and particular equipment and qualified technicians. This issue may be addressed in accordance with the standardized protocol previously described by our group 7 . Larger-sample, multicenter, and prospective comparative trials are called on for further identification.
Conclusion
In the present study, we retrospectively compared the long-term outcomes of ABGs and iTEBs in treating benign bone tumors and tumor-like lesions. Compared with ABGs, iTEBs were tagged with more rapid healing speed, better healing outcome, and lower complication and reoperation rates. Regardless of inconvenience in preparation, iTEBs were safe and effective in the reconstruction of bone defects caused by tumor curettage. Eventually, bone tissue engineering may provide promising strategies in repairing bone defects, especially for patients with limited autogenous bones.
Footnotes
Authors’ Contributions
PY analyzed the data and wrote the manuscript. JZ and QA collected the data. BY and MD prepared iTEB. FL, ZX, and TH performed the surgery. JX and TH conceived and designed this study. All authors read and approved the final manuscript.
Ethical Approval
Ethical approval to report this case was obtained from “the Medical Ethics Committee of Southwest Hospital.”
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the protocols approved by “the Medical Ethics Committee of Southwest Hospital.”
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Foundation of Southwest Hospital (SWH2016JCZD-10), the National Natural Science Foundation of China (Grant No. 81874009), and the Youth Development Project of Military Medical Science (16QNP097).
