Abstract
Mesenchymal stem cells (MSCs) are thought to have great potential in the therapy of acute liver injury. It is possible that these cells may be regulated by the stromal cell-derived factor-1 (SDF-1)/CXC chemokine receptor-4 (CXCR4) signaling axis, which has been shown to promote stem cells migration in the inflammation-associated diseases. However, the effects of SDF-1/CXCR4 axis on the MSCs-transplantation-based treatment for acute liver injury and the underlying mechanisms are largely unknown. In this study, we sought to determine whether SDF-1/CXCR4 would augment the therapeutic effect of bone marrow mesenchymal stem cells (BMSCs) by promoting their migration, which may result from activating the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway, in a rat acute liver injury model induced by lipopolysaccharide (LPS). We found that BMSCs transplantation markedly attenuated liver injury and improved the survival of LPS-treated rats. Of interest, overexpression of CXCR4 in BMSCs could substantially promote their migration both in vitro and in vivo, and result in even better therapeutic effects. This might be attributed to the activation of PI3K/Akt signaling pathway in BMSCs that is downstream of CXCR4, as demonstrated by the use of the CXCR4 antagonist AMD3100 and PI3K pathway inhibitor LY294002 assays in vitro and in vivo. Together, our results unraveled a novel molecular mechanism for the therapeutic effect of BMSCs for the treatment of acute liver injury, which may shed a new light on the clinical application of BMSCs for acute liver failure.
Keywords
Introduction
Sepsis threatens the life of ICU patients and is associated with high morbidity and mortality. It mainly happens because of systemic inflammation resulting from gram-negative and gram-positive bacteria, fungi, parasites, and viruses. In the past 30 years, the incidence rate of sepsis had been increasing year by year, along with the rising sepsis-related costs. A study showed that sepsis cost almost $15.4 billion every year, 4.3% of which are hospital costs. It is estimated that sepsis has become the sixth most common cause of hospitalization 1 . Previous reports revealed that inappropriate treatment would cause sepsis to develop into septic shock and multiple organ dysfunction syndrome (MODS), which results in greater difficulties in critical care and high mortality. It is well known that liver has a rich blood supply and is one of the most vulnerable organs to sepsis. Liver damage commonly reduces survival rate in sepsis patients. Sepsis-related liver damage often manifests syndromes such as liver microcirculation damage, liver cell energy metabolism disorders, liver cell degeneration, necrosis, liver cell inflammatory infiltration, etc 2 –4 .
Bone marrow mesenchymal stem cells (BMSCs) are a type of multipotent stem cells that possess distinct characteristics from those of hematopoietic stem cells, such as the capacity of transectoderm differentiation. BMSCs can proliferate and differentiate into osteoblasts, chondrocytes, and adipocytes in vitro under certain conditions. Moreover, with lower immunogenicity, BMSCs have fewer probability to be rejected after transplantation into the body. Accumulating evidences support BMSCs as a useful tool for the therapy of various inflammation-associated diseases. For example, BMSCs could reduce damage due to systemic reactive inflammatory response syndrome and lung injury induced by severe acute pancreatitis. BMSCs play a role in modulating pathological changes such as alveolar edema, hemorrhage, and even severe inflammatory exudation in the damaged lungs. They also inhibit the release of tumor necrosis factor-α and attenuate the extent of acute lung injury 5 . BMSCs could also reduce proximal renal tubular epithelial cell injury, and renal apoptosis improve renal function and reduce mortality in acute kidney injury mice 6 . Additionally, BMSCs have been found to prevent depression induced by traumatic brain injury 7 .
More and more evidences reveal that multiple cytokines and signaling pathways could regulate the therapeutic effects of BMSCs, both in vitro and in vivo. It has been demonstrated that stromal cell-derived factor-1 (SDF-1) and its receptor, CXC chemokine receptor-4 (CXCR4), which are two important cytokines under both physiological and pathological conditions, combine and play a crucial role in regulating the migration and proliferation of various types of stem cells, such as epidermal stem cells, cardiac stem cells, etc 8 . In a recent study of mesenchymal stem cells (MSCs) transplantation for the treatment of traumatic brain injury, SDF-1/CXCR4 signaling was found to be important in facilitating the migration of transplanted cells to the brain injury area. This is associated with a reduction in the number of apoptotic neurons and recovery of nerve function 9 . Other reports indicate the effects of SDF-1/CXCR4 axis on cardiogenic cells’ differentiation into pluripotent stem cells, which enhanced the host cardiogenic development 10 . On the other hand, the SDF-1/CXCR4 axis is also implicated in chronic stomach inflammation, chronic bronchitis, and with the formation and progress of various types of tumors 11 –13 . Alsayed et al 11 found that the SDF-1/CXCR4 axis could regulate the migration, adhesion, and invasion of tumor cells in the bone marrow. This important signaling axis has also been found to be related to the progression of colorectal cancer, endometrial cancer, and cholangiocarcinoma 14 –16 . Phosphoinositide 3-kinase (PI3K)/Akt, which is a major downstream pathway of SDF-1/CXCR4, has also been found to facilitate tumor progression, participate in the development of ischemic injury diseases, and regulate cell proliferation and migration 17 –19 . Our previous studies have demonstrated that BMSCs play an anti-inflammatory role in MODS rats induced by lipopolysaccharide (LPS) 20 . However, the underlying mechanisms of the effects of BMSCs are largely unknown. Since it is widely accepted that SDF-1/CXCR4 is associated with the migration of stem cells in vivo, in this study, a rat model of LPS-induced liver injury was employed to explore the molecular mechanism of BMSCs’ therapeutic effects on acute liver injury. This will give a deeper insight into more effective therapeutic strategy of BMSCs transplantation in clinical LPS-induced liver injury by gene intervening strategies associated with PI3K/Akt signaling pathway.
Materials and Methods
Construction of pLV-GFP-2A-CXCR4 Recombinant
The coding sequence of rat-CXCR4 gene was retrieved from GenBank, and the restriction enzyme digestion sites were analyzed and determined according to the sequence of pLV-GFP-2A, a lentiviral expression vector. The synthetic primers and templates were linked by polymerase chain reaction (PCR) amplification. A total of 10 µl connection products were extracted for transformation, monoclonal screening, and bacterial fluid amplification. Some colonies were selected for sequencing identification. The expression plasmid was prepared using the Endo-free Plasmid Mini kit II (200) (Omega, Norcross, GA, USA), and transfected into 293Ta lentivirus packaging cell line. After transfection, the expression level of CXCR4 gene was detected by real-time quantitative polymerase chain reaction (Q-PCR). Subsequently, the recombinant of lentiviral expression plasmid, packaged plasmid complex, and Lipo2000 diluent were mixed together and transfected into 293Ta cells for 48 h. Supernatant was collected, centrifuged, and filtered, and virus concentrate was harvested and stored in a −80°C refrigerator.
Isolation, Cultivation, and Identification of BMSCs
Female Sprague Dawley (SD) rats, aged 4–6 weeks, were anesthetized and sacrificed by cervical vertebra dislocation. The rats were soaked with 75% ethanol for 10 min, then the femur was dissected and separated and quickly brought to the ultra-clean table. The femur was cut on both ends, and the bone marrow in the central cavity was flushed out with serum-free Dulbecco’s modified Eagle’s medium/nutrient mixture F12 (DMEM/F12, 1:1) by using a 5-ml syringe and centrifuged at 200 × g for 5 min. Then, they were resuspended and expanded in DMEM/F12 medium supplemented with 10% fetal bovine serum and 1% penicillin (100 U/ml)/streptomycin (0.1 mg/ml) at 37°C in a 5% CO2 cell culture incubator. Twenty-four hours later, nonadherent cells were removed and fresh culture medium was added. The medium was changed thoroughly every 3 days. Upon reaching 80%–90% confluence, the adherent cells as passage 0 (P0) were trypsinized with 0.25% trypsin and 0.01% ethylenediaminetetraacetic acid and subcultured at 1:2 (volume) as the first generation (P1) in cell culture flask T-75 at 37°C in a 5% CO2 cell culture incubator. These procedures were repeated two times and the cultures were referred to as P2 and P3. According to phenotypic characterization of rat BMSCs biomarkers, these cells expressed high levels of MSC surface markers (CD90 and CD44) and low levels of hematopoietic surface markers (CD34 and CD45).The cells of third generation (P3) were identified by flow cytometry, then used for transplantation.
Lentivirus Transfection of BMSCs
The third-generation cells with good growth status were selected and inoculated into six-well plates with density of 1 × 105 cells per well. The next day, pLV-GFP-2A-CXCR4 recombinant and/or empty vector virus was dissolved on ice, respectively, and according to the multiplicity of infection (MOI) = 1, 10, 20, 50, 100, 150, and 200, lentivirus of corresponding volume and polybrene with final concentration of 6 μg/ml was added and cultured in an incubator with 5% CO2 at 37°C. The fluorescence intensity of each well of cultured cells was measured under the fluorescence microscope, and cells in the well with the strongest fluorescence were harvested. These were screened by flow cytometry and seeded to obtain a CXCR4 overexpressing BMSCs cell line (CXCR4-BMSCs).
TdT-mediated dUTP Nick-End Labeling Staining
BMSCs and CXCR4-BMSCs with good growth status were used in this experiment. After the cells were harvested and counted, the density of the cell suspension was adjusted to 1 × 105/ml, and a total of 100 µl cell suspension was added onto the cell slide. SDF-1, AMD3100 (CXCR4 antagonist), and LY294002 (PI3K pathway inhibitor) were added to each group. Cell slides of each group were taken out, fixed and stained, dried and photographed under a fluorescence microscope (DSX510i, OLYMPUS, Tokyo, Japan). A total of five visual fields were randomly selected for apoptosis rate calculation. The formula for apoptosis rate was: apoptosis rate = number of fluorescent cells/total number of cells.
Cell Proliferation Assay
Cell proliferation was detected with a Cell Counting Kit (CCK)-8 kit (Promega, Madison, WI, USA). The cells were divided into four groups: BMSC group, CXCR4-BMSC group (CXCR4-overexpression BMSCs), AMD3100 group (CXCR4-BMSCs + CXCR4 antagonist AMD3100), and LY294002 group (CXCR4-BMSCs + PI3K pathway inhibitor LY294002). BMSCs and CXCR4-BMSCs were cultured in 96-well plates, respectively. After the cells have adhered to the culture plate, AMD3100 (10 μg/ml, Abcam, Cambridge, UK) and LY294002 (15 μmol/L, Abcam) were added, respectively. Three wells were selected for each group at 12, 24, 48, and 72 h, respectively. The culture medium was replaced. Optical density value was measured at 450 nm.
Transwell Migration Assay
The transwell migration experiment was conducted using a transwell plate (8 μm, Millipore, Boston, MA, USA), to explore the effect of BMSC migration in different concentrations of SDF-1, with addition of DMEM–F12 medium (100 ng/ml) in the lower chamber. Migration efficiency was observed 12 h after addition of 1 × 104 BMSCs to the upper chamber. The migration experiment was divided into four groups. In each group, 1.5 ml DMEM–F12 medium containing 100 ng/ml of SDF-1 was added to the lower chamber. The upper chambers were seeded with BMSCs, CXCR4-BMSCs, 10 μg/ml AMD3100-treated CXCR4-BMSCs, and 15 μmol/L LY294002-treated CXCR4-BMSCs, respectively. The density of cells added in each chamber was 1 × 104, 500 µl in volume. Transwell plate was taken out after 12 h, and the cells were carefully scraped off with cotton swabs. Formaldehyde (4%) was used for fixation for 15 min, and the number of migrated cells was counted under the microscope.
Experimental Groups
SD rats were provided by the Animal Experimental Center of Kunming Medical University. Rat care was in full compliance with the Guide for the Care and Use of Laboratory Animals and the Use of Laboratory Animals in Research, and all studies were approved by the Animal Ethical Committee of Kunming Medical University. Healthy female SD rats, weighing 180–220 g, were fed with food and water ad libitum, and bred in 12 h/12 h light/dark relay under room temperature. They were randomly divided into six groups (n = 8 per group): I Control group: only sterile phosphate buffer saline was injected via the femoral vein at the same volume as capacity as with the other groups, II Liver injury group: (LPS injection + normal saline treatment), III BMSC group (LPS injection + green fluorescent protein [GFP]-BMSCs treatment), IV CXCR4-BMSC group (LPS injection+ CXCR4-overexpression BMSCs treatment), V AMD3100 group (LPS injection + GFP-BMSCs and CXCR4 antagonist AMD3100 treatment), and VI LY294002 group (LPS injection + GFP-BMSCs and PI3K pathway inhibitor LY294002 treatment). Rats in groups II–VI were treated with 1 mg/kg LPS (Sigma-Aldrich, St. Louis, MO, USA) injection via femoral vein, then GFP-BMSCs or CXCR4-BMSCs (2 × 106 cell/kg, 300 μl) were transplanted within 6 h following LPS injection via caudal vein, respectively. AMD3100 (10 mg/kg, Abcam) or LY294002 (40 mg/kg, Abcam) was intraperitoneal injected 30 min, respectively, before cell transplantation. The behavioral changes such as activity, appetite, and defecation of rats were observed, and the blood and liver tissues were collected 3 days after transplanting treatment.
Biochemical Assays and Histological Evaluation
The survival rate of the rats was calculated, and rats were sacrificed 3 days after modeling. The abdominal aortic blood was collected, and the serum levels of total bilirubin (Tbil), aspartate aminotransferase (AST), and alanine aminotransferase (ALT) were detected. After blood collection, normal saline was perfused into the heart, and the liver turned yellowish. The liver was removed and fixed in the 4% paraformaldehyde for 24 h, and paraffin embedding was conducted. Blocks were sectioned at 5 µm thickness. After paraffin section was dewaxed and hydrated, hematoxylin and eosin staining was used to observe the changes of liver structure and inflammatory response in each group under the microscope.
Immunohistochemical Staining
All lentiviruses used in this study carried the GFP gene and could express the GFP after transfection. Therefore, the number of cells migrated to the liver was indirectly detected by GFP staining. Paraffin sections of the liver were dewaxed, hydrated successively, and treated with 3% H2O2 for 10 min. They were then incubated with sodium citrate antigen retrieval solution for 5 min, followed by blocking serum for 30 min. Anti-GFP antibody (1:200, Proteintech, Chicago, IL, USA) was then added and incubated at 4°C overnight. This was followed by incubation with secondary antibody at 37°C for 20 min. Diaminobenzidine (DAB) was added for transparence and reacted for 2–5 min. The slides were then washed, restained with hematoxylin and eosin (H&E), dehydrated, mounted, and observed under the microscope.
Real-time Q-PCR
Three days after cell transplantation, the liver tissues were harvested using the same method as described earlier. Total RNA was extracted on ice, and the concentration was measured and calculated. Subsequently, cDNA, primer, and enzyme were mixed in a ratio of 1:2:10 and detected by real-time quantitative fluorescence PCR. Cycle threshold (Ct) values were obtained for analysis. The levels of mRNA were expressed with a relative quantitative method with 2−△△Ct. The primer sequences required in the experiment were: SDF-1 (forward 5’-TTTGAGAGCCATGTCGCCA-3’, reverse 5’-TGTCTGTTGTTGCTTTTCAGCC-3’), CXCR4 (forward 5’-GAGGCGTTTGGTGCTCCGGT-3’, reverse 5’-ACGGACAGGTGCAGCCGGTA-3’), PI3K (forward 5’-TCTCCGTAGCGGGGCACCAG-3’, reverse 5’-AACCAGCCAATATCTTCTGG-3’), Akt (forward 5’-ACCTCTGAGACCGACACCAG-3’, reverse 5’-AGGAGAACTGGGGAAAGTGC-3’), and β actin (forward 5’-CGTTGACATCCGTAAAGA-3’, reverse 5’-CTAGGAGCCAGGGCAGTAATC-3’).
Western Blotting
Fresh liver specimens were cut into small pieces using sterile scissors, with addition of RIPA buffer (Thermo Scientific, Rockford, IL, USA). The centrifugal parameter is 4°C, 12,500 RPM, 15 min. The supernatant was extracted with addition of five times loading buffer. Following denaturation, sodium dodecyl sulfate polyacrylamide gel electrophoresis was performed. After membrane transformation and primary antibody incubation, including SDF-1 (1:1,000, Abcam), CXCR4 (1:100, Abcam), PI3K (1:1,000, CST, Boston, MA, USA), and phospho-Akt (1:1,000, CST) antibody, enhanced chemiluminescence was employed to obtain the corresponding bands containing target protein. Image J software (National Institutes of Health, Bethesda, MD, USA) was used for data analysis.
Statistical Analysis
All quantitative data were expressed as mean ± standard error of the mean. One-way analysis of variance followed by post hoc Tukey’s test and chi-square test were used. A level of P < 0.05 was considered statistically significant.
Results
Establishment of BMSCs Overexpressing CXCR4
BMSCs were successfully transfected with CXCR4 gene after incubation with lentivirus for 72 h, as shown by green fluorescence. Fluorescence was the strongest when MOI = 150 (Fig. 1A, B). Q-PCR and western blotting showed that levels of CXCR4 mRNA and protein in the CXCR4-BMSC group were significantly higher than those of the BMSC group, respectively (Fig. 1C–E). Together, results confirm overexpression of the CXCR4 gene in BMSCs.
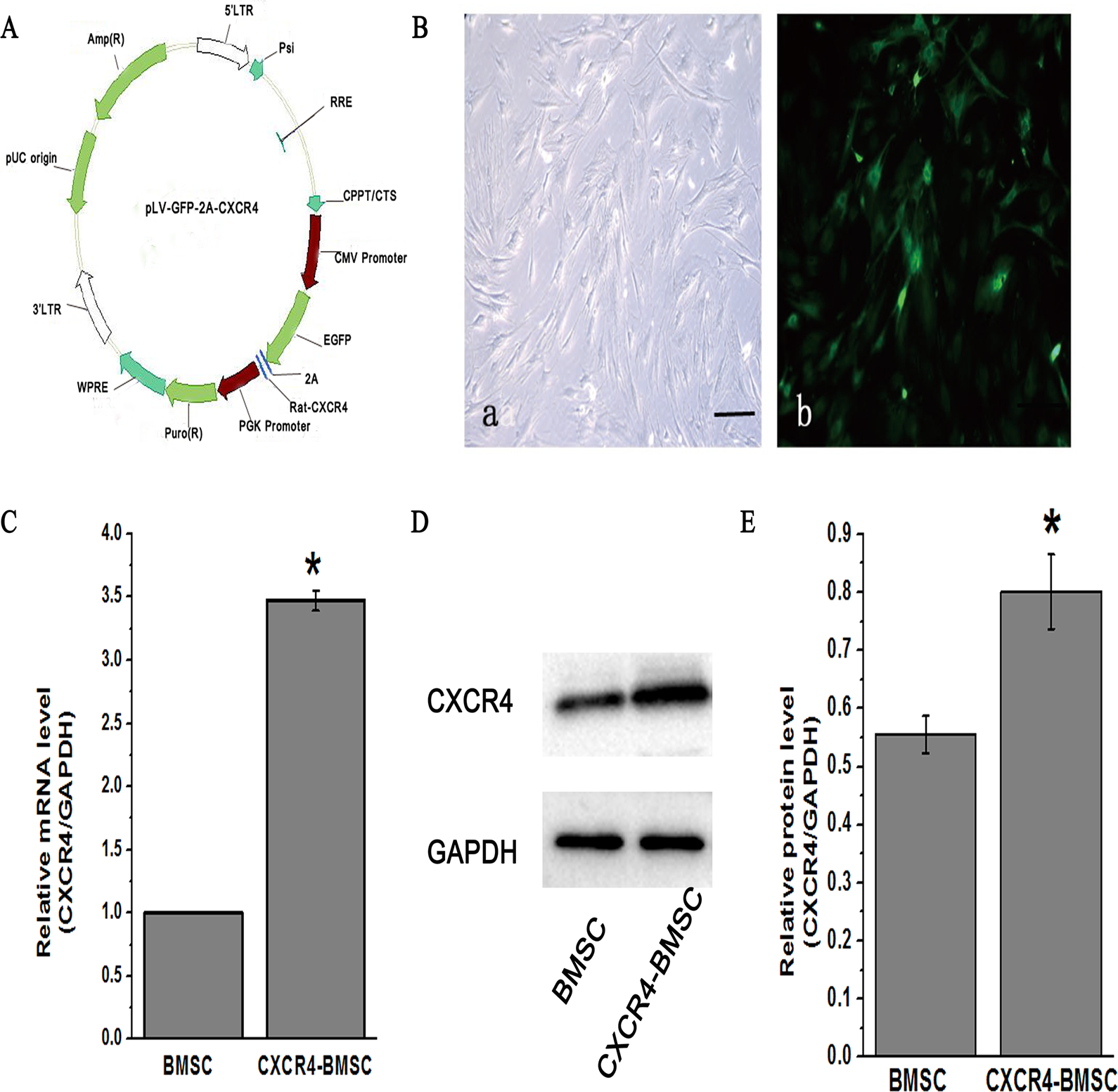
Characterization of CXCR4-BMSCs. (A) Plasmid mapping of pLV-GFP-2A-CXCR4. (B) Photomicrographs of BMSCs and transfected cells. At 72 h after the infection with the CXCR4 lentivirus detected by phase contrast microscopy and fluorescence microscope (×100, scale bar = 100 μm). (C) The levels of CXCR4mRNA in BMSC and CXCR4-BMSC group were analyzed by Q-PCR. (D) The levels of CXCR4 protein were detected by western blotting. (E) Quantitative analyses of CXCR4 protein levels were shown in the graph. *P < 0.05 versus BMSC group. BMSC: bone marrow mesenchymal stem cell; CXCR4: CXC chemokine receptor-4; Q-PCR: quantitative polymerase chain reaction.
CXCR4 Promoted BMSCs Proliferation and Inhibited Their Apoptosis
Compared with the BMSC group, the CXCR4-BMSC group had markedly lower apoptotic rate (P < 0.05). The apoptotic rate of AMD3100 (CXCR4 antagonist) group and LY294002 (PI3K pathway inhibitor) group was higher than that of CXCR4-BMSC group, respectively, although there was no significant difference (Fig. 2A, B). Cell proliferation assay revealed that BMSCs with CXCR4 overexpression have enhanced proliferation ability, which was remarkably reversed and further inhibited by AMD3100 and LY294002 treatment, respectively (Fig. 2C). These results suggest that CXCR4 gene overexpression plays a crucial role in improving BMSCs proliferation, while interference with CXCR4 markedly inhibited the proliferation of BMSCs.

Effects on apoptosis and proliferation of BMSCs induced by CXCR4 in vitro. (A) TUNEL staining in BMSC group, CXCR4-BMSC group, AMD3100 group, and LY294002 group (×200, scale bar = 50 μm). (B) Quantitative analyses of apoptosis rate were shown in the graph. (C) Cell proliferation by CCK8 assay was shown in the line graph. *P < 0.05 versus BMSC group. BMSC: bone marrow mesenchymal stem cell; CXCR4: CXC chemokine receptor-4; TUNEL: TdT-mediated dUTP Nick-End Labeling.
SDF-1/CXCR4 and PI3K/Akt Signaling Pathways Involved in BMSCs Migration
Compared with the BMSC group, the CXCR4-BMSC group induced by 100 ng/ml SDF-1 showed a higher number of cells migrating to the lower compartment, as counted in five visual fields (P < 0.05). In contrast, the numbers of cells migrating to the lower chamber in CXCR4-BMSCs that have been treated by AMD3100 or LY294002 were markedly fewer than that of CXCR4-BMSC group, respectively (P < 0.05) (Fig. 3A, B).

SDF-1/CXCR4 axis and PI3K/Akt signaling pathways were involved in BMSCs migration in vitro. (A) BMSCs were detected by transwell assays for 12 h in BMSC group, CXCR4-BMSC group, AMD3100 group, and LY294002 group (×100, scale bar = 100 μm) and quantitative analysis was shown in the graph (B). (C) Quantitative analyses of the mRNA levels of CXCR4, PI3K, and Akt in each group by Q-PCR. (D) Western blotting was performed on each group of cells and quantitative analysis was shown in the graph (E). *P < 0.05 versus BMSC group, #P < 0.05 versus CXCR4-BMSC group, △P < 0.05 versus AMD3100 group. BMSC: bone marrow mesenchymal stem cell; CXCR4: CXC chemokine receptor-4; PI3K: phosphoinositide 3-kinase; Q-PCR: quantitative polymerase chain reaction; SDF-1: stromal cell-derived factor-1.
As shown by Q-PCR, the mRNA expression levels of CXCR4, PI3K, and Akt in the CXCR4-BMSC group were significantly higher than those of BMSC group (P < 0.05), while AMD3100 and LY294002 treatment modulated the increase in expression and reversed and further inhibited the above effects (P < 0.05) (Fig. 3C). Western blotting analysis showed changes in protein expression that are consistent with the mRNA changes (Fig. 3D, E).
BMSCs Transplantation Improved the Survival Rate and Repaired Liver Damage
The survival rate of rats in each BMSCs transplantation group was significantly increased compared with the liver injury group (Fig. 4A). Moreover, assay of blood samples showed that Tbil, AST, and ALT levels in BMSC, CXCR4-BMSC, AMD3100, and LY294002 group were markedly lower than those of liver injury group (P < 0.05). The reduction in AST and ALT in the CXCR4-BMSC group was the most obvious among all the groups (Fig. 4B–D).
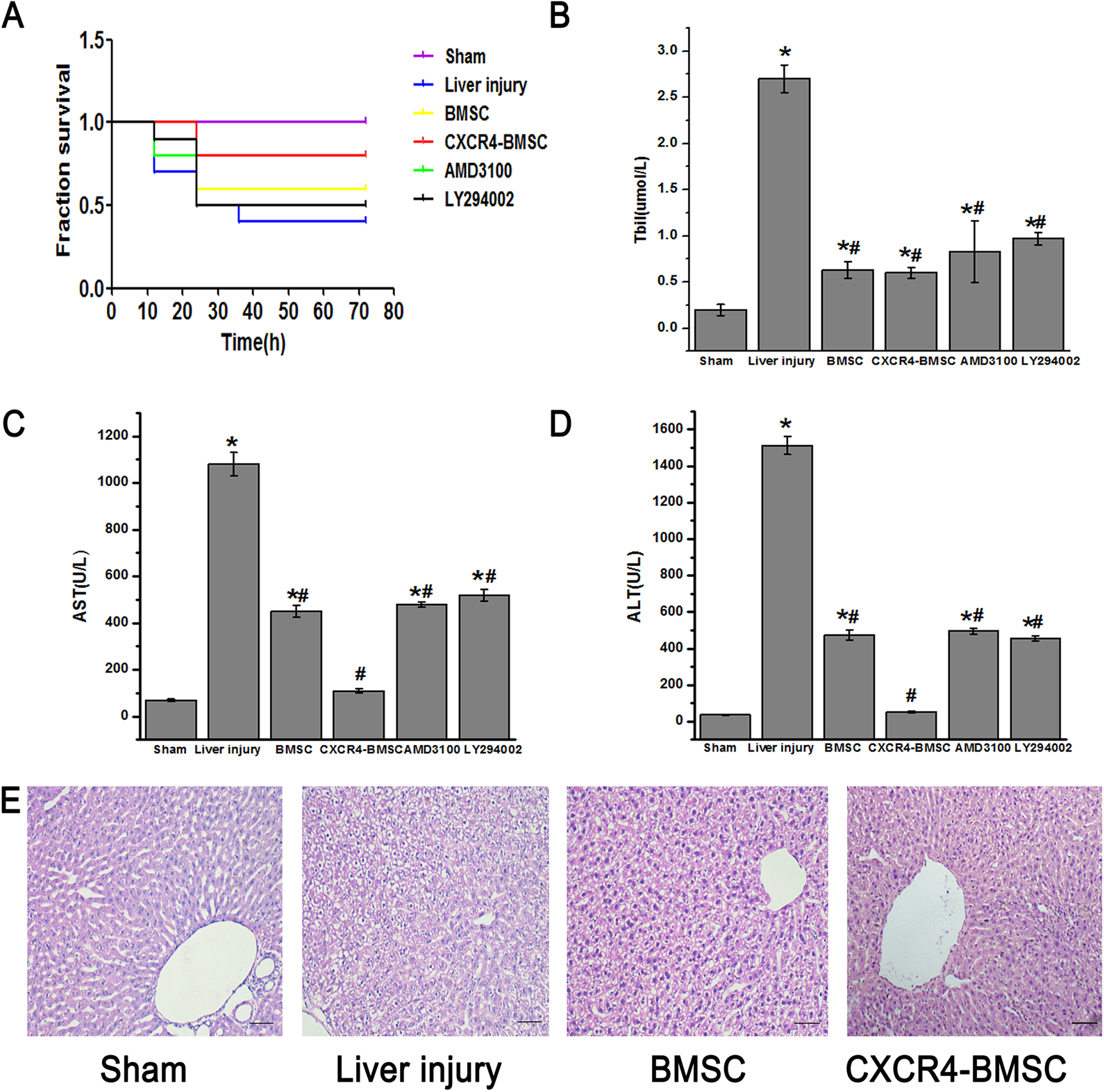
BMSCs transplantation improved the survival rate and liver repair in vivo. (A) The survival rate of rats in sham group, BMSC group, CXCR4-BMSC group, AMD3100 group, and LY294002 group. The Tbil, AST, and ALT levels in each group were shown in the graphs (B) to (D). (E) The H&E staining of each group, the sham group with clear hepatic lobule structure, and no inflammatory cells. A large number of liver cell degeneration, necrosis, and inflammatory cell infiltration can be seen in the liver injury group (×200, scale bar = 50 μm). *P < 0.05 versus sham group, #P < 0.05 versus liver injury group. ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMSC: bone marrow mesenchymal stem cell; CXCR4: CXC chemokine receptor-4; H&E: hematoxylin and eosin; Tbil: total bilirubin.
Additionally, H&E staining showed a large number of degenerative and necrotic hepatocytes in the liver of rats in the liver injury group, with obvious inflammatory cell infiltration and loss of hepatic sinus structure. These changes were significantly rescued by BMSCs transplantation both in the BMSC group and CXCR4-BMSC group, and liver pathological changes in the CXCR4-BMSC group were also markedly less than those of BMSC group (Fig. 4E).
SDF-1/CXCR4 and PI3K/Akt Pathway Regulated BMSCs’ Homing in Liver Injury Rats
GFP immunostained sections of the liver revealed that the average optical density of GFP-positive cells in CXCR4-BMSC group was the greatest among all the groups (P < 0.05). GFP immunohistochemical stain showed significantly lower average optical density in LY294002 group and AMD3100 group relative to the BMSC group (P < 0.05), respectively (Fig. 5A, B).

SDF-1/CXCR4 axis and PI3K/Akt pathway regulated BMSCs homing in liver injury rats. (A)The GFP staining of liver in BMSC group, CXCR4-BMSC group, AMD3100 group, and LY294002 group (×200, scale bar = 50 μm) and the optical density analyses of GFP staining were shown in the graph (B). (C) Quantitative analyses of the mRNA of SDF-1, CXCR4, PI3K, and Akt in each group by Q-PCR. (D) The liver protein expressions of SDF-1, CXCR4, PI3K, and Akt in each group were detected by western blotting and quantitative analyses were in the graph (E). *P < 0.05 versus BMSC group, △P < 0.05 versus AMD3100 group. BMSC: bone marrow mesenchymal stem cell; CXCR4: CXC chemokine receptor-4; GFP: green fluorescent protein; PI3K: phosphoinositide 3-kinase; Q-PCR: quantitative polymerase chain reaction; SDF-1: stromal cell-derived factor-1.
Q-PCR showed the mRNA expression levels of SDF-1, CXCR4, PI3K, and Akt in the CXCR4-BMSC group were substantially higher than those of BMSC group, respectively (P < 0.05). However, they were significantly less of mRNA levels of above key molecules in the AMD3100 group and LY294002 group compared with the CXCR4-BMSC group, respectively (P < 0.05). Additionally, compared with AMD3100 group, those in LY294002 group were further reduced (P < 0.05, Fig. 5C). Western blotting showed protein changes that were consistent with the mRNA findings (P < 0.05) (Fig. 5D, E
Discussion
It has been widely accepted that the SDF-1/CXCR4 axis could promote stem cells to migrate in the inflammation-associated diseases, but the effects of SDF-1/CXCR4 axis on the BMSCs based transplanting therapy for acute liver injury and the associated underlying mechanism remain unknown. Based on the above notion, we proposed that SDF-1/CXCR4 could improve the therapeutic effect of BMSCs on downregulating serum markers such as AST and ALT, and reducing mortality in rats with acute liver injury. This could be the basis for the pathological improvement found in this study, including attenuation of cellular necrosis and inflammation in liver cells, and promotion of recovery of hepatic lobule structure. The above therapeutic effects based on the roles of transplanted BMSCs were most likely dependent on the activation of PI3K/Akt signaling pathway in the rat acute liver injury model induced by LPS, which deepened the insight into the optimal stem cell transplantation study using gene interventional strategies. So, we told a complete story regarding the effects of BMSCs transplantation on the rescue of liver injury of rats which relied on liver function and pathology recovery tightly related to activation of the PI3K/Akt signaling pathway mediated by SDF-1/CXCR4 axis. Importantly, we elucidated both the material basis and molecular mechanism underlying BMSCs transplantation therapy using BMSCs. To our knowledge, it is a first and integrated report, and will open up a bright way for future liver injury clinical treatment by using BMSCs transplantation combining with gene interventional strategy.
In this study, it was found that SDF-1, as a kind of chemotactic factor, when under the condition of liver injury induced by LPS, was markedly upregulated in expression, which recruited BMSCs to migrate to the injured area and played roles in rescuing and improving the liver injury. This is a good interpretation for the synergetic changes of SDF-1, CXCR4, and the BMSCs migration into the liver injured area dramatically found in this study. Upon arrival at the injury site, BMSCs play an active role in attenuating liver injury, including significantly decreasing the size of the liver injury area, inhibiting liver cell apoptosis, and simultaneously promoting liver cell proliferation. These findings are accordance with previous reports of the positive effects of BMSCs in other pathological conditions 21 –34 . Thus, from this study, we extended the scope of pathological conditions that could benefit from BMSCs transplanting therapy to LPS or sepsis-induced acute liver injury. Furthermore, we used an antagonist of CXCR4 and an inhibitor of the PI3K/Akt pathway to elucidate possible roles of the interaction of SDF-1/CXCR4 and PI3K/Akt signaling axis in the beneficial effects of BMSCs transplanting. Results showed that the mRNA levels of SDF-1/CXCR4 in CXCR4-BMSCs transplanted group were markedly higher, which was consistent with the larger number of transplanted BMSCs homing in the liver. In contrast, the expression level of CXCR4 in the liver of AMD3100 group was found to be significantly lower, corresponding to fewer number of homing transplanted BMSCs. It is therefore likely that SDF-1/CXCR4 activation under liver injury not only greatly supported the BMSCs’ behavior for rescuing the liver injury but also regulated the homing of transplanted BMSCs.
PI3K/Akt is an important downstream pathway that could regulate the migration of various cells, including platelets and dental pulp stem cells, via the SDF-1/CXCR4 axis. It has been demonstrated that PI3K/Akt signaling plays an active and positive role in BMSCs transplanting for inflammation-associated and central nervous systemic diseases 35 –41 . Although SDF-1/CXCR4 is well known for its roles in regulating the migration and proliferation of various types of stem cells, there have been few reports regarding the relationship between PI3K/Akt and SDF-1/CXCR4, especially after liver injury under the condition of BMSCs transplantation 42 –47 . It has been found in this study that PI3K/Akt plays a crucial role in facilitating the migration of CXCR4-BMSCs based on the effects of its antagonist, AMD3100, in significantly reducing the migration of CXCR4-BMSCs. We also found that in the LY294002 intervention group, the PI3K/Akt pathway was inhibited and the number of homing of GFP-BMSCs transplanted to the liver was significantly reduced. Therefore, it is concluded that PI3K/Akt pathway serves as an important pathway, in tandem with SDF-1/CXCR4, which is its upstream pathway, during BMSCs transplantation to the liver. These results strongly suggest that both these signaling pathways are tightly associated with BMSC migration and liver function recovery in liver injured rats.
Taken together, our results strongly suggest that PI3K/Akt signaling pathway, activated synchronously with SDF-1/CXCR4, together play a crucial role in the migration and homing of transplanted BMSCs, thereby facilitating the repair of damaged liver after injury. Importantly, PI3K/Akt and SDF-1/CXCR4 are consistently activated in BMSCs during transplantation therapy in the liver injured rat model. In summary, BMSCs can effectively promote the recovery of rats with liver injury caused by LPS, and the mechanism might be related to the activation of PI3K/Akt mediated by SDF-1/CXCR4, and its distinctive role in promoting BMSCs migration and homing under BMSCs transplanting therapy.
Footnotes
Ethical Approval
All procedures involving animals in this study were approved by the Ethics Committee of Experimental Animals, Kunming Medical University, Kunming, China.
Statement of Human and Animal Rights
All the experimental procedures were carried out in accordance with the Ethics Committee of Experimental Animals, Kunming Medical University, Kunming, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (81901950, 81360289), Association Foundation Program of Yunnan Provincial Science and Technology Department and Kunming Medicine University (2017FE468(-180), 2019FE001(-009), 2018FE001(-078), 2015FB079), Basic Research Project of Yunnan Province (2019FB099), Yunnan Health Training Project of High Level Talents (H-2017060), and Xu Jun’s Expert Work Station (2017IC025).
