Abstract
Adult-derived human liver stem/progenitor cells (ADHLSCs) are a promising alternative to orthotopic liver transplantation in the treatment of inborn errors of metabolism. However, as is the case with many mesenchymal stromal cells, ADHLSCs have shown a low level of engraftment, which could be explained by the fact that they lack expression of selectin ligand and LFA-1 and only slightly express VLA- 4, molecules that have been shown to be involved in cell adhesion to the endothelium. In this paper, we have investigated strategies to increase their rolling and adhesion during the homing process by (1) adding a selectin ligand (Sialyl Lewis X) to their surface using biotinyl-N-hydroxy-succinimide–streptavidin bridges, and (2) protecting the adhesion proteins from trypsinization-induced damage using a thermosensitive polymer for cell culture and a nonenzymatic cell dissociation solution (CDS) for harvest. Despite increasing adhesion of ADHLSCs to E-selectin during an adhesion test in vitro performed under shear stress, the addition of Sialyl Lewis X did not increase adhesion to endothelial cells under the same conditions. Cultivating cells on a thermosensitive polymer and harvesting them with CDS increased their adhesion to endothelial cells under noninflammatory conditions, compared to the use of trypsin. However, we were not able to demonstrate any improvement in cell adhesion to the endothelium following culture on polymer and harvest with CDS, suggesting that alternative methods of improving engraftment still need to be evaluated.
Introduction
Mesenchymal stromal cells (MSCs) have been isolated, cultured, and characterized from diverse adult organs (bone marrow, adipose tissue, umbilical cord, peripheral blood, and liver) 1 –6 . Their regenerative and immunomodulatory properties are currently under evaluation in multiple clinical trials to treat a wide variety of diseases (cancer, inflammatory diseases, heart stroke, degenerative diseases, wound, and metabolic diseases) (www.clinicaltrials.gov). In 2007, Najimi et al. discovered a mesenchymal cell population called adult-derived human liver stem/progenitor cells (ADHLSCs), which has the capacity to differentiate into hepatocyte-like cells and repopulate the liver after hepatectomy in a murine model 7 . In addition, our group has demonstrated that ADHLSCs can inhibit hepatic stellate cells, which make them a potential candidate in the treatment of fibrosis 8 . ADHLSCs do not trigger tumor formation following transplantation and are currently in clinical trials for the treatment of inborn errors of metabolism (urea cycle disorders) and inflammatory disorders (acute-on-chronic liver disease) 9,10 . However, their level of engraftment in the recipient liver post transplantation remains fairly low, despite the fact that they seem poorly immunogenic in vitro 7,11 . This could be due to their adhesion profile, which is similar to that of other MSCs 11,12 .
We postulated that ADHLSCs, like MSCs from various organs, probably use homing and engraftment mechanisms similar to those used by leukocytes during inflammation 13 . The cells will first roll on the endothelium 14 , then firmly adhere to it 14 –16 , and finally transmigrate 17 to the targeted organ’s parenchyma. Contrary to leukocytes, MSCs and ADHLSCs do not express selectin ligands such as PSGL-1, which are involved in rolling. Alternatively, some groups have demonstrated that CD44, which is also expressed by ADHLSCs, could be used by MSCs to perform rolling; however, it would need to be fucosylated 15,16 . MSCs do not express LFA-1 either, which is used to bind ICAM-1 during the firm adhesion phase; only VLA-4 is expressed, which should allow binding to VCAM-1 and transmigration, but the expression is weak 14,18 . That is also the case for ADHLSCs 12 .
Because of the adhesion profile of MSCs, rolling and adhesion to the endothelium are considered as a weak point of their engraftment. Many groups have tried to improve engraftment by a variety of ways: fucosylating CD44 19 , adding Sialyl Lewis at the cell surface 16 for rolling, increasing the expression of CXCR4 by transfection 20 or culturing the cells in the presence of an inflammatory cocktail 21 , improving the expression of VLA-4 by transfection 22 , or priming the cells with extracellular matrix components 23 or with a proinflammatory cocktail 24 .
All of these studies have tried to either increase rolling on selectin, or increase firm adhesion. In this paper, we have investigated a combination of methods to increase rolling and adhesion of ADHLSCs to the liver endothelium during infusion. First, we added a selectin ligand to the surface of the cells by a biotinyl-N-hydroxy-succinimide (BNHS)/biotin streptavidin bridge, in order to increase rolling. Second, we decided to protect the adhesion proteins of the ADHLSCs from potential trypsinization-induced damage, as several groups have already shown trypsin to have a negative effect on MSCs adhesion 25 –27. To this end, we cultured the cells on a thermosensitive polymer (UpCell), which allows the harvest of the cells through a conformational change of the polymer resulting from a change in temperature, and added a nonenzymatic cell dissociation solution (CDS) to obtain a single cell suspension.
Materials and Methods
ADHLSC Isolation and Culture
ADHLSCs were obtained subsequent to the primary culture of the liver parenchymal fraction recovered after two-step collagenase perfusion of the organ, followed by filtration and low-speed centrifugation, as described elsewhere 28 . ADHLSCs were cultured on CellBIND® flasks (Corning®, Lasne, Belgium) in Dulbecco’s modified Eagle’s medium (DMEM) containing 4.5 g/l glucose (ThermoFisher Scientific, Erembodegem, Belgium), supplemented with 10% fetal calf serum (ThermoFisher Scientific) and 1% penicillin/streptomycin (P/S) (ThermoFisher Scientific), at 37°C under a fully humidified atmosphere (5% CO2). Upon reaching 80% confluence, the cells were lifted with 0.05% trypsin-EDTA (ThermoFisher Scientific) and replated at a density of 5,000 cells/cm2 and cultured until passage 5. The viability of the recovered cells was evaluated using the trypan blue dye exclusion method.
Alternative Culture and Harvest Methods
ADHLSCs were seeded at 5,000 cells/cm2 on a thermosensitive polymer, using the UpCellTM (ThermoFisher Scientific) technology. Upon reaching 70% to 80%, the dishes were left at room temperature (RT) with 10 ml of cold medium (DMEM with 10% fetal bovine serum [FBS], 1% P/S) for 30 min and the cells were harvested. Where indicated, the cold medium was replaced with 4 ml of cold nonenzymatic CDS (Sigma-Aldrich, Overijse, Belgium). The dishes were incubated for 15 min at RT, then the cells were harvested with 8 ml of DMEM with 10% FBS and 1% P/S. As a control (CTL), ADHLSCs were cultured on CellBIND® dishes and harvested either with trypsin, or with CDS as described above. Then the cells were centrifuged for 10 min at 1,200 rpm and resuspended in serum-free DMEM containing 4.5 g/l glucose (ThermoFisher Scientific) with 1% P/S (ThermoFisher Scientific). The concentration and viability of the cells were evaluated by the trypan blue exclusion method.
Prestoblue Viability and Proliferation Assay
ADHLSCs were seeded at 5,000 cells/cm2 on CellBIND® dishes or on a thermosensitive polymer, using the UpCellTM technology. Upon reaching 70% to 80% confluence, the prestoblue assay was performed by removing the culture medium and replacing it with complete medium containing a 1/10 dilution of the prestoBlue solution (ThermoFisher Scientific). After 24 h, the resulting fluorescence was measured using a VICTOR X4 2030 Multilabel Reader (Perkin Elmer, Zaventem, Belgium).
Ki67 Staining
ADHLSCs were seeded at 5,000 cells/cm2 on CellBIND® dishes or on a thermosensitive polymer, using the UpCellTM technology. Upon reaching 70% to 80% confluence, cells were fixed using 4% paraformaldehyde for 10 min, incubated with 3% hydrogen peroxide solution, rinsed twice with distilled water and incubated with 1% triton X-100 (Sigma-Aldrich) for 10 min. Samples were then washed twice with phosphate-buffered saline (PBS), incubated with 0.1% bovine serum albumin (BSA; Sigma-Aldrich) to block nonspecific staining, and incubated with mouse anti-human Ki67 antibody (DAKO, Agilent technologies, Diegem, Belgium) diluted 1/50 for 1 h. After three washes in PBS, the samples were incubated with secondary antibody (Envision, DAKO). Samples were finally washed with PBS, incubated with diaminobenzidine (DAB, DAKO) for 5 min, washed twice more, and incubated with Mayer’s hematoxylin for 10 min. After two final washes in distilled water, slides were mounted with glue, allowed to dry, and analyzed on a Leica DMIL inverted microscope (Leica, Diegem, Belgium).
Hepatogenic Differentiation
After one passage on thermosensitive polymer or on CellBIND® dishes, ADHLSCs were seeded at the density of 10,000 cells/cm2 in T25 Corning® BioCoat™ flasks in DMEM supplemented with 10% FBS and 1% P/S. After 3 d, the culture medium was switched to Iscove’s modified Dulbecco’s medium (IMDM) (ThermoFisher Scientific) and 1% P/S. Cells were differentiated according to Najimi et al. with some modifications 6 . Briefly, cells were incubated with 20 ng/ml of epidermal growth factor (Peprotech, London, UK) for 6 d. Then, cells were incubated with IMDM containing 20 ng/ml hepatocyte growth factor (Peprotech), 1% insulin-transferin-selenium (ThermoFisher Scientific) premix, 20 ng/ml oncostatin M (Peprotech), and 1 µM dexamethasone (Sigma-Aldrich) for 12 d. For each step, medium was changed every 3 d. At the end of the differentiation protocol, cells were harvested for CYP3A4 activity test or quantitative reverse transcription polymerase chain reaction (RT-qPCR) analysis of differentiation markers.
RT-qPCR
Total RNA was extracted using TriPure isolation reagent (Roche, Mannheim, Germany), following the manufacturer’s instructions. Briefly, cells were homogenized in TriPure reagent, mixed with chloroform, shaken vigorously for 15 s, and centrifuged at 12,000 × g for 15 min at 4°C. RNA in the upper aqueous phase was precipitated by isopropanol, washed in 75% ethanol, air-dried, and dissolved in RNase-free water. RNA samples were stored at −80°C after quantification with a NanoDrop 2000 spectrophotometer (ThermoFisher Scientific).
Complementary DNA was synthesized from 1 µg of total RNA by RT-PCR following DNAse treatment, using a high-capacity kit (Applied Biosystems, part of ThermoFisher Scientific). Vimentin, albumin, and CYP3A4 gene expression was then evaluated by real-time qPCR using Taqman® Gene Expression Assays (Hs00185584_m1, Hs00910225_m1, and Hs00604506_m1, respectively) and master Mix in a StepOnePlus thermocycler. The results were normalized to the housekeeping genes RPL37A (Hs01102345_m1) and TBP (Hs99999910_m1).
CYP3A4 Activity Test
The quality of the hepatogenic differentiation was evaluated using a CYP3A4 activity test according to the manufacturer’s instructions (Promega, Leiden, The Netherlands). Briefly, 100,000 cells from each condition were centrifuged, resuspended in phenol-free IMDM supplemented or not with luciferin-IPA substrate, and incubated for 4 h at 37°C under humidified atmosphere. Luciferase detection reagent was then added, and the mixture shaken for 5 min, and further incubated for 15 min before bioluminescence reading on a VICTOR X4 2030 Multilabel Reader.
Sialyl Lewisx (SLeX) Modification
The conjugation of biotinylated Sialyl Lewisx (BSLeX) to the ADHLSCs’ surface through biotin–streptavidin bridges was performed in PBS at RT. ADHLSCs were harvested with the different methods described earlier and washed with PBS. The resulting cell pellet was dispersed in sulfonated BNHS solution (1 mM, 1 ml), and allowed to incubate for 10 min at RT. The cells were then washed with PBS once to remove unattached and/or physically adsorbed BNHS from the cell surface. A streptavidin solution (50 μg/ml in PBS, 1 ml) (Sigma-Aldrich) was then used to treat the cells for 10 min at RT. The cells were washed with PBS. A BSleX solution (5 μg/ml in PBS, 1 ml) (Glycotech, Gaithersburg, MD, USA) was added to the streptavidin-conjugated cells, and the suspension was allowed to incubate for 10 min at room temperature. Finally, the cells were washed with PBS and resuspended in serum-free DMEM containing 4.5 g/l glucose (ThermoFisher Scientific) with P/S (ThermoFisher Scientific). The concentration and viability of the cells were evaluated by the trypan blue exclusion method.
Adhesion Test In Vitro
Ibidi μ-slides Luer 0.6 (Ibidi, Gräfelfing, Germany) were coated with either protein (VCAM-1 at 20 μg/ml, E-selectin at 5 µg/ml (R&D Systems, Abingdon, UK), or rat tail collagen type I at 50 μg/ml (BD Biosciences, Erembodegem, Belgium) overnight at RT, or with human umbilical vein endothelial cells (HUVECs) concentrated at 2 × 106 cells/ml and incubated for 18 to 24 h at 37°C 5% CO2, in the presence or absence of tumor necrosis factor alpha (TNF-α) 100 ng/ml (R&D Systems). Nonspecific protein-binding sites were blocked using DMEM 4.5 g/l glucose with 10% FBS and 1% P/S for 5 min. ADHLSCs harvested with the different conditions described earlier (CB trypsin, CB CDS, polymer, polymer CDS) with or without SLeX addition to the surface of the cells were resuspended at 0.5 × 106 cells/ml in serum-free media and perfused over protein- or HUVEC-coated slides at 0.5 dynes/cm2 to mimic physiological shear stress. ADHLSCs were injected for 2 min. Binding was maximized by stopping the flow for 4 min. The flow was then restarted with serum-free DMEM for 5 min, pictures were taken, and the number of cells remaining adherent was recorded over 30 fields. Cells were counted with the ImageJ software. Data are expressed as the mean adherent cell number by optic field. To confirm the interaction between VLA-4 and VCAM, a blocking anti-alpha 4 antibody was used at 50 µg of antibody for 1 × 106 cells incubated for 30 min at RT before perfusion into the μ-slides (R&D Systems). Untreated cells were used as a CTL.
Flow Cytometry
ADHLSCs harvested with the different methods were washed with PBS-BSA 1.5%. Nonspecific binding sites were blocked for 20 min in PBS–BSA 1.5% at 4°C, then the cells were stained with phycoerythrin (PE)-labeled antibodies against adhesion molecules (Table 1) for 30 min at 4°C, and washed twice with PBS–BSA 1.5%. Fluorescence was measured with a BD FACS Canto II cytometer (BD Biosciences, Erembodegem, Belgium) on 10,000 cells using the FACSDiva software. Data analyses were performed with the FlowJo software. The number of receptors per cell was determined using QuantiBRITETM (BD Biosciences), as recommended by the manufacturer.
Antibodies Used to Stain ADHLSCs After Different Harvesting Methods.

ADHLSC: adult-derived human liver stem cell.
Cell Adhesion to the Endothelium Ex Vivo
All animal procedures were performed in accordance to the Commission d’Ethique pour l’Expériementation Animale of the Université catholique de Louvain-approved protocols (approval 2018/UCL/MD/036).
ADHLSCs grown on CellBIND® or polymer were harvested with trypsin or CDS, respectively, enumerated using the trypan blue dye exclusion method and resuspended in culture medium at a concentration of 0.5 × 106 cells/ml. C57BL6 mice were anesthetized using ketamine and xylazine. An incision was made in the abdomen and organs were moved to the side to allow visualization of the portal vein. A butterfly was placed in the portal vein, and the inferior vena cava was sectioned. The liver was washed off blood by perfusing with PBS for 5 min using a syringe pump. Then 1 million cells were injected over a 2-min time period. Then the liver was washed with PBS for an additional 20 min. At the end of the experiment, the liver was harvested and placed in 4% paraformaldehyde. After overnight fixation at RT, samples were embedded in paraffin. Five micrometer sections were deparaffinized and rehydrated in successive baths of xylene (VWR, Oud-Heverlee, Belgium), isopropanol (Acros Organics, Thermo Fisher Scientific), and water. Samples were then incubated in citrate buffer (pH = 6) at 98°C for 1 h, washed with PBS, blocked with 5% PBS–BSA, and incubated with anti-human β-integrin antibody(Bioke, Leiden, The Netherlands) diluted 1/300 in 5% PBS–BSA overnight at 4°C. After three washes in PBS-Tween 0.5%, the samples were incubated with horseradish peroxidase-conjugated secondary antibody (DAKO) for 30 min at RT, washed another three times, and incubated with DAB at RT for 5 min. After washing in water, the samples were stained with Mayer’s hematoxylin for 3 min, and dehydrated in successive baths of water, isopropanol, and xylene, before mounting. Slides were scanned using the SCN400 slide scanner (Leica). Beta-integrin positive cells were then quantified using the visiopharm software (2IP imaging platform of the IREC Institute), distinguishing between positive cells that appeared to be in contact with the vessel or already in the parenchyma (adherent cells) and the cells that were circulating. Adhesion was expressed as a percentage of adherent cells.
Statistical Analyses
Each outcome was analyzed according to the following steps. First, a robust average of the technical replicates was computed for each donor and each condition. Then, a mixed-effect model was built to compare the different conditions encoded in a variable, which was included as a fixed effect of the model. A random patient effect was also introduced to model the interdonor variability. A logarithmic transformation was applied on each outcome in order to meet the assumptions of the statistical model (i.e., residuals with normal distribution and homogeneity of variance). The estimates of the coefficients of the model were back-transformed in order to compute the fold-change associated with each condition and compared to the reference group. Results were considered significant when the P-values were
Results
SLeX Can Efficiently Be Added to the Surface of ADHLSCs
We wanted to determine if adding SLeX groups to the surface of the cells would improve their rolling/adhesion potential. To this end, we first investigated the possibility of adding SLeX groups to the surface of the cells using BNHS, as previously described 16 . The efficiency of the linking between BNHS and the cells was assessed by incubating the cells that had been preincubated in the presence or absence of BNHS with different concentrations of streptavidin-fluorescein isothiocyanate (FITC). For cells preincubated with BNHS, the higher the streptavidin-FITC concentration, the higher the resulting fluorescence intensity (Fig. 1A). In contrast, cells that had not been preincubated with BNHS only showed background fluorescence regardless of the concentration of streptavidin-FITC. This result confirmed an efficient link between BNHS and the ADHLSCs.

Impact of SleX addition on adhesion to selectins. ADHLSCs were preincubated in the presence or absence of BNHS, then cells were incubated with different concentrations of streptavidin-FITC. The resulting fluorescence was evaluated using a victor X2 plate reader (fluorescence intensity per ADHLSC donor; circles, squares, and triangles represent different donors; n = 3) (A). Adhesion of SLeX+ and SLeX− ADHLSCs to P-selectin-coated µ-slides (B) and E-selectin µ-slides (C) (robust average of 30 optic fields per ADHLSC donor; circles, squares, and triangle represent different donors, n = 3).
SLeX Addition Improves Adhesion of ADHLSCs to E-Selectin but Not to P-Selectin
To evaluate the rolling/adhesion potential of ADHLSCs decorated with Sialyl Lewis X groups, we performed an adhesion test onto µslides coated with P- and E-selectin, using PBMCs as a positive CTL. Addition of SLeX did not improve adhesion of ADHLSCs to P-selectin-coated µ-slides under shear stress of 0.5 dyne/cm2 compared to ADHLSCs without SLeX (Fig. 1B). It did, however, significantly increase their adhesion to E-selectin-coated µ-slides by an 84.19-fold change (q < 0.01) (Fig. 1C).
Culture on a Thermosensitive Polymer has No Significant Effect on the Phenotypic Characteristics of ADHLSCs but Reduces ADHLSC Proliferation
Before assessing the effectiveness of the thermosensitive polymer in increasing adhesion, we first investigated the stability of the characteristics of ADHLSCs grown on UpCell™ for one passage. We found that ADHLSCs had the same morphology on UpCell™ than on CellBIND® (Fig. 2A), but appeared to grow slower on the thermosensitive polymer (Fig. 2A). This was confirmed by a reduction in proliferation as demonstrated by a reduced relative fluorescence in the Prestoblue Assay (Fig. 2B) and a lower percentage of Ki67-positive cells (Fig. 2C), as well as a lower yield, as shown in Table 2 (less than 50% ± 8.9% after polymer compared to CB trypsin). Moreover, the mesenchymal characteristics of ADHLSCs did not change after one passage on UpCell™, as demonstrated by their expression of CD73, CD90, and CD105 assessed by flow cytometry (Fig. 2D). Finally, we demonstrated that growth on the thermosensitive polymer did not affect ADHLSCs’ capacity to differentiate into hepatocyte-like cells, as shown by their cobblestone morphology (Fig. 3A), as well as their reduced expression of vimentin (Fig. 3B), and increased expression of albumin (Fig. 3C) and CYP3A4 (Fig. 3D) following differentiation, which were all comparable to cells grown on CellBIND®. In addition, their Cyp3A4 activity post-differentiation was not significantly different from that of ADHLSCs grown on CellBIND® (Fig. 2E).

Stability of ADHLSCs after culture on thermosensitive polymer: morphology, proliferation, and phenotype. Representative pictures of three ADHLSC donors grown on CellBIND® or on thermosensitive polymer at passage P5 (objective ×10) (A). Comparison of the proliferative capacity of ADHLSCs grown on CellBIND® or on thermosensitive polymer using the PrestoBlue assay (B) or Ki67 staining (C). Comparison of the expression of the MSC markers CD73, CD90, and CD105 by flow cytometry by ADHLSCs after culture on CellBIND® or on thermosensitive polymer (red histogram: isotype, blue histogram: marker of interest) (D).
Harvesting Data.

Comparison of CB trypsin, CB CDS, polymer alone and polymer CDS with respect to yield, cell viability, and ease of detachment of the cells (mean ± SD, n = 4, *P < 0.05, **P < 0.01, ***P < 0.001).
CB trypsin: cells grown on CellBIND® and harvested with trypsin; CB CDS: cells grown on CellBIND® and harvested with CDS; CDS: cell dissociation solution; polymer CDS: cells grown on the thermosensitive polymer and harvested with the CDS.

Stability of ADHLSCs after culture on thermosensitive polymer: hepatogenic differentiation capacity. Comparison of the hepatocyte-like differentiation capacity of ADHLSCs after culture on CellBIND® or on thermosensitive polymer: representative morphology (objective ×10, n = 4) (A), vimentin (B), albumin (C), and CYP3A4 expression (D) and CyP3A4 activity (fold change of differentiated/undifferentiated (E) ADHLSCs donor; circles, squares, diamonds, and triangles represent different donors, n = 4).
Comparison of Alternative Harvesting Methods
Our previous experiments comparing cells grown on CellBIND® or polymer revealed that cells grown on polymer were difficult to detach from each other (data not shown). Therefore, we tried combining growth on polymer to harvest with a nonenzymatic CDS. To investigate the efficiency of such alternative harvesting methods, cells grown on CellBIND® and harvested with trypsin (CB trypsin) were used as a CTL, and compared to (1) cells grown on the thermosensitive polymer (polymer) alone and (2) cells grown on the thermosensitive polymer and harvested with the CDS (polymer CDS) (condition of interest). In addition, to evaluate the contribution of CDS alone to the results, we also compared the previous conditions to cells grown on CellBIND® and harvested with CDS (CB CDS). First, we observed that CB trypsin remained the easiest and quickest method to harvest cells compared to all nonenzymatic alternative solutions. Furthermore, it was also easier to count the cells by the trypan blue exclusion method after CB trypsin than other methods, especially compared to polymer alone due to the cells’ tendency to create aggregates. Moreover, cell viability showed a decrease for all three alternative harvesting methods compared to CB trypsin. However, cell viability remained over 80% for all methods (Table 2). A more problematic issue was the yield obtained. Taking CB trypsin as a 100% reference, the yield dropped drastically for every other condition: 54.4% ± 24.6% for CB CDS, 50% ± 8.9% for polymer, and 46.4% ± 7.7% for polymer CDS (Table 2).
Impact of Culture on Thermosensitive Polymer and Harvest with CDS on the Adhesion of ADHLSCs to VCAM-1 and Collagen Type 1
Next, the capacity of the cells from different conditions to adhere to collagen type I and VCAM-1 was tested. ADHLSCs at 500,000 cells/ml were sent over an Ibidi luer slide 0.6 coated with VCAM-1 or collagen type I at 0.5 dyne/cm2 using a syringe pump. Adherent cells were counted over 20 to 30 optic fields after 5 min of rinsing and reported as number of cells per optic field. We found no significant improvement in cell adhesion to collagen type I-coated µ-slides following the use of the thermosensitive polymer and/or CDS (Fig. 4A).

Impact of different conditions of culture and harvesting on adhesion to VCAM-1 and collagen type I. Adhesion of ADHLSCs after different conditions of culture and harvesting on collagen type I-coated µ-slides (A) and VCAM-1-coated µ-slides (B) (robust average of 30 optic field per ADHLSC donor; circles, squares, diamonds, and triangles represent different donors; n = 4). Adhesion test on VCAM-1-coated µ-slides of ADHLSCs after CB trypsin and polymer CDS following blocking of integrin alpha-4 with a specific antibody (C).
Cells grown on polymer and harvested using CDS showed a 1.45-fold increase in adhesion to VCAM-1 compared to the CB trypsin CTL (Fig. 4B). However, this increase was not significant (q = 0.35) due to the fairly low number of donors. Similar to adhesion to collagen type I coating, there was no difference in adhesion to VCAM-1 coating between cells harvested after CB CDS and CB trypsin (fold change of 0.85 and 0.95 for adhesion to VCAM-1 and collagen type I, respectively). Culture on polymer and harvest with cold medium alone even seemed to decrease the adhesion to VCAM-1 and to collagen type I by a fold change of 0.51 and 0.72, respectively; however, this decrease was not significant (q = 0.14 and q = 0.44).
To investigate the specificity of cell adhesion to VCAM-1, the interactions with VLA-4 were blocked using an antibody against integrin alpha 4. As expected, this resulted in a significant inhibition of cell adhesion to VCAM-1 in both the CB trypsin and the polymer CDS conditions (fold change of 0.01, q < 0.01) (Fig. 4C).
Culture on the Thermosensitive Polymer Followed by Harvest Using CDS Improves the Adhesion of ADHLSCs to HUVECs Under Noninflammatory Conditions, but SLeX Addition Does Not
Next, we investigated the potential synergistic effect of culture on polymer followed by harvest with CDS and the addition of SLeX on adhesion. Cells cultivated under control (CB + trypsin) or alternative (polymer + CDS) conditions were either left untreated or incubated with BNHS followed by SLeX. Cells were then passed onto naive or activated HUVEC-coated μ-slides. As endothelial cells under normal conditions express very little E-selectin (Fig. 5A), we did not expect SLeX to improve ADHLSCs’ adhesion to HUVECs under naïve conditions. Indeed, we did not see any significant changes in adhesion to naïve HUVECs following these two harvesting conditions (Fig. 5B). On the other hand, culture on polymer and harvest with CDS increased the adhesion of ADHLSCs to naïve HUVECs compared to CB trypsin by a 5.36-fold change (q = 0.1) (Fig. 5B). Next, we investigated cell adhesion to HUVECs under inflammatory conditions by preincubating HUVECs with 100 ng/ml of TNF-α. First, we confirmed that HUVECs under inflammatory conditions have an upregulated expression of E-selectin and VCAM-1 compared to nonactivated HUVECs (Fig. 5A). Consistent with the results obtained on naïve HUVECs, we did not notice a significant change in cell adhesion after addition of SLeX (Fig. 5C). In fact, we observed a decrease for both harvesting methods after addition of SLeX (0.66-fold change for CB trypsin and 0.33-fold change for polymer CDS) (Fig. 5C), which was consistent with the slight decrease already noticeable on naive HUVEC following addition of SLeX to cells from the polymer CDS condition. On the other hand, cell adhesion to HUVEC under inflammatory conditions was naturally higher than adhesion to naïve HUVECs for both culture/harvesting methods, and there was no significant difference between polymer CDS and CB trypsin (fold change of 1.29, q = 0.52). To investigate the role of VLA-4 in ADHLSCs’ adhesion to HUVEC, we blocked the interactions between VLA-4 and VCAM-1 by preincubating ADHLSCs with an antibody blocking VLA-4. As expected based on the poor expression of VCAM-1 by naïve HUVECs, blocking VLA-4 using a specific antibody did not change the adhesion of ADHLSCs to naïve HUVECs regardless of the harvesting method, and the adhesion was still higher after polymer CDS by a 5.75-fold change compared to CB trypsin (q = 0.1) (Fig. 5D). On the other hand, adhesion of ADHLSCs to activated HUVEC drastically dropped for both conditions by a fold change of 0.22 for CB trypsin (q = 0.1) and 0.32 for polymer CDS (q = 0.06), but interestingly adhesion was higher for polymer CDS than CB trypsin after blocking VLA-4 by a fold change of 1.88 (q = 0.1) (Fig. 5D).
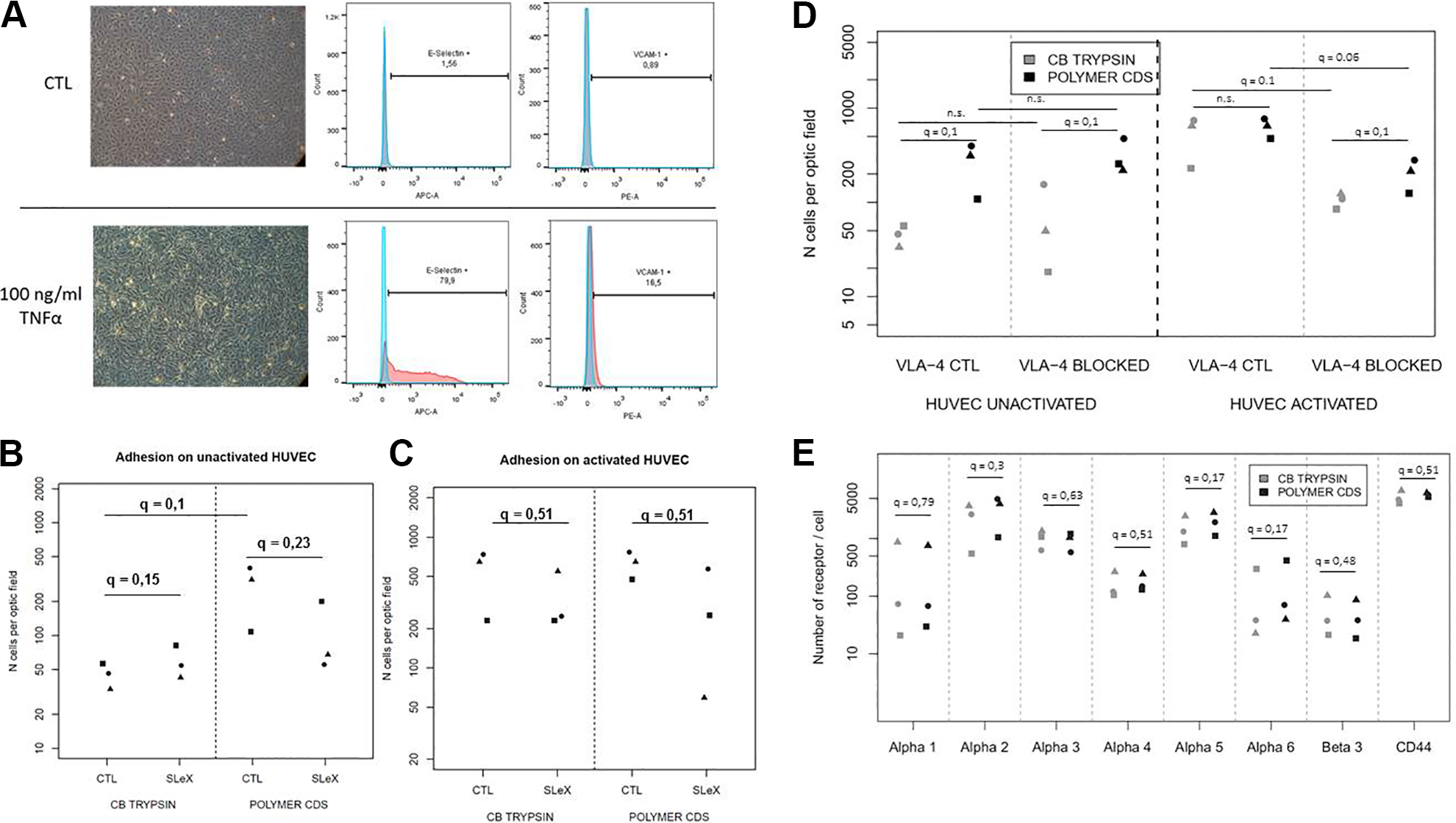
Impact of the different optimizations on adhesion to HUVEC and adhesion molecule expression. Morphology of HUVEC under control and inflammatory conditions (100 ng/ml of TNF-α) and their expression of E-selectin and VCAM-1 assessed by flow cytometry (A). Adhesion of ADHLSCs harvested after polymer CDS and CB trypsin with or without SLeX addition to naive HUVEC-coated µ-slides (B) and activated HUVEC-coated µ-slides (C) (robust average of 30 optic field per ADHLSC donor; circles, squares, and triangles represent different donors, n = 3). Comparison of adhesion between polymer CDS and CB trypsin on naïve or activated HUVECs in the presence or absence of an integrin alpha 4 blocking antibody (robust average of 30 optic field per ADHLSC donor; shapes round, square, and triangle represent different donors, n = 3) (D). Comparison of integrin expression between ADHLSCs harvested after CB trypsin and polymer CDS (number of receptors per ADHLSC donor; shapes round, square, and triangle represent different donors, n = 3) (E).
Culture on Polymer Followed by Harvest with CDS tends to Increase the Expression of Integrin Alpha 2, 5, and 6
The results of the adhesion tests have shown that ADHLSCs after polymer CDS have a higher adhesive capacity to naïve HUVECs than cells after CB and trypsin, which was not inhibited by blocking VLA-4. Moreover, blocking VLA-4 did not completely inhibit the adhesion of ADHLSCs cultivated on polymer and harvested with CDS to activated HUVECs. These results indicate that VL-4 may not be the only receptor mediating adhesion of ADHLSCs to activated and naive HUVECs. To determine if culture on polymer and harvest with CDS had a protective effect on the expression of adhesion molecules, we used the quantiBrite technology to assess the number of integrins and CD44 receptors present at the surface of the cell following both culture and harvesting methods (Fig. 5E). The expression of most adhesion molecules remained constant regardless of the harvesting method. However, we found an increase in the expression of the already highly expressed molecules such as integrin alpha 2 by 1.54-fold change, integrin alpha 5 by 1.31-fold change, and integrin alpha 6 following polymer and CDS. However, these increases were not significant.
Culture on Polymer Followed by Harvest with CDS Does Not Improve cell Adhesion to the Endothelium Ex Vivo
Finally, we wanted to determine whether culture on polymer and harvest with CDS would improve the adhesion of ADHLSCs to the endothelium. To remove confounding elements of an in vivo injection of cells such as the immune response, we perfused the liver with PBS before injecting the cells, and then washed the liver after cell injection to mimic blood flow. The liver was then harvested, fixed, and analyzed by immunohistochemistry using human β-integrin to detect ADHLSCs.The analysis took into consideration whether the cells were adherent (adhering to the endothelium or already in the parenchyma) or circulating. However, we were not able to demonstrate an increase in the percentage of adherent cells in the mice injected with cells grown on polymer and harvested with CDS when compared to the mice injected with cells grown on CB and harvested with trypsin (Fig. 6).

Impact of culture on polymer and harvest with CDS on cell adhesion to the liver endothelium. C57BL/6 mice were injected through the portal vein with 1 million ADHLSCs grown on CellBIND® and harvested with trypsin or grown on thermosensitive polymer and harvested with CDS as described in the Materials and Methods section. The liver was then harvested and fixed and paraffin-embedded sections were then analyzed for the expression of human β-integrin. A percentage of adherent cells was calculated based on the number of adherent cells (adherent and circulating) using the Visiopharm software.
Discussion
Mesenchymal stem/stromal cells have been proposed for the treatment of many diseases, targeting a variety of organs. Regardless of the targeted organs, many groups have reported a low engraftment level of MSCs, and have tried to improve it 7,18,23,24,29 –31 . In systemic infusion of MSCs, researchers are converging toward the hypothesis that the adhesion to the endothelium is the weak point of the engraftment process. In this study, we tried to improve both the cells’ rolling and their adhesion to the endothelium.
First, we have demonstrated that SLeX addition to cells by BNHS–streptavidin bridges increased adhesion/rolling to E-selectin, the selectin expressed on the endothelium. In fact, we found that in the absence of SleX, there was very little adhesion of ADHLSCs to E-selectin, and no adhesion to P-selectin, a result that is in line with those reported by Sheriff et al. showing that adhesion of bone marrow and umbilical cord-derived MSC to these selectins was barely detectable, even at a very low shear rate of 18 s−1 (equivalent to a shear stress of 0.25 dyne/cm2) 32 . Sarkar et al have previously shown that SleX addition decreased the velocity of BM-MSCs, both in a flow chamber assay using slides coated by P-selectin and in vivo, suggesting improved rolling 16 . Contrary to Sarkar et al, we did not observe any improvement of rolling/adhesion on P-selectin, but only on E-selectin. However, SLeX addition to ADHLSCs did not result in an increase in adhesion to activated or naive HUVECs, independently of the harvesting method (CB trypsin or polymer CDS). On the contrary, the addition of SLeX to ADHLSCs decreased their adhesion in three out of the four conditions tested. These results indicate that despite a tendency to increase the rolling of ADHLSCs, addition of SLeX has a negative impact on cell adhesion to endothelial cells. This effect was certainly due to the linking of BNHS, which is not specific and can link all surface proteins, including integrins and other adhesion molecules, and cause a steric hindrance between adhesion proteins of ADHLSCs and endothelial cells. Therefore, we feel that this simple method to enhance rolling has shown its limits. It might be worth trying the HCELL technique promoted by Sackstein et al 19 , which consists in using CD44, which is highly expressed by ADHLSCs 12 , and using an α-1,3-fucosyltransferase preparation to convert CD44 to HCELL. This would allow to avoid steric hindrance and to promote rolling without interfering with the adhesion capacity of ADHLSCs.
On the other hand, to increase cell firm adhesion to the endothelium, we investigated an alternative method to trypsinization and enzymatic dissociation, which have already been described by several groups as having a negative impact on surface protein expression and adhesion 25 –27,33 . We have tested alternative methods of culture and harvest for ADHLSCs, by growing the cells on a thermosensitive polymer and using a nonenzymatic CDS to harvest them. Garg et al have demonstrated that the use of a nonenzymatic cell dissociation buffer increased the expression of the chemokine receptors CCR5, CCR4, CXCR7, and CXCR3, and increased their migration capacity toward their respective ligands 25 . However, they observed a slight decrease in viability consistent with what was found in our study, which they explained by the autophagy of the MSCs after harvesting by CDS, demonstrated by an increase in the autophagy marker—monodansylcadaverine—by flow cytometry. Moreover, Brown et al. have shown that the concentration of trypsin alters the expression of integrins and decreases their adherence 26 . Here we observed that harvesting ADHLSCs with polymer CDS seemed to have a protective impact on highly expressed integrins compared to CB trypsin (Fig. 4B); however, the differences in expression remained nonsignificant for most of the integrins tested, which could be explained by the low number of donors used. Moreover, it is also possible that the trypsin will cut a part of the binding site of an integrin whereas the antibody will still recognize the integrin. Therefore, we may be able to see differences in cell adhesion but not in integrin expression following the use of polymer and CDS. A specific analysis of where the trypsin cuts on the three-dimensional structure of every adhesion molecule could be a key to answering this question. Nevertheless, using a thermosensitive polymer followed by CDS led to an increase in cell adhesion to naive HUVECs under shear stress. Interestingly, adhesion to naive HUVECs was not mediated by VLA-4, contrary to the results found by Steingen et al., the only other study to date that used naive endothelial cells and found a decrease in migration of MSCs after blocking VLA-4 34 . However, it has to be noted that in our hands, naïve HUVECs express very little VCAM-1; therefore, the lack of involvement of VLA-4 in the interactions between ADHLSCs and naïve HUVECs is not surprising. It would be interesting to see the VCAM-1 expression profile of the HUVECs used by Steingen et al. The results found on TNF-α-stimulated HUVECs were closer to what was expected, with a much higher adhesion and an involvement of VLA-4. Indeed, blocking VLA-4 significantly decreases ADHLSCs’ adhesion for both harvesting conditions tested. However, the decrease was more important for ADHLSCs harvested after CB trypsin than polymer CDS (Fig. 4E). Those results supported a potential beneficial effect of polymer CDS, but suggested the involvement of other receptors in addition to VLA-4. The precise molecules for which using polymer and CDS can be beneficial remain to be determined.
Alternative culture and harvest conditions led to a decrease in cell yield. For the CB CDS condition, the lower yield was probably due to remaining undetached cells because the cells had been plated on the same dishes at the same density and the same time as the CB trypsin; only the harvesting solution had changed. For conditions with the thermosensitive polymer, the lower yield is certainly due to the lower proliferation rate combined with the remaining undetached cells.
In addition, culture on polymer alone led to a decrease in cell adhesion to VCAM-1. This decrease was probably due to the aggregate formation that we noticed after culture on polymer. Indeed, it would likely be more difficult for cell aggregates to adhere to the coating compared to single cells, as most of the receptors involved would be hidden.
Despite a slight improvement in the adhesion of ADHLSCs to naïve HUVEC in vitro following culture on polymer and harvest with CDS, we were not able to show a similar improvement in the adhesion of the cells to the endothelium ex vivo.
Human β-integrin was used to detect ADHLSCs as it had been shown to be expressed on ADHLSCs and the antibody does not cross-react with mouse tissues (data not shown).
In these experiments, we chose to perfuse the liver with PBS before cell injection to avoid interference of factors unrelated to adhesion such as an immune response, or a procoagulant effect, as demonstrated previously 35 . In addition, we continuously perfused the liver with PBS following cell injection to mimic blood flow. Even if the timing of each phase could be further optimized, we were able to detect adherent cells in both conditions, so the negative results are not related to insufficient binding time.
Further, as demonstrated in our study, the use of polymer and CDS leads to an overall decrease in cell viability, which may be a problem to determine adhesion ex vivo or in vivo as the dead cells will be unable to adhere. A method to remove dead cells prior to injection such as a density gradient may be useful but difficult to add to a clinical protocol.
Together these results suggest that techniques aimed at improving cell rolling and adhesion such as the addition of SleX to the cell surface or culture on polymer do not improve the adhesion of ADHLSCs and it is questionable whether they would be useful with other MSCs. In addition, these techniques may be difficult to apply to large-scale cultures under good manufacturing practice (GMP) conditions from a technical point of view. Therefore, new alternatives need to be tested.
Although culture on polymer did not improve cell adhesion, this technique may still be of interest to grow cells as sheets that would be applied as a patch, rather than to grow individual cells, as these are difficult to separate from each other. This alternative is currently being tested in a number of applications.
Conclusion
Systemic infusions of MSCs have to circumvent engraftment issues to have a better impact on targeted diseases. Here we studied methods to increase rolling and adhesion of MSCs during the homing phase. We demonstrated that addition of SLeX by BNHS–streptavidin bridges increased rolling on E-selectin; however, it had the concomitant effect of decreasing adhesion to HUVECs, probably due to the random linking of BNHS. On the other hand, we investigated alternative harvesting techniques to trypsinization and demonstrated, during an adhesion test under shear stress, that the combination of a thermosensitive polymer and a CDS could increase cell adhesion to naive HUVECs in a VLA-4-independent manner. However, the same effect was not demonstrated when the cells were injected into the liver. Therefore, further research needs to be performed to find a viable alternative to the use of trypsin.
Footnotes
Ethical Approval
This study was approved by the ethics committees of the St Luc’s University Hospital and the Faculty of Medicine of the Université Catholique de Louvain (approval number JMM/sy/2010/12). Approval from the Belgian Ministry of Health was obtained for the hepatocytes and hepatic stem cells bank.
Statement of Human and Animal Rights
All the experimental procedures involving human tissue or cells were approved by the ethics committees of the St Luc's University Hospital and the Facuty of Medicine of the Université Catholique de Louvain (approval number JMM/sy/2010/12). All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of the University catholique de Louvain, Belgium, and approved by the Commission d’Ethique pour l’Expériementation Animale of the Université catholique de Louvain (approval 2018/UCL/MD/036).
Statement of Informed Consent
Written and signed informed consent was obtained from the donor or next of keen for each human liver used as ADHLSC source in the current study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Pr Etienne Sokal and Dr Mustapha Najimi are founders and scientific advisors for Promethera Biosciences and have founding shares and/or stock options. Dr Catherine Lombard and Joachim Ravau were paid in part by an unrestricted grant from Promethera Biosciences to UCL. Pierre-Edouard Dollet received research funding from Promethera Biosciences.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the FNRS (J.0040.13). Pierre-Edouard Dollet was paid by a grant from Promethera® Biosciences. Joachim Ravau and Catherine Lombard were paid by a PPP grant from the Région Wallonne (1318241) and an unrestricted grant from Promethera® Biosciences.
