Abstract
As the impairment of myocardial microenvironments due to coronary microembolization (CME) compromises the treatment effect of percutaneous coronary intervention and leads to adverse prognosis, we hypothesized that endothelial progenitor cells (EPCs) transplantation could improve cardiac function in the condition of CME.
Low- (2 × 105) and high- (2 × 106) dose rat bone marrow-derived EPCs were transplanted in a model of CME. To develop a CME model, rats were injected with autologous micro-blood-clots into the left ventricle. Echocardiograph was examined before and 1, 7, and 28 days after EPC transplantation; serum cardiac troponin I (cTNI), von Willebrand factor (vWF), and cardiac microRNA expression were examined one day after EPCs transplantation. Heart morphology and vascular endothelial growth factor (VEGF), vWF, and basic fibroblast growth factor (bFGF) expression were examined one day after EPC transplantation.
After 10 days of culture inductions, BM-EPCs have high purity as confirmed by flow cytometry. Cardiac function reflected by left ventricular ejection fraction significantly decreased after CME treatment and rescued by low-dose EPC. Compared to the sham group, cTNI and vWF serum levels increased significantly after CME treatment and rescued by low-dose EPC and high-dose EPC. Low-dose EPC treatment decreased myocardial necrosis and fibrosis and elevated cardiac expression of VEGF and vWF, while decreasing the cardiac expression of bFGF. Low-dose EPC treatment significantly suppressed cardiac expression of microRNA-19a but significantly enhanced microRNA-21, microRNA-214, and microRNA-486-3p expression. In conclusion, our results indicate that low-dose EPC transplantation may play a proangiogenic, antifibroblast, antifibrosis, and antinecrosis role and enhance cardiac function in a rat model of CME through a microRNA-related pathway.
Introduction
Coronary microembolization (CME) is encountered frequently in acute coronary syndromes (ACSs) and during the coronary interventions. CME can lead to a series of adverse cardiac events, such as myocardial contractile dysfunction, arrhythmias, myocardial macroinfarction, or microinfarction, and even death 1 . Considerable efforts have been made to prevent and treat CME, but with limited success 2 . Recently, some investigations showed that transplantation of autologous skeletal myoblasts can improve hemodynamics and left ventricular function in dogs with CME-induced chronic heart failure 3 . Bone marrow-derived stem cell (BMSC) transplantation could not improve cardiac function in CME, but BMSCs transplanted along with rosuvastatin upregulated the vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) expression, increased the capillary density, and improved the cardiac function in a rat model of CME 4 .
There is growing evidence that endothelial progenitor cells (EPCs) are involved in myocardial angiogenesis of ischemic cardiomyopathy patients 5 . In contrast to impaired functionality of BMSCs, EPCs retain a robust proangiogenic profile, both in vitro and in vivo, with therapeutic potential for targeting ischemic disease 5 . Unfortunately, it has not been elucidated whether EPC transplantation could improve cardiac function in CME. Recent studies have suggested that microRNAs (miRNAs) regulate the proliferation, migration, and angiogenesis of EPCs 6 . Therefore, miRNAs may have a role in the biology of EPCs. Specifically, microRNA-19a has been found to promote atherosclerosis through monocyte adhesion 7 , while microRNA-21 has been found to regulate the angiogenic activity of EPC 8 . MicroRNA-214 was identified as a regulator of cardiomyocyte Ca2+ homeostasis and survival during cardiac ischemic injury 9 ; microRNA-486-3P could differentiate patients with ACS from those with stable coronary heart disease 10 . Therefore, it would be interesting to investigate 1) whether EPCs transplantation could improve cardiac performance in case of impaired myocardial environment after CME and 2) whether miRNA is involved in this pathological process.
Materials and Methods
EPC Culture Identification and Counting
The isolation and culture of BM-EPCs were performed as described 11 . Rat bone marrow cells were obtained by flushing femurs and humeri with Dulbecco’s modified Eagle medium (DMEM)/F12 medium (Gibco, New York, NY, USA). BMSCs were then isolated by density gradient centrifugation with the Histopaque 1077 (Sigma-Aldrich, St. Louis, MO, USA) from bone marrow cells. After washing with red blood cell lysis buffer, bone marrow mononuclear cells were seeded into culture flasks in DMEM/F12 medium supplemented with 10% fetal bovine serum (Gibco). After 24 h, the plastic-adherent cells were removed, and the nonadherent cells were collected, washed, and replated into fibronectin-coated (10 μg/ml; BD Biosciences, San Jose, CA, USA) culture flasks with the inducing medium containing DMEM/F12 medium supplemented with 10% fetal bovine serum, 20 ng/ml VEGF, 5 ng/ml basic fibroblast growth factor, 5 ng/ml epidermal growth factor, 10 ng/ml insulin-like growth factor-1 (Peprotech, New Jersey, NJ, USA), and antibiotics (100 U/ml penicillin and 100 μg/ml streptomycin).
Flow Cytometry for Phenotypes of BM-EPCs
After 10 days of culture under inducing conditions, 2 × 106 BM-EPCs were incubated with the FcR blocking reagent (Miltenyi Biotec Inc., Auburn, CA, USA), the fluorescein isothiocyanate-conjugated rabbit anti-rat CD34 antibody (BD Biosciences), and the rabbit anti-rat vascular endothelial growth factor receptor 2 (VEGFR2) antibody (BD Biosciences) for 30 min at 4°C. Then, cells were incubated with phycoerythrin-conjugated goat anti-rabbit secondary antibody (BD Biosciences) for 30 min at 4°C. The phenotypic expression of BM-EPCs was analyzed by flow cytometry (FACS Scan flow cytometer; BD). BMSCs (2 × 106) cultured without induction for 10 days were used as a control.
Animal Modeling and Grouping
Male Wistar rats (weight, 220 to 240 g) were purchased from Vital River Co, Ltd. (Beijing, China) and kept under standard animal room conditions (temperature 21°C ± 1°C, humidity 55% to 60%) with food and water continuously available. Thirty-two rats were randomized into four groups: sham, CME, CME+EPC (low), and CME+EPC (high) (n = 8 per group). The present study established a CME model by injecting rat autologous micro-blood-clots (passed through a 300-mesh filter screen) into the left ventricle (LV). Briefly, a left lateral thoracotomy was undertaken in rats at the third and fifth intercostal space. The pericardium was opened, and the ascending aorta was exposed fully. A suspension of 300 μl microembolism was injected into the LV during 10-s occlusion of the ascending aorta. Rats in the CME+EPC (low) group received an injection of 2 × 105 EPC along with 300 μl microembolism, rats in the CME+EPC (high) group received an injection of 2 × 106 EPC along with 300 μl microembolism, and rats in the sham group received an injection of the same volume of normal saline only.
Cardiac Function Monitoring
Echocardiography was employed to assess cardiac function before, 1 day, 7 days, and 28 days after treatment. All of the echocardiographic examinations including the left ventricular ejection fraction (LVEF), the left ventricular fractional shortening (FS), the cardiac output, and the left ventricular end-diastolic diameter (LVEDD) of the rats in each group were performed at a probe frequency of 10 MHz by an experienced professional physician. All of the measures were expressed as the average of three heart beat cycles.
Tissue Sampling and Sample Treatment
After experimentation, the hearts were arrested by injecting 2 ml 10% potassium chloride into the tail vein. The hearts were isolated and cleaned with cold normal saline immediately. The atrial appendage and atrium cordis were removed, and parts of heart ventricle were rapidly frozen in liquid nitrogen and stored at −80°C for immunohistochemical staining of VEGF and bFGF. The others were fixed in 4% paraformaldehyde for 12 h, embedded in paraffin and serially sectioned into slices of 4-µm thickness for hematoxylin and eosin staining and Masson staining. The serum levels of cardiac troponin I (cTNI) and von Willebrand factor (vWF) were examined with ELISA kits (RTFI00698 and RTFI00252, ELISAGENIE, Dublin, Ireland) through tail vein blood.
RNA Extraction and Quantitative Reverse Transcriptase Polymerase Chain Reaction
Total RNA was extracted from cardiac tissue using the TRIzol reagent (Gibco) according to the protocols supplied by the manufacturers. The concentration of RNA was quantified by a NanoDrop (Thermo Fisher Scientific, Waltham, USA) and then subjected to reverse transcription using a cDNA reverse transcription kit (TaKaRa, Japan) according to the manufacturer’s instructions. Then, the obtained cDNA was subjected to RT-qPCR for microRNA-21, microRNA-19a, microRNA-214, and microRNA-486-3p mRNA using an SYBR Green I PCR kit (TaKaRa, Japan). The conditions for all of the RT-qPCR reactions were performed on the ABI PRISM 7500 system (Applied BioSystems, USA). The sequences of the primers were designed as follows: microRNA-21 forward: TCCACAACAGCAGTCGATGG and reverse: GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGACAGC; microRNA-19a forward: CCATGTGCAAATCTATGCAA and reverse: GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACTCAGTT; microRNA-214 forward: CAACAGCAGGCACAGAC and reverse: GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCTGCCT; microRNA-486-3p forward: TCCATCCTGTACTGAGCTGC and reverse: GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCTCGGG; U6-1 forward: TCGCTTCGGCAGCACATA and reverse: TTTGCGTGTCATCCTTGC. The relative quantification of miRNA expression was calculated using the 2−ΔΔCt method and was normalized to U6-1.
Statistical Analysis
All of the data are presented as mean ± SD. Differences between multiple groups were analyzed by one-way analyses of variance followed by Student–Neuman–Keuls or Dunnett tests, using Prism software (GraphPad Prism version 5.0). All of the experiments were performed independently in triplicate. Values of P <0.05 were considered to be statistically significant.
Results
Phenotype and Purity of BM-EPCs
After 10 days of culture inductions, BM-EPCs were adherent (Figure S1A). After 10 days, the expressions of CD34 and VEGFR2 were detected (Figure S1B). The coexpression level was 98.96% ± 1.35%, which was consistent with previous reports, suggesting high purity of EPC used in our experiment.
Changes in Cardiac Function
Compared to the sham group, LVEDD showed no significant difference in CME, CME+low-dose EPC, or CME+high-dose EPC (Figure 1A), whereas LVESD significantly increased 1 day after CME treatment comparing with the sham group (CME 0.316 ± 0.039 cm vs sham 0.188 ± 0.021 cm, P = 0.0021) and rescued by low-dose EPC (CME 0.316 ± 0.039 cm vs CME+EPC (low) 0.21 ± 0.023 pg/ml, P = 0.0093) (Figure 1B). Cardiac function reflected by LVEF significantly decreased 28 days after CME treatment comparing with sham group (CME 88.86% ± 1.89% vs sham 97.38% ± 2.27%, P < 0.0001) and rescued by low-dose EPC (CME 88.86% ± 1.89% vs CME+EPC (low) 93.88% ± 1.94%, P = 0.0115) (Figure 1C). Cardiac function reflected by LVFS significantly decreased 1 day after CME treatment comparing with sham group (CME 57.86% ± 4.73% vs sham 72.26% ± 5.47%, P = 0.0043) and rescued by low-dose EPC (CME 57.86% ± 4.73% vs CME+EPC (low) 68.8% ± 4.94%, P = 0.0265) (Figure 1C). These results suggest that pretreatment with low-dose EPC improved cardiac function in this rat model of CME.
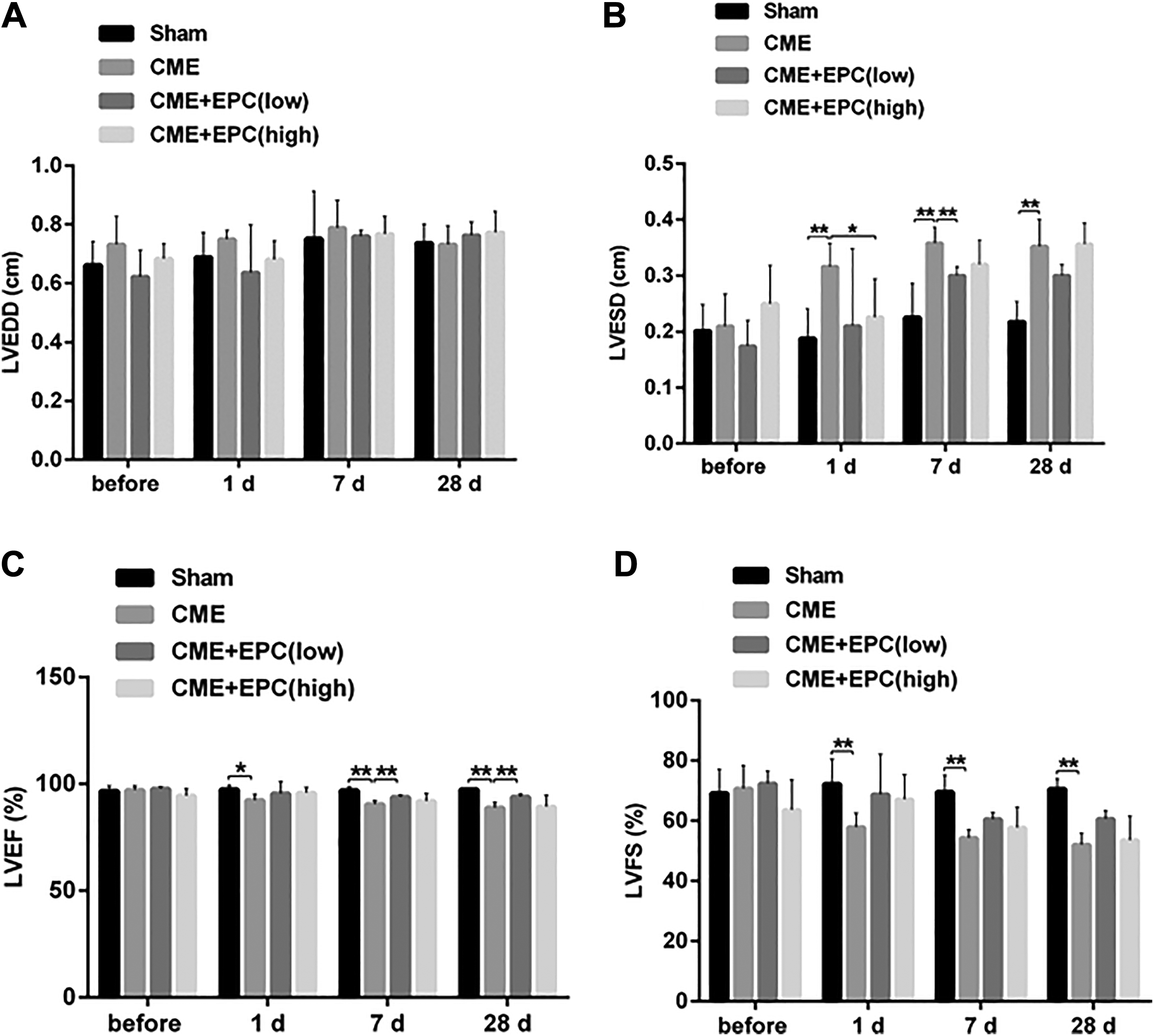
Effects of CME and EPC on cardiac function of rats. An echocardiography was employed to assess cardiac function before, 1 day, 7 days, and 28 days after treatment; A. LVEDD; B. LVESD; C. LVEF; D. LVFS. *P < 0.05, **P < 0.01. CME: coronary microembolization; EPC: endothelial progenitor cell; LVEDD: left ventricular end-diastolic diameter; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic diameter; LVFS: left ventricular fractional shortening.
Examination of Serum Markers
Compared to the sham group, cTNI serum level increased significantly one day after CME treatment (CME 34.1 ± 1.79 pg/ml vs sham 26.8 ± 1.46 pg/ml, P = 0.0017) and were rescued by low-dose EPC [CME 34.1 ± 1.79 pg/ml vs CME+EPC (low) 28.7 ± 1.82 pg/ml, P = 0.0108] (Figure 2A), vWF serum level increased significantly one day after CME treatment (CME 2,532 ± 179.5 pg/ml vs sham 1,196 ± 146.2 pg/ml, P < 0.0001) and were rescued by low-dose EPC and high-dose EPC [CME 2,532 ± 179.5 pg/ml vs CME + EPC (low) 1,276 ± 137.5 pg/ml, P < 0.0001] (Figure 2B). These results suggest that treatment with low-dose EPC reduced cardiac and vascular injury in the rat model of CME.

Effects of CME and EPC on serum markers. (A) cTNI; and (B) vWF serum levels were examined by ELISA before and 1 day after treatment. *P < 0.05, **P < 0.01. CME: coronary microembolization; cTNI: cardiac troponin I; EPC: endothelial progenitor cell; vWF: von Willebrand factor.
Morphology and Immunohistochemistry of Rat Hearts After CME and EPC Treatment
H & E staining discovered myocardial necrosis and swelling after CME treatment, while low-dose EPC treatment rescued myocardial damage (Figure 3A). Masson staining revealed cardiac fibrosis after CME treatment, which was significantly attenuated by low-dose EPC but not high-dose EPC (Figure 3B). Immunohistochemistry showed significantly decreased expression of VEGF and F8 after CME treatment, while low-dose EPC treatment elevated cardiac expression of VEGF and F8 (Figure 3C, E). In contrast, bFGF expression increased after CME treatment, while low-dose EPC treatment decreased the cardiac expression of bFGF (Figure 3D).
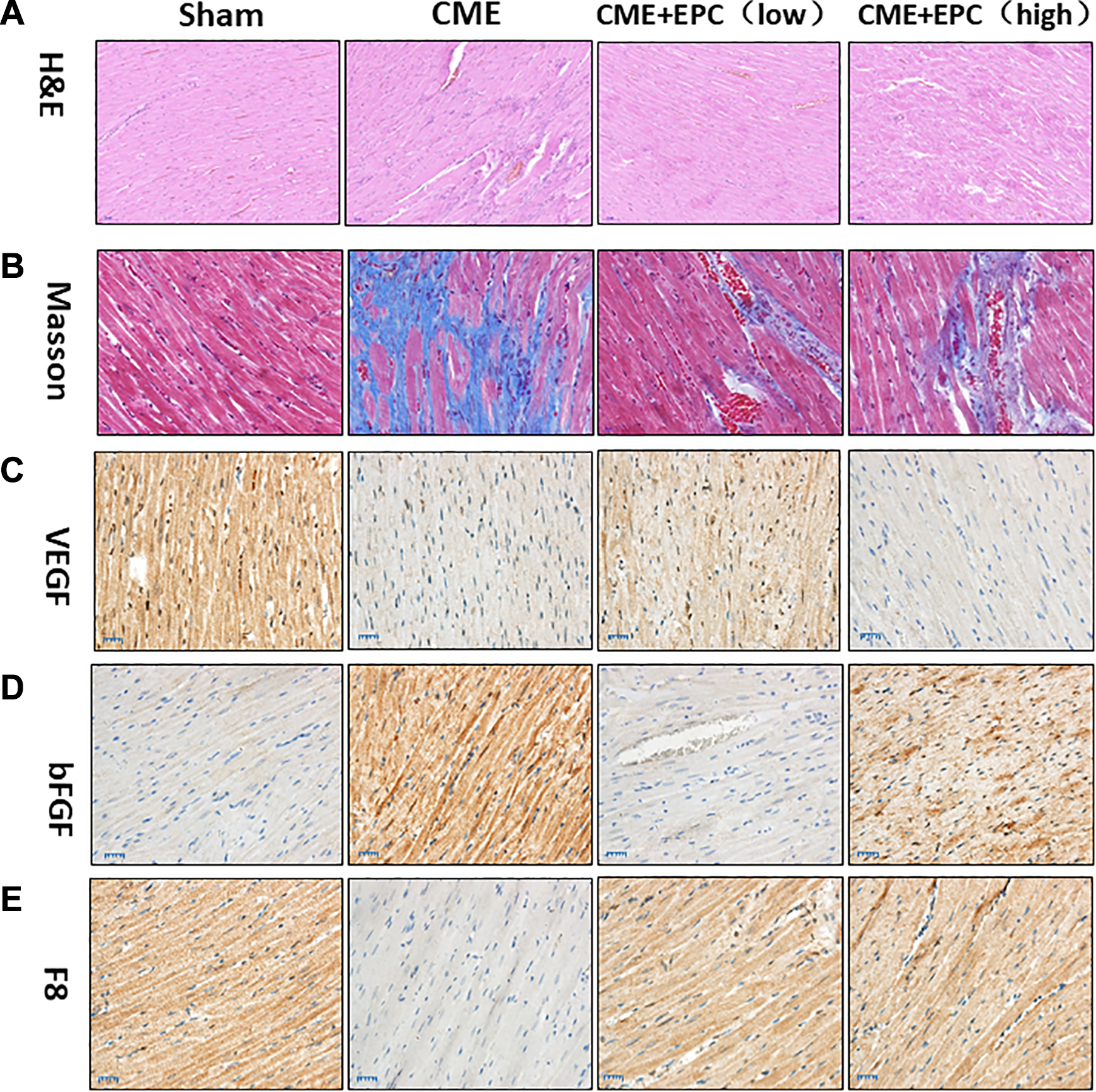
Morphology and immunohistochemistry of rat hearts after CME and EPC treatment. Representative images of (A) hematoxylin-eosin staining; (B) Masson’s trichrome staining, and immunohistochemical staining of (C) VEGF; (D) bFGF; (E) F8 28 days after treatment. Sham = sham surgery, CME = rats receiving CME surgery, CME+EPC (low) = rats receiving CME surgery as well as low-dose EPC transplantation, CME+EPC (high) = rats receiving CME surgery as well as high-dose EPC transplantation. Bars: 50 µm in A and C, Bars: 25 µm in D and E, Bars: 20 µm in B. bFGF: basic fibroblast growth factor; CME: coronary microembolization; EPC: endothelial progenitor cell; H&E: hematoxylin and eosin; VEGF: vascular endothelial growth factor.
Expression of miRNA
RT-qPCR analysis was performed to observe the expression of miRNA in the rat CME model. Compared with the sham group, CME treatment significantly enhanced expression of myocardial microRNA-19a (CME 1.92 ± 0.06 vs sham 1.00 ± 0.04, P < 0.0001) but significantly suppressed microRNA-21(CME 0.155 ± 0.03 vs sham 1.00 ± 0.04, P < 0.0001), microRNA-214 (CME 0.787 ± 0.06 vs sham 1.00 ± 0.04, P = 0.0105), and microRNA-486-3p (CME 0.655 ± 0.04 vs sham 1.00 ± 0.04, P < 0.0001) expression. Meanwhile, low-dose EPC treatment significantly suppressed cardiac expression of microRNA-19a [CME 1.92 ± 0.06 vs CME+EPC (low) 1.31 ± 0.06, P < 0.0001] but significantly enhanced microRNA-21[CME 0.155±0.03 vs CME+EPC (low) 0.798±0.03, P < 0.0001], microRNA-214 [CME 0.787 ± 0.06 vs CME+EPC (low) 0.966 ± 0.06, P = 0.0236], and microRNA-486-3p [CME 0.655 ± 0.04 vs CME+EPC (low) 0.955 ± 0.05, P = 0.0001] expression (Figure 4).

Effects of CME and EPC on cardiac microRNA expression. RT-qPCR was employed to assess cardiac microRNA expressions one day after treatment. (A) microRNA-19a expression; (B) microRNA-21 expression; (C) microRNA-214 expression; (D) microRNA-486-3p expression. *P < 0.05, **P < 0.01. CME: coronary microembolization; EPC: endothelial progenitor cell.
Discussion
The major findings of our study were that (1) the transplantation of low-dose (2 × 105) EPCs notably improved cardiac function in the pathological condition of CME; (2) the transplantation of low-dose (2 × 105) EPCs could significantly reduce cardiac injury, as revealed by serum cTNI and vWF levels; (3) low-dose EPC treatment decreased myocardial necrosis and fibrosis; (4) low-dose EPC treatment elevated cardiac expression of VEGF and F8 while decreasing the cardiac expression of bFGF; and (5) low-dose EPC treatment significantly suppressed cardiac expression of microRNA-19a but significantly enhanced microRNA-21, microRNA-214, and microRNA-486-3p expression. To the best of our knowledge, the present work is the first study to demonstrate that EPC transplantation can enhance cardiac function in the setting of CME.
According to the “seed and soil” hypotheses, the fate of transplanted cells in vivo is determined primarily by the donor cells and the myocardial microenvironment of the host. It has been demonstrated that the transplanted BMSCs did not survive well in the host myocardium, with cell death and inability to differentiate into active cell types being the major problems; this ultimately resulted in the limited therapeutic effects of transplanted BMSCs 12 . Whereas in 1997, Asahara et al. 13 proposed that purified CD34+ hematopoietic progenitor cells from adults can differentiate ex vivo into an endothelial phenotype. These cells were named “endothelial progenitor cells”, which showed expression of various endothelial markers, homing, and incorporating into neovessels at sites of ischemia. It has been well characterized that infusion of EPCs either isolated from the bone marrow or by ex vivo cultivation could augment capillary density and neovascularization of ischemic tissue 14 . According to previous reports, EPCs obtained by our procedure share the same characteristics with early outgrowth EPCs, including surface markers CD 133 11 , and possess the ability to migrate, proliferate, differentiate into endothelial cells, form tube-like structure in vitro 11,14,15 , and promote angiogenesis in vivo 16 . It has been shown that the proper amount of EPCs’ transplantation to promote angiogenesis in a rat model should be around 2 × 105 to 2 × 106, without obstruction of coronary microcirculation 4,17 – 20 . We found low-dose, but not high-dose, EPC transplantation exhibited proangiogenic (increased cardiac VEGF and F8 expression), antifibroblast (reduced cardiac bFGF expression), antifibrosis, and antinecrosis characteristics, and improved cardiac function as a result of the pathological process of CME.
CME is defined as coronary microcirculation thrombosis and microinfarction caused by atherosclerotic plaque rupture or microemboli in patients undergoing PCI 21 . CME mainly represents the unfavorable microenvironment that accompanies ACS and cannot be completely avoided during reperfusion therapy. In addition to possessing the pathophysiology of ACS, CME is characterized by acute microcirculation dysfunction and impaired myocardial perfusion as evidenced by “no-flow” or “low-flow,” which usually leads to an adverse prognosis. Our results showed that low-dose EPC treatment significantly suppressed cardiac expression of microRNA-19a, which has been found to promote atherosclerosis through monocyte adhesion. Low-dose EPC treatment also significantly enhanced microRNA-21, microRNA-214, and microRNA-486-3p expression, which was reported to regulate the angiogenic activity of EPC and cardiomyocyte survival during cardiac ischemic injury. Taken together, these results suggest that EPCs protect the heart from CME probably through miRNA-related mechanisms.
For better interpretation of the results, some limitations in this study should be acknowledged. First, the results were derived from a rat CME model, which was made by injecting autologous micro-blood-clots into the LV. Therefore, the current results may not be directly comparable with those obtained in vivo microembolization in patients. Additionally, we only investigated the short-term protective effects of EPCs transplantation. Thus, it is uncertain how long this protective effect would be sustained. Finally, this study is a proof of concept that EPCs transplantation may improve cardiac function in the condition of CME, only two different doses were included, and we will examine the impact of different doses ECPs in future studies.
In conclusion, our results indicate that low-dose EPCs transplantation may play a proangiogenic, antifibroblast, antifibrosis, and antinecrosis role and enhance cardiac function in a rat model of CME through an miRNA-related pathway.
Supplemental Material
Supplemental Material, FigureS1 - Transplantation of Endothelial Progenitor Cells in the Treatment of Coronary Artery Microembolism in Rats
Supplemental Material, FigureS1 for Transplantation of Endothelial Progenitor Cells in the Treatment of Coronary Artery Microembolism in Rats by Yajun Xue, Boda Zhou, Jian Wu, Guobin Miao, Kun Li, Siyuan Li, Jie Zhou, Yu Geng and Ping Zhang in Cell Transplantation
Supplemental Material
Supplemental Material, Figure_S2 - Transplantation of Endothelial Progenitor Cells in the Treatment of Coronary Artery Microembolism in Rats
Supplemental Material, Figure_S2 for Transplantation of Endothelial Progenitor Cells in the Treatment of Coronary Artery Microembolism in Rats by Yajun Xue, Boda Zhou, Jian Wu, Guobin Miao, Kun Li, Siyuan Li, Jie Zhou, Yu Geng and Ping Zhang in Cell Transplantation
Footnotes
Ethical Approval
Ethical approval to report this case was obtained from Animal Care and Use Committee of Beijing Tsinghua Changgung Hospital (2016-KY-21).
Statement of Human and Animal Rights
All of the procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Tsinghua University, China and approved by Animal Care and Use Committee of Beijing Tsinghua Changgung Hospital (2016-KY-21).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: We confirm that guidelines on animal rights and treatment have been met and any details of approval obtained are indicated within the text of the submitted manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the State Key Laboratory of Low-Dimensional Quantum Physics Open Project Fund (No. KF201608); Beijing Hospitals Authority Ascent Plan (No. DFL20190902).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
